In most mammals, the sex of the gonads is based on the fate of the supporting cell lineages, which arises from the proliferation of coelomic epithelium (CE) that surfaces on the bipotential genital ridge in both XY and XX embryos. Genetic studies and single-cell transcriptome analyses in mice have revealed the cellular and molecular events in the two-wave proliferation of the CE that produce the supporting cells. This proliferation contributes to the formation of the primary sex cords in the medullary region of both the testis and the ovary at the early phase of gonadal sex differentiation, as well as to that of the secondary sex cords in the cortical region of the ovary at the perinatal stage. To support gametogenesis, the testis forms seminiferous tubules in the medullary region, whereas the ovary forms follicles mainly in the cortical region. The medullary region in the ovary exhibits morphological and functional diversity among mammalian species that ranges from ovary-like to testis-like characteristics.
- sex differentiation
- testis
- ovary
- folliculogenesis
- cortex
- medulla
1. Early Gonadal Supporting Cell Development in Mice
- (1)(1)
-
Origin of Gonadal Supporting Cells from the Coelomic Epithelium
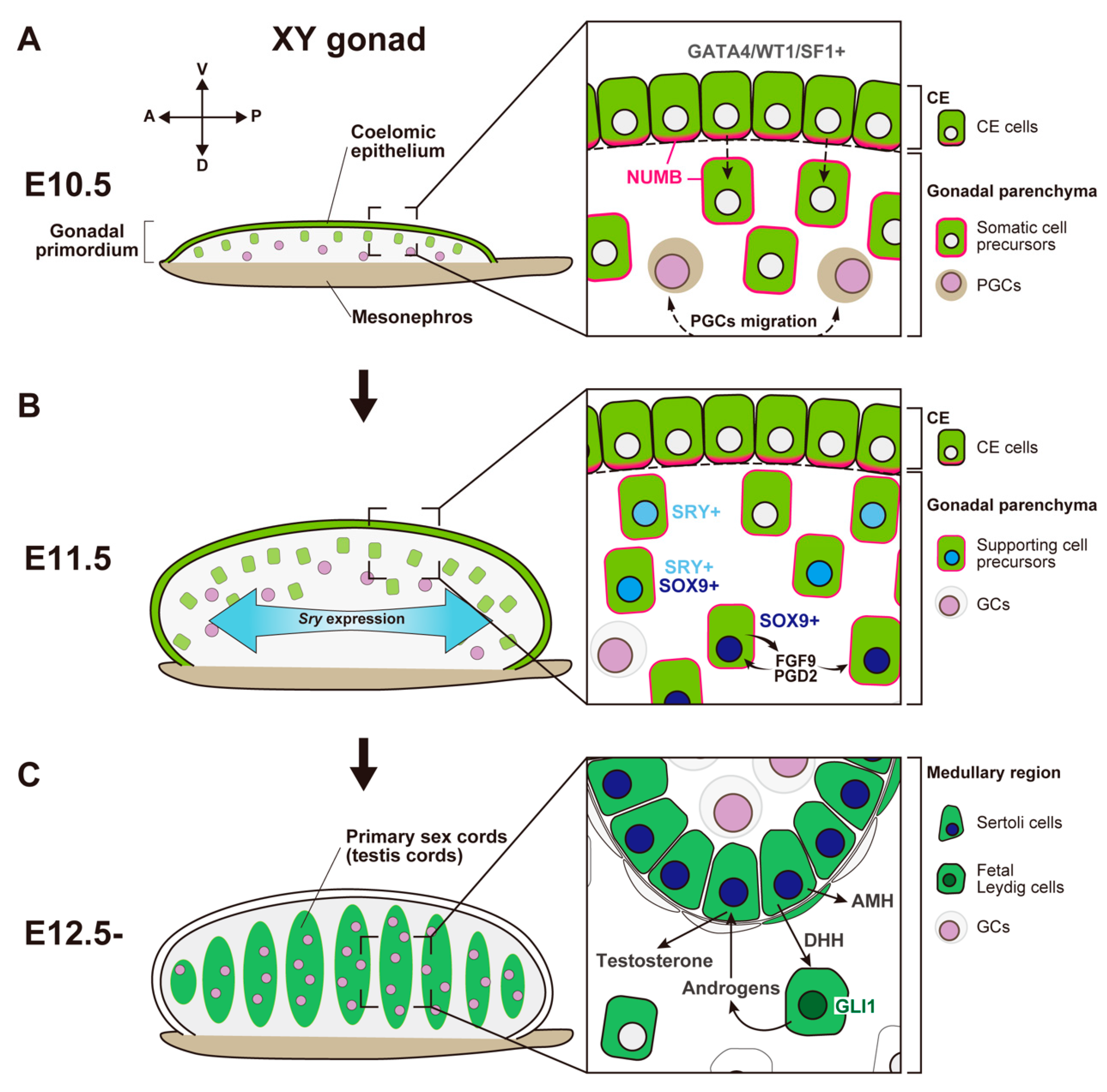 Figure 1. Schematic representation showing morphological change and somatic cell differentiation during the sex determination period, from the bipotential gonadal primordium at E10.5 to the differentiated testis at E12.5. (A) At E10.5, the gonadal primordium appears as a long and narrow structure composed of the coelomic epithelium (CE) and the migrated CE cells (somatic cell precursors). CE cells express GATA4/WT1/SF1, and their ingression and asymmetric cell division are primarily controlled by Notch signaling with the NUMB distribution (magenta) and Notch signaling. PGCs migration occurs around E10.5. (B) By E11.5, the gonadal primordium expands along the dorsoventral axis via the ingression of CE cells. Testis-specific Sry expression occurs in a center-to-pole manner along the anteroposterior axis. Beneath the CE, supporting cell precursors, SRY single- (cyan nuclei), SRY/SOX9 double- (blue nuclei), and SOX9 single- (deep-blue nuclei) positive cells, are distributed from the CE (dorsal)-to-mesonephric (ventral) side. SOX9-positive Sertoli cells secrete FGF9 and PGD2, and these factors upregulate and maintain Sox9 expression in the own and neighboring supporting cell precursors. (C) At E12.5, testis cords are formed by Sertoli cells and germ cells (GCs). SOX9-positive Sertoli cells secrete paracrine factors, such as AMH and DHH. DHH signaling activates its downstream factor, GLI1, in fetal Leydig cell progenitors. Fetal Leydig cells with activated GLI1 (deep-green nuclei) produce androgens, which are converted to testosterone by Sertoli cells and then induce proper differentiation of the internal and external genital tract.
Figure 1. Schematic representation showing morphological change and somatic cell differentiation during the sex determination period, from the bipotential gonadal primordium at E10.5 to the differentiated testis at E12.5. (A) At E10.5, the gonadal primordium appears as a long and narrow structure composed of the coelomic epithelium (CE) and the migrated CE cells (somatic cell precursors). CE cells express GATA4/WT1/SF1, and their ingression and asymmetric cell division are primarily controlled by Notch signaling with the NUMB distribution (magenta) and Notch signaling. PGCs migration occurs around E10.5. (B) By E11.5, the gonadal primordium expands along the dorsoventral axis via the ingression of CE cells. Testis-specific Sry expression occurs in a center-to-pole manner along the anteroposterior axis. Beneath the CE, supporting cell precursors, SRY single- (cyan nuclei), SRY/SOX9 double- (blue nuclei), and SOX9 single- (deep-blue nuclei) positive cells, are distributed from the CE (dorsal)-to-mesonephric (ventral) side. SOX9-positive Sertoli cells secrete FGF9 and PGD2, and these factors upregulate and maintain Sox9 expression in the own and neighboring supporting cell precursors. (C) At E12.5, testis cords are formed by Sertoli cells and germ cells (GCs). SOX9-positive Sertoli cells secrete paracrine factors, such as AMH and DHH. DHH signaling activates its downstream factor, GLI1, in fetal Leydig cell progenitors. Fetal Leydig cells with activated GLI1 (deep-green nuclei) produce androgens, which are converted to testosterone by Sertoli cells and then induce proper differentiation of the internal and external genital tract.- (2)
-
SRY-Mediated Primary Sex Determination
2. Molecular and Cellular Events in Ovarian Somatic Cells
- (1)
-
Female Fate Determination in Somatic Supporting Cells in the Early Phase of Ovarian Development
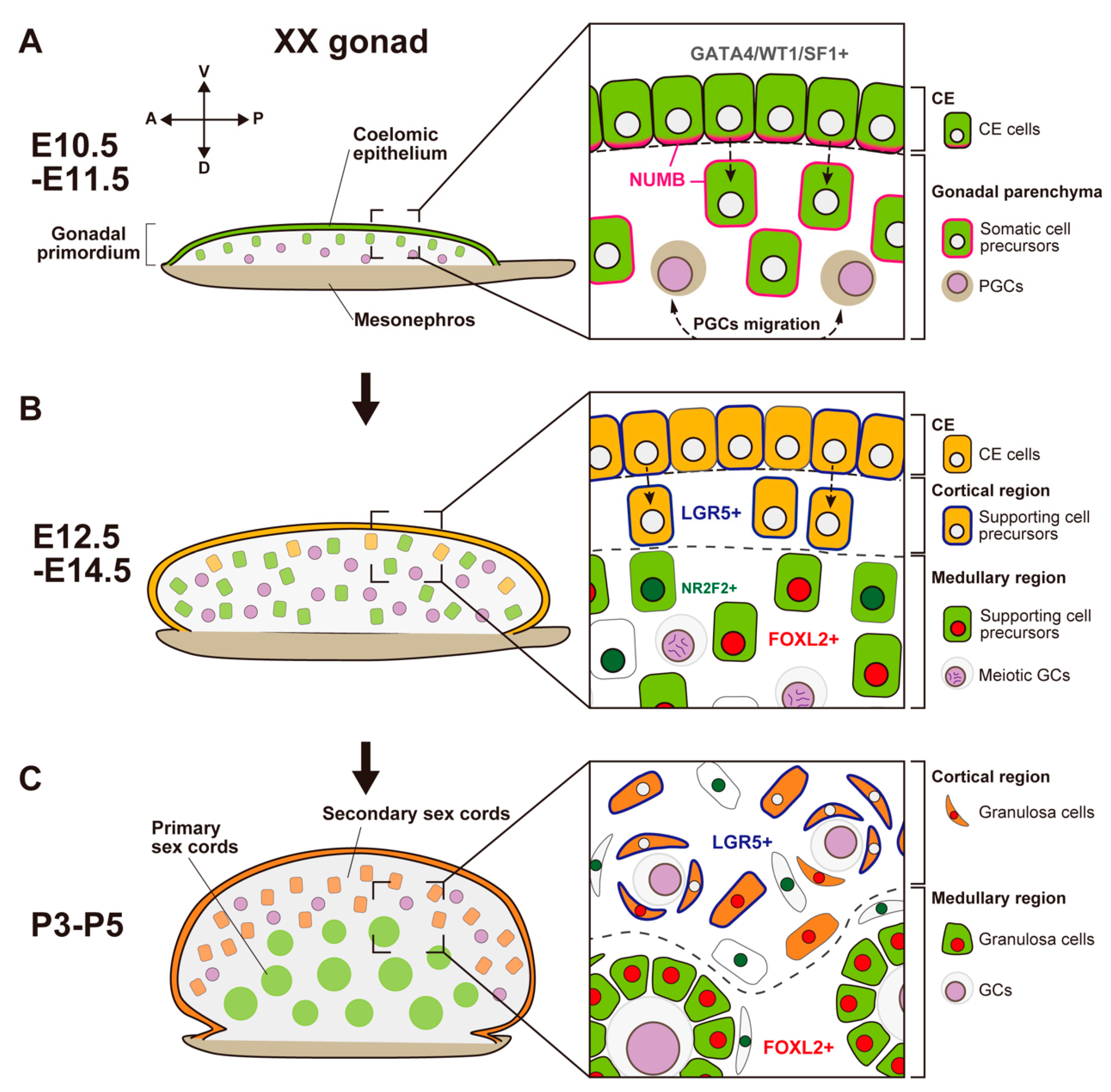
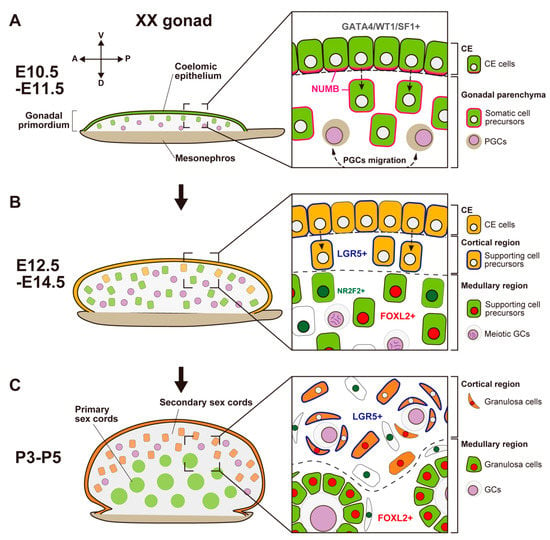 Figure 2. Schematic representation showing morphological changes and somatic cell differentiation along the cortical-medullary axis during fetal and postnatal stages, from the bipotential gonadal primordium at E10.5 to the differentiated ovary around P5. (A) At E10.5, the CE cells proliferate and ingress into the gonadal parenchyma, controlled by the NUMB distribution (magenta) and Notch signaling, to form primary sex cords without sexual dimorphism. (B) At E12.5, supporting cell precursors in the medullary region (green cells) express an early ovarian factor, FOXL2 (red). In contrast to testis differentiation, the proliferation and ingression of the CE in XX gonads continues after E12.5, leading to the formation of secondary sex cords in the cortical region (orange cells). Supporting cell precursors in secondary sex cords, including CE cells, express the transmembrane receptor LGR5 (blue). LGR5 and FOXL2 expression is mutually exclusive, and it exhibits a gradient from the cortex (CE side) to the medulla (mesonephric side). Other somatic cells (i.e., interstitial precursor cells) also exclusively express NR2F2 (deep green). Germ cells (GCs) initiate meiosis at E12.5 (deep purple, condensed chromosomes). (C) At postnatal stages, FOXL2-positive granulosa cells in primary sex cords in the medullary region contribute to the formation of the first wave of follicles. In the cortical region, granulosa cells that originate from LGR5-positive cells (orange cells) form the secondary sex cords and primordial follicle pools. After sexual maturity, these follicles develop according to the estrous cycle.In mouse XX gonads, the ovarian-specific transcriptional program begins around E11.5 [77,92,93,94][45][58][59][60]. Forkhead box L2 (FOXL2) is an early ovarian factor that is essential for the fate and phenotype of granulosa cells [95,96,97,98][61][62][63][64]. FOXL2 expression is induced later than the activation of the WNT4/β-catenin pathway [77][45]; the loss of Ctnnb1 (β-catenin) decreases FOXL2 expression, whereas the stabilization of β-catenin in XY gonads induces FOXL2 expression [90][65]. These results suggest that RSPO1/WNT4/β-catenin signaling is essential for FOXL2 upregulation in granulosa cells.There are functional differences between mice and other mammalian species in relation to the role of Foxl2 and related genes in ovarian development. In goats, FOXL2 is a key female sex-determining gene [105,106][66][67]. FOXL2 is a gene responsible for polled intersex syndrome (PIS), which leads to XX female-to-male sex reversal associated with the absence of horn growth [106][67]. In the fetal XX gonads of FOXL2 knock-out goats, SOX9 and DMRT1 are upregulated in Sertoli-like cells, forming testicular cords [105][66]. In mice, in contrast, the loss of Foxl2 in XX gonads does not induce an appreciable sex-reversal phenotype at the fetal stage and birth, although it results in the transdifferentiation of granulosa cells into Sertoli-like cells in the postnatal stage [96,98,107][62][64][68].
Figure 2. Schematic representation showing morphological changes and somatic cell differentiation along the cortical-medullary axis during fetal and postnatal stages, from the bipotential gonadal primordium at E10.5 to the differentiated ovary around P5. (A) At E10.5, the CE cells proliferate and ingress into the gonadal parenchyma, controlled by the NUMB distribution (magenta) and Notch signaling, to form primary sex cords without sexual dimorphism. (B) At E12.5, supporting cell precursors in the medullary region (green cells) express an early ovarian factor, FOXL2 (red). In contrast to testis differentiation, the proliferation and ingression of the CE in XX gonads continues after E12.5, leading to the formation of secondary sex cords in the cortical region (orange cells). Supporting cell precursors in secondary sex cords, including CE cells, express the transmembrane receptor LGR5 (blue). LGR5 and FOXL2 expression is mutually exclusive, and it exhibits a gradient from the cortex (CE side) to the medulla (mesonephric side). Other somatic cells (i.e., interstitial precursor cells) also exclusively express NR2F2 (deep green). Germ cells (GCs) initiate meiosis at E12.5 (deep purple, condensed chromosomes). (C) At postnatal stages, FOXL2-positive granulosa cells in primary sex cords in the medullary region contribute to the formation of the first wave of follicles. In the cortical region, granulosa cells that originate from LGR5-positive cells (orange cells) form the secondary sex cords and primordial follicle pools. After sexual maturity, these follicles develop according to the estrous cycle.In mouse XX gonads, the ovarian-specific transcriptional program begins around E11.5 [77,92,93,94][45][58][59][60]. Forkhead box L2 (FOXL2) is an early ovarian factor that is essential for the fate and phenotype of granulosa cells [95,96,97,98][61][62][63][64]. FOXL2 expression is induced later than the activation of the WNT4/β-catenin pathway [77][45]; the loss of Ctnnb1 (β-catenin) decreases FOXL2 expression, whereas the stabilization of β-catenin in XY gonads induces FOXL2 expression [90][65]. These results suggest that RSPO1/WNT4/β-catenin signaling is essential for FOXL2 upregulation in granulosa cells.There are functional differences between mice and other mammalian species in relation to the role of Foxl2 and related genes in ovarian development. In goats, FOXL2 is a key female sex-determining gene [105,106][66][67]. FOXL2 is a gene responsible for polled intersex syndrome (PIS), which leads to XX female-to-male sex reversal associated with the absence of horn growth [106][67]. In the fetal XX gonads of FOXL2 knock-out goats, SOX9 and DMRT1 are upregulated in Sertoli-like cells, forming testicular cords [105][66]. In mice, in contrast, the loss of Foxl2 in XX gonads does not induce an appreciable sex-reversal phenotype at the fetal stage and birth, although it results in the transdifferentiation of granulosa cells into Sertoli-like cells in the postnatal stage [96,98,107][62][64][68].- (2)
-
Secondary Population of Granulosa Cells in the Cortical Region of the OvaryIn contrast to mouse XY gonads, the CE in XX gonads continuously exhibits proliferation, ingression, and expansion [102][69]. This contributes to the formation of ovigerous cords that consist of female germ cells and the surrounding pregranulosa cells by the perinatal stage. These cords are regarded as secondary sex cords or ovarian cords (Figure 2B).In the fetal to adult stages, LGR4 and LGR5, receptors for RSPO1, are expressed in the cortical region of XX gonads, including the proliferative region of the ovarian surface epithelium (Figure 2B) [115,116][70][71]; the expression of LGR5 after E12.5 is dependent on RSPO1/WNT4/β-catenin signaling [116][71]. LGR4 and LGR5 are markers for tissue stem cells, such as stem cells in the mammary gland, intestine, and hair follicle [117,118,119,120][72][73][74][75]. LGR4 and LGR5 are also markers of stem/progenitor cells in the ovarian surface epithelium, which generate new granulosa cells that contribute to cortical follicles until birth (Figure 2B) [116,121][71][76]. In the postnatal ovary after the cessation of granulosa cell recruitment, Lgr5 is restricted to stem cells in the ovarian surface epithelium; moreover, Lgr5 promotes the regenerative repair of ovulatory wounds in the adult ovary [115][70]. In both XX and XY gonads, RSPO1/WNT4/β-catenin signaling is involved in the proliferation of the CE in the early stage of gonadogenesis [74][42]. In contrast, the expression levels of LGR4 and LGR5 are higher in XX gonads compared with XY gonads after E12.5; this timing coincides with the ovarian-specific proliferation of the CE throughout the fetal and perinatal stages (Figure 2B,C).
- (3)
-
Cortical–Medullary Regionality of Folliculogenesis Waves
3. Diversity of Ovarian Organogenesis along the Cortical–Medullary Axis
In the testis developmental pathway, the male-specific gene cascade (SRY, SOX9, and AMH) is highly conserved among mammals. In contrast, ovarian organogenesis—such as the formation of secondary sex cords, the initiation of meiosis in germ cells, and the timing of folliculogenesis—exhibits considerable diversity among mammalian species. To understand this diversity, it may be helpful to focus on the regionalization of the medullary region (the primary sex cords) and the cortical region (the secondary sex cords), which correspond to the region of future folliculogenesis in the ovary (Figure 3A). Indeed, the morphological characteristics of the ovarian medullary region exhibit considerable diversity among mammal species. Figure 3. Sexual differentiation of bipotential gonads and the histological variations of ovaries in the indicated species. (A) Bipotential gonads differentiate into testes or ovaries. In mammalian testes, primary sex cords form tubular structures (seminiferous tubules (deep blue) with germ cells (yellow dots)) and the rete testis (cyan), which provide a route for continuous sperm transportation from the seminiferous tubules to the efferent ducts. Interstitial components are shown in gray. Mammalian ovaries commonly develop secondary sex cords with germ cells in the cortical region as a site of future folliculogenesis (orange; yellow dots, germ cells). However, the morphological characteristics of primary sex cords in the medullary region of the ovary show considerable diversity among mammals. (B) In the ovaries of goats and cows, secondary sex cords in the cortical region form during the fetal period. Germ cells in the medullary region disappear and the primary sex region regresses. In the ovaries of mice, secondary sex cords in the cortical region are not distinct in the fetal period; germ cells in the medullary region are maintained and develop immediately after birth as the first wave of follicles (pale-orange circles with yellow dots). In the ovaries of spotted hyenas and most mole species, the medullary region develops as a male-like tissue with Leydig-cell-like steroidogenic cells (deep blue). In the ovaries of horses, the cortical region (in which folliculogenesis occurs) is surrounded by the well-developed medulla and forms a unique structure known as the ovulation fossa.In the ovaries of goats and cows, species with a longer gestating period (Capra hircus, 150 days; Bos taurus, 280 days), distinct secondary sex cords in the cortical region are formed during the fetal period; folliculogenesis occurs before birth. In contrast, the few germ cells in the medullary region immediately disappear during the fetal period, and the primary sex cords regress (Figure 3B) [136][87]. Horses (Equus caballus) have large developed medullary region in the ovary. The equine ovary has a unique structure that includes a concave cortical region known as the ovulation fossa [2,136][2][87]. The cortical region is restricted to the central area enclosed within a dense, richly vascularized connective tissue casing, which corresponds to medulla [138][88]; folliculogenesis is limited to the central cortical region (Figure 3B). In some mammalian species, ovarian development exhibits a unique pattern, with an ovotestis-like structure containing ovarian tissue in the cortical region and a testis-like structure in the medullary region. In the ovary of the spotted hyena (Crocuta crocuta; gestation period, 110 days), the medullary region is separated from the cortical region by a connective tissue boundary during the mid-gestation period. In the medullary region, a cluster of cells expresses 3βHSD; these cells are regarded as Leydig cell-like steroidogenic cells (Figure 3B) [139][89]. In contrast, AMH expression is not present in the cortical or medullary region, suggesting that the supporting cells are not masculinized [139][89]. In the ovaries of most species of moles, secondary sex cords are formed earlier than the testicular cords in male moles [140][90]. Additionally, a large medullary region develops that encompasses Leydig-like cells and a testicular cord-like structure [140][90]; this structure does not exhibit the expression of SOX9 or AMH (Figure 3B) [141][91].
Figure 3. Sexual differentiation of bipotential gonads and the histological variations of ovaries in the indicated species. (A) Bipotential gonads differentiate into testes or ovaries. In mammalian testes, primary sex cords form tubular structures (seminiferous tubules (deep blue) with germ cells (yellow dots)) and the rete testis (cyan), which provide a route for continuous sperm transportation from the seminiferous tubules to the efferent ducts. Interstitial components are shown in gray. Mammalian ovaries commonly develop secondary sex cords with germ cells in the cortical region as a site of future folliculogenesis (orange; yellow dots, germ cells). However, the morphological characteristics of primary sex cords in the medullary region of the ovary show considerable diversity among mammals. (B) In the ovaries of goats and cows, secondary sex cords in the cortical region form during the fetal period. Germ cells in the medullary region disappear and the primary sex region regresses. In the ovaries of mice, secondary sex cords in the cortical region are not distinct in the fetal period; germ cells in the medullary region are maintained and develop immediately after birth as the first wave of follicles (pale-orange circles with yellow dots). In the ovaries of spotted hyenas and most mole species, the medullary region develops as a male-like tissue with Leydig-cell-like steroidogenic cells (deep blue). In the ovaries of horses, the cortical region (in which folliculogenesis occurs) is surrounded by the well-developed medulla and forms a unique structure known as the ovulation fossa.In the ovaries of goats and cows, species with a longer gestating period (Capra hircus, 150 days; Bos taurus, 280 days), distinct secondary sex cords in the cortical region are formed during the fetal period; folliculogenesis occurs before birth. In contrast, the few germ cells in the medullary region immediately disappear during the fetal period, and the primary sex cords regress (Figure 3B) [136][87]. Horses (Equus caballus) have large developed medullary region in the ovary. The equine ovary has a unique structure that includes a concave cortical region known as the ovulation fossa [2,136][2][87]. The cortical region is restricted to the central area enclosed within a dense, richly vascularized connective tissue casing, which corresponds to medulla [138][88]; folliculogenesis is limited to the central cortical region (Figure 3B). In some mammalian species, ovarian development exhibits a unique pattern, with an ovotestis-like structure containing ovarian tissue in the cortical region and a testis-like structure in the medullary region. In the ovary of the spotted hyena (Crocuta crocuta; gestation period, 110 days), the medullary region is separated from the cortical region by a connective tissue boundary during the mid-gestation period. In the medullary region, a cluster of cells expresses 3βHSD; these cells are regarded as Leydig cell-like steroidogenic cells (Figure 3B) [139][89]. In contrast, AMH expression is not present in the cortical or medullary region, suggesting that the supporting cells are not masculinized [139][89]. In the ovaries of most species of moles, secondary sex cords are formed earlier than the testicular cords in male moles [140][90]. Additionally, a large medullary region develops that encompasses Leydig-like cells and a testicular cord-like structure [140][90]; this structure does not exhibit the expression of SOX9 or AMH (Figure 3B) [141][91].
References
- Russell, L.D.; Ettlin, R.A.; Sinha hikim, A.P.; Clegg, E.D. Histological and Histopathological Evaluation of the Testis, 1st ed.; Cache River Press: Clearwater, FL, USA, 1990.
- Dellmann, H.; Brown, E.M. Textbook of Veterinary Histology; LEA & FEBIGER: Philadelphia, PA, USA, 1976.
- Capel, B. The battle of the sexes. Mech. Dev. 2000, 92, 89–103.
- Hatano, O.; Takakusu, A.; Nomura, M.; Morohashi, K. Identical origin of adrenal cortex and gonad revealed by expression profiles of Ad4BP/SF-1. Genes Cells 1996, 1, 663–671.
- Ikeda, Y.; Shen, W.H.; Ingraham, H.A.; Parker, K.L. Developmental expression of mouse steroidogenic factor-1, an essential regulator of the steroid hydroxylases. Mol. Endocrinol. 1994, 8, 654–662.
- Harikae, K.; Miura, K.; Kanai, Y. Early gonadogenesis in mammals: Significance of long and narrow gonadal structure. Dev. Dyn. 2013, 242, 330–338.
- Svingen, T.; Koopman, P. Building the mammalian testis: Origins, differentiation, and assembly of the component cell populations. Genes Dev. 2013, 27, 2409–2426.
- Wilhelm, D.; Yang, J.X.; Thomas, P. Mammalian sex determination and gonad development. Curr. Top. Dev. Biol. 2013, 106, 89–121.
- Wainwright, E.N.; Svingen, T.; Ng, E.T.; Wicking, C.; Koopman, P. Primary cilia function regulates the length of the embryonic trunk axis and urogenital field in mice. Dev. Biol. 2014, 395, 342–354.
- Karl, J.; Capel, B. Sertoli Cells of the Mouse Testis Originate from the Coelomic Epithelium. Dev. Biol. 1998, 203, 323–333.
- Lin, Y.; Barske, L.; Defalco, T.; Capel, B. Numb regulates somatic cell lineage commitment during early gonadogenesis in mice. Development 2017, 144, 1607–1618.
- Kanai, Y.; Hiramatsu, R.; Matoba, S.; Kidokoro, T. From SRY to SOX9: Mammalian Testis Differentiation. J. Biochem. 2005, 138, 13–19.
- Kashimada, K.; Koopman, P. Sry: The master switch in mammalian sex determination. Development 2010, 137, 3921–3930.
- Sinclair, A.H.; Berta, P.; Palmer, M.S.; Hawkins, J.R.; Griffiths, B.L.; Smith, M.J.; Foster, J.W.; Frischauf, A.M.; Lovell-Badge, R.; Goodfellow, P.N. A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature 1990, 346, 240–244.
- Koopman, P.; Gubbay, J.; Vivian, N.; Goodfellow, P.; Lovell-Badge, R. Male development of chromosomally female mice transgenic for Sry. Nature 1991, 351, 117–121.
- Miyawaki, S.; Kuroki, S.; Maeda, R.; Okashita, N.; Koopman, P.; Tachibana, M. The mouse Sry locus harbors a cryptic exon that is essential for male sex determination. Science 2020, 370, 121–124.
- Albrecht, K.H.; Eicher, E.M. Evidence That Sry Is Expressed in Pre-Sertoli Cells and Sertoli and Granulosa Cells Have a Common Precursor. Dev. Biol. 2001, 240, 92–107.
- Bullejos, M.; Koopman, P. Spatially dynamic expression of Sry in mouse genital ridges. Dev. Dyn. 2001, 221, 201–205.
- Lovell-Badge, R.; Robertson, E. XY female mice resulting from a heritable mutation in the primary testis-determining gene, Tdy. Development 1990, 109, 635–646.
- Sekido, R.; Lovell-Badge, R. Sex determination involves synergistic action of SRY and SF1 on a specific Sox9 enhancer. Nature 2008, 453, 930–934.
- Sekido, R.; Bar, I.; Narváez, V.; Penny, G.; Lovell-Badge, R. SOX9 is up-regulated by the transient expression of SRY specifically in Sertoli cell precursors. Dev. Biol. 2004, 274, 271–279.
- Kidokoro, T.; Matoba, S.; Hiramatsu, R.; Fujisawa, M.; Kanai-Azuma, M.; Taya, C.; Kurohmaru, M.; Kawakami, H.; Hayashi, Y.; Kanai, Y.; et al. Influence on spatiotemporal patterns of a male-specific Sox9 activation by ectopic Sry expression during early phases of testis differentiation in mice. Dev. Biol. 2005, 278, 511–525.
- Barrionuevo, F.; Bagheri-Fam, S.; Klattig, J.; Kist, R.; Taketo, M.M.; Englert, C.; Scherer, G. Homozygous inactivation of Sox9 causes complete XY sex reversal in mice. Biol. Reprod. 2006, 74, 195–201.
- Kim, Y.; Kobayashi, A.; Sekido, R.; Dinapoli, L.; Brennan, J.; Chaboissier, M.; Poulat, F.; Behringer, R.R.; Lovell-Badge, R.; Capel, B. Fgf9 and Wnt4 Act as Antagonistic Signals to Regulate Mammalian Sex Determination. PLoS Biol. 2006, 4, e187.
- Hiramatsu, R.; Harikae, K.; Tsunekawa, N.; Kurohmaru, M.; Matsuo, I.; Kanai, Y. FGF signaling directs a center-to-pole expansion of tubulogenesis in mouse testis differentiation. Development 2010, 137, 303–312.
- Wilhelm, D.; Hiramatsu, R.; Mizusaki, H.; Widjaja, L.; Combes, A.N.; Kanai, Y.; Koopman, P. SOX9 Regulates Prostaglandin D Synthase Gene Transcription in Vivo to Ensure Testis Development. J. Biol. Chem. 2007, 282, 10553–10560.
- Moniot, B.; Declosmenil, F.; Barrionuevo, F.; Scherer, G.; Aritake, K.; Malki, S.; Marzi, L.; Cohen-Solal, A.; Georg, I.; Klattig, J.; et al. The PGD2 pathway, independently of FGF9, amplifies SOX9 activity in Sertoli cells during male sexual differentiation. Development 2009, 136, 1813–1821.
- Stévant, I.; Neirjinck, Y.; Borel, C.; Escoffier, J.; Smith, L.B.; Antonarakis, S.E.; Dermitzakis, E.T.; Nef, S. Deciphering cell lineage specification during male sex determination with single-cell RNA sequencing. Cell Rep. 2018, 22, 1589–1599.
- Stévant, I.; Kühne, F.; Greenfield, A.; Chaboissier, M.; Dermitzakis, E.T.; Nef, S. Dissecting Cell Lineage Specification and Sex Fate Determination in Gonadal Somatic Cells Using Single-Cell Transcriptomics. Cell Rep. 2019, 26, 3272–3283.e3.
- Mayère, C.; Regard, V.; Perea-Gomez, A.; Bunce, C.; Neirijnck, Y.; Djari, C.; Bellido-Carreras, N.; Sararols, P.; Reeves, R.; Greenaway, S.; et al. Origin, specification and differentiation of a rare supporting-like lineage in the developing mouse gonad. Sci. Adv. 2022, 8, eabm0972.
- Major, A.T.; Estermann, M.A.; Smith, C.A. Anatomy, Endocrine Regulation, and Embryonic Development of the Rete Testis. Endocrinology 2021, 162, bqab04.
- McKey, J.; Anbarci, D.N.; Bunce, C.; Ontiveros, A.E.; Behringer, R.R.; Capel, B. Integration of mouse ovary morphogenesis with developmental dynamics of the oviduct, ovarian ligaments, and rete ovarii. bioRxiv 2022, 11, e81088.
- Brennan, J.; Tilmann, C.; Capel, B. Pdgfr-alpha mediates testis cord organization and fetal Leydig cell development in the XY gonad. Genes Dev. 2003, 17, 800–810.
- Behringer, R.R.; Cate, R.L.; Froelick, G.J.; Palmiter, R.D.; Brinster, R.L. Abnormal sexual development in transgenic mice chronically expressing Müllerian inhibiting substance. Nature 1990, 345, 167–170.
- Behringer, R.R.; Finegold, M.J.; Cate, R.L. Müllerian-inhibiting substance function during mammalian sexual development. Cell 1994, 79, 415–425.
- Mishina, Y.; Rey, R.; Finegold, M.J.; Matzuk, M.M.; Josso, N.; Cate, R.L.; Behringer, R.R. Genetic analysis of the Müllerian-inhibiting substance signal transduction pathway in mammalian sexual differentiation. Genes Dev. 1996, 10, 2577–2587.
- Imbeaud, S.; Carré-Eusèbe, D.; Rey, R.; Belville, C.; Josso, N.; Picard, J.Y. Molecular genetics of the persistent müllerian duct syndrome: A study of 19 families. Hum. Mol. Genet. 1994, 3, 125–131.
- Imbeaud, S.; Faure, E.; Lamarre, I.; Mattéi, M.G.; di Clemente, N.; Tizard, R.; Carré-Eusèbe, D.; Belville, C.; Tragethon, L.; Tonkin, C.; et al. Insensitivity to anti-müllerian hormone due to a mutation in the human anti-müllerian hormone receptor. Nat. Genet. 1995, 11, 382–388.
- Knebelmann, B.; Boussin, L.; Guerrier, D.; Legeai, L.; Kahn, A.; Josso, N.; Picard, J.Y. Anti-Müllerian hormone Bruxelles: A nonsense mutation associated with the persistent Müllerian duct syndrome. Proc. Natl. Acad. Sci. USA 1991, 88, 3767–3771.
- Yao, H.H.C.; Whoriskey, W.; Capel, B. Desert Hedgehog/Patched 1 signaling specifies fetal Leydig cell fate in testis organogenesis. Genes Dev. 2002, 16, 1433–1440.
- Li, Y.; Zheng, M.; Lau, Y.C.C. The Sex-Determining Factors SRY and SOX9 Regulate Similar Target Genes and Promote Testis Cord Formation during Testicular Differentiation. Cell Rep. 2014, 8, 723–733.
- Chassot, A.; Bradford, S.T.; Auguste, A.; Gregoire, E.P.; Pailhoux, E.; De Rooij, D.G.; Schedl, A.; Chaboissier, M. WNT4 and RSPO1 together are required for cell proliferation in the early mouse gonad. Development 2012, 139, 4461–4472.
- Carmon, K.S.; Gong, X.; Lin, Q.; Thomas, A.; Liu, Q. R-spondins function as ligands of the orphan receptors LGR4 and LGR5 to regulate Wnt/beta-catenin signaling. Proc. Natl. Acad. Sci. USA 2011, 108, 11452–11457.
- Munger, S.C.; Aylor, D.L.; Syed, H.A.; Magwene, P.M.; Threadgill, D.W.; Capel, B. Elucidation of the transcription network governing mammalian sex determination by exploiting strain-specific susceptibility to sex reversal. Genes Dev. 2009, 23, 2521–2536.
- Munger, S.C.; Natarajan, A.; Looger, L.L.; Ohler, U.; Capel, B. Fine Time Course Expression Analysis Identifies Cascades of Activation and Repression and Maps a Putative Regulator of Mammalian Sex Determination. PLoS Genet. 2013, 9, e1003630.
- Jameson, S.A.; Natarajan, A.; Cool, J.; Defalco, T.; Maatouk, D.M.; Mork, L.; Munger, S.C.; Capel, B. Temporal Transcriptional Profiling of Somatic and Germ Cells Reveals Biased Lineage Priming of Sexual Fate in the Fetal Mouse Gonad. PLoS Genet. 2012, 8, e1002575.
- Mizusaki, H.; Kawabe, K.; Mukai, T.; Ariyoshi, E.; Kasahara, M.; Yoshioka, H.; Swain, A.; Morohashi, K. Dax-1 (dosage-sensitive sex reversal-adrenal hypoplasia congenita critical region on the X chromosome, gene 1) gene transcription is regulated by Wnt4 in the female developing gonad. Mol. Endocrinol. 2003, 17, 507–519.
- Yao, H.H.C.; Matzuk, M.M.; Jorgez, C.J.; Menke, D.B.; Page, D.C.; Swain, A.; Capel, B. Follistatin operates downstream of Wnt4 in mammalian ovary organogenesis. Dev. Dyn. 2004, 230, 210–215.
- Kim, B.; Kim, Y.; Cooke, P.S.; Rüther, U.; Jorgensen, J.S. The fused toes locus is essential for somatic-germ cell interactions that foster germ cell maturation in developing gonads in mice. Biol. Reprod. 2011, 84, 1024–1032.
- Liu, C.F.; Liu, C.; Yao, H.H. Building pathways for ovary organogenesis in the mouse embryo. Curr. Top. Dev. Biol. 2010, 90, 263–290.
- Chassot, A.A.; Gregoire, E.P.; Lavery, R.; Taketo, M.M.; de Rooij, D.G.; Adams, I.R.; Chaboissier, M.C. RSPO1/β-catenin signaling pathway regulates oogonia differentiation and entry into meiosis in the mouse fetal ovary. PLoS ONE 2011, 6, e25641.
- Nicol, B.; Yao, N.C. Gonadal Identity in the Absence of Pro-Testis Factor SOX9 and Pro-Ovary Factor Beta-Catenin in Mice. Biol. Reprod. 2015, 93, 35.
- Vainio, S.; Heikkilaè, M.; Kispert, A.; Chin, N.; Mcmahon, A.P. Female development in mammals is regulated by Wnt-4 signalling. Nature 1999, 397, 405–409.
- Jeays-Ward, K.; Hoyle, C.; Brennan, J.; Dandonneau, M.; Alldus, G.; Capel, B.; Swain, A. Endothelial and steroidogenic cell migration are regulated by WNT4 in the developing mammalian gonad. Development 2003, 130, 3663–3670.
- Jordan, B.K.; Shen, J.H.; Olaso, R.; Ingraham, H.A.; Vilain, E. Wnt4 overexpression disrupts normal testicular vasculature and inhibits testosterone synthesis by repressing steroidogenic factor 1/beta-catenin synergy. Proc. Natl. Acad. Sci. USA 2003, 100, 10866–10871.
- Chassot, A.; Ranc, F.; Gregoire, E.P.; Roepers-Gajadien, H.L.; Taketo, M.M.; Camerino, G.; De Rooij, D.G.; Schedl, A.; Chaboissier, M. Activation of β-catenin signaling by Rspo1 controls differentiation of the mammalian ovary. Hum. Mol. Genet. 2008, 17, 1264–1277.
- Tomizuka, K.; Horikoshi, K.; Kitada, R.; Sugawara, Y.; Iba, Y.; Kojima, A.; Yoshitome, A.; Yamawaki, K.; Amagai, M.; Inoue, A.; et al. R-spondin1 plays an essential role in ovarian development through positively regulating Wnt-4 signaling. Hum. Mol. Genet. 2008, 17, 1278–1291.
- Nef, S.; Schaad, O.; Stallings, N.R.; Cederroth, C.R.; Pitetti, J.; Schaer, G.; Malki, S.; Dubois-Dauphin, M.; Boizet-Bonhoure, B.; Descombes, P.; et al. Gene expression during sex determination reveals a robust female genetic program at the onset of ovarian development. Dev. Biol. 2005, 287, 361–377.
- Beverdam, A.; Koopman, P. Expression profiling of purified mouse gonadal somatic cells during the critical time window of sex determination reveals novel candidate genes for human sexual dysgenesis syndromes. Hum. Mol. Genet. 2006, 15, 417–431.
- Chen, H.; Palmer, J.S.; Thiagarajan, R.D.; Dinger, M.E.; Lesieur, E.; Chiu, H.; Schulz, A.; Spiller, C.; Grimmond, S.M.; Little, M.H.; et al. Identification of Novel Markers of Mouse Fetal Ovary Development. PLoS ONE 2012, 7, e41683.
- Schmidt, D.; Ovitt, C.E.; Anlag, K.; Fehsenfeld, S.; Gredsted, L.; Treier, A.C.; Treier, M. The murine winged-helix transcription factor Foxl2 is required for granulosa cell differentiation and ovary maintenance. Development 2004, 131, 933–942.
- Uda, M.; Ottolenghi, C.; Crisponi, L.; Garcia, J.E.; Deiana, M.; Kimber, W.; Forabosco, A.; Cao, A.; Schlessinger, D.; Pilia, G. Foxl2 disruption causes mouse ovarian failure by pervasive blockage of follicle development. Hum. Mol. Genet. 2004, 13, 1171–1181.
- Ottolenghi, C.; Pelosi, E.; Tran, J.; Colombino, M.; Douglass, E.; Nedorezov, T.; Cao, A.; Forabosco, A.; Schlessinger, D. Loss of Wnt4 and Foxl2 leads to female-to-male sex reversal extending to germ cells. Hum. Mol. Genet. 2007, 16, 2795–2804.
- Uhlenhaut, N.H.; Jakob, S.; Anlag, K.; Eisenberger, T.; Sekido, R.; Kress, J.; Treier, A.; Klugmann, C.; Klasen, C.; Holter, N.I.; et al. Somatic Sex Reprogramming of Adult Ovaries to Testes by FOXL2 Ablation. Cell 2009, 139, 1130–1142.
- Maatouk, D.M.; Dinapoli, L.; Alvers, A.; Parker, K.L.; Taketo, M.M.; Capel, B. Stabilization of beta-catenin in XY gonads causes male-to-female sex-reversal. Hum. Mol. Genet. 2008, 17, 2949–2955.
- Boulanger, L.; Pannetier, M.; Gall, L.; Allais-Bonnet, A.; Elzaiat, M.; Le Bourhis, D.; Daniel, N.; Richard, C.; Cotinot, C.; Ghyselinck, N.B.; et al. FOXL2 is a female sex-determining gene in the goat. Curr. Biol. 2014, 24, 404–408.
- Pailhoux, E.; Vigier, B.; Chaffaux, S.; Servel, N.; Taourit, S.; Furet, J.P.; Fellous, M.; Grosclaude, F.; Cribiu, E.P.; Cotinot, C.; et al. A 11.7-kb deletion triggers intersexuality and polledness in goats. Nat. Genet. 2001, 29, 453–458.
- Ottolenghi, C.; Omari, S.; Garcia-Ortiz, J.E.; Uda, M.; Crisponi, L.; Forabosco, A.; Pilia, G.; Schlessinger, D. Foxl2 is required for commitment to ovary differentiation. Hum. Mol. Genet. 2005, 14, 2053–2062.
- Mork, L.; Maatouk, D.M.; Mcmahon, J.A.; Guo, J.J.; Zhang, P.; Mcmahon, A.P.; Capel, B. Temporal Differences in Granulosa Cell Specification in the Ovary Reflect Distinct Follicle Fates in Mice. Biol. Reprod. 2012, 86, 37.
- Ng, A.; Tan, S.; Singh, G.; Rizk, P.; Swathi, Y.; Tan, T.Z.; Huang, R.Y.; Leushacke, M.; Barker, N. Lgr5 marks stem/progenitor cells in ovary and tubal epithelia. Nat. Cell Biol. 2014, 16, 745–757.
- Rastetter, R.H.; Bernard, P.; Palmer, J.S.; Chassot, A.; Chen, H.; Western, P.S.; Ramsay, R.G.; Chaboissier, M.; Wilhelm, D. Marker genes identify three somatic cell types in the fetal mouse ovary. Dev. Biol. 2014, 394, 242–252.
- Barker, N.; van Es, J.H.; Kuipers, J.; Kujala, P.; van den Born, M.; Cozijnsen, M.; Haegebarth, A.; Korving, J.; Begthel, H.; Peters, P.J.; et al. Identification of stem cells in small intestine and colon by marker gene Lgr5. Nature 2007, 449, 1003–1007.
- Jaks, V.; Barker, N.; Kasper, M.; van Es, J.H.; Snippert, H.J.; Clevers, H.; Toftgard, R. Lgr5 marks cycling, yet long-lived, hair follicle stem cells. Nat. Genet. 2008, 40, 1291–1299.
- Barker, N.; Clevers, H. Leucine-rich repeat-containing G-protein-coupled receptors as markers of adult stem cells. Gastroenterology 2010, 138, 1681–1696.
- Plaks, V.; Brenot, A.; Lawson, D.A.; Linnemann, J.R.; Van Kappel, E.C.; Wong, K.C.; de Sauvage, F.; Klein, O.D.; Werb, Z. Lgr5-expressing cells are sufficient and necessary for postnatal mammary gland organogenesis. Cell Rep. 2013, 3, 70–78.
- Zheng, W.; Zhang, H.; Gorre, N.; Risal, S.; Shen, Y.; Liu, K. Two classes of ovarian primordial follicles exhibit distinct developmental dynamics and physiological functions. Hum. Mol. Genet. 2014, 23, 920–928.
- Tam, P.P.; Snow, M.H. Proliferation and migration of primordial germ cells during compensatory growth in mouse embryos. J. Embryol. Exp. Morphol. 1981, 64, 133–147.
- Lei, L.; Spradling, A.C. Mouse primordial germ cells produce cysts that partially fragment prior to meiosis. Development 2013, 140, 2075–2081.
- Bowles, J.; Knight, D.; Smith, C.; Wilhelm, D.; Richman, J.; Mamiya, S.; Yashiro, K.; Chawengsaksophak, K.; Wilson, M.J.; Rossant, J.; et al. Retinoid signaling determines germ cell fate in mice. Science 2006, 312, 596–600.
- Koubova, J.; Menke, D.B.; Zhou, Q.; Capel, B.; Griswold, M.D.; Page, D.C. Retinoic acid regulates sex-specific timing of meiotic initiation in mice. Proc. Natl. Acad. Sci. USA 2006, 103, 2474–2479.
- Menke, D.B.; Koubova, J.; Page, D.C. Sexual differentiation of germ cells in XX mouse gonads occurs in an anterior-to-posterior wave. Dev. Biol. 2003, 262, 303–312.
- Koubova, J.; Hu, Y.C.; Bhattacharyya, T.; Soh, Y.Q.; Gill, M.E.; Goodheart, M.L.; Hogarth, C.A.; Griswold, M.D.; Page, D.C. Retinoic acid activates two pathways required for meiosis in mice. PLoS Genet. 2014, 10, e1004541.
- Pepling, M.E.; Spradling, A.C. Mouse ovarian germ cell cysts undergo programmed breakdown to form primordial follicles. Dev. Biol. 2001, 234, 339–351.
- Perez, G.I.; Robles, R.; Knudson, C.M.; Flaws, J.A.; Korsmeyer, S.J.; Tilly, J.L. Prolongation of ovarian lifespan into advanced chronological age by Bax-deficiency. Nat. Genet. 1999, 21, 200–203.
- Alton, M.; Taketo, T. Switch from BAX-dependent to BAX-independent germ cell loss during the development of fetal mouse ovaries. J. Cell Sci. 2007, 120, 417–424.
- Greenfeld, C.R.; Pepling, M.E.; Babus, J.K.; Furth, P.A.; Flaws, J.A. BAX regulates follicular endowment in mice. Reproduction 2007, 133, 865–876.
- McGeady, T.A.; Quinn, P.J.; Fitzpatrick, E.S.; Ryan, M.T.; Kilroy, D.; Lonergan, P. Veterinary Embryology, 2nd ed.; John Wiley & Sons: Chichester, UK, 2017.
- Dyce, K.M.; Sack, W.O.; Wensing, C.J.G. Textbook of Veterinary Anatomy, 4th ed.; Saunders/Elsevier: St. Louis, MO, USA, 2010.
- Browne, P.; Place, N.J.; Vidal, J.D.; Moore, I.T.; Cunha, G.R.; Glickman, S.E.; Conley, A.J. Endocrine differentiation of fetal ovaries and testes of the spotted hyena (Crocuta crocuta): Timing of androgen-independent versus androgen-driven genital development. Reproduction 2006, 132, 649–659.
- Jiménez, R. Ovarian organogenesis in mammals: Mice cannot tell us everything. Sex. Dev. 2009, 3, 291–301.
- Carmona, F.D.; Lupiáñez, D.G.; Real, F.M.; Burgos, M.; Zurita, F.; Jiménez, R. SOX9 is not required for the cellular events of testicular organogenesis in XX mole ovotestes. J. Exp. Zool. B Mol. Dev. Evol. 2009, 312, 734–748.
