Hierarchical structures are an essential part of numerous types of architecture in nature. They are defined as the presence of different structural elements with different length scales in a single body [1]. This different length scale gives each hierarchical structure its “order, n” and characteristic properties [2]. The higher the (n) the more sophisticated hierarchical structures; where n = 0 refers to continuum materials with only a single length scale. Noteworthy, several composites are considered low-ordered hierarchical structures. The idea of building blocks for hierarchical structures intersects perfectly with the modularity concept in click chemistry. Click chemistry is a powerful tool for constructing nano, micro and macro structures through two different approaches: (A) the first approach: through direct crosslinking of (pico-building blocks) monomers give a final micro/macro structure such as hydrogels [9,14]; (B) the second approach: through nano-building blocks formation using click chemistry (e.g., dendrimers and dendrons) followed by connecting and crosslinking those formed nano-building blocks again using click chemistry to form bigger structures
- click chemistry
- hierarchial structures
- 0D/nano/micro building blocks
1. Click Chemistry
| Advantage | Explanation |
|---|---|
| High Selectivity | - |
| High yield | - |
| Mild conditions | Usage of mild solvents and reactants |
| Fast reaction | The exothermicity of both thermodynamic and kinetics of the reaction allowed fast and stable final products |
| Minimum byproducts | - |
| Easy purification | No need for complicated chromatography or other purification techniques |
| Modularity | The ability of forming diverse library of chemical structures from simple chemical modules |
| Regiospecific | Minimum stereoisomers are being formed |
Historically, the first reaction to being identified as click chemistry is the copper-catalyzed azide-alkyne reaction to form a 5-membered ring. Nowadays, the click chemistry approach is being handled as a toolbox that different groups worldwide are adding to and modifying with different new reactions. So far, four categories have been identified to represent the different click chemistry reactions.
2. Hierarchical Structures and Click Chemistry
For any hierarchical structures in nature there have been repeated smaller building blocks which have been arranged in a certain manner to provide the required functionality for this hierarchical construction.
Moreover, click chemistry acts through different pathways for building higher structures either alone or in combination with different assisting chemistries, techniques, and tools. While in the first approach mentioned above, the role of click chemistry involves connecting the different building blocks, using the spatial hindrance and the chemical natures of the involved compounds to give the final topological and internal structures of the final product. In the second approach, click chemistry is used as an assisting tool for the fabrication techniques, such as STAMP-Lithography, SLA 3D printing, phase separation, self-assembly, etc. Noteworthy, during the is review, the following considerations have been taken: 0D is the xyz < 100 nm, 1D xy < 100nm and 2D x or y < 100 nm [2][15]. Additionally, there has been no differentiation between micro and higher dimensions. Moreover, as molecules, atoms and monomers do not fall into any of those categories, a general term to describe them was used: “Pico building blocks”.2.1. The First Approach
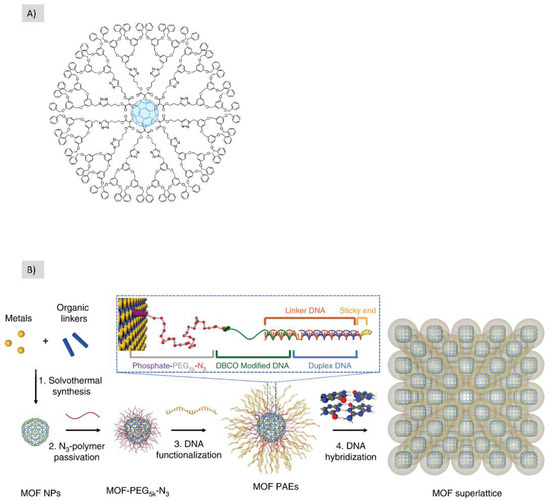
All the micro/macro structures to be constructed must pass first through the nanoscale level. However, what is meant in this category is the whole continuous process (click orthogonal), i.e., from the pico/0D nano to the micro/macro without the involvement of intermediate processing and modifications. In 2020, Hassan et al. published an excellent review article discussing the construction of hierarchical structures from MOF (Metal/covalent Organic frame) using click chemistry (Figure 15) [3][16]. The significant potential of clickable MOF for building high precision hierarchical structures, porosity control, the transformation of internal and external structures and post-synthesis modifications (PSM) opened a new horizon in the field of bio/material science.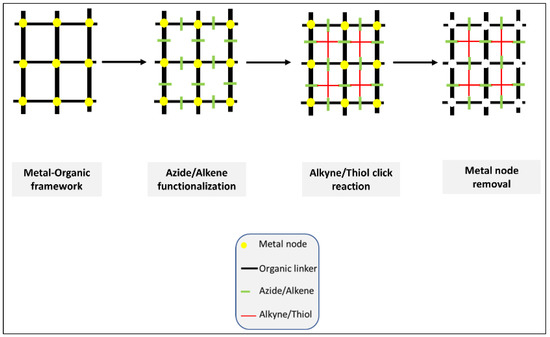
2.2. The Second Approach
2.2.1. Picometer ➔ 0D-Nanometer
For such small dimensions of controlled fabrication, a significant number of articles and review articles have already been published covering the usage of click chemistry to synthesize 0D structures: dendrimers, dendrons, hyperbranched polymer [5][32], single chain nanoparticles [6][33] and sequence-controlled polymers [7][34]. Other reactions which do not include a construction frame behind it, e.g., conjugation for labelling or targeting purposes have also been shown in literature such as nanoparticles [8][35], buckyballs [9][36], micelles [10][37] and microparticles [11][38].2.2.2. 0D/1D-Nanometer ➔ 2D-Nanometer
The idea of creating a unified theory covering the use of 0D-nanobuilding blocks for building higher structures was a detailed cornerstone theoretical work of Tomalia et al. 2009 [12][10]. In his published works, Tomalia suggested a similar system to the periodic atomic table system for higher structures (0D) [13][39]. The dendrimers were used as the explanatory model for his idea. The 0D nanoscale building blocks were categorized into two main classes, hard and soft nanostructures, and under each category six subcategories were proposed based on compositional/architectural considerations as shown in the following (Figure 37).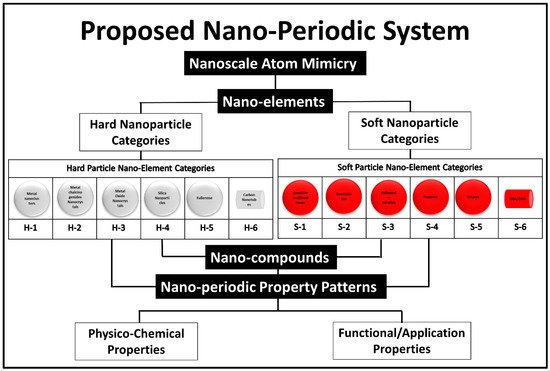
Soft/Soft Nano Building Blocks
Hard/Hard Nano Building Blocks
For electro/photocatalytic applications, alkyne-azide click chemistry showed a very efficient potential to form mono/multilayers, hetero/homo structure on different substrates using metallic, semiconductor and dielectric nanoparticles. Upadhyay et al. showed even further control over the packing ratio and crystal unit by controlling the number of click groups on the substrates, substrate surface roughness and the solvent used [15][50]. Williams and Teplyakov showed another metal/metal assembly, in which azide-terminated Si nanoparticles and alkyne-terminated Si nanoparticles were added alternatively to a gold substrate using click chemistry to form nearly 100% coverage of the substrate [16][51]. For self-healing polymers applications, Le Neindre and Nicolaÿ, used a thiolated copolymer with silver nanoparticles as crosslinkers [17][52]. The self-healing potential was attributed to the reversible thiolates exchange that takes place over the Ag nanoparticles.Soft/Hard Nano Building Blocks
Fullerene and dendrimers represent different types of hard and soft building blocks, respectively. The clicking between those two types both dendrimers in the core and fullerene in the shells or the opposites has been published in the literature [18][19][56,57]. While Hahn et al. 2012 used fullerene as a peripheral decoration of the polypropylene simine (PPI) dendrimers, Nierengarten 2017, showed different dendrimers using fullerene hexa-adduct core building blocks bearing twelve equivalent clickable groups (Figure 410A). Their post-modification allows for the introduction of twelve equivalent peripheral subunits.