Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 3 by Conner Chen and Version 2 by Conner Chen.
The approach to equine anti-doping is focused on the targeted detection of prohibited substances. However, as new substances are rapidly being developed, the need for complimentary methods for monitoring is crucial to ensure the integrity of the racing industry is upheld. Lipidomics is a growing field involved in the characterisation of lipids, their function and metabolism in a biological system. Different lipids have various biological effects throughout the equine system including platelet aggregation and inflammation.
- lipidomics
- review
- analytical
- equine
- anti-doping
1. Biomarkers for Equine Anti-Doping
The current approach to equine anti-doping is focused on the targeted detection of prohibited substances [1]. However, as new substances are rapidly being developed, the need for complimentary methods of monitoring is important to ensure the integrity of the racing industry is upheld [1]. The use of biomarkers for the detection of doping abuse is a significant advancement for sports anti-doping. Teale et al. [2] define biomarkers as an “individual biological parameter or substance (metabolite, protein or transcript); the concentration of which is indicative of the use or abuse of a drug or therapy”. With the discovery of novel biomarkers for detecting doping abuse, the potential exists for a larger number of drugs to be indirectly detected and over longer periods of time. However, with indirect detection, there is the possibility of the method not being specific and the increased likelihood of inconsistent results [3]. An “omics” approach may provide an alternative to direct detection of doping as maintaining a contemporary scope of testing makes direct detection continually difficult due to availability of reference materials [5, 6][4][5]. The use of metabolomics has been utilised in many different laboratories to measure metabolites at low levels relative to time-related biological responses of a drug administration[3, 5, 6][4][5][6]. This provides a framework for non-targeted detection, particularly for drugs that have a short half-life but long lasting effect on any individual system[5] [4].
2. Lipids
Lipidomics is a growing field involved in the characterisation of lipids, their function and metabolism in a biological system [47][58]. Lipids are non-polar molecules with a diverse chemistry and functionality [69][710]. In conjunction with carbohydrates, lipids are the main energy source for equine striated muscles [811]. There are a number of different classes of lipids including monounsaturated fatty acids (MUFAs) and polyunsaturated fatty acids (PUFAs) [912][1013]. MUFAs are lipids that have a single double bond present in the compound and usually only exist in seeds or marine organisms, however, are naturally rare [912]. PUFAs comparatively contain more than one double bond, are more commonly found [912] and have various biological effects including platelet aggregation and inflammation [1114]. In animals, common PUFAs include arachidonic acid (AA), eicosapentaenoic acid (EPA) and docosahexaenoic acid (DHA) [912]. The most relevant and important oxygenated products for the racing industry are lipids known as the eicosanoids [912]. Eicosanoids are a large subclass predominately defined by the 20 carbon chain containing over 100 lipid mediators including prostaglandins, thromboxanes, leukotrienes, hydroxy fatty acids and lipoxins [912][1215] with the majority derived from AA, an omega-6 fatty acid [1316]. Eicosanoids are believed to act as inflammatory mediators since they have the ability to mimic inflammatory symptoms and decrease in the presence of anti-inflammatory drugs [58][1114]. Disruption of eicosanoids can cause a range of inflammatory pathological conditions including asthma, chronic obstructive pulmonary disease, fevers, pain, a range of cardiovascular diseases and cancers [1215]. Eicosanoids are synthesised at the site of injury in order to control and regulate the inflammatory response [47]. AA is released from membrane phospholipids through the activation of phospholipase A2 enzyme (PLA2) [1114]. It can be further converted into other eicosanoids in the cascade (Figure 1). In a non-targeted sense, it should be possible to utilise the AA cascade to determine which lipids are being affected following drug administration. This cascade includes prostaglandin D2 (PGD2) [1417], prostaglandin E2 (PGE2) [1518], prostaglandin F2α (PGF2α) [47], thromboxane B2 (TXB2) [1619], 11-dehydro thromboxane B2 (11-Dehydro TXB2) [1720], 6-keto prostaglandin F1α (6-Keto PGF1α) [1821], 15(S)-hydroxyeicosatetraenoic acid (15-HETE) [47], 5(S)-hydroxyeicosatetraenoic acid (5-HETE) [1922], leukotriene B4 (LTB4) [2023][2124][2225], leukotriene D4 (LTD4) [2023][2225] and leukotriene E4 (LTE4) [2023][2225]. The analogues of AA are also of interest including arachidonoyl ethanolamide (AEA) [2326][2427] and oleoyl ethanolamide (OEA) [2326].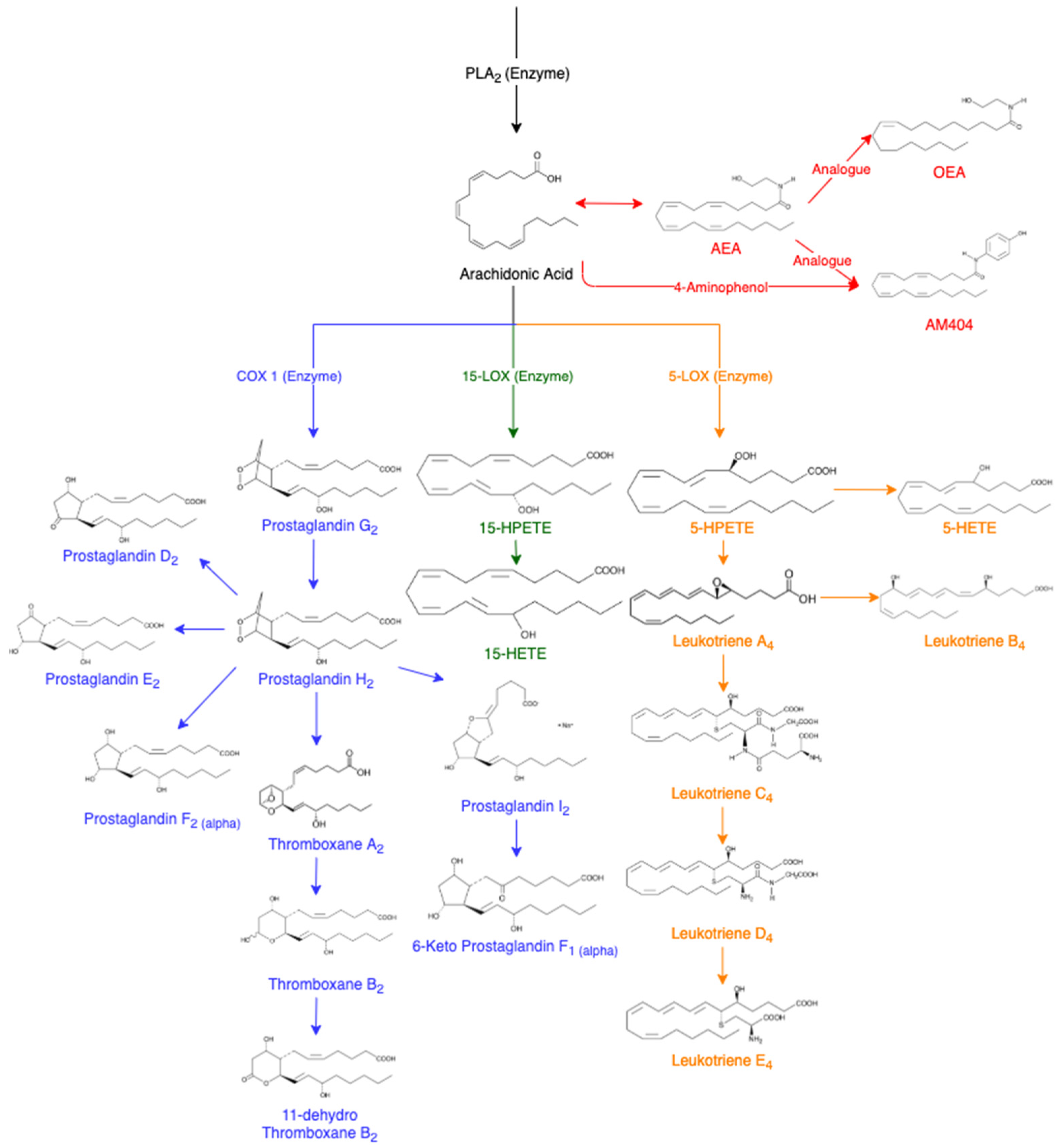
Figure 1. Arachidonic Acid Cascade, adapted from various sources [47][5][118][14][15][16][17][18][19][20][21][22][23][24][25][26][27][28][29]. Abbreviations of eicosanoids: arachidonoyl ethanolamide (AEA), oleoyl ethanolamide (OEA), prostaglandin (PG), thromboxane (Tx), hydroperoxyeicosatetraenoic acids (HPETES), hydroxyeicosatetraenoic acid (HETE) and leukotriene (LT).
References
- Fragkaki, A.G.; Kioukia-Fougia, N.; Kiousi, P.; Kioussi, M.; Tsivou, M. Challenges in detecting substances for equine anti-doping. Drug Test. Anal. 2017, 9, 1291–1303.
- Teale, P.; Barton, C.; Driver, P.M.; Kay, R.G. Biomarkers: Unrealized potential in sports doping analysis. Bioanalysis 2009, 1, 1103–1118.
- Narduzzi, L.; Dervilly, G.; Audran, M.; Le Bizec, B.; Buisson, C. A role for metabolomics in the antidoping toolbox? Drug Test. Anal. 2020, 12, 677–690.
- Mangal, D.; Uboh, C.E.; Soma, L.R. Analysis of bioactive eicosanoids in equine plasma by stable isotope dilution reversed-phase liquid chromatography/multiple reaction monitoring mass spectrometry. Rapid Commun. Mass Spectrom. 2011, 25, 585–598. Stojiljkovic, N.; Paris, A.; Garcia, P.; Popot, M.-A.; Bonnaire, Y.; Tabet, J.-C.; Junot, C. Evaluation of horse urine sample preparation methods for metabolomics using LC coupled to HRMS. Bioanalysis 2014, 6, 785–803.
- Dass, C. Characterization of Lipids. In Fundamentals of Contemporary Mass Spectrometry; John Wiley & Sons: New York, NY, USA, 2007; pp. 423–451. Reichel, C. OMICS-strategies and methods in the fight against doping. Forensic Sci. Int. 2011, 213, 20.
- López-Bascón, M.A.; Calderón-Santiago, M.; Díaz-Lozano, A.; Camargo, A.; López-Miranda, J.; Priego-Capote, F. Development of a qualitative/quantitative strategy for comprehensive determination of polar lipids by LC–MS/MS in human plasma. Anal. Bioanal. Chem. 2020, 412, 489–498. Narduzzi, L.; Dervilly, G.; Audran, M.; Le Bizec, B.; Buisson, C. A role for metabolomics in the antidoping toolbox? Drug Test. Anal. 2020, 12, 677–690.
- Koelmel, J.P.; Li, X.; Stow, S.M.; Sartain, M.J.; Murali, A.; Kemperman, R.; Tsugawa, H.; Takahashi, M.; Vasiliou, V.; Bowden, J.A.; et al. Lipid Annotator: Towards Accurate Annotation in Non-Targeted Liquid Chromatography High-Resolution Tandem Mass Spectrometry (LC-HRMS/MS) Lipidomics Using A Rapid and User-Friendly Software. Metabolites 2020, 10, 101. Mangal, D.; Uboh, C.E.; Soma, L.R. Analysis of bioactive eicosanoids in equine plasma by stable isotope dilution reversed-phase liquid chromatography/multiple reaction monitoring mass spectrometry. Rapid Commun. Mass Spectrom. 2011, 25, 585–598.
- Nolazco Sassot, L.; Villarino, N.F.; Dasgupta, N.; Morrison, J.J.; Bayly, W.M.; Gang, D.; Sanz, M.G. The lipidome of Thoroughbred racehorses before and after supramaximal exercise. Equine Vet. J. 2019, 51, 696–700. Dass, C. Characterization of Lipids. In Fundamentals of Contemporary Mass Spectrometry; John Wiley & Sons: New York, NY, USA, 2007; pp. 423–451.
- Harwood, J.L.; Frayn, K.N.; Murphy, D.J.; Michell, R.H.; Gurr, M.I. Lipids: Biochemistry, Biotechnology and Health; John Wiley & Sons, Incorporated: Hoboken, UK, 2016. López-Bascón, M.A.; Calderón-Santiago, M.; Díaz-Lozano, A.; Camargo, A.; López-Miranda, J.; Priego-Capote, F. Development of a qualitative/quantitative strategy for comprehensive determination of polar lipids by LC–MS/MS in human plasma. Anal. Bioanal. Chem. 2020, 412, 489–498.
- Fahy, E.; Cotter, D.; Sud, M.; Subramaniam, S. Lipid classification, structures and tools. Biochim. Et Biophys. Acta. Mol. Cell Biol. Lipids 2011, 1811, 637–647. Koelmel, J.P.; Li, X.; Stow, S.M.; Sartain, M.J.; Murali, A.; Kemperman, R.; Tsugawa, H.; Takahashi, M.; Vasiliou, V.; Bowden, J.A.; et al. Lipid Annotator: Towards Accurate Annotation in Non-Targeted Liquid Chromatography High-Resolution Tandem Mass Spectrometry (LC-HRMS/MS) Lipidomics Using A Rapid and User-Friendly Software. Metabolites 2020, 10, 101.
- Granström, E. The arachidonic acid cascade. Inflammation 1984, 8, S15–S25. Nolazco Sassot, L.; Villarino, N.F.; Dasgupta, N.; Morrison, J.J.; Bayly, W.M.; Gang, D.; Sanz, M.G. The lipidome of Thoroughbred racehorses before and after supramaximal exercise. Equine Vet. J. 2019, 51, 696–700.
- Thakare, R.; Chhonker, Y.S.; Gautam, N.; Nelson, A.; Casaburi, R.; Criner, G.; Dransfield, M.T.; Make, B.; Schmid, K.K.; Rennard, S.I.; et al. Simultaneous LC–MS/MS analysis of eicosanoids and related metabolites in human serum, sputum and BALF. Biomed. Chromatogr. 2018, 32, e4102. Harwood, J.L.; Frayn, K.N.; Murphy, D.J.; Michell, R.H.; Gurr, M.I. Lipids: Biochemistry, Biotechnology and Health; John Wiley & Sons, Incorporated: Hoboken, UK, 2016.
- Toewe, A.; Balas, L.; Durand, T.; Geisslinger, G.; Ferreirós, N. Simultaneous determination of PUFA-derived pro-resolving metabolites and pathway markers using chiral chromatography and tandem mass spectrometry. Anal. Chim. Acta 2018, 1031, 185–194. Fahy, E.; Cotter, D.; Sud, M.; Subramaniam, S. Lipid classification, structures and tools. Biochim. Et Biophys. Acta. Mol. Cell Biol. Lipids 2011, 1811, 637–647.
- Giles, H.; Leff, P. The biology and pharmacology of PGD2. Prostaglandins 1988, 35, 277–300. Granström, E. The arachidonic acid cascade. Inflammation 1984, 8, S15–S25.
- Jackson, C.A.; Colahan, P.T.; Rice, B. Use of a Commercially Available Prostaglandin E2 Enzyme-Linked Immunosorbent Assay for Non-Steroidal Anti-Inflammatory Drug Screening. In Proceedings of the 16th International Conference of Racing Analysts and Veterinarians, Tokyo, Japan, 21–27 October 2006; Volume 16, pp. 477–482. Thakare, R.; Chhonker, Y.S.; Gautam, N.; Nelson, A.; Casaburi, R.; Criner, G.; Dransfield, M.T.; Make, B.; Schmid, K.K.; Rennard, S.I.; et al. Simultaneous LC–MS/MS analysis of eicosanoids and related metabolites in human serum, sputum and BALF. Biomed. Chromatogr. 2018, 32, e4102.
- Lees, P.; Ewins, C.P.; Taylor, J.B.O.; Sedgwick, A.D. Serum thromboxane in the horse and its inhibition by aspirin, phenylbutazone and flunixin. Br. Vet. J. 1987, 143, 462–476. Toewe, A.; Balas, L.; Durand, T.; Geisslinger, G.; Ferreirós, N. Simultaneous determination of PUFA-derived pro-resolving metabolites and pathway markers using chiral chromatography and tandem mass spectrometry. Anal. Chim. Acta 2018, 1031, 185–194.
- Lopez, L.R.; Guyer, K.E.; Torre, I.G.D.L.; Pitts, K.R.; Matsuura, E.; Ames, P.R. Platelet thromboxane (11-dehydro-Thromboxane B2) and aspirin response in patients with diabetes and coronary artery disease. World J. Diabetes 2014, 5, 115–127. Giles, H.; Leff, P. The biology and pharmacology of PGD2. Prostaglandins 1988, 35, 277–300.
- Johnson, R.A.; Morton, D.R.; Kinner, J.H.; Gorman, R.R.; McGuire, J.C.; Sun, F.F.; Whittaker, N.; Bunting, S.; Salmon, J.; Moncada, S.; et al. The chemical structure of prostaglandin X (prostacyclin). Prostaglandins 1976, 12, 915–928. Jackson, C.A.; Colahan, P.T.; Rice, B. Use of a Commercially Available Prostaglandin E2 Enzyme-Linked Immunosorbent Assay for Non-Steroidal Anti-Inflammatory Drug Screening. In Proceedings of the 16th International Conference of Racing Analysts and Veterinarians, Tokyo, Japan, 21–27 October 2006; Volume 16, pp. 477–482.
- Connolly, P.J.; Wetter, S.K.; Beers, K.N.; Hamel, S.C.; Chen, R.H.K.; Wachter, M.P.; Ansell, J.; Singer, M.M.; Steber, M.; Ritchie, D.M.; et al. N-Hydroxyurea and hydroxamic acid inhibitors of cyclooxygenase and 5-lipoxygenase. Bioorganic Med. Chem. Letters 1999, 9, 979–984. Lees, P.; Ewins, C.P.; Taylor, J.B.O.; Sedgwick, A.D. Serum thromboxane in the horse and its inhibition by aspirin, phenylbutazone and flunixin. Br. Vet. J. 1987, 143, 462–476.
- Samuelsson, B.; Dahlen, S.-E.; Lindgren, J.A.; Rouzer, C.A.; Serhan, C.N. Leukotrienes and lipoxins: Structures, biosynthesis, and biological effects. Science 1987, 237, 1171–1176. Lopez, L.R.; Guyer, K.E.; Torre, I.G.D.L.; Pitts, K.R.; Matsuura, E.; Ames, P.R. Platelet thromboxane (11-dehydro-Thromboxane B2) and aspirin response in patients with diabetes and coronary artery disease. World J. Diabetes 2014, 5, 115–127.
- McMillan, R.M.; Foster, S.J. Leukotriene B4 and inflammatory disease. Agents Actions 1988, 24, 114–119. Johnson, R.A.; Morton, D.R.; Kinner, J.H.; Gorman, R.R.; McGuire, J.C.; Sun, F.F.; Whittaker, N.; Bunting, S.; Salmon, J.; Moncada, S.; et al. The chemical structure of prostaglandin X (prostacyclin). Prostaglandins 1976, 12, 915–928.
- Goodman, L.; Coles, T.B.; Budsberg, S. Leukotriene inhibition in small animal medicine. J. Vet. Pharmacol. Ther. 2008, 31, 387–398. Connolly, P.J.; Wetter, S.K.; Beers, K.N.; Hamel, S.C.; Chen, R.H.K.; Wachter, M.P.; Ansell, J.; Singer, M.M.; Steber, M.; Ritchie, D.M.; et al. N-Hydroxyurea and hydroxamic acid inhibitors of cyclooxygenase and 5-lipoxygenase. Bioorganic Med. Chem. Letters 1999, 9, 979–984.
- Felder, C.C.; Briley, E.M.; Axelrod, J.; Simpson, J.T.; Mackie, K.; Devane, W.A. Anandamide, an endogenous cannabimimetic eicosanoid, binds to the cloned human cannabinoid receptor and stimulates receptor-mediated signal transduction. Proc. Natl. Acad. Sci. USA 1993, 90, 7656–7660. Samuelsson, B.; Dahlen, S.-E.; Lindgren, J.A.; Rouzer, C.A.; Serhan, C.N. Leukotrienes and lipoxins: Structures, biosynthesis, and biological effects. Science 1987, 237, 1171–1176.
- Beltramo, M.; Stella, N.; Calignano, A.; Lin, S.Y.; Makriyannis, A.; Piomelli, D. Functional role of high-affinity anandamide transport, as revealed by selective inhibition. Science 1997, 277, 1094–1097. McMillan, R.M.; Foster, S.J. Leukotriene B4 and inflammatory disease. Agents Actions 1988, 24, 114–119.
- Sharma, C.V.; Long, J.H.; Shah, S.; Rahman, J.; Perrett, D.; Ayoub, S.S.; Mehta, V. First evidence of the conversion of paracetamol to AM404 in human cerebrospinal fluid. J. Pain Res. 2017, 10, 2703–2709. Goodman, L.; Coles, T.B.; Budsberg, S. Leukotriene inhibition in small animal medicine. J. Vet. Pharmacol. Ther. 2008, 31, 387–398.
- Högestätt, E.D.; Jönsson, B.A.G.; Ermund, A.; Andersson, D.A.; Björk, H.; Alexander, J.P.; Cravatt, B.F.; Basbaum, A.I.; Zygmunt, P.M. Conversion of Acetaminophen to the Bioactive N-Acylphenolamine AM404 via Fatty Acid Amide Hydrolase-dependent Arachidonic Acid Conjugation in the Nervous System. J. Biol. Chem. 2005, 280, 31405–31412. Felder, C.C.; Briley, E.M.; Axelrod, J.; Simpson, J.T.; Mackie, K.; Devane, W.A. Anandamide, an endogenous cannabimimetic eicosanoid, binds to the cloned human cannabinoid receptor and stimulates receptor-mediated signal transduction. Proc. Natl. Acad. Sci. USA 1993, 90, 7656–7660.
- Ricciotti, E.; FitzGerald, G.A. Prostaglandins and Inflammation. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 986–1000. Beltramo, M.; Stella, N.; Calignano, A.; Lin, S.Y.; Makriyannis, A.; Piomelli, D. Functional role of high-affinity anandamide transport, as revealed by selective inhibition. Science 1997, 277, 1094–1097.
- Wang, B.; Wu, L.; Chen, J.; Dong, L.; Chen, C.; Wen, Z.; Hu, J.; Fleming, I.; Wang, D.W. Metabolism pathways of arachidonic acids: Mechanisms and potential therapeutic targets. Signal Transduct. Target. Ther. 2021, 6, 94. Sharma, C.V.; Long, J.H.; Shah, S.; Rahman, J.; Perrett, D.; Ayoub, S.S.; Mehta, V. First evidence of the conversion of paracetamol to AM404 in human cerebrospinal fluid. J. Pain Res. 2017, 10, 2703–2709.
- Högestätt, E.D.; Jönsson, B.A.G.; Ermund, A.; Andersson, D.A.; Björk, H.; Alexander, J.P.; Cravatt, B.F.; Basbaum, A.I.; Zygmunt, P.M. Conversion of Acetaminophen to the Bioactive N-Acylphenolamine AM404 via Fatty Acid Amide Hydrolase-dependent Arachidonic Acid Conjugation in the Nervous System. J. Biol. Chem. 2005, 280, 31405–31412.
- Ricciotti, E.; FitzGerald, G.A. Prostaglandins and Inflammation. Arterioscler. Thromb. Vasc. Biol. 2011, 31, 986–1000.
- Wang, B.; Wu, L.; Chen, J.; Dong, L.; Chen, C.; Wen, Z.; Hu, J.; Fleming, I.; Wang, D.W. Metabolism pathways of arachidonic acids: Mechanisms and potential therapeutic targets. Signal Transduct. Target. Ther. 2021, 6, 94.
More
