In nature, interactions between bacteria and microalgae play an indispensable role in maintaining the integrity of the aquatic ecosystem through networks of interactions such as competition and mutualism. In fact, in the wild, the growth of algae is consistently associated with the growth of other microorganisms, especially bacteria. Axenic culture systems and sterilization of culture media in large-scale production of microalga are not economically feasible. Therefore, the characterization of associated heterotrophs in algae culture systems is an important step since bacteria may use compounds excreted by algae, increasing the availability of trace elements and solubility of nutrients, making them more bioavailable for microalgae. In addition, they can help to reduce the saturation of dissolved O2]. In microalgae cultivation, it is well known that dissolved O2 can attain inhibitory levels.
- microalgae
- bacteria
- bioproducts
1. Microalgae-Bacteria Interaction
1.1. Microbial Interactions through Quorum Sensing
1.2. Oxygen and Carbon Dioxide Exchanges
The co-cultures of bacterium and algae may be effective in detoxifying inorganic and organic pollutants and removing nutrients from wastewater if compared to the activity of these microorganisms individually. Photosynthesis by cyanobacteria and eukaryotic algae provides oxygen, an essential factor for heterotrophic bacteria that degrade pollutants. Sequentially, the bacteria help the photoautotrophic growth of the collaborators, furnishing carbon dioxide and stimulating factors [14][17] and decreasing the oxygen concentration in the culture medium [15]. This relationship is interesting because microalgae can be part of a circular economy, since it allows the use of CO2 coming from industrial processes, like distilleries (CO2 from alcoholic fermentation and sugarcane bagasse burning), cement industry (CO2 from burning of energy source and CaCO3 decomposition), energy industry (CO2 from burning of energy source) as well as from aerobic and anaerobic treatment of wastewaters, as a carbon source to produce microalgae biomass. Simultaneously, the oxygen produced by phytoplankton may support the necessity of aerobic processes in these industries. The oxygen produced by microalgae could be used in an integrated process involving aerobic depuration of wastewaters, with a production of bacteria which could be used in different applications, like agricultural inoculants, among others, depending on the species of microorganism cultivated [16][45].2. Effects of Interactions
2.1. Inhibitory Effect by Metabolites on Algae and Bacteria
Although several species of bacteria have a beneficial effect on algae growth, some bacterial species may also inhibit microalgae by producing extracellular algaecide compounds [17][22]. This inhibitory effect helps to control the proliferation of harmful algae in bodies of water [15]. Some bacteria can induce the lysis of microalgal cells. For instance, the Kordia algicida secretes an algaecide protease that hinders the growth of several diatomaceous marine species [18][46]. Bacteria can compete with microalgae for limiting nutrients, such as nitrogen and phosphorus, when they grow together in an organic carbon source. Due to the higher growth rate of bacteria than microalgae in this condition, their propagation would consume more nutrients, limiting the growth of eukaryotic photosynthetic microorganisms due to a lack of nutrients [19][25]. For instance, when Acinetobacter sp. was inoculated in the exponential phase, a substantial drop in the growth of Botryococcus braunii was observed. This bacterium, which presented a negative interaction with B. braunii, produces AHL signaling molecules involved in bacterial quorum sensing, which were found in the non-axenic culture of microalgae [20][47]. From these considerations, it is important to evaluate the environmental conditions of the processes where the presence of undesirable bacteria is minimized and preferentially suppressed to maximize the microalgal growth. Otherwise, favoring beneficial bacteria within an appropriate threshold is also a determinant for optimizing microalgae growth.2.2. Bacteria That Promote the Growth of Microalgae
Normally, non-pathogenic bacteria from several species have been found in microalgae cultivations with beneficial effects on their growth. It has been shown that bacteria may modify the growth of phytoplankton, accumulating biomass and increasing cell productivity, which is of particular interest for industrial production. The bacteria of the genera Alteromonas and Muricauda allowed the most significant accumulation of Dunaliella biomass due to the increase in the availability of nitrogen for microalgae. However, more research is needed to understand the mechanisms behind these interactions [21][49]. In the cultivation of Chlorella prototecoides in synthetic wastewater media in co-culture with Brevundimonas diminuta with light intensities of 75 and 130 μmol photons m−2s−1, the μ values in non-axenic conditions were at least five times higher than in cultivations without co-culture. Thus, under these conditions, the addition of Brevundimonas diminuta was able to provide higher growth rates of C. protothecoides with more efficient nutrient removal [22][50]. A substantial promotion in the growth of phytoplankton has been described because the bacterium produces indole-3-acetic acid (IAA) [15]. The Achromobacter sp. produces IAA, which promotes the growth of Haematococcus pluvialis, with an increase in chlorophyll and cell concentrations [23][51]. In fact, the bacterial population in low concentrations can improve the microalgae metabolism by releasing factors that promote growth or reduce the concentration of O2 in the medium, preventing this gas from reaching an inhibitory concentration. As a consequence of higher microalgae growth in co-culture with bacteria, there is a higher removal of nutrients from the cultivation medium, which is particularly important in the tertiary treatment of wastewater since nitrogen and phosphorus are constituents of the microalgae biomass. In a study using synthetic wastewater with a semi-continuous process, the immobilized consortium of Chlorella vulgaris and Azospirillum brasilense led to an increase in the uptake of ammonium by culture [24][52]. Thiamine and tryptophan released by C. sorokiniana are signaling molecules that may be used by A. brasilense to synthesize and secrete another signaling molecule, indole-3-acetic acid, which promotes microalgae growth. The occurrence of signaling compounds, such as thiamine and tryptophan in the exudates of Chlorella sorokiniana, supports the mutualistic interaction of this photosynthetic microorganism with A. brasilense [25][53].2.3. Supply of Nutrients
Microalgae may increase bacterial activity by secreting extracellular molecules such as lipids, proteins, and nucleic acids that serve as nutrients for bacterial growth. In this sense, dead microalgae cells can also provide nutrients for the growth of bacterial cells [15]. Croft et al. (2005) showed that vitamin B12 is an important molecule in algae metabolism, the main cofactor for methionine synthase, which depends on vitamin B12. They also observed that cobalamin auxotrophy had appeared numerous times throughout evolution processes, probably related to the presence or absence of vitamin B12-dependent enzymes. An example of this symbiosis is the case of bacteria of the genus Halomonas, that supply cobalamin for the microalgae Amphidinium operculatum [26][57]. Pseudomonas sp., on the other hand, produced a glycoprotein that performed as a growth factor for Asterionella glacialis [27][58]. In another research, the growth of Chlorella sp was shown to be improved due to the release of riboflavin by E. coli [28][59].2.4. Modification of the Composition of Microalgae in Co-Culture
The association of Rhizobium sp. KB10 with B. braunii increased algae growth by nine times and improved the oleate content, used to produce biodiesel [29][61]. Inoculation of the bacterial strain Rhizobium 10II in the cultivation of Ankistrodesmus sp. strain SP2-15 increased by 30% the chlorophyll content in the microalgae biomass, and the lipid productivity was up to 112 g.m−2d−1 on the sixth day of cultivation [30][62]. The co-cultivation of Chlamydomonas reinhardtii with Bradyrhizobium japonicum improved the growth of the microalgae by 3.9 times, reaching lipid contents 26% higher and increasing Fe-hydrogenase activity and H2 production [31][63].
2.5. Flocculant Activity by Bacteria
The activity of bio-flocculant depends on the growth phase of the bacteria, being enzymatic activities related to the formation of bio-flocculants observed during the stationary growth phase. Although the production of bacterial bio-flocculant is beneficial for improving the formation of large flakes of microalgae and bacteria, the additional cost related to the carbon source required for the growth of these bacteria still remains a challenge [32][67], which evidence that organic by-products could be used to produce such bacteria, thus diminishing the cost of the process [33][68]. Exopolysaccharides and pyruvic and uronic acids are important for cell adhesion. In addition, factors such as the sources of nitrogen and carbon and the ratio between these two elements influence the production of bio-flocculants. The bio-flocculant produced by the Paenibacillus polymyxa exhibited high efficiency for the flocculation of C. vulgaris and Scenedesmus sp. [32][67]. Bacteria such as Flavobacterium, Terrimonas, and Sphingobacterium and their extracellular polymeric substances may help to increase the flocculating activity of algae such as C. vulgaris, resulting in sedimentable flakes [34][69].2.6. Microalgae Co-Immobilization by Bacteria
Microalgae are part of the organisms attached to filters in wastewater treatment plants, where the wastewater percolates during the treatment process. In these filters, enzymes or whole cells may be immobilized, including microalgae cells, which serve to obtain more biomass and for removing macronutrients since the production of oxygen by the algae improves the aerobic degradation of these substances. Moreover, the consumption of CO2 and the production of exopolysaccharides by microalgae can increase the bacterial growth rate, as CO2 and the production of growth-promoting substances by bacteria can improve microalgae growth. However, bacteria and microalgae may produce substances that hinder the growth of the other co-immobilized organism. Besides, the increase in pH and oxygen concentration in the medium, due to photosynthetic activity, can reduce bacterial growth in the system with the co-immobilization of bacteria and algae [35][70].3. Microalgae as Potential Raw Material for Bioproducts
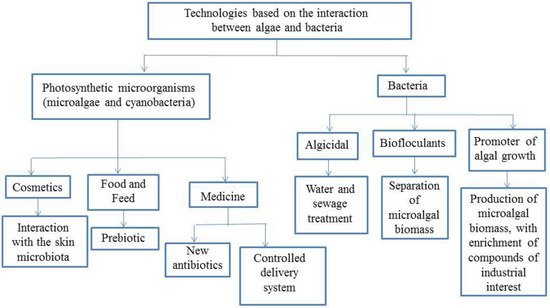
Considering the information on the interaction of microalgae and bacteria, besides the high potential of using microalgal biomass as a source of carbohydrates or fatty acids for energy production, food, cosmetic, and pharmaceutical industries, one could develop products in which microalgae, or their components could be used to confer special properties to them (Figure 12).