Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Conner Chen and Version 1 by TURKI ABUALAIT.
Epilepsy is a long-term neurological condition that results in recurrent seizures. Approximately 30% of patients with epilepsy have drug-resistant epilepsy (DRE). The ketogenic diet (KD) is considered an effective alternative treatment for epileptic patients.
- epilepsy
- drug-resistant epilepsy
- biomarkers
- parameters
1. Epilepsy
Epilepsy is bimodally distributed with two peaks at both extremes of life: it is highest in the first year, then incidence drops to adult levels by the age of 10, before incidence rises again in people over the age of 85 years [10][1]. Incidence is higher in low-income countries, and usually above 80–100 per 100,000 persons per year for unknown reasons, but sub-standard health-delivery system, poor hygiene, lack of basic sanitation, and a higher risk of infections and traumatic brain injury may contribute [11,12][2][3].
Epilepsy is defined as: (1) two unprovoked seizures occurring more than 24 h 24 apart; or (2) a single unprovoked seizure if recurrence risk is high (i.e., >60% over the next 10 years) or (3) a diagnosis of an epilepsy syndrome [12][3]. Figure 1 shows the etiologies of epilepsy at various ages [13][4].
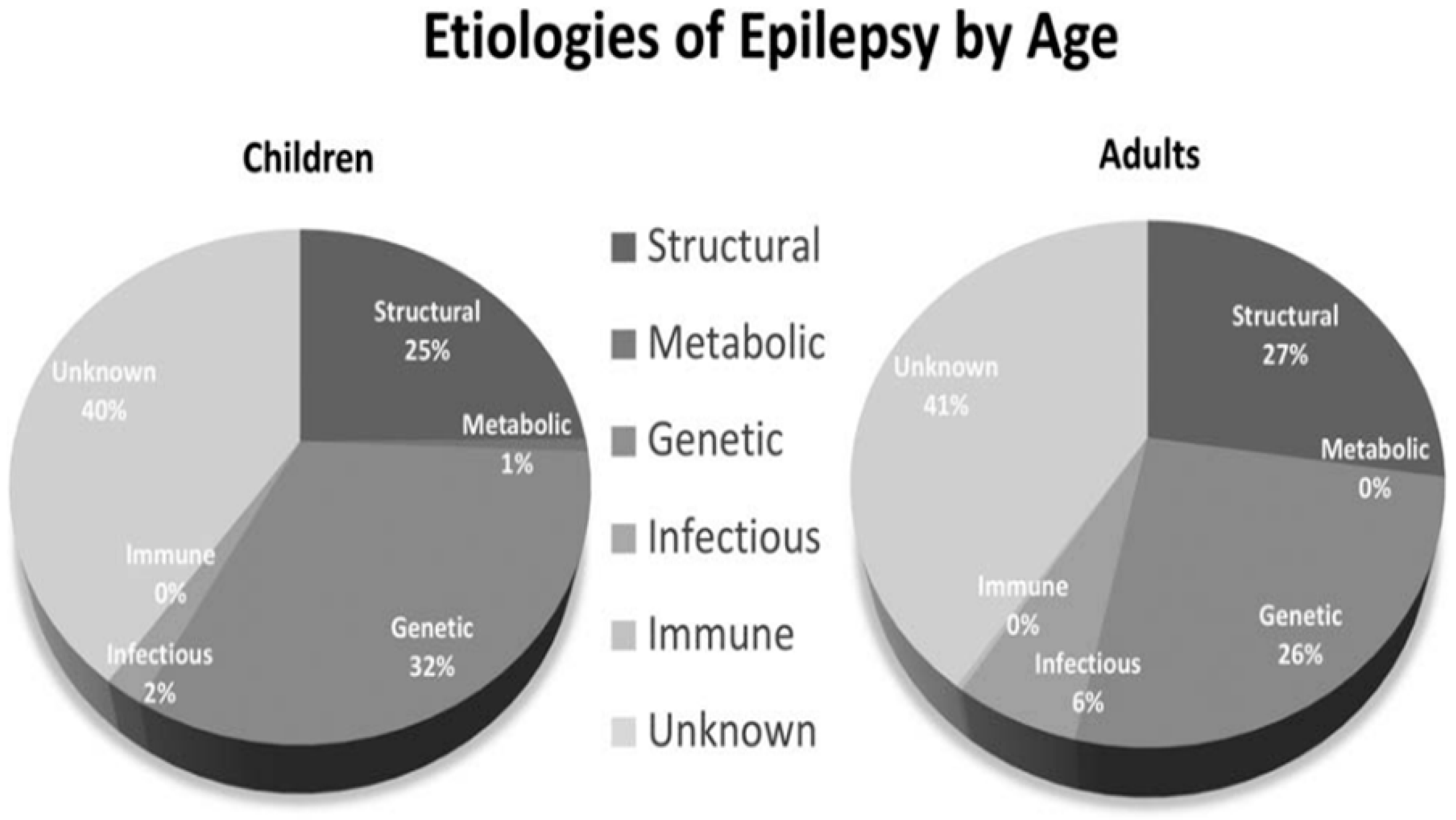
Figure 1.
Etiologies of epilepsy by age.
The process converting a non-epileptic brain into one capable of generating spontaneous, recurrent seizures is known as “Epileptogenesis”. The process is conceptualized to result from an imbalance between excitatory and inhibitory activity within a neuronal network, becomes more disposed to fire in an excessive, hypersynchronous, oscillatory manner which when sustained, disrupts normal neuronal processing, and is capable of recruiting other neuronal networks [10,11][1][2].
During the past several decades, neuroimaging, genomics, and molecular biology have substantially improved our knowledge of the pathophysiology of seizures and epilepsy [14,15][5][6]. Seizure is the main incident indicator found in epilepsy that is related with high persistence pulse, emitted from a set of neurons [16][7]. Seizures can present in various forms; seizures can present with motor symptoms or behavioral changes. Seizures can also happen with the patient aware or unaware [17][8].
2. Prevalence
Epilepsy affects 1–2% of people worldwide [18,19,20][9][10][11]. It is estimated that 23 million Asians suffer from epilepsy, while only 3.3 million Africans are affected by this disease [10][1]. Epilepsy affects both males and females of all ages. Focal seizures are common in children and adults [10][1].
3. Diagnosis
After carful history taking and examination, techniques such as neuroimaging, neurophysiological studies, and lab tests are used in diagnosing epileptic seizures and related disorders [21][12].
4. Mortality
Epilepsy death rates are relatively high in the USA and UK [22][13]. One study has linked epilepsy to 15% of deaths, and it remains unclear how to lessen this risk, while other studies reported 87.5% mortalities due to non-epileptic reasons [23][14]. Age, generalized seizures, and other independent variables raise the risk of death among epileptic patients [23,24][14][15].
5. Epilepsy Therapy
Epilepsy patients must be managed in the aim to become seizure-free. The type of epileptic syndrome determines treatment, as does the patient’s age, gender, and acceptance [25][16].
6. Medicines
Anti-epileptic drugs (AEDs) work by boosting neurotransmitters or decreasing excitatory processes [26][17]. In the US, phenytoin is considered a frequently used AED. Unfortunately, its metabolic role in the liver and random pharmacokinetics is not fruitful for older individuals [26][17].
Although there are several therapies, the treatment of epilepsy is based mainly on drugs, which, depending on the year of coming onto the market are classified as first, second, or third generation. The new-generation (third generation) AEDs may offer better tolerability, milder adverse effects, less drug interactions and improved pharmacokinetic characteristics compared to the conventional AEDs. For this reason, the New-generation AEDs may be used earlier in epileptic patients. Further head-to-head comparisons are needed to determine the exact position of New-generation AEDs relative to conventional AEDs, because, despite advancements and the development of New-generation AEDs, a third of patients with epilepsy remain refractory to pharmacotherapy [27][18].
Nanomaterials or nanomedicine, especially biosensor-based methods, can facilitate the analysis of these agents with unique advantages such as rapid analysis, sensitivity, selectivity, and low cost. Additionally, various chemical and biological modifiers to improve the sensitivity and selectivity of the sensor have been also been categorized [28][19]. These new molecules have been developed in order to provide a pharmaceutical profile and tolerance superior to the previously available drugs, and it is forecast that as their use increases, their true potential and profile will be widen. Furthermore, for the first time in Paediatric Epileptology, the extrapolation of the efficacy data in adults have been also been used (together with specific safety and pharmacokinetic studies in the paediatric population), in order to speed up their approval for use in the child population in upcoming years [29][20].
7. Surgery
Epileptic surgery is a potentially curative treatment for children with refractory seizures. Early epileptic surgery has been emphasized to treat medically intractable epilepsy in children. Seizure reduction results in remarkable developmental and cognitive improvements. Prolonged invasive extraoperative electroencephalography (EEG) or stereoEEG monitoring with depth electrodes and/or subdural grids are usually used for patients with nonlesional MRI or discordant EEG epileptogenic zones [31][21]. Epileptic surgery is among the most successful methods to achieve a seizure-free status [32,33][22][23]. Approximately 50%–80% of patients became seizure-free after surgery [34][24].
8. Dietary Treatment
In drug-resistant epilepsy, diet alteration is an alternative non-pharmacological option to treat epileptic seizures and is widely used to treat glucose transporter type 1 deficiency syndrome (GLUT1 DS), pyruvate dehydrogenase deficiency [35][25]. The ketoge KDnic diet (KD) has long been used to treat epileptic seizures [36][26]. The potential of the KD to control epileptic seizures has been known about for a century in medical and research institutes. Additionally, efforts are being made to recognize the KD’s therapeutic role in treating acute and severe metabolic disorders [37][27].
9. Ketogenic Diet
KD is defined as a diet containing a high amount of fat, low in carbohydrates, and with adequate protein content. It was first designed in the 1920s to treat seizures and supplies energy through ketone bodies (KBs) to the brain when the glucose level is lower in the body [38][28]. There are three main KBs: β-hydroxybutyrate (BHB), acetoacetate (ACA), and acetone. KBs act as fuel elements and are mainly formed from fatty acids by the liver during starvation and exercise [39][29]. The medium-chain triglycerides diet (MCTD) consists of high fat content with low glycemic index (LGI) [40][30]. Ketogenesis is a metabolic process that provides the body with an alternative form of energy through the production of KBs [41][31]. In ketogenesis, acetyl-CoA derived from β-oxidation of fatty acids is converted into KBs in the mitochondrial matrix of liver cells and then these ketone bodies are carried to the extrahepatic tissues for alternative energy sources. Adenosine has long been linked to metabolic and neural activity, and studies have proven that a ketogenic diet suppresses seizures by increasing inhibitory effects mediated through adenosine A1 receptors [8,42][32][33].
The ketone bodies, which are derived from fatty acid oxidation and usually produced in fasting state or on high-fat diets, have broad neuroprotective effects [43][34]. It is also suggested that the insulin sensitivity increased during a Ketogenic meal [44][35]. Furthermore, the neuroprotection and homeostasis also promotes the activation of inhibitory adenosine A1 receptors (A1Rs) by dephosphorylating extracellular ATP to adenosine [45][36]. Also, it activates GIRKs, which are G protein-coupled inwardly rectifying K+ channels. KATP channels activation may also be linked to A1R activation by a KD [46][37]. Another molecular relationship exists between the KD and γ-aminobutyric acid (GABA) levels and KATP channel activation through GABAB receptors. KATP channels activation has also been reported by other stimulants such as xanthine, diazoxide, etc. KATP channels play basic roles in nerve, muscle, epithelial, and endocrine tissue physiology and their direct activation regulates pancreatic islet β-cell membrane potential, calcium influx, and insulin secretion, and rectifies drug targets for metabolic disorders of glucose homeostasis [47][38]. Enhanced PIP3 signaling in pro-opiomelanocortin (POMC) neurons causes a KATP channel activation that leads to diet-sensitive obesity. In a mice study, a POMC neurons showed a marked hyperpolarization and a reduction in basal firing rate due to increased ATP-sensitive potassium (KATP) channel activity as well. The KATP blocker (e.g., tolbutamide) restored electrical activity and leptin-evoked firing of POMC neurons in mice. These data indicate that PIP3-mediated signals are critical regulators of the melanocortin system via modulation of KATP channels [48][39]. In another study it was well documented that KATP channel blockers control glucagon secretion by distinct mechanisms i.e., a direct stimulation of α-cells involving a [Ca2+]c rise and an indirect inhibition mediated by somatostatin. By closing α-cell KATP channels, sulfonylureas depolarize α-cells, increase [Ca2+]c, and stimulate glucagon secretion. However, their effects also involve an indirect inhibitory effect via somatostatin (SST) secreted by δ-cells on the glucose concentration [49,50][40][41].
Reactive oxygen species (ROS) may be reduced by metabolic modifications, improving seizure resistance (ROS). Fructose 1,6-bisphosphate administered to rats shifts glucose consumption to the pentose phosphate pathway [51][42].
10. Types of KD
KD is widely used to treat patients with refractory epilepsy or those individuals unfit for surgical management [52][43]. There are four types of KD [53][44].
Classic KD: In the classic KD, the ratio of fat and carbohydrates is 4:1. This ratio can be altered to 3:1 for moderate metabolism activity [7][45].
Medium Chain Triglyceride (MCTD): This modified Atkin diet includes high production of KBs than any other class of fats, such as long-chain triglycerides (LCT) [1][46]. It can lower the intake of fatty acids due to its ketogenic properties and greater carbohydrate and protein content due to its ketogenic properties because it contains high fat content (60%) and lower carbohydrate and protein ratio. Moreover, it also leads to marked alterations in brain energy metabolism, with ketone bodies partly replacing glucose as fuel. Though the phenomena is still not completely understood, it is reported that the ketone body acetone has anticonvulsant activity and could play a role in the seizure protection afforded by the diet [53][44]. In addition to acute seizure protection, the ketogenic diet provides protection against the development of spontaneous recurrent seizures in models of chronic epilepsy, and it has neuroprotective properties in diverse models of neurodegenerative disease [54,55][47][48]. The MCTD diet is more flexible for children than other KD because it increases the growth rate, decreases the requirement for other micronutrients, and has a lower cholesterol ratio [53][44].
Low glycemic index treatment (LGIT): LGIT is a non-restrictive treatment that consists of a diet with an increased amount of fat (60%), a high amount of protein (20 to 30%), and 10% carbohydrates [7][45]. It comprises foods with a low glycemic index (i.e., mutton, few fruits, dairy food) [55][48]. The fat:carbohydrate:protein ratio is about 1:6:0. There are no restrictions on diet and calories intake. Although LGIT represent fewer KBs than another KD, it is a better-tolerated diet.
Modified Atkins Diet (MAD): MAD constitutes 65% fat content, 25% protein, and a low carbohydrate intake (10%). The fat ratio is high in MAD and is considered the most savory form of KD and acceptable for adults or individuals suffering from behavioral issues [56][49]. There is no specific amount restriction of liquid or protein intake, but the carbohydrates amount is fixed as 10 to 20 g/day in infants and 15 to 20 g/day in youngsters. It is also recommended to take an appropriate calcium supplementation and a KD [57,58][50][51] because sufficient vitamins and minerals are normally found in a well-balanced diet. However, due to the limited quantities of fruits, vegetables, enriched grains, and foods containing calcium in the KD, supplementation is essential, especially vitamins B and C. Previous study also suggests that there is little vitamin D and calcium in KD and evidence for decreased Vitamin D levels in children with epilepsy, and therefore both vitamin D and calcium should be supplemented [59][52].
11. Biochemistry of KD
At the beginning of KD utilization, blood glucose becomes low and stabilizes, by which insulin release stops, and the body goes into a catabolic condition [53][44]. If insulin is depleted further through a KD and the gluconeogenesis process does not favor the metabolic reactions, free fatty acids (FFAs) are utilized by the body and provide power to the brain as the primary energy reservoir [39][29]. The brain can use KBs produced by the oxidation of FFAs in the absence of glucose [7,55,60][45][48][53]. Although the brain demands less glucose when KB levels are between 2 and 4 mM in the blood, these KBs might only meet up to 60% of brain activity demand [41][31]. After entering into mitochondria, fatty acids are separated and converted into acetyl-CoA by the action of β-oxidation. Large amounts of acetyl-CoA are generated when the liver deletes FFAs excessively by applying classic KD [61][54]. Oversupply of acetyl-CoA initiates the production of KBs through ketogenesis. Less insulin stimulates enzymatic transformation that results in ACA production from two molecules of acetyl-CoA that are further converted into acetone bodies or BHB [62][55]. These are the three KBs generated in the bloodstream during a prolonged period of KD application. These KBs can either be moved to the brain through monocarboxylate transporter 1 (MCT-1) [36][26] or can be removed via urination. After entering the brain, KBs are again changed into acetyl-CoA that reaches the tricarboxylic acid (TCA) cycle [60][53]. One of the significant roles of KBs is that it generates more ATP molecules than glucose, establishing metabolic activity even in starvation or caloric limitation [63][56]. Ketogenesis is a functional modification to a malnourishment condition or a diet consisting of lower carbohydrate function as KBs present in the bloodstream in the range of 2–5 mM for 1–2 weeks [64][57]. Ketogenesis never implicates a change in acid-base stability or reducing blood pH. By constantly raising the amount of KBs in the bloodstream and the presence of lipids and blood glucose, the alleged state of beneficial ketosis is established by KD introduction [41][31]. Ketosis is a naturally occurring process in response to low glucose supply and is involved in the continuity of human life. It acts as an alternative fuel reservoir for brain and muscle tissues, compensating for glucose deficiency [36][26].References
- Beghi, E. The Epidemiology of Epilepsy. Neuroepidemiology 2020, 54, 185–191.
- Thijs, R.D.; Surges, R.; O‘Brien, T.J.; Sander, J.W. Epilepsy in adults. Lancet 2019, 393, 689–701.
- Perucca, P.; Bahlo, M.; Berkovic, S.F. The Genetics of Epilepsy. Annu. Rev. Genomics. Hum. Genet. 2020, 21, 205–230.
- Falco-Walter, J. Epilepsy-Definition, Classification, Pathophysiology, and Epidemiology. Semin. Neurol. 2020, 40, 617–623.
- Pressler, R.M.; Cilio, M.R.; Mizrahi, E.M.; Moshé, S.L.; Nunes, M.L.; Plouin, P.; Vanhatalo, S.; Yozawitz, E.; de Vries, L.S.; Puthenveettil Vinayan, K.; et al. The ILAE classification of seizures and the epilepsies: Modification for seizures in the neonate. Position paper by the ILAE Task Force on Neonatal Seizures. Epilepsia 2021, 62, 615–628.
- Scheffer, I.E.; Berkovic, S.; Capovilla, G.; Connolly, M.B.; French, J.; Guilhoto, L.; Hirsch, E.; Jain, S.; Mathern, G.W.; Moshé, S.L.; et al. ILAE classification of the epilepsies: Position paper of the ILAE Commission for Classification and Terminology. Epilepsia 2017, 58, 512–521.
- Baumgartner, C.; Koren, J.; Britto-Arias, M.; Schmidt, S.; Pirker, S. Epidemiology and pathophysiology of autonomic seizures: A systematic review. Clin. Auton. Res. 2019, 29, 137–150.
- Fisher, R.S.; Cross, J.H.; French, J.A.; Higurashi, N.; Hirsch, E.; Jansen, F.E.; Lagae, L.; Moshé, S.L.; Peltola, J.; Roulet Perez, E.; et al. Operational classification of seizure types by the International League Against Epilepsy: Position Paper of the ILAE Commission for Classification and Terminology. Epilepsia 2017, 58, 522–530.
- Singh, G.; Sander, J.W. The global burden of epilepsy report: Implications for low- and middle-income countries. Epilepsy Behav. 2020, 105, 106949.
- Jovel, C.A.E.; Salazar, S.R.; Rodríguez, C.R.; Mejía, F.E.S. Factors associated with quality of life in a low-income population with epilepsy. Epilepsy Res. 2016, 127, 168–174.
- Nazir, N.; Sabri, A.A.; Ahmad, N.; Akram, M.N.; Hussain, H.A.; Rasool, A.G. Epidemiological study of epilepsy in Faisalabad. Prof. Med. J. 2020, 27, 2608–2612.
- Subota, A.; Khan, S.; Josephson, C.B.; Manji, S.; Lukmanji, S.; Roach, P.; Wiebe, S.; Buchhalter, J.; Federico, P.; Teskey, G.C.; et al. Signs and symptoms of the postictal period in epilepsy: A systematic review and meta-analysis. Epilepsy Behav. 2019, 94, 243–251.
- DeGiorgio, C.M.; Curtis, A.; Carapetian, A.; Hovsepian, D.; Krishnadasan, A.; Markovic, D. Why are epilepsy mortality rates rising in the United States? A population-based multiple cause-of-death study. BMJ Open 2020, 10, e035767.
- Mula, M. Emerging drugs for focal epilepsy. Expert Opin. Emerg. Drugs 2018, 23, 243–249.
- Quintana, M.; Sánchez-López, J.; Mazuela, G.; Santamarina, E.; Abraira, L.; Fonseca, E.; Seijo, I.; Álvarez-Sabin, J.; Toledo, M. Incidence and mortality in adults with epilepsy in northern Spain. Acta Neurol. Scand. 2021, 143, 27–33.
- Nabbout, R.; Kuchenbuch, M. Impact of predictive, preventive and precision medicine strategies in epilepsy. Nat. Rev. Neurol. 2020, 16, 674–688.
- Krasowski, M.D.; McMillin, G.A. Advances in anti-epileptic drug testing. Clin. Chim. Acta 2014, 436, 224–236.
- de Biase, S.; Nilo, A.; Bernardini, A.; Gigli, G.L.; Valente, M.; Merlino, G. Timing use of novel anti-epileptic drugs: Is earlier better? Expert Rev. Neurother. 2019, 19, 945–954.
- Mobed, A.; Shirafkan, M.; Charsouei, S. Biosensors technology for anti-epileptic drugs. Clin. Chim. Acta 2022, 533, 175–182.
- Málaga, I.; Sánchez-Carpintero, R.; Roldán, S.; Ramos-Lizana, J.; García-Peñas, J.J. New anti-epileptic drugs in paediatrics. An. Pediatría (English Ed.) 2019, 91, e1–e415.
- Peng, S.J.; Wong, T.T.; Huang, C.C.; Chang, H.; Hsieh, K.L.C.; Tsai, M.L.; Yang, Y.S.; Chen, C.L. Quantitative analysis of intraoperative electrocorticography mirrors histopathology and seizure outcome after epileptic surgery in children. J. Formos. Med. Assoc. 2021, 120, 1500–1511.
- Consales, A.; Casciato, S.; Asioli, S.; Barba, C.; Caulo, M.; Colicchio, G.; Cossu, M.; de Palma, L.; Morano, A.; Vatti, G.; et al. The surgical treatment of epilepsy. Neurol. Sci. 2021, 42, 2249–2260.
- Helmstaedter, C.; Beeres, K.; Elger, C.E.; Kuczaty, S.; Schramm, J.; Hoppe, C. Cognitive outcome of pediatric epilepsy surgery across ages and different types of surgeries: A monocentric 1-year follow-up study in 306 patients of school age. Seizure 2020, 77, 86–92.
- Numis, A.L.; da Gente, G.; Sherr, E.H.; Glass, H.C. Whole-exome sequencing with targeted analysis and epilepsy after acute symptomatic neonatal seizures. Pediatr. Res. 2022, 91, 896–902.
- Janmohamed, M.; Brodie, M.J.; Kwan, P. Pharmacoresistance—Epidemiology, mechanisms, and impact on epilepsy treatment. Neuropharmacology 2020, 168, 107790.
- Zarnowska, I.M. Therapeutic Use of the Ketogenic Diet in Refractory Epilepsy: What We Know and What Still Needs to Be Learned. Nutrients 2020, 12, 2616.
- Tekin, E.; Serdaroğlu, F.M.; Şahin, Ş.; Taşdemir, H.A. Ketogenic diet experience at Ondokuz Mayıs University. Neurol. Sci. 2021, 42, 2481–2485.
- Li, R.J.; Liu, Y.; Liu, H.Q.; Li, J. Ketogenic diets and protective mechanisms in epilepsy, metabolic disorders, cancer, neuronal loss, and muscle and nerve degeneration. J. Food Biochem. 2020, 44, 13140.
- Dabek, A.; Wojtala, M.; Pirola, L.; Balcerczyk, A. Modulation of Cellular Biochemistry, Epigenetics and Metabolomics by Ketone Bodies. Implications of the Ketogenic Diet in the Physiology of the Organism and Pathological States. Nutrients 2020, 12, 788.
- Wells, J.; Swaminathan, A.; Paseka, J.; Hanson, C. Efficacy and Safety of a Ketogenic Diet in Children and Adolescents with Refractory Epilepsy—A Review. Nutrients 2020, 12, 1809.
- Dhillon, K.K.; Gupta, S. Biochemistry, Ketogenesis; StatPearls Publishers; StatPearls . 2022. Available online: https://www.ncbi.nlm.nih.gov/pubmed/29630231 (accessed on 10 February 2022).
- Ko, A.; Kwon, H.E.; Kim, H.D. Updates on the ketogenic diet therapy for pediatric epilepsy. Biomed. J. 2022, 45, 19–26.
- Masino, S.A.; Li, T.; Theofilas, P.; Sandau, U.S.; Ruskin, D.N.; Fredholm, B.B.; Geiger, J.D.; Aronica, E.; Boison, D. A ketogenic diet suppresses seizures in mice through adenosine A1 receptors. J. Clin. Investig. 2011, 121, 2679–2683.
- Yang, H.; Shan, W.; Zhu, F.; Wu, J.; Wang, Q. Ketone Bodies in Neurological Diseases: Focus on Neuroprotection and Underlying Mechanisms. Front. Neurol. 2019, 10, 585.
- Rosenbaum, M.; Hall, K.D.; Guo, J.; Ravussin, E.; Mayer, L.S.; Reitman, M.L.; Smith, S.R.; Walsh, B.T.; Leibel, R.L. Glucose and Lipid Homeostasis and Inflammation in Humans Following an Isocaloric Ketogenic Diet. Obesity 2019, 27, 971–981.
- Dunwiddie, T.V.; Diao, L.; Proctor, W.R. Adenine Nucleotides Undergo Rapid, Quantitative Conversion to Adenosine in the Extracellular Space in Rat Hippocampus. J. Neurosci. 1997, 17, 7673–7682.
- Kawamura, M.; Ruskin, D.N.; Masino, S.A. Metabolic Autocrine Regulation of Neurons Involves Cooperation among Pannexin Hemichannels, Adenosine Receptors, and KATP Channels. J. Neurosci. 2010, 30, 3886–3895.
- Mao, X.-Y.; Zhou, H.-H.; Jin, W.-L. Redox-Related Neuronal Death and Crosstalk as Drug Targets: Focus on Epilepsy. Front. Neurosci. 2019, 13, 512.
- Plum, L. Enhanced PIP3 signaling in POMC neurons causes KATP channel activation and leads to diet-sensitive obesity. J. Clin. Investig. 2006, 116, 1886–1901.
- Singh, B.; Khattab, F.; Chae, H.; Desmet, L.; Herrera, P.L.; Gilon, P. KATP channel blockers control glucagon secretion by distinct mechanisms: A direct stimulation of α-cells involving a c rise and an indirect inhibition mediated by somatostatin. Mol. Metab. 2021, 53, 101268.
- Yang, H.Q.; Echeverry, F.A.; ElSheikh, A.; Gando, I.; Arredondo, S.A.; Samper, N.; Cardozo, T.; Delmar, M.; Shyng, S.L.; Coetzee, W.A. Subcellular trafficking and endocytic recycling of K ATP channels. Am. J. Physiol. Physiol. 2022, 322, C1230–C1247.
- Lutas, A.; Yellen, G. The ketogenic diet: Metabolic influences on brain excitability and epilepsy. Trends Neurosci. 2013, 36, 32–40.
- Ułamek-Kozioł, M.; Czuczwar, S.J.; Januszewski, S.; Pluta, R. Ketogenic Diet and Epilepsy. Nutrients 2019, 11, 2510.
- Kayode, O.T.; Rotimi, D.E.; Afolayan, O.A.; Kayode, A.A. Ketogenic diet: A nutritional remedy for some metabolic disorders. J. Educ. Health Sport. 2020, 10, 180–188.
- D’Andrea Meira, I.; Romão, T.T.; Pires do Prado, H.J.; Krüger, L.T.; Pires, M.E.P.; da Conceição, P.O. Ketogenic Diet and Epilepsy: What We Know So Far. Front. Neurosci. 2019, 13, 5–13.
- Verrotti, A.; Iapadre, G.; Di Francesco, L.; Zagaroli, L.; Farello, G. Diet in the Treatment of Epilepsy: What We Know So Far. Nutrients 2020, 12, 2645.
- Hartman, A.L.; Gasior, M.; Vining, E.P.; Rogawski, M.A. The Neuropharmacology of the Ketogenic Diet. Pediatr. Neurol. 2007, 36, 281–292.
- Murakami, M.; Tognini, P. Molecular Mechanisms Underlying the Bioactive Properties of a Ketogenic Diet. Nutrients 2022, 14, 782.
- Molteberg, E.; Thorsby, P.M.; Kverneland, M.; Iversen, P.O.; Selmer, K.K.; Nakken, K.O.; Taubøll, E. Effects of modified Atkins diet on thyroid function in adult patients with pharmacoresistant epilepsy. Epilepsy Behav. 2020, 111, 107285.
- Cervenka, M.C.; Barron, B.J.; Kossoff, E.H.; Zahava Turner, R.D. The Ketogenic and Modified Atkins Diets: Treatments for Epilepsy and Other Disorders. 2016. Available online: https://ebookcentral.proquest.com/lib/unc/detail.action?docID=4429634 (accessed on 10 February 2022).
- Dou, X.; Xu, X.; Mo, T.; Chen, H.; Wang, Z.; Li, X.; Jia, S.; Wang, D. Evaluation of the seizure control and the tolerability of ketogenic diet in Chinese children with structural drug-resistant epilepsy. Seizure 2022, 94, 43–51.
- Kossoff, E.H.; Zupec-Kania, B.A.; Amark, P.E.; Ballaban-Gil, K.R.; Christina Bergqvist, A.G.; Blackford, R.; Buchhalter, J.R.; Caraballo, R.H.; Helen Cross, J.; Dahlin, M.G.; et al. Optimal clinical management of children receiving the ketogenic diet: Recommendations of the International Ketogenic Diet Study Group. Epilepsia 2009, 50, 304–317.
- Feinman, R.D. The biochemistry of low-carbohydrate and ketogenic diets. Curr. Opin. Endocrinol. Diabetes Obes. 2020, 27, 261–268.
- Longo, R.; Peri, C.; Cricrì, D.; Coppi, L.; Caruso, D.; Mitro, N.; De Fabiani, E.; Crestani, M. Ketogenic Diet: A New Light Shining on Old but Gold Biochemistry. Nutrients 2019, 11, 2497.
- Bruci, A.; Tuccinardi, D.; Tozzi, R.; Balena, A.; Santucci, S.; Frontani, R.; Mariani, S.; Basciani, S.; Spera, G.; Gnessi, L.; et al. Very Low-Calorie Ketogenic Diet: A Safe and Effective Tool for Weight Loss in Patients with Obesity and Mild Kidney Failure. Nutrients 2020, 12, 333.
- Stubbs, B.J.; Koutnik, A.P.; Goldberg, E.L.; Upadhyay, V.; Turnbaugh, P.J.; Verdin, E.; Newman, J.C. Investigating Ketone Bodies as Immunometabolic Countermeasures against Respiratory Viral Infections. Med 2020, 1, 43–65.
- Morris, G.; Puri, B.K.; Carvalho, A.; Maes, M.; Berk, M.; Ruusunen, A.; Olive, L. Induced Ketosis as a Treatment for Neuroprogressive Disorders: Food for Thought? Int. J. Neuropsychopharmacol. 2020, 23, 366–384.
More
