The potential use of nanomaterials in medicine offers opportunities for novel therapeutic approaches to treating complex disorders. For that reason, a new branch of science, named nanotoxicology, which aims to study the dangerous effects of nanomaterials on human health and on the environment, has recently emerged. However, the toxicity and risk associated with nanomaterials are unclear or not completely understood. The development of an adequate experimental strategy for assessing the toxicity of nanomaterials may include a rapid/express method that will reliably, quickly, and cheaply make an initial assessment. One possibility is the characterization of the hemocompatibility of nanomaterials, which includes their hemolytic activity as a marker.
- nanomaterials
- nanoparticles
- red blood cells
- hemocompatibility
1. Introduction
2. Methods for Assessment of Nanomaterials’ Hemotoxicity
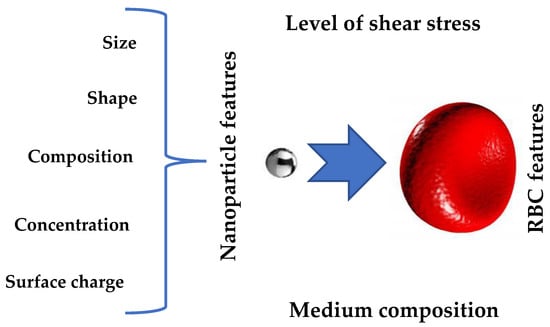
Nano-toxicology is a fast-developing area of nanoscience and nanotechnology. Current studies on the toxic effects of NPs, aiming to identifying the mechanisms of their harmful effects, are carried out in cell culture and animal models [41][42][43][44][45][46][47][48][49][50][54,55,57,59,157,158,159,160,161,162]. The toxicity of NPPS has received special attention [50][51][52][53][162,163,164,165]. These particles can be easily synthesized in a wide range of sizes, and their surfaces can be given different functionality [44][59]. Thus, they are ideally suited as a model for studying the effect of particle surface characteristics on various biological parameters both in vitro and in vivo. Sarma and colleagues [41][54] have analyzed the cytotoxic and genotoxic potential of NPPS on human peripheral lymphocytes (in vitro), while Loos et al. [44][59] have summarized information regarding the effect of functionalized (positively and negatively charged) NPPS on macrophages and THP-1 cells (in vitro). These studies indicate that while polystyrene is non-toxic, functionalized nanoparticles may behave differently than bulk material, and surface chemistry plays a critical role in determining the effect of NPPS on various cells. The toxicity of NPPS was also analyzed in vivo in animal models [47][48][49][50][159,160,161,162]. Fan et al. [48][160] observed the accumulation of fluorescent NPPS in various organs of mice after oral ingestion, including in the liver, kidney, spleen, and pancreas. The main mechanism of damage to the internal organs was the impairment of liver function and lipid metabolism. Yasin and colleagues also identified the striking hepatoxicity of NPPS (in a dose-dependent manner) [50][162] in rats. In addition, a recent in vivo study showed that PSNPs induced reproductive toxicity [49][161] in mice, caused fetal growth restriction, and significantly impaired cholesterol metabolism in both the mice’s placenta and the fetus [47][159]. However, the toxicity and risk associated with the use of NPs still need to be understood in their entirety [54][95]. The development of an adequate experimental strategy for estimating NPs’ toxicity should include the choice between in vitro (cell lines) and in vivo (animal models) methods or a combination of both, as both methods have advantages and disadvantages. The NP toxic effects on individual cell components and tissues are more accessible for in vitro analysis, while in vivo models enable the assessment of NP toxicity for individual organs or the whole organism [51][163]. It seems more logical to first test NP toxicity on cells, and if toxic effects are clearly demonstrated, this may spare the need for animal testing, in accordance with the global trend of reducing the number of animal experiments [55][56][97,98]. The rapid growth of nanomedicine and the development of more and more new NPs make in vivo toxicity tests undesirable on both ethical and financial grounds, creating an urgent need to develop in vitro cell-based assays that accurately predict in vivo toxicity and facilitate safe nanotechnology. Of all the cell types [52][164] that can be used to assess the toxicity of nanomaterials, the choice of RBC as a target cell seems to be the most useful. As noted above, irrespective of their use, source, and route of exposure, NPs enter the bloodstream and interact with RBCs, the major cellular component in the circulation (4–5 million RBCs per 1 μL of blood), producing a negative effect on their functionality. As RBCs are well characterized, accessible, and easy to manipulate, they make an excellent candidate for being the target cells for nanotoxicity assessment. Numerous studies have examined the NP-RBC interaction, focusing on the hemolytic potential of NPs [53][57][49,165], suggesting that this is the critical test of NP safety [58][59][75,166]. Although hemolysis tests have been conducted with various NPs, comparing results across studies is difficult due to the variability of protocols implemented for particle characterization and hemolysis testing [60][52]. The American Society for Testing and Materials (ASTM) published (2008) a standard test protocol for the assessment of NPs’ hemolytic properties [59][166], which determines the percentage of hemoglobin (Hb) released after NP-RBC interaction. The hemolytic assay has proven to be a promising test for surveying nanomaterial toxicity [61][167] due to its low cost, good reproducibility, and quick results [62][77]. To date, hemolytic activity has even been demonstrated with therapeutic NPs in vitro [63][64][65][73,168,169] and in vivo [66][67][170,171], indicating the potential adverse effects of NPs, which may limit their applications in nanomedicine. Cho et al. [68][172] studied the nanotoxicity of a panel of NPs (CeO2, TiO2, carbon black, SiO2, NiO, Co3O4, Cr2O3, CuO, and ZnO). The authors compared the acute lung inflammogenicity in a rat model with in vitro toxicity. For in vitro testing, eight different cell-based assays were used, including epithelial cells, monocytic/macrophage cells, human erythrocytes, and combined culture. Cytotoxicity in differentiated peripheral blood mononuclear cells was the most accurate, demonstrating 89% accuracy and 11% false negative results in predicting acute pulmonary inflammation. However, only hemolysis tests demonstrated a 100% match with lung inflammation at all NP concentrations. Other in vitro cellular assays showed a weaker correlation with in vivo inflammatory activity. An analysis of the related literature supports the finding that NP-induced hemolytic activity can assess in vivo NP toxicity and has been proposed as a critical test in determining NP hemocompatibility [58][59][62][69][75,77,166,173]. However, despite the attempts to develop a unified protocol to determine NPs’ hemolytic activity, the measurement conditions used by various research groups still differ significantly [60][52]. For a universal protocol, it is necessary to consider that forming a protein corona around NP inhibits its effective hemolytic activity. In addition, the interaction between a red cell and a nanoparticle in the bloodstream occurs under flow-induced mechanical stress, which can cause RBC deformation [70][174] and stimulate NP hemolytic activity [71][47]. Thus, it would be appropriate to test NP hemocompatibility under mechanical stress conditions in a medium supplemented by plasma proteins or in the plasma itself (and not in a buffer, as is customary in many laboratories). The ability of an NP to change RBC properties can be expressed as an alteration in its functionality and, in its extreme form, as the destruction of the cell [38][72][73][38,175,176]. Therefore, other properties of red cells, such as their aggregability, deformability, and adhesion to EC, should be considered alternative markers to NP hemolytic activity [38][72][73][38,175,176]. All of the mentioned studies demonstrate the protective role of the protein corona formed on the nanomaterial’s surface, improving the NP hemocompatibility and providing promising options for the design of therapeutic nanomaterials without prohibitive toxic effects. Thus, itwe can be summarized that the NPs’ characteristics and the plasma composition are the dominant factors determining the NPs’ hemocompatibility. Additional factors that can affect the NP hemolytic ability inclue the properties of the RBCs themselves and the presence of mechanical stress (Figure 2). For these reasons, when developing a protocol for testing the hemolytic activity of NPs, it is necessary to consider all four factors.