Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Camila Xu and Version 1 by Chaiyavat Chaiyasut.
Probiotics are proven adjuvant therapeutic agents for various acute and chronic infections, cancer, inflammatory diseases, and cognitive and psychiatric disorders.
- probiotics
- psychobiotics
- cognition
- Parkinson’s disease
1. Introduction
Probiotics consumption is increasing globally because of their health benefits [1]. Probiotics are proven adjuvant therapeutic agents for various acute and chronic infections, cancer [2], inflammatory diseases [3], and cognitive and psychiatric disorders [4]. Approximately 20% of people worldwide suffer from mental health disorders such as depression and anxiety [5]. Psychologically good mental health can be described as the status of the individual’s well-being. Generally, psychological research carried out in rodents focuses on stress, anxiety, and motivation [6].
A panel of experts in the International Scientific Association for Probiotics and Prebiotics (ISAPP) officially described probiotics as “live microorganisms which, when administered in adequate amounts, confer a health benefit on the host” [7]. In 2013, a novel class of probiotics emerged as “psychobiotics.” Dinan and colleagues described psychobiotics as “live organisms that produce health benefits in psychiatric illness patients when ingested in adequate amounts” [8]. Sarkar and colleagues mentioned psychotropics and antibiotics for mental health disorders could be considered psychobiotics [6]. Psychobiotics work through synthesizing, distributing, and regulating the neurotransmitters such as gamma-aminobutyric acid (GABA), serotonin (5-HT), glutamate and brain-derived neurotrophic factor (BDNF) in executing and regulating the neural processes such as learning, memory, mood, and other cognitive functions [9].
Psychobiotics influence the central nervous system (CNS) through the gut–brain axis with the support of immune, neural, and metabolic pathways [9]. Psychobiotics are efficient in treating neurodegenerative and neurodevelopmental disorders by improving cognitive functions in Alzheimer’s’ disease (AD), motor functions in Parkinson’s disease (PD) and improve autism spectrum disorder (ASD) symptoms [9]. Many approaches have emerged to manage mental health through psychobiotics and dietary supplementation.
2. Probiotics and Brain Health
Physical well-being is important for mental well-being and vice versa. Psychobiotics influence brain–bacteria communications through the enteric nervous system (ENS) and immune system and exert anxiolytic, anti-depressant properties resulting in enormous changes in the cognitive and emotional parameters [6]. Bacterial genera such as Lactobacillus, Bifidobacterium, Enterococcus, Streptococcus and Escherichia commonly influence the bidirectional interactions of the brain and gut system through the production of neurochemicals; they are used as psychobiotics. Unlike conventional probiotics, psychobiotics can synthesize or stimulate the production of various neurotransmitters, anti-inflammatory cytokines, and gastric endocrine hormones [10]. Balancing and maintaining an individual’s physical and emotional well-being requires two-way communication between the gut and brain, mainly regulated by the gut microbiota. Gut microbiota (GM) is important in neurodevelopmental and neuropsychiatric disorders [10]. The studies on psychobiotics and psychological illness are very limited. However, employing psychobiotics for treating neuropsychiatric diseases is growing as a new field of interest among neurological researchers: psychobiotics used in the treatment of stress, anxiety and depression and other mood disorders. It was found that psychobiotics reduced the cognitive reactivity to negative mood and improved depressive symptoms, anxiety, and stress response [11,12][11][12]. The microbial infection could affect mental processes, and the metabolically active multifaceted healthy intestinal microbiota provides positive mental health benefits [8]. Logan and Katzman first used probiotics as an adjunct therapy in managing major depressive disorder (MDD). In MDD, gut microflora becomes altered, and the levels of Lactobacilli and Bifidobacterium are lower, which could change the gut functions through elevated pro-inflammatory cytokines and oxidative stress. Probiotics could lower the pro-inflammatory cytokines, reduce oxidative stress, and increase BDNF [13]. GABA is the inhibitory neurotransmitter that plays a significant role in physiological and psychological functions. GABA receptors are important for normal behaviour. More specifically, GABAB receptors play an important role in mood and anxiety disorders. Any changes in the expression of GABA receptors can cause anxiety and depression along with bowel disorders as comorbid conditions [14]. Lactobacillus rhamnosus (L. rhamnosus) reduced anxiety in stress-induced hyperthermia (SIH), elevated plus maze test (EPM) and forced swim test (FST) in mice. L. rhamnosus altered the expression of GABAB1b receptors in the brain with an increase in cingulate and prelimbic regions and a decrease in the hippocampus, amygdala, and locus coeruleus. GABAAα2 was reduced in the prefrontal cortex and amygdala and increased in the hippocampus compared to the control mice. L. rhamnosus reduced stress-induced corticosterone elevation and depression-related behaviours [14]. The results indicated that L. rhamnosus could change neural functions, which causes behavioural and neurological effects. Thus, it can be considered in therapeutic applications against depression. Gut disorders with comorbid psychiatric conditions increase gut permeability, enhance lipopolysaccharide (LPS) translocation and increase depressive symptoms. Lactobacillus farciminis suppress acute psychological stress-induced gut permeability by attenuating the hypothalamus–pituitary–adrenal (HPA) axis in rats [15]. The oral administration of Lactobacillus increases the GABA, N-acetyl aspartate, and glutamate in the brain of mice [16]. Bifidobacterium longum (B. longum) 1714 enhanced the behaviour and cognitive performance in stressed mice [17]. Psychobiotics benefit neuropsychiatric disorders such as schizophrenia, Tourette’s syndrome, attention deficit hyperactivity disorder (ADHD), AD, PD, ASD, stress, depression, and anxiety [10]. In addition to neurodegenerative diseases, neurologic injuries such as traumatic brain injury (TBI), ischemic stroke, spinal cord injury (SCI), and haemorrhagic cerebrovascular lesions could cause gut dysbiosis. In another way, any changes in GM can produce proinflammatory cytokines and clotting factors, increasing the risk of neurological injuries [18]. The interactions between the gut microbiota, gut and CNS are familiarly known as the microbiota–gut–brain axis (MGBA). Dysregulation in MGBA could cause intestinal disorders, stress, anxiety, depression, and other psychiatric disorders [14]. The emotional state depends on the gastrointestinal (GI) tract function. MGBA dysregulation can cause GI, neuropsychological, and metabolic disorders. The gut–brain interaction can be identified through the relationship between gut dysbiosis and GI and CNS disorders [19]. MGBA dysregulation increases intestinal permeability, promotes the proinflammatory phase and causes CNS injuries. The systemic inflammation might result in secondary CNS injuries [20]. High differentiation and migration of immune cells to the CNS can cause maladaptive CNS inflammation [21], resulting in TBI, SCI, strokes, and brain tumours [22] (Figure 21).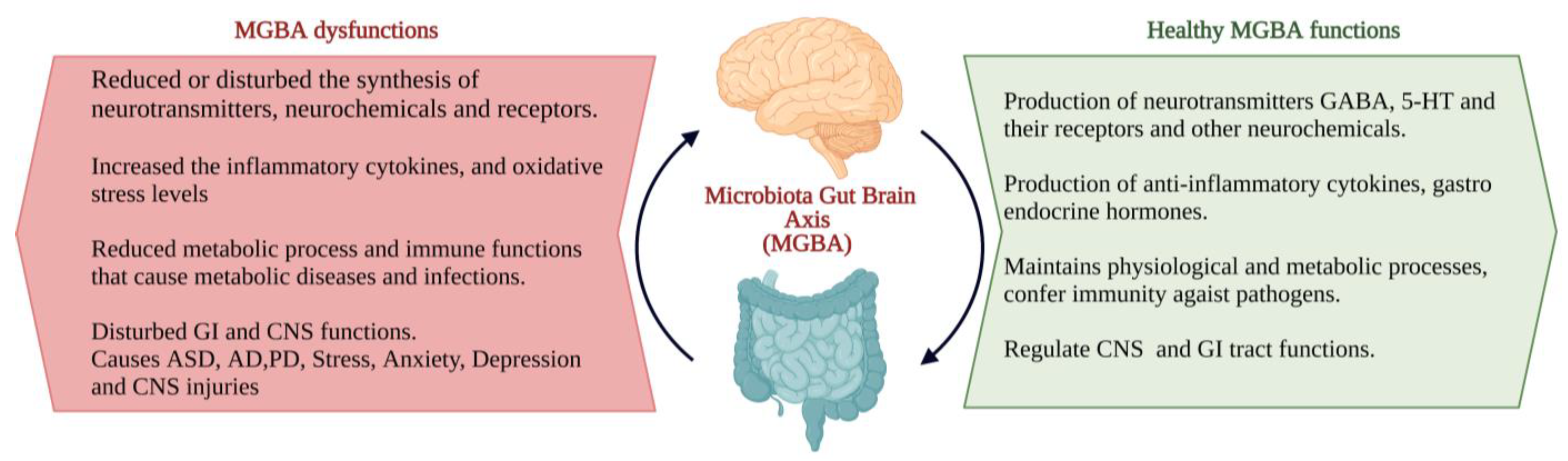
References
- Holzapfel, W.H.; Schillinger, U. Introduction to pre- and probiotics. Food Res. Int. 2002, 35, 109–116.
- Nazir, Y.; Hussain, S.A.; Abdul Hamid, A.; Song, Y. Probiotics and their potential preventive and therapeutic role for cancer, high serum cholesterol, and allergic and HIV diseases. BioMed. Res. Int. 2018, 2018, 3428437.
- de Souza, M.; Barbalho, S.; Goulart, R.; de Carvalho, A. The current and future role of drugs and probiotics in the management of inflammatory bowel disease. JSBM 2015, 3, 76–85.
- Alagiakrishnan, K.; Halverson, T. Microbial therapeutics in neurocognitive and psychiatric disorders. J. Clin. Med. Res. 2021, 13, 439–459.
- Bear, T.L.K.; Dalziel, J.E.; Coad, J.; Roy, N.C.; Butts, C.A.; Gopal, P.K. The role of the gut microbiota in dietary interventions for depression and anxiety. Adv. Nutr. 2020, 11, 890–907.
- Sarkar, A.; Lehto, S.M.; Harty, S.; Dinan, T.G.; Cryan, J.F.; Burnet, P.W.J. Psychobiotics and the manipulation of bacteria-gut-brain signals. Trends Neurosci. 2016, 39, 763–781.
- Hill, C.; Guarner, F.; Reid, G.; Gibson, G.R.; Merenstein, D.J.; Pot, B.; Morelli, L.; Canani, R.B.; Flint, H.J.; Salminen, S.; et al. Expert consensus document. The International Scientific Association for Probiotics and Prebiotics consensus statement on the scope and appropriate use of the term probiotic. Nat. Rev. Gastroenterol. Hepatol. 2014, 11, 506–514.
- Dinan, T.G.; Stanton, C.; Cryan, J.F. Psychobiotics: A novel class of psychotropic. Biol. Psychiatry 2013, 74, 720–726.
- Cheng, L.H.; Liu, Y.W.; Wu, C.C.; Wang, S.; Tsai, Y.C. Psychobiotics in mental health, neurodegenerative and neurodevelopmental disorders. J. Food Drug Anal. 2019, 27, 632–648.
- Sharma, R.; Gupta, D.; Mehrotra, R.; Mago, P. Psychobiotics: The next-generation probiotics for the brain. Curr. Microbiol. 2021, 78, 449–463.
- Messaoudi, M.; Violle, N.; Bisson, J.F.; Desor, D.; Javelot, H.; Rougeot, C. Beneficial psychological effects of a probiotic formulation (Lactobacillus helveticus R0052 and Bifidobacterium longum R0175) in healthy human volunteers. Gut Microbes 2011, 2, 256–261.
- Mohammadi, A.A.; Jazayeri, S.; Khosravi-Darani, K.; Solati, Z.; Mohammadpour, N.; Asemi, Z.; Adab, Z.; Djalali, M.; Tehrani-Doost, M.; Hosseini, M.; et al. The effects of probiotics on mental health and hypothalamic–pituitary–adrenal axis: A randomized, double-blind, placebo-controlled trial in petrochemical workers. Nutr. Neurosci. 2016, 19, 387–395.
- Logan, A.C.; Katzman, M. Major depressive disorder: Probiotics may be an adjuvant therapy. Med. Hypotheses 2005, 64, 533–538.
- Bravo, J.A.; Forsythe, P.; Chew, M.V.; Escaravage, E.; Savignac, H.M.; Dinan, T.G.; Bienenstock, J.; Cryan, J.F. Ingestion of Lactobacillus strain regulates emotional behavior and central GABA receptor expression in a mouse via the vagus nerve. Proc. Natl. Acad. Sci. USA 2011, 108, 16050–16055.
- Ait-Belgnaoui, A.; Durand, H.; Cartier, C.; Chaumaz, G.; Eutamene, H.; Ferrier, L.; Houdeau, E.; Fioramonti, J.; Bueno, L.; Theodorou, V. Prevention of gut leakiness by a probiotic treatment leads to attenuated HPA response to an acute psychological stress in rats. Psychoneuroendocrinology 2012, 37, 1885–1895.
- Janik, R.; Thomason, L.A.M.; Stanisz, A.M.; Forsythe, P.; Bienenstock, J.; Stanisz, G.J. Magnetic resonance spectroscopy reveals oral Lactobacillus promotion of increases in brain GABA, N-acetyl aspartate and glutamate. Neuroimage 2016, 125, 988–995.
- Allen, A.P.; Hutch, W.; Borre, Y.E.; Kennedy, P.J.; Temko, A.; Boylan, G.; Murphy, E.; Cryan, J.F.; Dinan, T.G.; Clarke, G. Bifidobacterium longum 1714 as a translational psychobiotic: Modulation of stress, electrophysiology and neurocognition in healthy volunteers. Transl. Psychiatry. 2016, 6, e939.
- Panther, E.J.; Dodd, W.; Clark, A.; Lucke-Wold, B. Gasterointestinal microbiome and neurological injury. Biomedicines 2022, 10, 500.
- Carabotti, M.; Scirocco, A.; Maselli, M.A.; Severi, C. The gut-brain axis: Interactions between enteric microbiota, central and enteric nervous systems. Ann. Gastroenterol. 2015, 28, 203–209.
- Li, X.J.; You, X.Y.; Wang, C.Y.; Li, X.L.; Sheng, Y.Y.; Zhuang, P.W.; Zhang, Y.J. Bidirectional Brain-gut-microbiota Axis in increased intestinal permeability induced by central nervous system injury. CNS Neurosci. Ther. 2020, 26, 783–790.
- Agirman, G.; Yu, K.B.; Hsiao, E.Y. Signaling inflammation across the gut-brain axis. Science 2021, 374, 1087–1092.
- Willman, J.; Willman, M.; Reddy, R.; Fusco, A.; Sriram, S.; Mehkri, Y.; Charles, J.; Goeckeritz, J.; Lucke-Wold, B. Gut microbiome and neurosurgery: Implications for treatment. Clin. Transl. Discov. 2022, 2, e139.
- Sasselli, V.; Pachnis, V.; Burns, A.J. The enteric nervous system. Dev. Biol. 2012, 366, 64–73.
- Thangaleela, S.; Sivamaruthi, B.S.; Kesika, P.; Bharathi, M.; Chaiyasut, C. Nasal microbiota, olfactory health, neurological disorders, and aging-A review. Microorganisms 2022, 10, 1405.
- Marchesi, J.R.; Ravel, J. The vocabulary of microbiome research: A proposal. Microbiome 2015, 3, 31.
- Mayer, E.A.; Tillisch, K.; Gupta, A. Gut/brain axis and the microbiota. J. Clin. Investig. 2015, 125, 926–938.
- Bik, E.M.; Ugalde, J.A.; Cousins, J.; Goddard, A.D.; Richman, J.; Apte, Z.S. Microbial biotransformations in the human distal gut. Br. J. Pharmacol. 2018, 175, 4404–4414.
- Kang, S.S.; Jeraldo, P.R.; Kurti, A.; Miller, M.E.; Cook, M.D.; Whitlock, K.; Goldenfeld, N.; Woods, J.A.; White, B.A.; Chia, N.; et al. Diet and exercise orthogonally alter the gut microbiome and reveal independent associations with anxiety and cognition. Mol. Neurodegener. 2014, 9, 36.
- Lyte, M. Probiotics function mechanistically as delivery vehicles for neuroactive compounds: Microbial endocrinology in the design and use of probiotics. BioEssays 2011, 33, 574–581.
- Rijkers, G.T.; Bengmark, S.; Enck, P.; Haller, D.; Herz, U.; Kalliomaki, M.; Kudo, S.; Lenoir-Wijnkoop, I.; Mercenier, A.; Myllyluoma, E.; et al. Guidance for substantiating the evidence for beneficial effects of probiotics: Current status and recommendations for future research. J. Nutr. 2010, 140, 671S–676S.
More
