Antibodies (Abs) are important immune mediators and powerful diagnostic markers in a wide range of infectious diseases. Understanding the humoral immunity or the development of effective antibodies against SARS-CoV-2 is a prerequisite for limiting disease burden in the community and aids in the development of new diagnostic, therapeutic, and vaccination options. Antibody testing showed the potential in adding important diagnostic value to the routine diagnosis and clinical management of COVID-19. They could also play a critical role in COVID-19 surveillance, allowing for a better understanding of the full scope of the disease. The development of several vaccines and the success of passive immunotherapy suggest that anti-SARS-CoV-2 antibodies have the potential to be used in the treatment and prevention of SARS-CoV-2 infection.
- COVID-19
- SARS-CoV-2
- antibodies
- testing
- serodiagnostic
- immunotherapy
- vaccine
1. Introduction
2. Key Proteins of SARS-CoV-2
Understanding the behavior of SARS-CoV-2 key proteins is crucial for developing diagnostic tests, vaccines, and therapeutics. The genome of SARS-CoV-2 encodes both structural and nonstructural proteins. Spike (S), nucleocapsid (N), membrane (M), and envelope (E) are the four key structural proteins that have the potential to be targeted by the antibody response (Figure 1) [9][4]. The choice of antigen is crucially important in utilizing the virus-specific antibodies for detecting SARS-CoV-2 infection and aiding the development of therapeutics and vaccine candidates. Achieving high sensitivity and specificity when developing antibody tests is mainly dependent on the selection of the diagnostic antigens [10][5]. However, focusing on the proteins required for viral entry would have a significant impact on the development of any therapeutics or vaccination strategies; the E and M proteins have an essential role in viral assembly [11][6], and the N protein has been proved to be necessary for viral RNA synthesis (Figure 1) [12][7].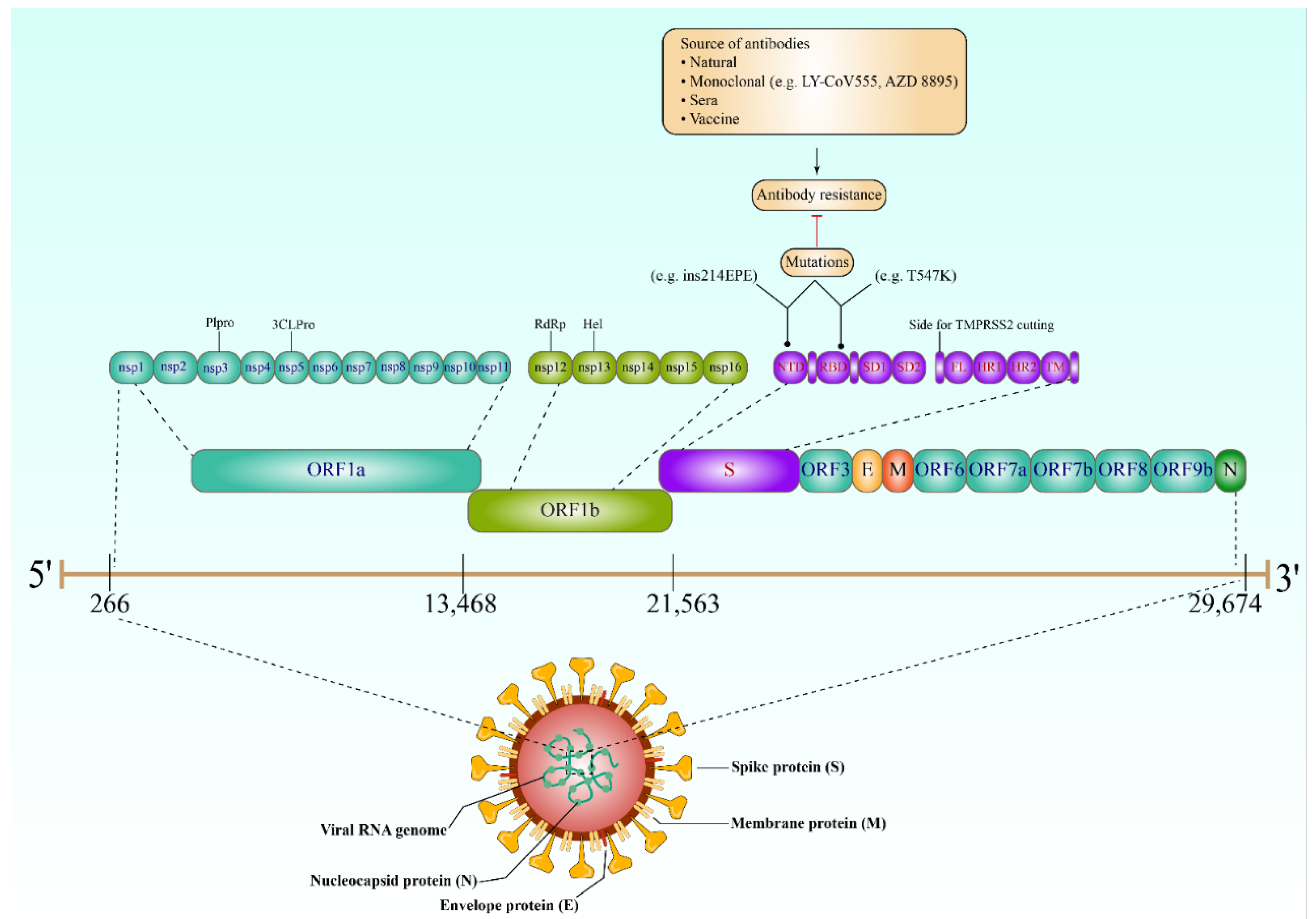
3. Antibody Response against SARS-CoV-2
4. The Role of Antibodies in SARS-CoV-2 Diagnosis
4.1. COVID-19 Antibody Tests
Unlike molecular techniques, antibody tests (also known as serological assays) rely on the detection of either viral antigenic proteins or diagnostically detectable antibodies which are created during the immune response to SARS-CoV-2 infection [27][10]. The main advantage of SARS-CoV-2 antigen tests is that the results—detecting viral antigens in throat or nasal swab samples—can be obtained in minutes [28][11]. For this reason, using antigen tests is practical, particularly for a large number of people. However, the gradual decline in viral load over time may cause difficulty in detecting viral antigens [29][12]. Instead, measuring the antibodies produced during SARS-CoV-2 infection has shown great potential for the indirect detection of the virus in a larger time window. In addition, antibody testing can play a crucial role in SARS-CoV-2 contact tracing, surveillance, and epidemiological efforts [30][13]. Many serological assays have been developed to detect SARS-CoV-2-specific IgM, IgG, and IgA, despite the uncertainty about using these different isotypes individually or in combination [31][14]. However, none of these antibody isotypes have been clearly identified as the optimal option in the scenario of COVID-19, even though the accurate interpretation of serodiagnostic tests currently depends on the type of antibodies being detected. Indeed, many serological platforms have been utilized to measure the presence of SARS-CoV-2 antibodies and/or antigens. These platforms generally take the form of chemiluminescence immunoassays (CLIA) [32[15][16],33], enzyme-linked immunosorbent assay (ELISA) [21[17][18],34], fluorescence immunoassays (FIA), rapid diagnostic tests (RDTs) and neutralization assays [35,36][19][20].4.2. Implications of Seroconversion in SARS-CoV-2 Antibody Tests
The time required by the host immune system to develop an antibody response significantly affects the capability of the serological tests to confirm SARS-CoV infection immediately after a person contracts the virus or during the early stage of infection [56][21]. Understanding the timing of seroconversion is crucial in determining the optimal time points for specimen collection [57][22]. Accordingly, establishing or planning any diagnostic protocol involving antibody tests should consider seroconversion time because it has a pivotal role in the efficacy of serodiagnostic tests. Similar to patients with SARS-CoV, the majority of COVID-19 patients seroconvert after seven days of contracting the virus. The median seroconversion times for IgM and IgG were 12 and 14 days, respectively (Figure 2) [29,58,59][12][23][24]. Even though in some patients, IgM and IgG were detected as early as four or five days (Figure 2) [60,61][25][26].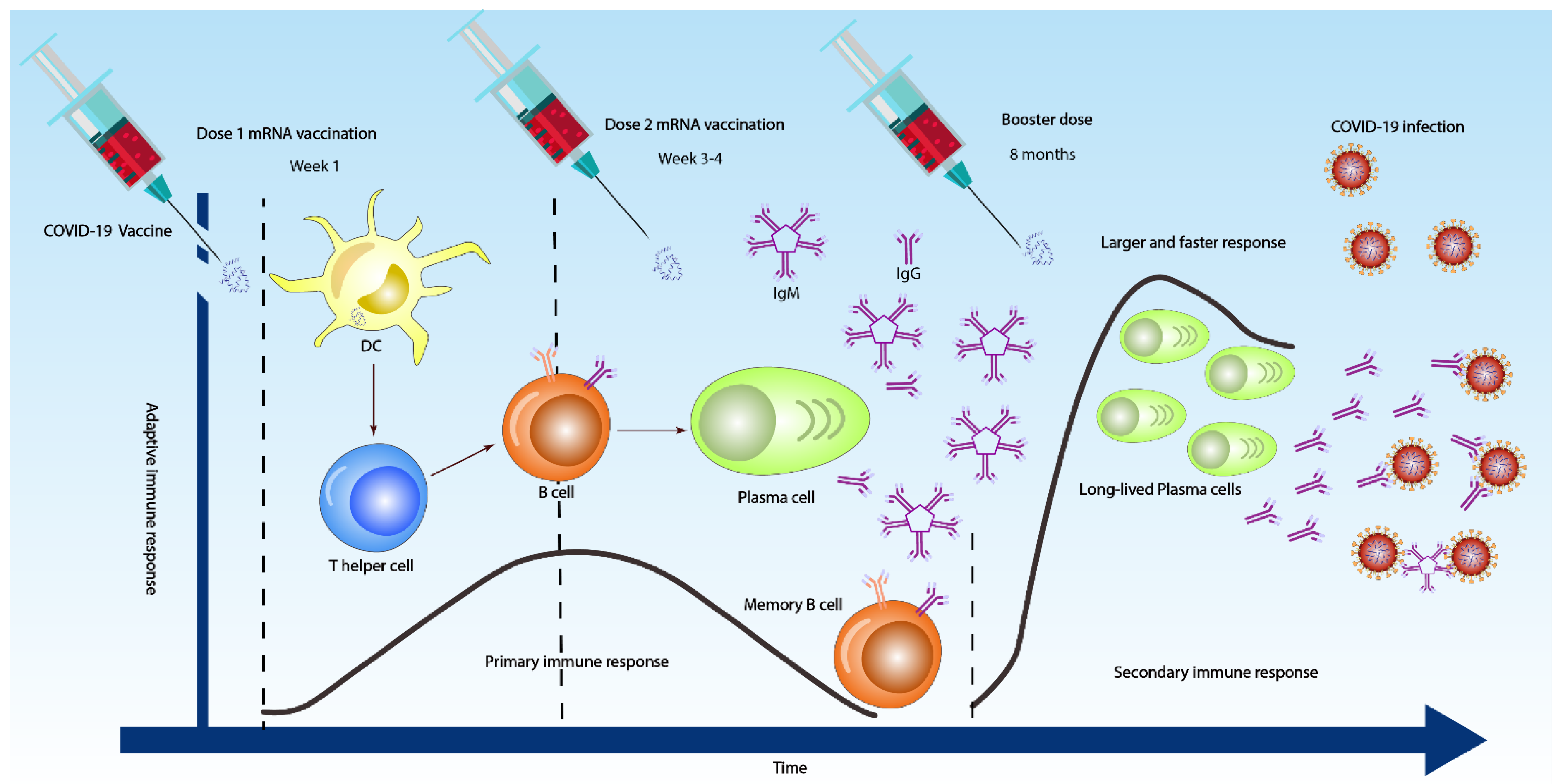
4.3. Antibody Tests at an Individual Level
4.4. Population Serological Testing
Serological testing can help identify who is infected or exposed and who is immune by assuming protective immunity. Accordingly, population-based serological information will be helpful for officials in making decisions about lifting or enforcing any control measures. The potential of this test in determining the accurate number of infected people in a large population has been tested [83,84][37][38]. Population seropositivity indicates that the number of people positive for anti-SARS-CoV-2 antibodies is much higher than that of the reported cases [84,85][38][39]. Population-based serological surveillance has been carried out around the world, including countries in Europe [86,87][40][41], America [88,89][42][43], Asia [90,91][44][45], and Africa [92,93][46][47]. Indeed, healthcare workers are the population most targeted for serological surveillance due to their high risk of SARS-CoV-2 infection, with ELISA being the most commonly used diagnostic tool for detecting anti-SARS-CoV-2 antibodies.
5. Antibody Tests and Seroprotection
5.1. Antibodies in SARS-CoV-2 Vaccine Development
Immunity against SARS-CoV-2 infection is normally acquired in one of two ways. Contracting the virus typically ends in natural immunity for a certain period, and vaccination is another way to become resistant. The entry of SARS-CoV-2 into the human cell is the first step of the infection and one of the most crucial processes in the virus’ life cycle. As a result, it is a prime target for vaccinations and therapeutics. The virus enters the cells of the lung, gastrointestinal tract (GI tract), kidneys, liver, heart, and other organs through the binding of the S protein’s receptor-binding domain (RBD) to its target host receptor, the angiotensin-converting enzyme 2 (ACE2), and a host protease known as transmembrane serine protease 2 (TMPRSS2), which facilitates the cleavage of the S glycoprotein, allowing viral access into the host cells [99,100][48][49]. Therefore, one of the main goals of SARS-CoV-2 vaccine development is to generate neutralizing antibodies that block virus entry or prevent membrane fusion. Despite the fact that the post-vaccination immune response has several components, including innate, humoral, cellular, and cytokine responses, immunological surveillance that measures antibody response is fundamental for assessing the efficacy of all SARS-CoV-2 vaccines. In particular, measuring the level of circulating anti-S-RBD antibodies could provide important information on SARS-CoV-2 acquired immunity [102][50].5.2. Mechanism of Antibody-Mediated Protection
The role of antibodies in resistance to SARS-CoV-2 infection was explored. Understanding the properties and mechanisms by which antibodies provide protection is essential to defining immunity. Infection or vaccination history may have a role in providing protection against the subsequent infection, and several studies have provided evidence for such protective associations [105][51]. Upon infection, pre-existing antibodies bind to the surface of SARS-CoV-2 virus particles and lead to the neutralization of the viral spike. Neutralizing antibodies are critical for the efficacy of any SARS-CoV-2 vaccine. Over the past two years, SARS-CoV-2-neutralizing antibodies have been developed for preventive or therapeutic uses [106,107,108][52][53][54]. Most of the neutralizing antibodies target the S protein; their neutralization potency and breadth vary according to recognition epitopes. These findings have prompted an intense effort to identify potential immunodominant epitopes that are recognized by broadly neutralizing the antibodies that could be used as templates for SARS-CoV-2 vaccine design.5.3. Prevaccination Antibody Screening
Notably, limited-resource countries (LRCs) remain at the back of the line when it comes to new technologies, infrastructure, and public health control measures such as vaccines. Evidently, COVID-19 mass vaccination is currently not applicable in most of the LRCs, where a lack of resources is placing enormous pressure on governments to accelerate the mass vaccination strategies. Accordingly, the establishment of new strategies that maximize the number of individuals who get vaccines without losing the efficacy of immune protection is urgently needed. The limited availability of authorized SARS-CoV-2 vaccines has led to widespread consideration of a single vaccine dose for people with past SARS-CoV-2 infection [110][55].6. Resistance to SARS-CoV-2 Antibodies
Resistance to the SARS-CoV-2’s antibody is now a living fact due to the introduction of new virus variants, and this places an enormous load on the vaccination process. As such, recent vaccines have been developed to contradict the virus that was first discovered in late 2019 in Wuhan, China [116][56]. However, emerged variants such as the South African-Beta (B.1.351) and the UK-Alpha (B.1.1.7) have demonstrated extensive mutations in their S proteins, and these variants have been demonstrated to be highly contiguous [117][57]. Indeed, almost all monoclonal antibodies directed against the S protein’s N-terminal domain failed to recognize Alpha, although antibodies directed against the receptor-binding region were more effective [118][58]. Nonetheless, the variation exhibited reduced affinity for plasma from patients who have recovered from SARS-CoV-2 or sera from those who have been immunized against SARS-CoV-2. Both monoclonal antibodies directed against the N-terminal domain and several separate monoclonal antibodies directed against the receptor-binding motif are very ineffective against the mutated Beta, and this resistance might have been acquired from the mutated E484K substitution. Furthermore, Beta is much more resistant to neutralization by convalescent plasma (9.4-fold) and serum (10.3–12.4-fold) from BNT162b2-immunized individuals [118][58]. Furthermore, the Beta SARS-CoV-2 variants, which had various changes in their S proteins, were resistant to 17 neutralizing monoclonal antibodies as well as sera from convalescent patients and vaccinated mice, which were unable to neutralize the variants [119][59]. Subsequent studies indicated that certain Omicron variants are now resistant to antibodies elicited by vaccine doses. Remarkably, Omicron S proteins evaded blockage by antibodies obtained from persons inoculated with the BioNTech-Pfizer vaccine (BNT162b2) or convalescent patients with 12- to 44-fold more efficiency than the Delta (B.1.617.2) variant S protein [120][60]. Furthermore, the antibodies ReGN10933, REGN10987, and JS016 and serum from vaccinated participants neutralized the S protein of numerous wild-type variations, including Alpha, Beta, Gamma, and Delta. Regn10987 antibodies, for example, were the most effective in neutralizing the aforementioned variations. In addition, the Regn10933 and JS016 antibodies were both effective, although their reactions to the S protein were quite different [125][61]. These two antibodies, however, were unable to neutralize the Beta version. Unexpectedly, none of these antibodies neutralized the Omicron variant [125][61]. This result implies that these antibodies cannot be used to combat the ongoing Omicron variant pandemic or any developing variation. Despite immunocompromised individuals potentially being more susceptible to SARS-CoV-2 viruses with unusual manifestations, prolonged immunosuppression treatment may provide some defense from severe COVID-19 disease consequences. However, the possibility of immunocompromised people acquiring abnormally severe COVID-19 is currently elusive [126][62]. Recent evidence shows that immunocompromised people may benefit greatly from convalescent plasma therapy [127[63][64][65][66],128,129,130], and that mutations and newly emerged virus strains will lead to more severe complications in the group. New variants, however, can appear in this patient population as a result of the selection pressure brought on by a severe viral infection [131][67]. The majority of people who have severe SARS-CoV-2 have immune system issues, thus the virus may live on for a very long period. Patients with impaired immune systems have been demonstrated to have varying SARS-CoV-2 evolution patterns [131][67]. How selection forces and evolutionary processes interact during chronic infection is an issue that has yet to be resolved. With that in mind, due to mutations (Q493KRBD, Q493K/RRBD) in the S protein in immunocompromised individuals, antibodies that were recovered from a healthy COVID-19 convalescent donor were shown to be ineffective in providing protection against SARS-CoV-2. In particular, the Q493KRBD mutation 15-fold reduced the effectiveness of REGN10933 pseudotype neutralization, but the Q493K/RRBD mutations almost completely imparted resistance to healthy COVID-19 convalescent donor IgG [132][68].7. Limitations of Antibody Tests
Although antibody tests are useful in COVID-19 case management and in vaccinations effectiveness, a number of drawbacks occur. The significant limitation is that antibodies may be present at undetectable levels in early days, thereby influencing the potency of any serodiagnostic tests and their effectiveness for diagnosing SARS-CoV-2 infection [135,136][69][70]. In this situation, a false-negative serological result from individuals with replicating and shedding viruses can have serious public health consequences [68][31]. Another limitation is the unknown duration at which IgM or IgG antibodies remain detectable after the virus has been cleared from the body. Furthermore, variations in antigens and methodologies used in IgM and IgG detection kits are essential, and they affect the sensitivity and specificity of the tests [137][71]. Unfortunately, the inaccuracy of antibody tests is unavoidable and will inevitably lead to false-positive or -negative results and disease misclassifications, particularly if these tests are not properly conducted and interpreted [138][72]. Moreover, the proven cross-reactivity of SARS-CoV-2 antibody tests with other coronaviruses is difficult to avoid [73][35]. Indeed, increasing the levels and duration of antibodies after SARS-CoV-2 vaccination to provide full protection and diagnostic opportunity against the current variants and those that can emerge in the future is still the primary objective in the upcoming studies.References
- Chowdhury, M.A.; Hossain, N.; Kashem, M.A.; Shahid, M.A.; Alam, A. Immune response in COVID-19: A review. J. Infect. Public Health 2020, 13, 1619–1629.
- Scourfield, D.O.; Reed, S.G.; Quastel, M.; Alderson, J.; Bart, V.M.; Teijeira Crespo, A.; Jones, R.; Pring, E.; Richter, F.C.; The Oxford-Cardiff COVID-19 Literature Consortium; et al. The role and uses of antibodies in COVID-19 infections: A living review. Oxf. Open Immunol. 2021, 2, iqab003.
- Mallano, A.; Ascione, A.; Flego, M. Antibody Response against SARS-CoV-2 Infection: Implications for Diagnosis, Treatment and Vaccine Development. Int. Rev. Immunol. 2021, 41, 393–413.
- Khailany, R.A.; Safdar, M.; Ozaslan, M. Genomic characterization of a novel SARS-CoV-2. Gene Rep. 2020, 19, 100682.
- Ching, L.; Chang, S.P.; Nerurkar, V.R. COVID-19 Special Column: Principles Behind the Technology for Detecting SARS-CoV-2, the Cause of COVID-19. Hawaii J. Health Soc. Welf. 2020, 79, 136.
- Naqvi, A.A.T.; Fatima, K.; Mohammad, T.; Fatima, U.; Singh, I.K.; Singh, A.; Atif, S.M.; Hariprasad, G.; Hasan, G.M.; Hassan, M.I. Insights into SARS-CoV-2 Genome, Structure, Evolution, Pathogenesis and Therapies: Structural Genomics Approach. Biochim. Biophys. Acta Mol Basis Dis. 2020, 1, 165878.
- Chang, C.-k.; Sue, S.-C.; Yu, T.-h.; Hsieh, C.-M.; Tsai, C.-K.; Chiang, Y.-C.; Lee, S.-j.; Hsiao, H.-h.; Wu, W.-J.; Chang, W.-L. Modular organization of SARS coronavirus nucleocapsid protein. J. Biomed. Sci. 2006, 13, 59–72.
- Dispinseri, S.; Secchi, M.; Pirillo, M.F.; Tolazzi, M.; Borghi, M.; Brigatti, C.; De Angelis, M.L.; Baratella, M.; Bazzigaluppi, E.; Venturi, G.; et al. Neutralizing antibody responses to SARS-CoV-2 in symptomatic COVID-19 is persistent and critical for survival. Nat. Commun. 2021, 12, 2670.
- Amanat, F.; Stadlbauer, D.; Strohmeier, S.; Nguyen, T.H.; Chromikova, V.; McMahon, M.; Jiang, K.; Arunkumar, G.A.; Jurczyszak, D.; Polanco, J. A serological assay to detect SARS-CoV-2 seroconversion in humans. Nat. Med. 2020, 26, 1033–1036.
- Carter, L.J.; Garner, L.V.; Smoot, J.W.; Li, Y.; Zhou, Q.; Saveson, C.J.; Sasso, J.M.; Gregg, A.C.; Soares, D.J.; Beskid, T.R. Assay techniques and test development for COVID-19 diagnosis. ACS Cent. Sci. 2020, 6, 591–605.
- Scohy, A.; Anantharajah, A.; Bodéus, M.; Kabamba-Mukadi, B.; Verroken, A.; Rodriguez-Villalobos, H. Low performance of rapid antigen detection test as frontline testing for COVID-19 diagnosis. J. Clin. Virol. 2020, 129, 104455.
- To, K.K.-W.; Tsang, O.T.-Y.; Leung, W.-S.; Tam, A.R.; Wu, T.-C.; Lung, D.C.; Yip, C.C.-Y.; Cai, J.-P.; Chan, J.M.-C.; Chik, T.S.-H. Temporal profiles of viral load in posterior oropharyngeal saliva samples and serum antibody responses during infection by SARS-CoV-2: An observational cohort study. Lancet Infect. Dis. 2020, 20, 565–574.
- Udugama, B.; Kadhiresan, P.; Kozlowski, H.N.; Malekjahani, A.; Osborne, M.; Li, V.Y.; Chen, H.; Mubareka, S.; Gubbay, J.B.; Chan, W.C. Diagnosing COVID-19: The disease and tools for detection. ACS Nano 2020, 14, 3822–3835.
- Ma, H.; Zeng, W.; He, H. Serum IgA, IgM, and IgG Responses in COVID-19. Cell Mol Immunol. 2020, 17, 773–775.
- Cai, X.-F.; Chen, J.; Long, Q.-X.; Deng, H.-J.; Liu, P.; Fan, K.; Liao, P.; Liu, B.-Z.; Wu, G.-C.; Chen, Y.-K. A Peptide-Based Magnetic Chemiluminescence Enzyme Immunoassay for Serological Diagnosis of Coronavirus Disease 2019. J. Infect. Dis. 2020, 222, 189–193.
- Infantino, M.; Grossi, V.; Lari, B.; Bambi, R.; Perri, A.; Manneschi, M.; Terenzi, G.; Liotti, I.; Ciotta, G.; Taddei, C. Diagnostic accuracy of an automated chemiluminescent immunoassay for anti-SARS-CoV-2 IgM and IgG antibodies: An Italian experience. J. Med. Virol. 2020, 92, 1671–1675.
- Guo, L.; Ren, L.; Yang, S.; Xiao, M.; Chang, D.; Yang, F.; Dela Cruz, C.S.; Wang, Y.; Wu, C.; Xiao, Y. Profiling early humoral response to diagnose novel coronavirus disease (COVID-19). Clin. Infect. Dis. 2020, 71, 778–785.
- Liu, W.; Liu, L.; Kou, G.; Zheng, Y.; Ding, Y.; Ni, W.; Wang, Q.; Tan, L.; Wu, W.; Tang, S. Evaluation of nucleocapsid and spike protein-based enzyme-linked immunosorbent assays for detecting antibodies against SARS-CoV-2. J. Clin. Microbiol. 2020, 58, e00461-20.
- Chen, Z.; Zhang, Z.; Zhai, X.; Li, Y.; Lin, L.; Zhao, H.; Bian, L.; Li, P.; Yu, L.; Wu, Y. Rapid and Sensitive Detection of anti-SARS-CoV-2 IgG, Using Lanthanide-Doped Nanoparticles-Based Lateral Flow Immunoassay. Anal. Chem. 2020, 92, 7226–7231.
- Stephen, M.; Hahn, M.D.; Commissioner of Food and Drugs—Food and Drug Administration. Coronavirus (COVID-19) Update: FDA Authorizes First Antigen Test to Help in the Rapid Detection of the Virus that Causes COVID-19 in Patients. Available online: https://www.fda.gov/news-events/press-announcements/coronavirus-covid-19-update-fda-authorizes-first-antigen-test-help-rapid-detection-virus-causes (accessed on 26 July 2022).
- Zhao, J.; Yuan, Q.; Wang, H.; Liu, W.; Liao, X.; Su, Y.; Wang, X.; Yuan, J.; Li, T.; Li, J. Antibody responses to SARS-CoV-2 in patients of novel coronavirus disease 2019. Clin. Infect. Dis. 2020, 71, 2027–2034.
- Fu, Y.; Li, Y.; Guo, E.; He, L.; Liu, J.; Yang, B.; Li, F.; Wang, Z.; Li, Y.; Xiao, R.; et al. SARS-CoV-2 Viral Dynamic Shedding, Seroconversion, and Diseases Severity Among 3192 Patients Hospitalized with COVID-19: A Large Case Series Study in Wuhan. Lancet 2020. Available online: https://ssrn.com/abstract=3594600 or http://dx.doi.org/10.2139/ssrn.3594600 (accessed on 2 July 2022).
- Wölfel, R.; Corman, V.M.; Guggemos, W.; Seilmaier, M.; Zange, S.; Müller, M.A.; Niemeyer, D.; Jones, T.C.; Vollmar, P.; Rothe, C. Virological assessment of hospitalized patients with COVID-2019. Nature 2020, 581, 465–469.
- Wu, L.-P.; Wang, N.-C.; Chang, Y.-H.; Tian, X.-Y.; Na, D.-Y.; Zhang, L.-Y.; Zheng, L.; Lan, T.; Wang, L.-F.; Liang, G.-D. Duration of antibody responses after severe acute respiratory syndrome. Emerg. Infect. Dis. 2007, 13, 1562.
- Zhang, W.; Du, R.-H.; Li, B.; Zheng, X.-S.; Yang, X.-L.; Hu, B.; Wang, Y.-Y.; Xiao, G.-F.; Yan, B.; Shi, Z.-L. Molecular and serological investigation of 2019-nCoV infected patients: Implication of multiple shedding routes. Emerg. Microbes Infect. 2020, 9, 386–389.
- Pan, Y.; Li, X.; Yang, G.; Fan, J.; Tang, Y.; Zhao, J.; Long, X.; Guo, S.; Zhao, Z.; Liu, Y. Serological immunochromatographic approach in diagnosis with SARS-CoV-2 infected COVID-19 patients. J. Infect. 2020, 81, e28–e32.
- West, R.; Kobokovich, A.; Connell, N.; Gronvall, G.K. COVID-19 Antibody Tests: A Valuable Public Health Tool with Limited Relevance to Individuals. Trends Microbiol. 2020, 29, 214–223.
- Hoffman, T.; Nissen, K.; Krambrich, J.; Rönnberg, B.; Akaberi, D.; Esmaeilzadeh, M.; Salaneck, E.; Lindahl, J.; Lundkvist, Å. Evaluation of a COVID-19 IgM and IgG rapid test; an efficient tool for assessment of past exposure to SARS-CoV-2. Infect. Ecol. Epidemiol. 2020, 10, 1754538.
- Xiang, F.; Wang, X.; He, X.; Peng, Z.; Yang, B.; Zhang, J.; Zhou, Q.; Ye, H.; Ma, Y.; Li, H. Antibody detection and dynamic characteristics in patients with COVID-19. Clin. Infect. Dis. 2020, 71, 1930–1934.
- CDC. Interim Guidelines for COVID-19 Antibody. Testing: What are the implications? Available online: https://stacks.cdc.gov/view/cdc/91257 (accessed on 1 July 2022).
- Abbasi, J. The promise and peril of antibody testing for COVID-19. JAMA 2020, 323, 1881–1883.
- Cassaniti, I.; Novazzi, F.; Giardina, F.; Salinaro, F.; Sachs, M.; Perlini, S.; Bruno, R.; Mojoli, F.; Baldanti, F. Performance of VivaDiag COVID-19 IgM/IgG Rapid Test is inadequate for diagnosis of COVID-19 in acute patients referring to emergency room department. J. Med. Virol. 2020, 92, 1724–1727.
- Li, Z.; Yi, Y.; Luo, X.; Xiong, N.; Liu, Y.; Li, S.; Sun, R.; Wang, Y.; Hu, B.; Chen, W. Development and clinical application of a rapid IgM-IgG combined antibody test for SARS-CoV-2 infection diagnosis. J. Med. Virol. 2020, 92, 1724–1727.
- WHO. Advice on the Use of Point-of-Care Immunodiagnostic Tests for COVID-19; Scientific Brief. 2020. Available online: https://www.who.int/news-room/commentaries/detail/advice-on-the-use-of-point-of-care-immunodiagnostic-tests-for-covid-19 (accessed on 26 July 2022).
- Lv, H.; Wu, N.C.; Tsang, O.T.-Y.; Yuan, M.; Perera, R.A.; Leung, W.S.; So, R.T.; Chan, J.M.C.; Yip, G.K.; Chik, T.S.H. Cross-reactive antibody response between SARS-CoV-2 and SARS-CoV infections. Cell Rep. 2020, 31, 107725.
- Jacofsky, D.; Jacofsky, E.M.; Jacofsky, M. Understanding antibody testing for COVID-19. J. Arthroplast. 2020, 35, S74–S81.
- Bendavid, E.; Mulaney, B.; Sood, N.; Shah, S.; Ling, E.; Bromley-Dulfano, R.; Lai, C.; Weissberg, Z.; Saavedra, R.; Tedrow, J. COVID-19 Antibody Seroprevalence in Santa Clara County, California. Int. J. Epidemiol. 2021, 50, 410–419.
- Doi, A.; Iwata, K.; Kuroda, H.; Hasuike, T.; Nasu, S.; Kanda, A.; Nagao, T.; Nishioka, H.; Tomii, K.; Morimoto, T. Estimation of seroprevalence of novel coronavirus disease (COVID-19) using preserved serum at an outpatient setting in Kobe, Japan: A cross-sectional study. Clin. Epidemiology Glob. Heal. 2021, 11, 100747.
- Shakiba, M.; Nazari, S.S.H.; Mehrabian, F.; Rezvani, S.M.; Ghasempour, Z.; Heidarzadeh, A. Seroprevalence of COVID-19 virus infection in Guilan province, Iran. Emerg. Infect Dis. 2020, 27, 636–638.
- Pollán, M.; Pérez-Gómez, B.; Pastor-Barriuso, R.; Oteo, J.; Hernán, M.A.; Pérez-Olmeda, M.; Sanmartín, J.L.; Fernández-García, A.; Cruz, I.; de Larrea, N.F. Prevalence of SARS-CoV-2 in Spain (ENE-COVID): A nationwide, population-based seroepidemiological study. Lancet 2020, 396, 535–544.
- Stringhini, S.; Wisniak, A.; Piumatti, G.; Azman, A.S.; Lauer, S.A.; Baysson, H.; De Ridder, D.; Petrovic, D.; Schrempft, S.; Marcus, K. Seroprevalence of anti-SARS-CoV-2 IgG antibodies in Geneva, Switzerland (SEROCoV-POP): A population-based study. Lancet 2020, 396, 313–319.
- Silveira, M.F.; Barros, A.J.; Horta, B.L.; Pellanda, L.C.; Victora, G.D.; Dellagostin, O.A.; Struchiner, C.J.; Burattini, M.N.; Valim, A.R.; Berlezi, E.M. Population-based surveys of antibodies against SARS-CoV-2 in Southern Brazil. Nat. Med. 2020, 26, 1196–1199.
- Havers, F.P.; Reed, C.; Lim, T.; Montgomery, J.M.; Klena, J.D.; Hall, A.J.; Fry, A.M.; Cannon, D.L.; Chiang, C.-F.; Gibbons, A.; et al. Seroprevalence of antibodies to SARS-CoV-2 in 10 sites in the United States, 23 March–12 May 2020. JAMA Intern. Med. 2020, 180, 1576–1586.
- Xu, X.; Sun, J.; Nie, S.; Li, H.; Kong, Y.; Liang, M.; Hou, J.; Huang, X.; Li, D.; Ma, T. Seroprevalence of immunoglobulin M and G antibodies against SARS-CoV-2 in China. Nat. Med. 2020, 26, 1193–1195.
- Noh, J.Y.; Seo, Y.B.; Yoon, J.G.; Seong, H.; Hyun, H.; Lee, J.; Lee, N.; Jung, S.; Park, M.-J.; Song, W. Seroprevalence of anti-SARS-CoV-2 antibodies among outpatients in southwestern Seoul, Korea. J. Korean Med. Sci. 2020, 35, e311.
- Adetifa, I.M.; Uyoga, S.; Gitonga, J.N.; Mugo, D.; Otiende, M.; Nyagwange, J.; Karanja, H.K.; Tuju, J.; Wanjiku, P.; Aman, R. Temporal trends of SARS-CoV-2 seroprevalence in transfusion blood donors during the first wave of the COVID-19 epidemic in Kenya. Nat. Commun. 2021, 12, 3966.
- George, J.A.; Khoza, S.; Mayne, E.; Dlamini, S.; Kone, N.; Jassat, W.; Chetty, K.; Centner, C.; Pillay, T.; Maphayi, M. Sentinel seroprevalence of SARS-CoV-2 in Gauteng Province, South Africa, August–October 2020. S. Afr. Med. J. 2021, 111, 1078–1083.
- Peng, R.; Wu, L.-A.; Wang, Q.; Qi, J.; Gao, G.F. Cell entry by SARS-CoV-2. Trends Biochem. Sci. 2021, 46, 848–860.
- Alzain, A.A.; Elbadwi, F.A.; Alsamani, F.O. Discovery of novel TMPRSS2 inhibitors for COVID-19 using in silico fragment-based drug design, molecular docking, molecular dynamics, and quantum mechanics studies. Inform. Med. Unlocked 2022, 29, 100870.
- Lo Sasso, B.; Agnello, L.; Giglio, R.V.; Gambino, C.M.; Ciaccio, A.M.; Vidali, M.; Ciaccio, M. Longitudinal analysis of anti-SARS-CoV-2 S-RBD IgG antibodies before and after the third dose of the BNT162b2 vaccine. Sci. Rep. 2022, 12, 8679.
- Huang, A.T.; Garcia-Carreras, B.; Hitchings, M.D.T.; Yang, B.; Katzelnick, L.C.; Rattigan, S.M.; Borgert, B.A.; Moreno, C.A.; Solomon, B.D.; Trimmer-Smith, L.; et al. A systematic review of antibody mediated immunity to coronaviruses: Kinetics, correlates of protection, and association with severity. Nat. Commun. 2020, 11, 4704.
- Park, Y.-J.; Marco, A.D.; Starr, T.N.; Liu, Z.; Pinto, D.; Walls, A.C.; Zatta, F.; Zepeda, S.K.; Bowen, J.E.; Sprouse, K.R.; et al. Antibody-mediated broad sarbecovirus neutralization through ACE2 molecular mimicry. Science 2022, 375, 449–454.
- Hansen, J.; Baum, A.; Pascal, K.E.; Russo, V.; Giordano, S.; Wloga, E.; Fulton, B.O.; Yan, Y.; Koon, K.; Patel, K. Studies in humanized mice and convalescent humans yield a SARS-CoV-2 antibody cocktail. Science 2020, 369, 1010–1014.
- Shi, R.; Shan, C.; Duan, X.; Chen, Z.; Liu, P.; Song, J.; Song, T.; Bi, X.; Han, C.; Wu, L. A human neutralizing antibody targets the receptor-binding site of SARS-CoV-2. Nature 2020, 584, 120–124.
- Frieman, M.; Harris, A.D.; Herati, R.S.; Krammer, F.; Mantovani, A.; Rescigno, M.; Sajadi, M.M.; Simon, V. SARS-CoV-2 vaccines for all but a single dose for COVID-19 survivors. EBioMedicine 2021, 68, 103401.
- Harvey, W.T.; Carabelli, A.M.; Jackson, B.; Gupta, R.K.; Thomson, E.C.; Harrison, E.M.; Ludden, C.; Reeve, R.; Rambaut, A.; Peacock, S.J.; et al. SARS-CoV-2 variants, spike mutations and immune escape. Nat. Rev. Microbiol. 2021, 19, 409–424.
- Faria, N.R.; Mellan, T.A.; Whittaker, C.; Claro, I.M.; Candido, D.d.S.; Mishra, S.; Crispim, M.A.E.; Sales, F.C.S.; Hawryluk, I.; McCrone, J.T.; et al. Genomics and epidemiology of the P.1 SARS-CoV-2 lineage in Manaus, Brazil. Science 2021, 372, 815–821.
- Wang, P.; Nair, M.S.; Liu, L.; Iketani, S.; Luo, Y.; Guo, Y.; Wang, M.; Yu, J.; Zhang, B.; Kwong, P.D.; et al. Antibody resistance of SARS-CoV-2 variants B.1.351 and B.1.1.7. Nature 2021, 593, 130–135.
- Li, Q.; Nie, J.; Wu, J.; Zhang, L.; Ding, R.; Wang, H.; Zhang, Y.; Li, T.; Liu, S.; Zhang, M.; et al. SARS-CoV-2 501Y.V2 variants lack higher infectivity but do have immune escape. Cell 2021, 184, 2362–2371.e2369.
- Hoffmann, M.; Krüger, N.; Schulz, S.; Cossmann, A.; Rocha, C.; Kempf, A.; Nehlmeier, I.; Graichen, L.; Moldenhauer, A.S.; Winkler, M.S.; et al. The Omicron variant is highly resistant against antibody-mediated neutralization: Implications for control of the COVID-19 pandemic. Cell 2022, 185, 447–456.e411.
- Kim, S.J.; Yao, Z.; Marsh, M.C.; Eckert, D.M.; Kay, M.S.; Lyakisheva, A.; Pasic, M.; Bansal, A.; Birnboim, C.; Jha, P.; et al. Homogeneous surrogate virus neutralization assay to rapidly assess neutralization activity of anti-SARS-CoV-2 antibodies. Nat. Commun. 2022, 13, 3716.
- Lauterio, A.; Valsecchi, M.; Santambrogio, S.; De Carlis, R.; Merli, M.; Calini, A.; Centonze, L.; Buscemi, V.; Bottiroli, M.; Puoti, M.; et al. Successful recovery from severe COVID-19 pneumonia after kidney transplantation: The interplay between immunosuppression and novel therapy including tocilizumab. Transpl. Infect. Dis. Off. J. Transplant. Soc. 2020, 22, e13334.
- Thompson, M.A.; Henderson, J.P.; Shah, P.K.; Rubinstein, S.M.; Joyner, M.J.; Choueiri, T.K.; Flora, D.B.; Griffiths, E.A.; Gulati, A.P.; Hwang, C.; et al. Association of Convalescent Plasma Therapy With Survival in Patients With Hematologic Cancers and COVID-19. JAMA Oncol. 2021, 7, 1167–1175.
- Betrains, A.; Godinas, L.; Woei, A.J.F.; Rosseels, W.; Van Herck, Y.; Lorent, N.; Dierickx, D.; Compernolle, V.; Meyfroidt, G.; Vanderbeke, L.; et al. Convalescent plasma treatment of persistent severe acute respiratory syndrome coronavirus-2 (SARS-CoV-2) infection in patients with lymphoma with impaired humoral immunity and lack of neutralising antibodies. Br. J. Haematol. 2021, 192, 1100–1105.
- Fung, M.; Nambiar, A.; Pandey, S.; Aldrich, J.M.; Teraoka, J.; Freise, C.; Roberts, J.; Chandran, S.; Hays, S.R.; Bainbridge, E.; et al. Treatment of immunocompromised COVID-19 patients with convalescent plasma. Transpl. Infect. Dis. Off. J. Transplant. Soc. 2021, 23, e13477.
- Ljungquist, O.; Lundgren, M.; Iliachenko, E.; Månsson, F.; Böttiger, B.; Landin-Olsson, M.; Wikén, C.; Rosendal, E.; Överby, A.K.; Wigren, B.J.; et al. Convalescent plasma treatment in severely immunosuppressed patients hospitalized with COVID-19: An observational study of 28 cases. Infect. Dis. 2022, 54, 283–291.
- Evolutionary insight into the emergence of SARS-CoV-2 variants of concern. Nat. Med. 2022, 28, 1357–1358.
- Clark, S.A.; Clark, L.E.; Pan, J.; Coscia, A.; McKay, L.G.A.; Shankar, S.; Johnson, R.I.; Brusic, V.; Choudhary, M.C.; Regan, J.; et al. SARS-CoV-2 evolution in an immunocompromised host reveals shared neutralization escape mechanisms. Cell 2021, 184, 2605–2617.e2618.
- Binnicker, M.J. Challenges and Controversies to Testing for COVID-19. J. Clin.Microbiol. 2020, 58, e01695-20.
- Zhao, J.; Liao, X.; Wang, H.; Wei, L.; Xing, M.; Liu, L.; Zhang, Z. Early virus clearance and delayed antibody response in a case of COVID-19 with a history of co-infection with HIV-1 and HCV. Clin. Infect. Dis. 2020, 71, 2233–2235.
- Xie, J.; Ding, C.; Li, J.; Wang, Y.; Guo, H.; Lu, Z.; Wang, J.; Zheng, C.; Jin, T.; Gao, Y. Characteristics of patients with coronavirus disease (COVID-19) confirmed using an IgM-IgG antibody test. J. Med. Virol. 2020, 92, 2004–2010.
- Ismail, A.A. Serological tests for COVID-19 antibodies: Limitations must be recognized. Ann. Clin. Biochem. 2020, 57, 274–276.
