Infertility is an increasing global public health issue that affects up to 16% of couples of reproductive age. According to the World Health Organization (WHO), infertility is a disease of the reproductive system defined by the failure to achieve a clinical pregnancy after 12 months of regular, unprotected sexual intercourse. Half of infertility cases are due to female factors, which are generally attributed to hormonal, functional or anatomical dysfunction of the organs of the reproductive tract. Moreover, the age of the female is of great importance because of its physiological and genetic influence on conception, which includes a reduced ovarian follicular pool, perturbations in ovulation and increased meiotic errors within the oocyte. As for male factors, these are responsible for 20–30% of infertility cases. The most important factors are hormonal deficits, physical causes, sexually transmitted problems, and genetic factors. Nevertheless, the origin of about 40% of male infertility is unknown. Many studies have investigated the potential use of melatonin in assisted reproductive techniques (ART) to improve success rates. These techniques include oocyte manipulation, artificial insemination, in vitro fertilization (IVF), and embryo culture and transfer.
- oxidative stress
- melatonin
- infertility
- assisted reproductive techniques
- oocyte quality
1. Sources of Reactive Oxygen Species (ROS) in Assisted Reproductive Techniques (ART)
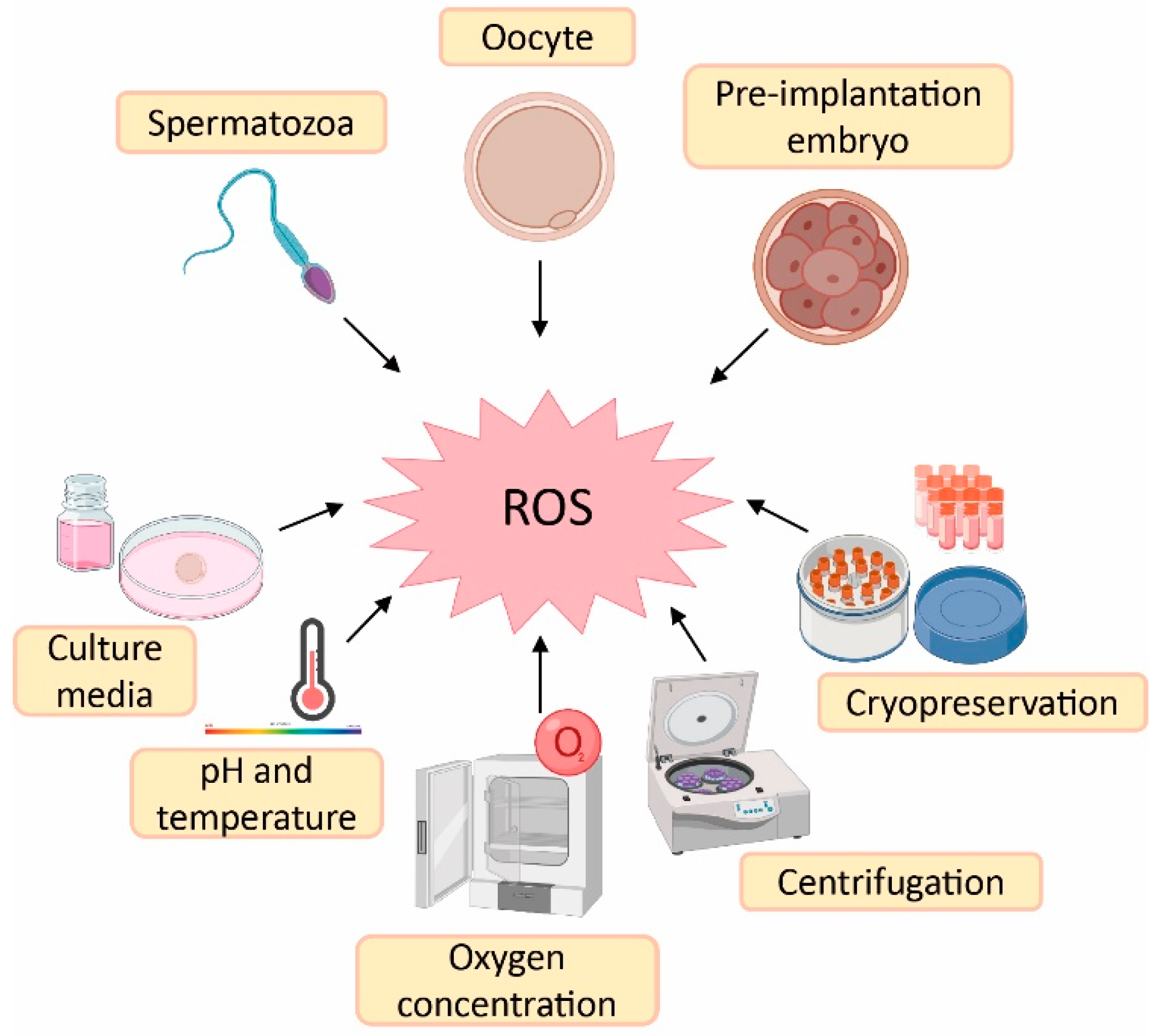
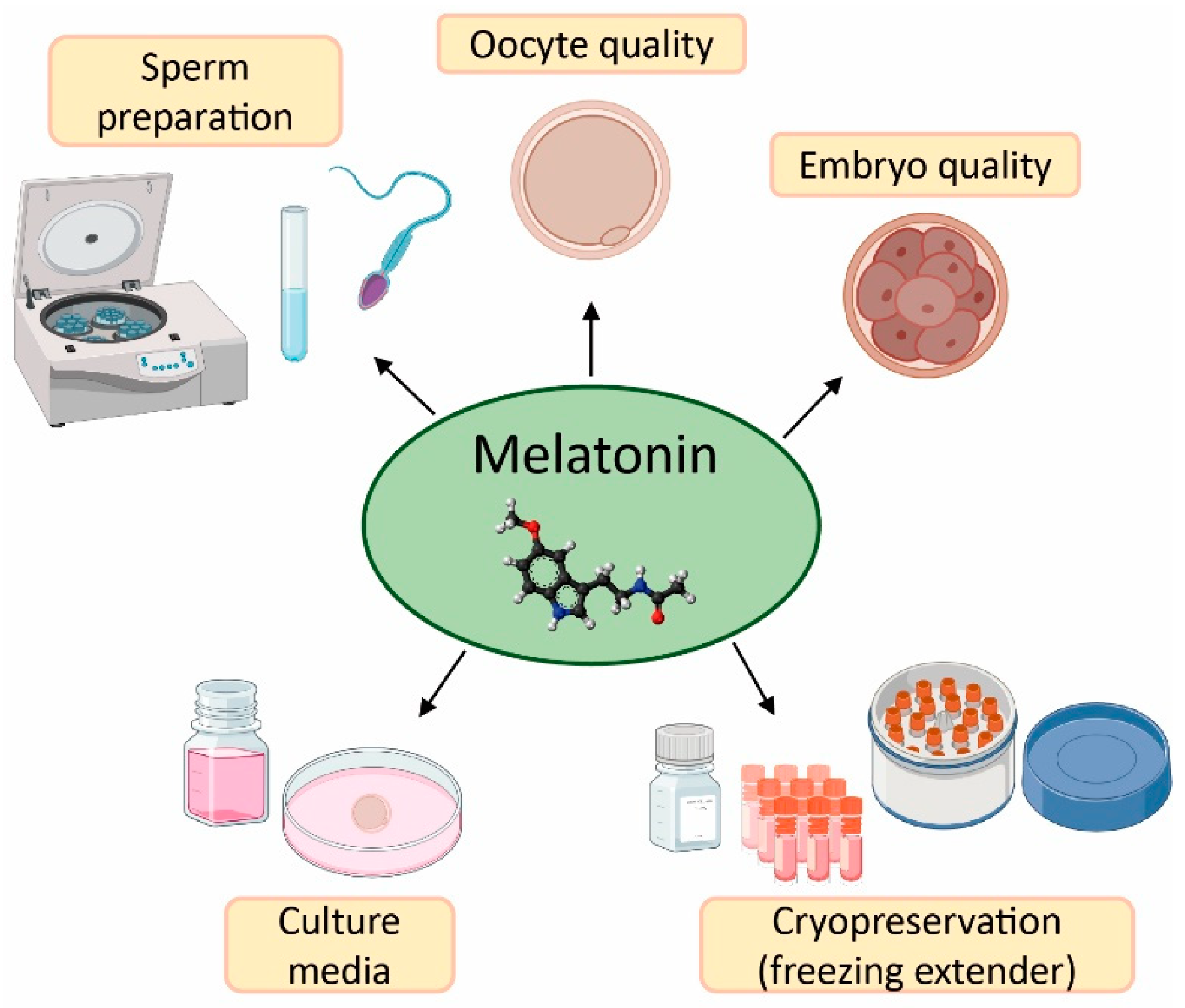
2. Effect of Melatonin in Oocyte Quality and Embryo Quality
3. Application of Melatonin in Sperm Preparation for ART
4. Melatonin as Protective Agent in Gametes Cryopreservation
References
- Aitken, R.J.; Jones, K.T.; Robertson, S.A. Reactive Oxygen Species and Sperm Function-in Sickness and in Health. J. Androl. 2012, 33, 1096–1106.
- Haldar, C.; Chowdhury, J.P. Management of Ovarian Functions by Melatonin. J. Reprod. Healthc. Med. 2021, 2, 16.
- Agarwal, A.; Durairajanayagam, D.; du Plessis, S.S. Utility of Antioxidants during Assisted Reproductive Techniques: An Evidence Based Review. Reprod. Biol. Endocrinol. 2014, 12, 112.
- Lampiao, F. Free Radicals Generation in an in Vitro Fertilization Setting and How to Minimize Them. World J. Obstet. Gynecol. 2012, 1, 29.
- Agarwal, A.; Durairajanayagam, D.; Virk, G.; Du Plessis, S.S. Sources of ROS in ART. In Strategies to Ameliorate Oxidative Stress during Assisted Reproduction; Agarwal, A., Durairajanayagam, D., Virk, G., Du Plessis, S.S., Eds.; Springer: Cham, Switzerland, 2014; pp. 3–22. ISBN 978-3-319-10258-0.
- Will, M.A.; Clark, N.A.; Swain, J.E. Biological PH Buffers in IVF: Help or Hindrance to Success. J. Assist. Reprod. Genet. 2011, 28, 711–724.
- Mantikou, E.; Bontekoe, S.; van Wely, M.; Seshadri, S.; Repping, S.; Mastenbroek, S. Low Oxygen Concentrations for Embryo Culture in Assisted Reproductive Technologies. Hum. Reprod. Update 2013, 19, 209.
- Kasterstein, E.; Strassburger, D.; Komarovsky, D.; Bern, O.; Komsky, A.; Raziel, A.; Friedler, S.; Ron-El, R. The Effect of Two Distinct Levels of Oxygen Concentration on Embryo Development in a Sibling Oocyte Study. J. Assist. Reprod. Genet. 2013, 30, 1073–1079.
- Agarwal, A.; Majzoub, A. Role of Antioxidants in Assisted Reproductive Techniques. World J. Mens. Health 2017, 35, 77.
- Di Santo, M.; Tarozzi, N.; Nadalini, M.; Borini, A. Human Sperm Cryopreservation: Update on Techniques, Effect on DNA Integrity, and Implications for ART. Adv. Urol. 2012, 2012, 854837.
- Majzoub, A.; Agarwal, A. Antioxidants in Sperm Cryopreservation. In Male Infertility: Contemporary Clinical Approaches, Andrology, ART and Antioxidants; Parekattil, S.J., Esteves, S.C., Agarwal, A., Eds.; Springer: Cham, Switzerland, 2020; pp. 671–678. ISBN 978-3-03-032299-1.
- Hashim, F.; Tvrdá, E.; Greifová, H.; Lukáč, N. Effect of Vitamins on the Quality of Insemination Doses of Bulls. J. Microbiol. Biotechnol. Food Sci. 2017, 7, 242–247.
- Fatemi, F.; Mohammadzadeh, A.; Sadeghi, M.R.; Akhondi, M.M.; Mohammadmoradi, S.; Kamali, K.; Lackpour, N.; Jouhari, S.; Zafadoust, S.; Mokhtar, S.; et al. Role of Vitamin E and D3 Supplementation in Intra-Cytoplasmic Sperm Injection Outcomes of Women with Polycystic Ovarian Syndrome: A Double Blinded Randomized Placebo-Controlled Trial. Clin. Nutr. ESPEN 2017, 18, 23–30.
- Takeo, S.; Sato, D.; Kimura, K.; Monji, Y.; Kuwayama, T.; Kawahara-Miki, R.; Iwata, H. Resveratrol Improves the Mitochondrial Function and Fertilization Outcome of Bovine Oocytes. J. Reprod. Dev. 2014, 60, 92–99.
- Gadani, B.; Bucci, D.; Spinaci, M.; Tamanini, C.; Galeati, G. Resveratrol and Epigallocatechin-3-Gallate Addition to Thawed Boar Sperm Improves in Vitro Fertilization. Theriogenology 2017, 90, 88–93.
- Wang, Y.; Zhang, M.; Chen, Z.J.; Du, Y. Resveratrol Promotes the Embryonic Development of Vitrified Mouse Oocytes after in Vitro Fertilization. Vitr. Cell. Dev. Biol.-Anim. 2018, 54, 430–438.
- Ardeshirnia, R.; Zandi, M.; Sanjabi, M.R. The Effect of Quercetin on Fertility of Frozen-Thawed Ram Epididymal Spermatozoa. S. Afr. J. Anim. Sci. 2017, 47, 237.
- Tamura, H.; Tanabe, M.; Jozaki, M.; Taketani, T.; Sugino, N. Antioxidative Action of Melatonin and Reproduction. Glycative Stress Res. 2019, 6, 192–197.
- Fernando, S.; Wallace, E.M.; Vollenhoven, B.; Lolatgis, N.; Hope, N.; Wong, M.; Lawrence, M.; Lawrence, A.; Russell, C.; Leong, K.; et al. Melatonin in Assisted Reproductive Technology: A Pilot Double-Blind Randomized Placebo-Controlled Clinical Trial. Front. Endocrinol. 2018, 9, 545.
- Lord, T.; Nixon, B.; Jones, K.T.; Aitken, R.J. Melatonin Prevents Postovulatory Oocyte Aging in the Mouse and Extends the Window for Optimal Fertilization in Vitro. Biol. Reprod. 2013, 88, 1–9.
- Lin, T.; Lee, J.E.; Kang, J.W.; Oqani, R.K.; Cho, E.S.; Kim, S.B.; Il Jin, D. Melatonin Supplementation during Prolonged in Vitro Maturation Improves the Quality and Development of Poor-Quality Porcine Oocytes via Anti-Oxidative and Anti-Apoptotic Effects. Mol. Reprod. Dev. 2018, 85, 665–681.
- Marques, T.C.; da Silva Santos, E.C.; Diesel, T.O.; Leme, L.O.; Martins, C.F.; Dode, M.A.N.; Alves, B.G.; Costa, F.P.H.; de Oliveira, E.B.; Gambarini, M.L. Melatonin Reduces Apoptotic Cells, SOD2 and HSPB1 and Improves the in Vitro Production and Quality of Bovine Blastocysts. Reprod. Domest. Anim. 2018, 53, 226–236.
- Wang, F.; Tian, X.Z.; Zhou, Y.H.; Tan, D.X.; Zhu, S.E.; Dai, Y.P.; Liu, G.S. Melatonin Improves the Quality of in Vitro Produced (IVP) Bovine Embryos: Implications for Blastocyst Development, Cryotolerance, and Modifications of Relevant Gene Expression. PLoS ONE 2014, 9, e93641.
- Li, Q.; Geng, X.D.; Zheng, W.; Tang, J.; Xu, B.; Shi, Q.H. Current Understanding of Ovarian Aging. Sci. China Life Sci. 2012, 55, 659–669.
- Tamura, H.; Kawamoto, M.; Sato, S.; Tamura, I.; Maekawa, R.; Taketani, T.; Aasada, H.; Takaki, E.; Nakai, A.; Reiter, R.J.; et al. Long-Term Melatonin Treatment Delays Ovarian Aging. J. Pineal Res. 2017, 62, e12381.
- Tamura, H.; Jozaki, M.; Tanabe, M.; Shirafuta, Y.; Mihara, Y.; Shinagawa, M.; Tamura, I.; Maekawa, R.; Sato, S.; Taketani, T.; et al. Importance of Melatonin in Assisted Reproductive Technology and Ovarian Aging. Int. J. Mol. Sci. 2020, 21, 1135.
- Batiioǧlu, A.S.; Şahin, U.; Grlek, B.; Öztrk, N.; Ünsal, E. The Efficacy of Melatonin Administration on Oocyte Quality. Gynecol. Endocrinol. 2012, 28, 91–93.
- Nishihara, T.; Hashimoto, S.; Ito, K.; Nakaoka, Y.; Matsumoto, K.; Hosoi, Y.; Morimoto, Y. Oral Melatonin Supplementation Improves Oocyte and Embryo Quality in Women Undergoing in Vitro Fertilization-Embryo Transfer. Gynecol. Endocrinol. 2014, 30, 359–362.
- Jahromi, B.N.; Sadeghi, S.; Alipour, S.; Parsanezhad, M.E.; Alamdarloo, S.M. Effect of Melatonin on the Outcome of Assisted Reproductive Technique Cycles in Women with Diminished Ovarian Reserve: A Double-Blinded Randomized Clinical Trial. Iran. J. Med. Sci. 2017, 42, 73–78.
- Espino, J.; Macedo, M.; Lozano, G.; Ortiz, A.; Rodríguez, C.; Rodríguez, A.B.; Bejarano, I. Impact of Melatonin Supplementation in Women with Unexplained Infertility Undergoing Fertility Treatment. Antioxidants 2019, 8, 338.
- Valeri, C.; Sbracia, G.; Selman, H.; Antonini, G.; Pacchiarotti, A. Beneficial Effects of Melatonin on Oocytes and Embryo Quality in Aged IVF Patients. In Human Reproduction; OXFORD UNIV PRESS: Oxford, UK, 2015; Volume 30, p. 57.
- Jiménez-Tuñón, J.M.; Trilles, P.P.; Molina, M.G.; Duvison, M.H.; Pastor, B.M.; Martín, P.S.; Martín, F.S.; Sánchez-Borrego, R. A Double-Blind, Randomized Prospective Study to Evaluate the Efficacy of Previous Therapy with Melatonin, Myo-Inositol, Folic Acid, and Selenium in Improving the Results of an Assisted Reproductive Treatment. Clin. Med. Insights Ther. 2017, 9, 1179559X17742902.
- Bejarano, I.; Monllor, F.; Marchena, A.M.; Ortiz, A.; Lozano, G.; Jiménez, M.I.; Gaspar, P.; García, J.F.; Pariente, J.A.; Rodríguez, A.B.; et al. Exogenous Melatonin Supplementation Prevents Oxidative Stress-Evoked DNA Damage in Human Spermatozoa. J. Pineal Res. 2014, 57, 333–339.
- Henkel, R.R.; Schill, W.B. Sperm Preparation for ART. Reprod. Biol. Endocrinol. 2003, 1, 108.
- Mehta, A.; Sigman, M. Identification and Preparation of Sperm for Art. Urol. Clin. N. Am. 2014, 41, 169–180.
- Perumal, P.; Vupru, K.; Rajkhowa, C. Effect of Addition of Trehalose on the Liquid Storage (5 °C) of Mithun (Bos Frontalis) Semen. Indian J. Anim. Res. 2013, 49, 837–846.
- Dehghani-Mohammadabadi, M.; Salehi, M.; Farifteh, F.; Nematollahi, S.; Arefian, E.; Hajjarizadeh, A.; Parivar, K.; Nourmohammadi, Z. Melatonin Modulates the Expression of BCL-Xl and Improve the Development of Vitrified Embryos Obtained by IVF in Mice. J. Assist. Reprod. Genet. 2014, 31, 453–461.
- Cheuquemán, C.; Arias, M.E.; Risopatrón, J.; Felmer, R.; Álvarez, J.; Mogas, T.; Sánchez, R. Supplementation of IVF Medium with Melatonin: Effect on Sperm Functionality and in Vitro Produced Bovine Embryos. Andrologia 2015, 47, 604–615.
- El-Raey, M.; Badr, M.R.; Assi, M.M.; Rawash, Z.M. Effect of Melatonin on Buffalo Bull Sperm Freezability, Ultrastructure Changes and Fertilizing Potentials. Assiut Vet. Med. J. 2015, 61, 201–208.
- Perumal, P.; Chang, S.; Sangma, C.T.R.; Savino, N.; Khate, K. Effect of Melatonin on Mobility and Velocity Parameters of Mithun (Bos Frontalis) Semen Preserved in Liquid State (5OC). J. Exp. Biol. Agric. Sci. 2016, 4, S95–S102.
- Thongrueang, N.; Chaibangyang, N.; Chanapiwat, P.; Kaeoket, K. Effects of Adding Melatonin on the Quality Offrozen-Thawed Boar Semen. J. Appl. Anim. Sci. 2017, 10, 47–56.
- Gutiérrez-Añez, J.C.; Henning, H.; Lucas-Hahn, A.; Baulain, U.; Aldag, P.; Sieg, B.; Hensel, V.; Herrmann, D.; Niemann, H. Melatonin Improves Rate of Monospermic Fertilization and Early Embryo Development in a Bovine IVF System. PLoS ONE 2021, 16, e0256701.
- Su, G.; Wu, S.; Wu, M.; Wang, L.; Yang, L.; Du, M.; Zhao, X.; Su, X.; Liu, X.; Bai, C.; et al. Melatonin Improves the Quality of Frozen Bull Semen and Influences Gene Expression Related to Embryo Genome Activation. Theriogenology 2021, 176, 54–62.
- Pang, Y.W.; Sun, Y.Q.; Jiang, X.L.; Huang, Z.Q.; Zhao, S.J.; Du, W.H.; Hao, H.S.; Zhao, X.M.; Zhu, H.B. Protective Effects of Melatonin on Bovine Sperm Characteristics and Subsequent in Vitro Embryo Development. Mol. Reprod. Dev. 2016, 83, 993–1002.
- Li, C.Y.; Hao, H.S.; Zhao, Y.H.; Zhang, P.P.; Wang, H.Y.; Pang, Y.W.; Du, W.H.; Zhao, S.J.; Liu, Y.; Huang, J.M.; et al. Melatonin Improves the Fertilization Capacity of Sex-Sorted Bull Sperm by Inhibiting Apoptosis and Increasing Fertilization Capacitation via MT1. Int. J. Mol. Sci. 2019, 20, 3921.
- Stival, C.; Puga Molina, L.D.C.; Paudel, B.; Buffone, M.G.; Visconti, P.E.; Krapf, D. Sperm Capacitation and Acrosome Reaction in Mammalian Sperm. Adv. Anat. Embryol. Cell Biol. 2016, 220, 93–106.
- Gimeno-Martos, S.; Casao, A.; Yeste, M.; Cebrián-Pérez, J.A.; Muiño-Blanco, T.; Pérez-Pé, R. Melatonin Reduces CAMP-Stimulated Capacitation of Ram Spermatozoa. Reprod. Fertil. Dev. 2019, 31, 420–431.
- Casao, A.; Mendoza, N.; Pérez-Pé, R.; Grasa, P.; Abecia, J.A.; Forcada, F.; Cebrián-Pérez, J.A.; Muino-Blanco, T. Melatonin Prevents Capacitation and Apoptotic-like Changes of Ram Spermatozoa and Increases Fertility Rate. J. Pineal Res. 2010, 48, 39–46.
- Fujinoki, M. Melatonin-Enhanced Hyperactivation of Hamster Sperm. Reproduction 2008, 136, 533–541.
- Sohrabi, G.; Malmir, M.; Ghafarizadeh, A. Ameliorative Effect of Melatonin versus the Passage of Time and Lipid Peroxidation on Sperm Motility in Asthenotratospermic Men. Jorjani Biomed. J. 2021, 9, 44–54.
- Monllor, F.; Espino, J.; Marchena, A.M.; Ortiz, A.; Lozano, G.; García, J.F.; Pariente, J.A.; Rodríguez, A.B.; Bejarano, I. Melatonin Diminishes Oxidative Damage in Sperm Cells, Improving Assisted Reproductive Techniques. Turkish J. Biol. 2017, 41, 881–889.
- Ortiz, A.; Espino, J.; Bejarano, I.; Lozano, G.M.; Monllor, F.; García, J.F.; Pariente, J.A.; Rodríguez, A.B. High Endogenous Melatonin Concentrations Enhance Sperm Quality and Short-Term in Vitro Exposure to Melatonin Improves Aspects of Sperm Motility. J. Pineal Res. 2011, 50, 132–139.
- Du Plessis, S.S.; Hagenaar, K.; Lampiao, F. The in Vitro Effects of Melatonin on Human Sperm Function and Its Scavenging Activities on NO and ROS. Andrologia 2010, 42, 112–116.
- Espino, J.; Bejarano, I.; Ortiz, A.; Lozano, G.M.; García, J.F.; Pariente, J.A.; Rodríguez, A.B. Melatonin as a Potential Tool against Oxidative Damage and Apoptosis in Ejaculated Human Spermatozoa. Fertil. Steril. 2010, 94, 1915–1917.
- Espino, J.; Ortiz, A.; Bejarano, I.; Lozano, G.M.; Monllor, F.; García, J.F.; Rodríguez, A.B.; Pariente, J.A. Melatonin Protects Human Spermatozoa from Apoptosis via Melatonin Receptor- and Extracellular Signal-Regulated Kinase-Mediated Pathways. Fertil. Steril. 2011, 95, 2290–2296.
- Mir, S.M.; Aliarab, A.; Goodarzi, G.; Shirzad, M.; Jafari, S.M.; Qujeq, D.; Samavarchi-Tehrani, S.; Asadi, J. Melatonin: A Smart Molecule in the DNA Repair System. Cell Biochem. Funct. 2022, 40, 4–16.
- Huleihel, M.; Lunenfeld, E. Approaches and Technologies in Male Fertility Preservation. Int. J. Mol. Sci. 2020, 21, 5471.
- Sun, T.C.; Li, H.Y.; Li, X.Y.; Yu, K.; Deng, S.L.; Tian, L. Protective Effects of Melatonin on Male Fertility Preservation and Reproductive System. Cryobiology 2020, 95, 1–8.
- Tiwari, S.; Dewry, R.K.; Srivastava, R.; Nath, S.; Mohanty, T.K. Targeted Antioxidant Delivery Modulates Mitochondrial Functions, Ameliorates Oxidative Stress and Preserve Sperm Quality during Cryopreservation. Theriogenology 2022, 179, 22–31.
- Ugur, M.R.; Saber Abdelrahman, A.; Evans, H.C.; Gilmore, A.A.; Arifiantini, R.I.; Purwantara, B.; Kaya, A.; Memili, E. Advances in Cryopreservation of Bull Sperm. Front. Vet. Sci. 2019, 6, 268.
- Kopeika, J.; Thornhill, A.; Khalaf, Y. The Effect of Cryopreservation on the Genome of Gametes and Embryos: Principles of Cryobiology and Critical Appraisal of the Evidence. Hum. Reprod. Update 2015, 21, 209–227.
- Said, T.M.; Gaglani, A.; Agarwal, A. Implication of Apoptosis in Sperm Cryoinjury. Reprod. Biomed. Online 2010, 21, 456–462.
- Lemma, A. Effect of Cryopreservation on Sperm Quality and Fertility. In Artificial Insemination in Farm Animals; Manafi, M., Ed.; IntechOpen: London, UK, 2011; pp. 190–216. ISBN 9782367030029.
- Grötter, L.G.; Cattaneo, L.; Marini, P.E.; Kjelland, M.E.; Ferré, L.B. Recent Advances in Bovine Sperm Cryopreservation Techniques with a Focus on Sperm Post-Thaw Quality Optimization. Reprod. Domest. Anim. 2019, 54, 655–665.
- Cruz, M.H.C.; Leal, C.L.V.; da Cruz, J.F.; Tan, D.X.; Reiter, R.J. Role of Melatonin on Production and Preservation of Gametes and Embryos: A Brief Review. Anim. Reprod. Sci. 2014, 145, 150–160.
- Ashrafi, I.; Kohram, H.; Ardabili, F.F. Antioxidative Effects of Melatonin on Kinetics, Microscopic and Oxidative Parameters of Cryopreserved Bull Spermatozoa. Anim. Reprod. Sci. 2013, 139, 25–30.
- El-Raey, M.; Badr, M.R.; Rawash, Z.M.; Darwish, G.M. Evidences for the Role of Melatonin as a Protective Additive during Buffalo Semen Freezing. Am. J. Anim. Vet. Sci. 2014, 9, 252–262.
- Inyawilert, W.; Rungruangsak, J.; Liao, Y.J.; Tang, P.C.; Paungsukpaibool, V. Melatonin Supplementation Improved Cryopreserved Thai Swamp Buffalo Semen. Reprod. Domest. Anim. 2021, 56, 83–88.
- Chaudhary, S.C.; Aeksiri, N.; Wanangkarn, A.; Liao, Y.J.; Inyawilert, W. Effects of Melatonin on Cryopreserved Semen Parameters and Apoptosis of Thai Swamp Buffalo Bull (Bubalus Bubalis) in Different Thawing Conditions. Adv. Anim. Vet. Sci. 2021, 9, 238–245.
- Fadl, A.M.; Ghallab, A.R.M.; Abou-Ahmed, M.M.; Moawad, A.R. Melatonin Can Improve Viability and Functional Integrity of Cooled and Frozen/Thawed Rabbit Spermatozoa. Reprod. Domest. Anim. 2021, 56, 103–111.
- Lançoni, R.; Celeghini, E.C.C.; Alves, M.B.R.; Lemes, K.M.; Gonella-Diaza, A.M.; Oliveira, L.Z.; Arruda, R.P. Melatonin Added to Cryopreservation Extenders Improves the Mitochondrial Membrane Potential of Postthawed Equine Sperm. J. Equine Vet. Sci. 2018, 69, 78–83.
- Divar, M.R.; Azari, M.; Mogheiseh, A.; Ghahramani, S. Supplementation of Melatonin to Cooling and Freezing Extenders Improves Canine Spermatozoa Quality Measures. BMC Vet. Res. 2022, 18, 86.
- Succu, S.; Berlinguer, F.; Pasciu, V.; Satta, V.; Leoni, G.G.; Naitana, S. Melatonin Protects Ram Spermatozoa from Cryopreservation Injuries in a Dose-Dependent Manner. J. Pineal Res. 2011, 50, 310–318.
- Appiah, M.O.; He, B.; Lu, W.; Wang, J. Antioxidative Effect of Melatonin on Cryopreserved Chicken Semen. Cryobiology 2019, 89, 90–95.
- Chen, X.J.; Zhang, Y.; Jia, G.X.; Meng, Q.G.; Bunch, T.D.; Liu, G.S.; Zhu, S.E.; Zhou, G.B. Effect of Melatonin Supplementation on Cryopreserved Sperm Quality in Mouse. Cryo-Letters 2016, 37, 115–122.
- Yu, K.; Deng, S.L.; Sun, T.C.; Li, Y.Y.; Liu, Y.X. Melatonin Regulates the Synthesis of Steroid Hormones on Male Reproduction: A Review. Molecules 2018, 23, 447.
- Karimfar, M.H.; Niazvand, F.; Haghani, K.; Ghafourian, S.; Shirazi, R.; Bakhtiyari, S. The Protective Effects of Melatonin against Cryopreservation-Induced Oxidative Stress in Human Sperm. Int. J. Immunopathol. Pharmacol. 2015, 28, 69–76.
- Najafi, A.; Adutwum, E.; Yari, A.; Salehi, E.; Mikaeili, S.; Dashtestani, F.; Abolhassani, F.; Rashki, L.; Shiasi, S.; Asadi, E. Melatonin Affects Membrane Integrity, Intracellular Reactive Oxygen Species, Caspase3 Activity and AKT Phosphorylation in Frozen Thawed Human Sperm. Cell Tissue Res. 2018, 372, 149–159.
- Pariz, J.R.; Raneá, C.; Monteiro, R.A.C.; Evenson, D.P.; Drevet, J.R.; Hallak, J. Melatonin and Caffeine Supplementation Used, Respectively, as Protective and Stimulating Agents in the Cryopreservation of Human Sperm Improves Survival, Viability, and Motility after Thawing Compared to Traditional TEST-Yolk Buffer. Oxid. Med. Cell. Longev. 2019, 2019, 6472945.
- Liang, T.; Motan, T. Mature Oocyte Cryopreservation for Fertility Preservation. In Advances in Experimental Medicine and Biology; Karimi-Busheri, F., Weindfeld, M., Eds.; Springer: Berlin/Heidelberg, Germany, 2016; Volume 951, pp. 155–161. ISBN 9783319454559.
- Schattman, G.L. Cryopreservation of Oocytes. N. Engl. J. Med. 2015, 373, 1755–1760.
- Dunselman, G.A.; Vermeulen, N.; Becker, C.; Calhaz-Jorge, C.; D’Hooghe, T.; nd De Bie, B. Management of Women with Endometriosis: Guideline of the European Society of Human Reproduction and Embryology. Eur. Soc. Hum. Reprod. Embryol. 2013, 82, 1–97.
- Tang, Y.; Zhang, Y.; Liu, L.; Yang, Y.; Wang, Y.; Xu, B. Glycine and Melatonin Improve Preimplantation Development of Porcine Oocytes Vitrified at the Germinal Vesicle Stage. Front. Cell Dev. Biol. 2022, 10, 856486.
- Aghaz, F.; Vaisi-Raygani, A.; Khazaei, M.; Arkan, E. Enhanced Cryoprotective Effect of Melatonin and Resveratrol by Coencapsulation: Improved in Vitro Development of Vitrified-Warmed Mouse Germinal Vesicle Oocytes. Biopreserv. Biobank. 2021, 19, 184–193.
- Zhang, Z.; Mu, Y.; Ding, D.; Zou, W.; Li, X.; Chen, B.; Leung, P.C.K.; Chang, H.M.; Zhu, Q.; Wang, K.; et al. Melatonin Improves the Effect of Cryopreservation on Human Oocytes by Suppressing Oxidative Stress and Maintaining the Permeability of the Oolemma. J. Pineal Res. 2021, 70, e12707.
- Doroudi, R.; Changizi, Z.; Nematollahi-Mahani, S.N. Effects of Melatonin and Human Follicular Fluid Supplementation of in Vitro Maturation Medium on Mouse Vitrified Germinal Vesicle Oocytes: A Laboratory Study. Int. J. Reprod. Biomed. 2021, 19, 889–898.
- Qin, J.; Guo, S.; Yang, J.; Qazi, I.H.; Pan, B.; Lv, T.; Zang, S.; Fang, Y.; Zhou, G. Melatonin Promotes in Vitro Development of Vitrified-Warmed Mouse GV Oocytes, Potentially by Modulating Phosphorylation of Drp1. Front. Vet. Sci. 2021, 8, 752001.
- Guo, S.; Yang, J.; Qin, J.; Qazi, I.H.; Pan, B.; Zang, S.; Lv, T.; Deng, S.; Fang, Y.; Zhou, G. Melatonin Promotes in Vitro Maturation of Vitrified-Warmed Mouse Germinal Vesicle Oocytes, Potentially by Reducing Oxidative Stress through the Nrf2 Pathway. Animals 2021, 11, 2324.
- Yang, J.; Guo, S.; Pan, B.; Qazi, I.H.; Qin, J.; Zang, S.; Han, H.; Meng, Q.; Zhou, G. Melatonin Promotes in Vitro Maturation of Vitrified-Warmed Mouse GV Oocytes Potentially by Modulating MAD2 Protein Expression of SAC Component through MTRs. Cryobiology 2021, 102, 82–91.
- Wu, Z.; Pan, B.; Qazi, I.H.; Yang, H.; Guo, S.; Yang, J.; Zhang, Y.; Zeng, C.; Zhang, M.; Han, H.; et al. Melatonin Improves in Vitro Development of Vitrified-Warmed Mouse Germinal Vesicle Oocytes. Cells 2019, 8, 1009.
- Pan, B.; Qazi, I.H.; Guo, S.; Yang, J.; Qin, J.; Lv, T.; Zang, S.; Zhang, Y.; Zeng, C.; Meng, Q.; et al. Melatonin Improves the First Cleavage of Parthenogenetic Embryos from Vitrified–Warmed Mouse Oocytes Potentially by Promoting Cell Cycle Progression. J. Anim. Sci. Biotechnol. 2021, 12, 84.
- Pan, B.; Yang, H.; Wu, Z.; Qazi, I.H.; Liu, G.; Han, H.; Meng, Q.; Zhou, G. Melatonin Improves Parthenogenetic Development of Vitrified–Warmed Mouse Oocytes Potentially by Promoting G1/S Cell Cycle Progression. Int. J. Mol. Sci. 2018, 19, 4029.
- Gao, L.; Du, M.; Zhuan, Q.; Luo, Y.; Li, J.; Hou, Y.; Zeng, S.; Zhu, S.; Fu, X. Melatonin Rescues the Aneuploidy in Mice Vitrified Oocytes by Regulating Mitochondrial Heat Product. Cryobiology 2019, 89, 68–75.
- Lee, A.R.; Hong, K.; Choi, S.H.; Park, C.; Park, J.K.; Lee, J.I.; Bang, J.I.; Seol, D.W.; Lee, J.E.; Lee, D.R. Anti-Apoptotic Regulation Contributes to the Successful Nuclear Reprogramming Using Cryopreserved Oocytes. Stem Cell Reports 2019, 12, 545–556.
- Zhang, Y.; Li, W.; Ma, Y.; Wang, D.; Zhao, X.; Zeng, C.; Zhang, M.; Zeng, X.; Meng, Q.; Zhou, G. Improved Development by Melatonin Treatment after Vitrification of Mouse Metaphase II Oocytes. Cryobiology 2016, 73, 335–342.
- Clérico, G.; Taminelli, G.; Veronesi, J.C.; Polola, J.; Pagura, N.; Pinto, C.; Sansinena, M. Mitochondrial Function, Blastocyst Development and Live Foals Born after ICSI of Immature Vitrified/Warmed Equine Oocytes Matured with or without Melatonin. Theriogenology 2021, 160, 40–49.
- Li, W.; Cheng, K.; Zhang, Y.; Meng, Q.; Zhu, S.; Zhou, G. No Effect of Exogenous Melatonin on Development of Cryopreserved Metaphase II Oocytes in Mouse. J. Anim. Sci. Biotechnol. 2015, 6, 42.
