In order to overcome the serious complications associated with COVID-19 and to prevent SARS-CoV-2 entry into the host cell, it is necessary to repurpose drugs with a broad medicinal application as soon as they become available. It is in this pattern that Nigella sativa seeds manifest their extensive therapeutic effects, which have been reported to be particularly effective in the treatment of skin diseases, jaundice, and gastrointestinal problems. One important component of these seeds is thymoquinone (TQ), which has a wide range of beneficial properties, including antioxidant and anti-inflammatory properties, as well as antibacterial and parasitic properties, in addition to anticarcinogenic, antiallergic, and antiviral properties.
- COVID-19
- thymoquinone
- complications
1. Possible Curative Efficacy of TQ for the Inflammation and Multiple Organ Failure Associated with COVID-19

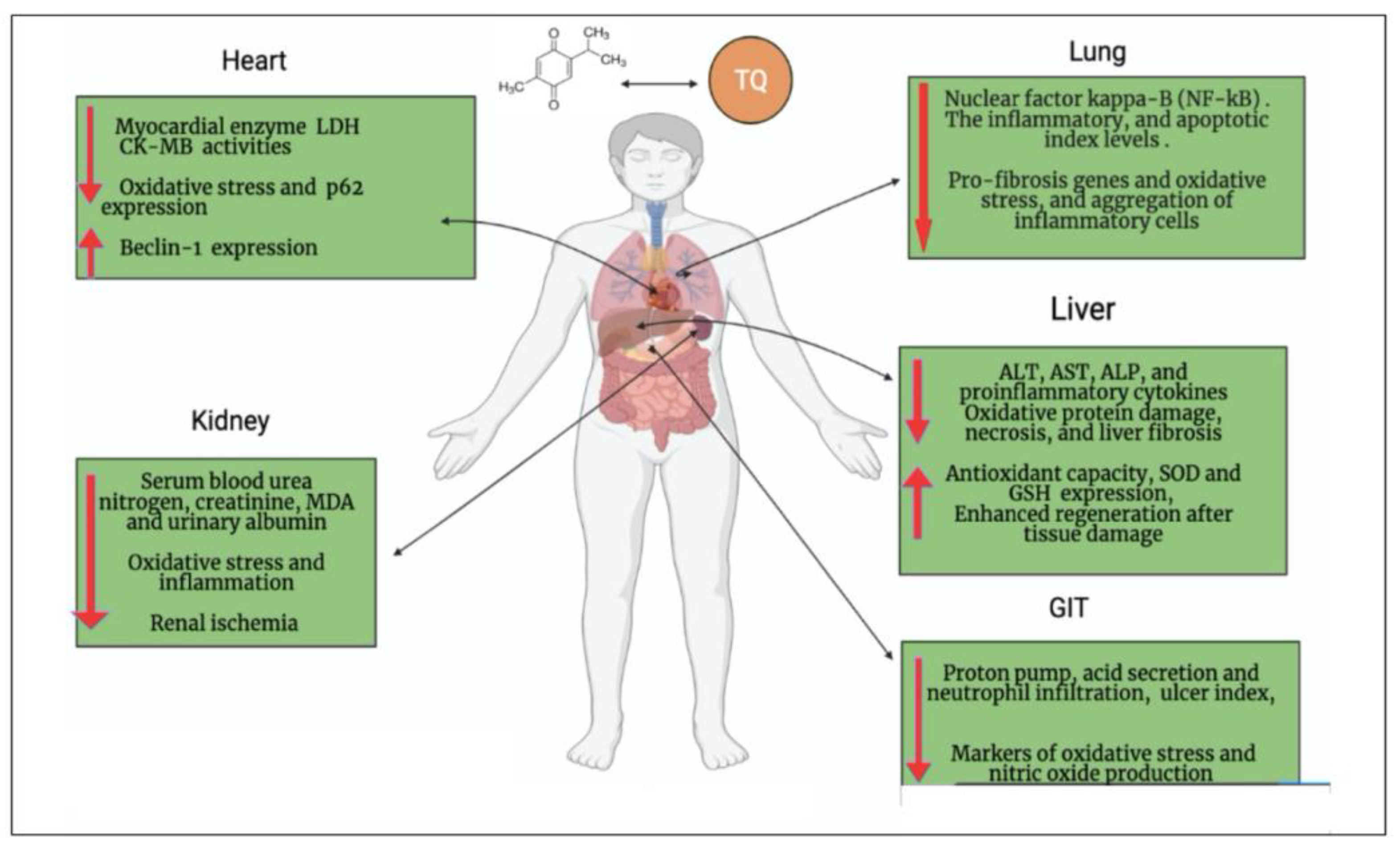
2. Oxidative Stress Associated with COVID-19 and the Antioxidant Effect of TQ
3. Cardiopulmonary Protective Effect of TQ
4. Neuroprotective Effect of TQ can Overcome the Neurologic/Cognitive Manifestations Associated with COVID-19
5. Hepatorenal Protective Effect of TQ against COVID-19
6. Gastrointestinal Dysfunction Associated with COVID-19 and the Gastroprotective Effect of TQ
| COVID-19 Complications | Thymoquinone | References |
|---|---|---|
| Inflammation and cytokine release syndrome (CRS) |
|
[22,[30,931,32]][10][11][12] |
| Oxidative damage |
|
[39,40][20][21] |
| Cardiac injury |
|
[41,47,48,49][22][28][29][30] |
| Pulmonary damage |
|
[53,54,55][34][35][36] |
| Neurological disease |
|
[60,61,62][41][42][43] |
| Liver injury |
|
[67,[68,4869]][49][50] |
| Kidney injury |
|
[72,74,75][53][55][56] |
| GIT injury |
|
[78][59] |
References
- Panigrahy, D.; Gilligan, M.M.; Huang, S.; Gartung, A.; Cortes-Puch, I.; Sime, P.J.; Phipps, R.P.; Serhan, C.N.; Hammock, B.D. Inflammation resolution: A dual-pronged approach to averting cytokine storms in COVID-19? Cancer Metastasis Rev. 2020, 39, 337–340.
- Kell, D.B.; Heyden, E.L.; Pretorius, E. The Biology of Lactoferrin, an Iron-Binding Protein That Can Help Defend Against Viruses and Bacteria. Front. Immunol. 2020, 11, 1221.
- Bunt, S.K.; Yang, L.; Sinha, P.; Clements, V.K.; Leips, J.; Ostrand-Rosenberg, S. Reduced inflammation in the tumor microenvironment delays the accumulation of myeloid-derived suppressor cells and limits tumor progression. Cancer Res. 2007, 67, 10019–10026.
- Shah, A. Novel Coronavirus-Induced NLRP3 Inflammasome Activation: A Potential Drug Target in the Treatment of COVID-19. Front. Immunol. 2020, 11, 1021.
- Barkat, M.A.; Ahmad, J.; Khan, M.A.; Beg, S.; Ahmad, F.J. Insights into the targeting potential of thymoquinone for therapeutic intervention against triple-negative breast cancer. Curr. Drug Targets 2018, 19, 70–80.
- Ranjbar, M.; Rahimi, A.; Baghernejadan, Z.; Ghorbani, A.; Khorramdelazad, H. Role of CCL2/CCR2 axis in the pathogenesis of COVID-19 and possible Treatments: All options on the Table. Int. Immunopharmacol. 2022, 113, 109325.
- Gibellini, L.; De Biasi, S.; Paolini, A.; Borella, R.; Boraldi, F.; Mattioli, M.; Lo Tartaro, D.; Fidanza, L.; Caro-Maldonado, A.; Meschiari, M.; et al. Altered bioenergetics and mitochondrial dysfunction of monocytes in patients with COVID-19 pneumonia. EMBO Mol. Med. 2020, 12, e13001.
- Yang, L.; Nilsson-Payant, B.E.; Han, Y.; Jaffré, F.; Zhu, J.; Wang, P.; Zhang, T.; Redmond, D.; Houghton, S.; Møller, R.; et al. Cardiomyocytes recruit monocytes upon SARS-CoV-2 infection by secreting CCL2. Stem Cell Rep. 2021, 16, 2274–2288.
- Liu, Y.; Liao, R.; Qiang, Z.; Zhang, C. Pro-inflammatory cytokine-driven PI3K/Akt/Sp1 signalling and H2S production facilitates the pathogenesis of severe acute pancreatitis. Biosci. Rep. 2017, 37, BSR20160483.
- Arjumand, S.; Shahzad, M.; Shabbir, A.; Yousaf, M.Z. Thymoquinone attenuates rheumatoid arthritis by downregulating TLR2, TLR4, TNF-alpha, IL-1, and NFkappaB expression levels. Biomed. Pharmacother. 2019, 111, 958–963.
- Houghton, P.J.; Zarka, R.; de las Heras, B.; Hoult, J.R. Fixed oil of Nigella sativa and derived thymoquinone inhibit eicosanoid generation in leukocytes and membrane lipid peroxidation. Planta Med. 1995, 61, 33–36.
- Koshak, A.E.; Koshak, E.A.; Mobeireek, A.F.; Badawi, M.A.; Wali, S.O.; Malibary, H.M.; Atwah, A.F.; Alhamdan, M.M.; Almalki, R.A.; Madani, T.A. Nigella sativa for the treatment of COVID-19: An open-label randomized controlled clinical trial. Complement. Ther. Med. 2021, 61, 102769.
- Liu, X.; Chen, Z. The pathophysiological role of mitochondrial oxidative stress in lung diseases. J. Transl. Med. 2017, 15, 207.
- Jean, S.S.; Lee, P.I.; Hsueh, P.R. Treatment options for COVID-19: The reality and challenges. J. Microbiol. Immunol. Infect. 2020, 53, 436–443.
- Olsson, M.; Zhivotovsky, B. Caspases and cancer. Cell Death Differ. 2011, 18, 1441–1449.
- Khan, M.A.; Tania, M.; Wei, C.; Mei, Z.; Fu, S.; Cheng, J.; Xu, J.; Fu, J. Thymoquinone inhibits cancer metastasis by downregulating TWIST1 expression to reduce epithelial to mesenchymal transition. Oncotarget 2015, 6, 19580–19591.
- Ismail, M.; Al-Naqeep, G.; Chan, K.W. Nigella sativa thymoquinone-rich fraction greatly improves plasma antioxidant capacity and expression of antioxidant genes in hypercholesterolemic rats. Free Radic. Biol. Med. 2010, 48, 664–672.
- Elbarbry, F.; Ragheb, A.; Marfleet, T.; Shoker, A. Modulation of hepatic drug metabolizing enzymes by dietary doses of thymoquinone in female New Zealand White rabbits. Phytother. Res. 2012, 26, 1726–1730.
- Periyanayagam, S.; Arumugam, G.; Ravikumar, A.; Ganesan, V.S. Thymoquinone ameliorates NLRP3-mediated inflammation in the pancreas of albino Wistar rats fed ethanol and high-fat diet. J. Basic Clin. Physiol. Pharmacol. 2015, 26, 623–632.
- Holgersen, E.M.; Gandhi, S.; Zhou, Y.; Kim, J.; Vaz, B.; Bogojeski, J.; Bugno, M.; Shalev, Z.; Cheung-Ong, K.; Goncalves, J.; et al. Transcriptome-Wide Off-Target Effects of Steric-Blocking Oligonucleotides. Nucleic Acid Ther. 2021, 31, 392–403.
- Goncalves, T.L.; Erthal, F.; Corte, C.L.; Muller, L.G.; Piovezan, C.M.; Nogueira, C.W.; Rocha, J.B. Involvement of oxidative stress in the pre-malignant and malignant states of cervical cancer in women. Clin. Biochem. 2005, 38, 1071–1075.
- Guo, T.; Fan, Y.; Chen, M.; Wu, X.; Zhang, L.; He, T.; Wang, H.; Wan, J.; Wang, X.; Lu, Z. Cardiovascular Implications of Fatal Outcomes of Patients With Coronavirus Disease 2019 (COVID-19). JAMA Cardiol. 2020, 5, 811–818.
- Parvu, S.; Müller, K.; Dahdal, D.; Cosmin, I.; Christodorescu, R.; Duda-Seiman, D.; Man, D.; Sharma, A.; Dragoi, R.; Baneu, P.; et al. COVID-19 and cardiovascular manifestations. Eur. Rev. Med. Pharmacol. Sci. 2022, 26, 4509–4519.
- Lindner, D.; Fitzek, A.; Bräuninger, H.; Aleshcheva, G.; Edler, C.; Meissner, K.; Scherschel, K.; Kirchhof, P.; Escher, F.; Schultheiss, H.P.; et al. Association of Cardiac Infection With SARS-CoV-2 in Confirmed COVID-19 Autopsy Cases. JAMA Cardiol. 2020, 5, 1281–1285.
- Shi, S.; Qin, M.; Shen, B.; Cai, Y.; Liu, T.; Yang, F.; Gong, W.; Liu, X.; Liang, J.; Zhao, Q.; et al. Association of Cardiac Injury With Mortality in Hospitalized Patients With COVID-19 in Wuhan, China. JAMA Cardiol. 2020, 5, 802–810.
- Shah, K.S.; Yang, E.H.; Maisel, A.S.; Fonarow, G.C. The Role of Biomarkers in Detection of Cardio-toxicity. Curr. Oncol. Rep. 2017, 19, 42.
- Zilinyi, R.; Czompa, A.; Czegledi, A.; Gajtko, A.; Pituk, D.; Lekli, I.; Tosaki, A. The Cardioprotective Effect of Metformin in Doxorubicin-Induced Cardiotoxicity: The Role of Autophagy. Molecules 2018, 23, 1184.
- Su, Y.; Qu, Y.; Zhao, F.; Li, H.; Mu, D.; Li, X. Regulation of autophagy by the nuclear factor kappaB signaling pathway in the hippocampus of rats with sepsis. J. Neuroinflammation 2015, 12, 116.
- Ojha, S.; Azimullah, S.; Mohanraj, R.; Sharma, C.; Yasin, J.; Arya, D.S.; Adem, A. Thymoquinone Protects against Myocardial Ischemic Injury by Mitigating Oxidative Stress and Inflammation. Evid. Based Complement. Altern. Med. 2015, 2015, 143629.
- Khan, M.A.; Younus, H. Potential Implications of Black Seed and its Principal Constituent Thymoquinone in the Treatment of COVID-19 Patients. Curr. Pharm. Biotechnol. 2021, 22, 1315–1324.
- Singhal, T. A Review of Coronavirus Disease-2019 (COVID-19). Indian J. Pediatr. 2020, 87, 281–286.
- Kanter, M. Thymoquinone attenuates lung injury induced by chronic toluene exposure in rats. Toxicol. Ind. Health 2011, 27, 387–395.
- Pourgholamhossein, F.; Sharififar, F.; Rasooli, R.; Pourgholi, L.; Nakhaeipour, F.; Samareh-Fekri, H.; Iranpour, M.; Mandegary, A. Thymoquinone effectively alleviates lung fibrosis induced by paraquat herbicide through down-regulation of pro-fibrotic genes and inhibition of oxidative stress. Environ. Toxicol. Pharmacol. 2016, 45, 340–345.
- Colak, M.; Kalemci, S.; Alpaydin, A.O.; Karacam, V.; Meteoglu, I.; Yilmaz, O.; Itil, B.O. Efficacy of thymoquinone in the treatment of experimental lipopolysaccharide-induced acute lung injury. Kardiochirurgia Torakochirurgia Pol. 2020, 17, 65–69.
- Boskabady, M.; Khazdair, M.R.; Bargi, R.; Saadat, S.; Memarzia, A.; Mohammadian Roshan, N.; Hosseini, M.; Askari, V.R.; Boskabady, M.H. Thymoquinone Ameliorates Lung Inflammation and Pathological Changes Observed in Lipopolysaccharide-Induced Lung Injury. Evid. Based Complement. Altern. Med. 2021, 2021, 6681729.
- Khazdair, M.R.; Ghafari, S.; Sadeghi, M. Possible therapeutic effects of Nigella sativa and its thymoquinone on COVID-19. Pharm. Biol. 2021, 59, 696–703.
- Lu, Y.; Li, X.; Geng, D.; Mei, N.; Wu, P.Y.; Huang, C.C.; Jia, T.; Zhao, Y.; Wang, D.; Xiao, A.; et al. Cerebral Micro-Structural Changes in COVID-19 Patients—An MRI-based 3-month Follow-up Study. EClinicalMedicine 2020, 25, 100484.
- Filatov, A.; Sharma, P.; Hindi, F.; Espinosa, P.S. Neurological Complications of Coronavirus Disease (COVID-19): Encephalopathy. Cureus 2020, 12, e7352.
- Farkhondeh, T.; Samarghandian, S.; Azimin-Nezhad, M.; Samini, F. Effect of chrysin on nociception in formalin test and serum levels of noradrenalin and corticosterone in rats. Int. J. Clin. Exp. Med. 2015, 8, 2465–2470.
- Cárdenas-Rodríguez, N.; Bandala, C.; Vanoye-Carlo, A.; Ignacio-Mejía, I.; Gómez-Manzo, S.; Hernández-Cruz, E.Y.; Pedraza-Chaverri, J.; Carmona-Aparicio, L.; Hernández-Ochoa, B. Use of Antioxidants for the Neuro-Therapeutic Management of COVID-19. Antioxidants 2021, 10, 971.
- Wang, Y.; Gao, H.; Zhang, W.; Zhang, W.; Fang, L. Thymoquinone inhibits lipopolysaccharide-induced inflammatory mediators in BV2 microglial cells. Int. Immunopharmacol. 2015, 26, 169–173.
- Mosley, R.L.; Benner, E.J.; Kadiu, I.; Thomas, M.; Boska, M.D.; Hasan, K.; Laurie, C.; Gendelman, H.E. Neuroinflammation, Oxidative Stress and the Pathogenesis of Parkinson’s Disease. Clin. Neurosci. Res. 2006, 6, 261–281.
- Alhebshi, A.H.; Gotoh, M.; Suzuki, I. Thymoquinone protects cultured rat primary neurons against amyloid beta-induced neurotoxicity. Biochem. Biophys. Res. Commun. 2013, 433, 362–367.
- Parohan, M.; Yaghoubi, S.; Seraji, A. Liver injury is associated with severe coronavirus disease 2019 (COVID-19) infection:A systematic review and meta-analysis of retrospective studies. Hepatol. Res. 2020, 50, 924–935.
- Abd El Rhman, M.M.; Shafik, N.S.; Maher, A.; Hemdan, S.B.; Abd Elhamed, R.M. Liver Injuries in COVID-19 infected patients. Sohag Med. J. 2019, 12, 223–230.
- Maizels, R.M.; McSorley, H.J. Regulation of the host immune system by helminth parasites. J. Allergy Clin. Immunol. 2016, 138, 666–675.
- Nili-Ahmadabadi, A.; Tavakoli, F.; Hasanzadeh, G.; Rahimi, H.; Sabzevari, O. Protective effect of pretreatment with thymoquinone against Aflatoxin B(1) induced liver toxicity in mice. Daru 2011, 19, 282–287.
- Bai, T.; Yang, Y.; Wu, Y.L.; Jiang, S.; Lee, J.J.; Lian, L.H.; Nan, J.X. Thymoquinone alleviates thioacetamide-induced hepatic fibrosis and inflammation by activating LKB1-AMPK signaling pathway in mice. Int. Immunopharmacol. 2014, 19, 351–357.
- Raghunandhakumar, S.; Paramasivam, A.; Senthilraja, S.; Naveenkumar, C.; Asokkumar, S.; Binuclara, J.; Jagan, S.; Anandakumar, P.; Devaki, T. Thymoquinone inhibits cell proliferation through regulation of G1/S phase cell cycle transition in N-nitrosodiethylamine-induced experimental rat hepatocellular carcinoma. Toxicol. Lett. 2013, 223, 60–72.
- Nagi, M.N.; Almakki, H.A. Thymoquinone supplementation induces quinone reductase and glutathione transferase in mice liver: Possible role in protection against chemical carcinogenesis and toxicity. Phytother. Res. 2009, 23, 1295–1298.
- Gabarre, P.; Dumas, G.; Dupont, T.; Darmon, M.; Azoulay, E.; Zafrani, L. Acute kidney injury in critically ill patients with COVID-19. Intensive Care Med. 2020, 46, 1339–1348.
- Li, A.; Zhang, W.; Zhang, L.; Liu, Y.; Li, K.; Du, G.; Qin, X. Elucidating the time-dependent changes in the urinary metabolome under doxorubicin-induced nephrotoxicity. Toxicol. Lett. 2020, 319, 204–212.
- Fouda, A.M.; Daba, M.H.; Dahab, G.M.; Sharaf El-Din, O.A. Thymoquinone ameliorates renal oxidative damage and proliferative response induced by mercuric chloride in rats. Basic Clin. Pharmacol. Toxicol. 2008, 103, 109–118.
- Sayed-Ahmed, M.M.; Nagi, M.N. Thymoquinone supplementation prevents the development of gentamicin-induced acute renal toxicity in rats. Clin. Exp. Pharmacol. Physiol. 2007, 34, 399–405.
- Guo, L.P.; Liu, S.X.; Yang, Q.; Liu, H.Y.; Xu, L.L.; Hao, Y.H.; Zhang, X.Q. Effect of Thymoquinone on Acute Kidney Injury Induced by Sepsis in BALB/c Mice. Biomed Res. Int. 2020, 2020, 1594726.
- Ince, S.; Kucukkurt, I.; Demirel, H.H.; Turkmen, R.; Sever, E. Thymoquinone attenuates cypermethrin induced oxidative stress in Swiss albino mice. Pestic. Biochem. Physiol. 2012, 104, 229–235.
- Su, S.; Shen, J.; Zhu, L.; Qiu, Y.; He, J.S.; Tan, J.Y.; Iacucci, M.; Ng, S.C.; Ghosh, S.; Mao, R.; et al. Involvement of digestive system in COVID-19: Manifestations, pathology, management and challenges. Ther. Adv. Gastroenterol. 2020, 13, 1756284820934626.
- Randhawa, M.A.; Alenazy, A.K.; Alrowaili, M.G.; Basha, J. An active principle of Nigella sativa L., thymoquinone, showing significant antimicrobial activity against anaerobic bacteria. J. Intercult. Ethnopharmacol. 2017, 6, 97–101.
- Goel, S.; Mishra, P. Thymoquinone inhibits biofilm formation and has selective antibacterial activity due to ROS generation. Appl. Microbiol. Biotechnol. 2018, 102, 1955–1967.
