Polyphenols consumption has been associated to a lower risk of cardiovascular diseases (CVDs) notably through nitric oxide (NO)- and estrogen receptor α (ERα)-dependent pathways. Among polyphenolic compounds, chalcones have been suggested to prevent endothelial dysfunction and hypertension. However, the involvement of both the NO and the ERα pathways for the beneficial vascular effects of chalcones has never been demonstrated. In this study, we aimed to identify chalcones with high vasorelaxation potential and to characterize the signaling pathways in relation with ERα signaling and NO involvement. The evaluation of vasorelaxation potential was performed by myography on wild-type (WT) and ERα knock-out (ERα-KO) mice aorta in presence or in absence of the eNOS inhibitor Nω-nitro-L-arginine methyl ester (L-NAME). Among the set of chalcones that were synthesized, four exhibited a strong vasorelaxant effect (more than 80% vasorelaxation) while five compounds have shown a 60% relief of the pre-contraction and four compounds led to a lower vasorelaxation. We were able to demonstrate that the vasorelaxant effect of two highly active chalcones was either ERα-dependent and NO-independent or ERα-independent and NO-dependent.
- Polyphenols
- chalcones
- structure-activity relationships
- endothelium
- vasodilation
- cardiovascular diseases
1. Chemistry and cLogP
3
6
8
10
8
11
13
17
2
[3]
18
[4]
12
14
’
13
15
3
[5]
16
17
18
6
14
Scheme 1.
3
Table 1.
|
Compound |
R2’ |
R4’ |
R6’ |
R3 |
R4 |
R5 |
cLogP |
|
3 |
OH |
OH |
H |
OCH3 |
OH |
OCH3 |
2.42 |
|
6 |
11
16
17
3
12
14
18
20
3
2
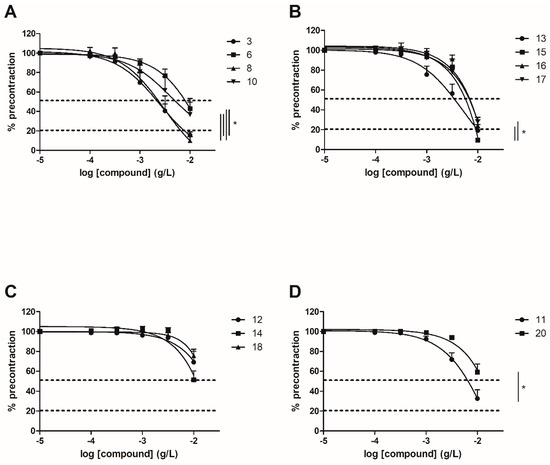
Figure 1.
A
2’
4’
6’
B
2’
4’
3
6’
3
C
2’
4’
3
6’
3
D
2’
4’
3
6’
y
x
3
8
13
15
6
10
11
16
17
12
14
18
20
N
p
Table 2.
|
Compound |
Potency (EC50, g/L) WT |
Efficacy (Emax, %) |
||||||||||||
|
WT |
WT with L-NAME |
ERα KO |
ERα KO with L-NAME |
|||||||||||
|
3 |
OH |
OH |
OH |
OCH3 |
2.44 × 10 |
OH |
OCH3 |
1.80 |
||||||
-3 |
84.28 ± 3.24a,d |
77.62 ± 4.92 |
56.23 ± 4.91£ |
43.22 ± 6.10$ |
8 |
OH |
||||||||
|
6 |
2.12 × 10-3 |
OH |
57.10 ± 10.40c,d |
H |
33.92 ± 12.66 | OCH3 |
60.69 ± 10.75 |
OCH3 |
38.15 ± 10.91 |
OCH3 |
2.75 |
|||
|
10 |
OH |
OH |
||||||||||||
|
8 |
2.99 × 10-3 |
89.96 ± 4.13b,c |
OH |
OCH3 |
OCH3 |
OCH3 |
2.13 |
|||||||
86.69 ± 3.47 | 79.73 ± 3.71 |
69.76 ± 7.99 |
11 |
OCH3 |
||||||||||
|
10 |
4.08 × 10 |
OCH3 |
-3 |
63.29 ± 12.79a,b |
H |
44.91 ± 15.07 |
H |
52.83 ± 12.88 | OH |
43.33 ± 10.37 |
H |
3.20 |
||
|
12 |
||||||||||||||
|
11 |
OH |
OCH3 |
H |
OCH3 |
OH |
OCH3 |
2.99 |
|||||||
1.93 × 10-2 |
67.35 ± 8.94g |
49.06 ± 9.47 |
74.81 ± 2.07 |
36.40 ± 5.81 |
13 |
OCH3 |
OCH3 |
H |
||||||
|
12 |
NA | OCH | 3 |
30.95 ± 13.23 | OH |
NA |
OCH3 |
NA |
2.84 |
|||||
NA |
14 |
OH |
OCH3 |
H |
OCH | |||||||||
|
13 | 3 | OCH |
5.06 × 10-2 |
80.86 ± 6.11 | 3 |
19.85 ± 7.81* |
81.96 ± 2.46 |
26.96 ± 6.72 |
OCH3 |
3.33 |
||||
|
15 |
OCH3 |
|||||||||||||
|
14 |
NA |
OCH3 |
48.38 ± 7.75 | H |
NA |
OCH3 |
4.64 ± 9.95 |
OCH3 |
NA |
OCH3 |
3.17 |
|||
|
16 |
||||||||||||||
|
15 |
OCH3 |
NA |
OCH3 |
90.59 ± 1.56e,f | OCH3 |
76.07 ± 3.72 | OCH3 |
OCH3 |
OCH3 |
3.13 |
||||
79.78 ± 5.51 | 76.74 ± 5.68 |
17 |
OCH3 |
OCH3 |
OCH3 |
OCH3 |
OH |
OCH3 |
2.79 |
|||||
|
16 |
NA |
75.89 ± 5.99f |
NA |
74.91 ± 10.15 |
NA |
18 |
OH |
OCH3 |
OCH3 |
OCH3 |
OH |
OCH3 |
||
|
17 |
NA |
72.16 ± 4.59e |
NA | 2.97 | ||||||||||
86.88 ± 4.40 | NA |
20 |
OCH3 |
OCH3 |
H |
OCH3 |
OCH2 | |||||||
|
18 |
NA | COOH |
24.34 ± 4.60 |
NA |
OCH3 |
2.46 |
||||||||
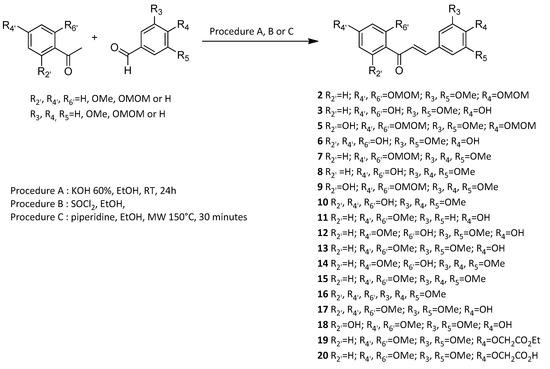
2. Evaluation of Vasorelaxant Activity
3
8
13
15
3
6
10
NA | |||||
NA | |||||
20 | |||||
NA |
40.77 ± 8.22g |
NA |
NA |
NA |
3. Involvement of ERα and NO Pathways
3.1. Evaluation of Vasorelaxant Activity on ERα KO Mice Aorta
3
3
6
8
10
13
15
16
17
3
6
8
10
13
15
16
17
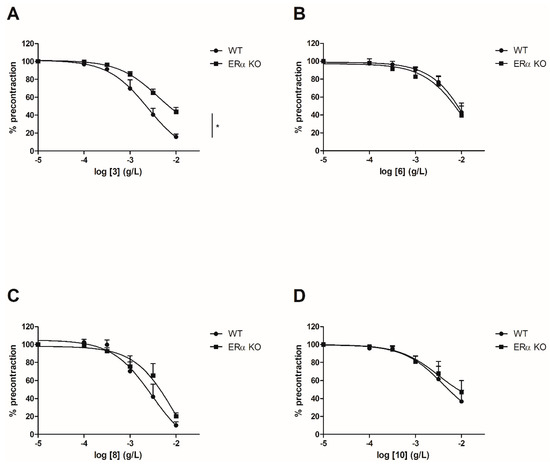
Figure 2.
3
A
6
B
8
C
10
D
y
x
3
6
8
10
N
p
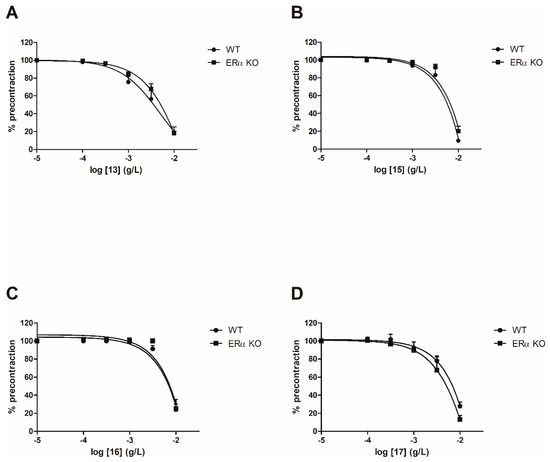
Figure 3.
13
A
15
B
16
C
17
D
y
x
13
15
16
17
N
3.2. Evaluation of Vasorelaxant Activity in the Presence of
Nω-nitro-L-arginine methyl ester
(L-NAME)
−4
3
8
13
15
13
−2
3
8
−2
15
13
3
8
15
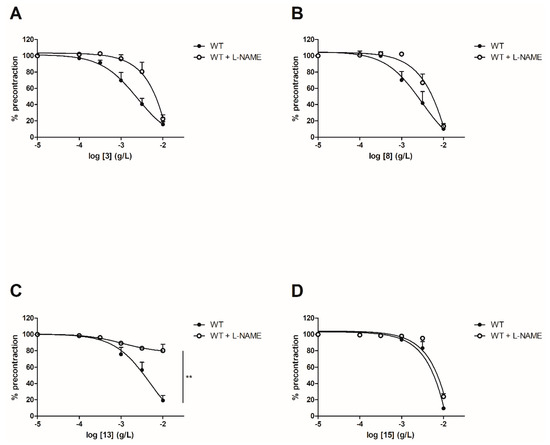
Figure 4.
3
A
8
B
13
C
15
D
y
x
13
−2
3
8
15
N
p
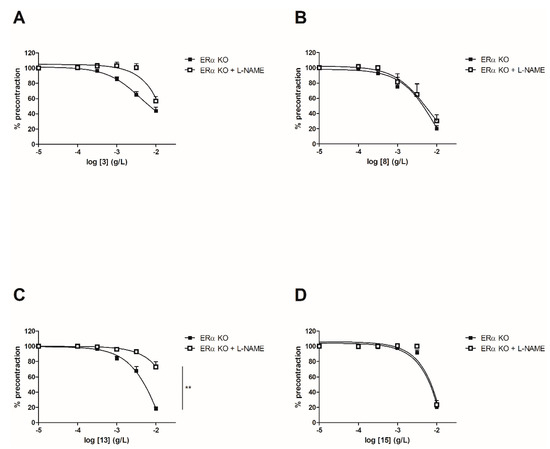
Figure 5.
3
A
8
B
13
C
15
D
y
x
13
3
8
15
N
p < 0.01 (Mann-Whitney).
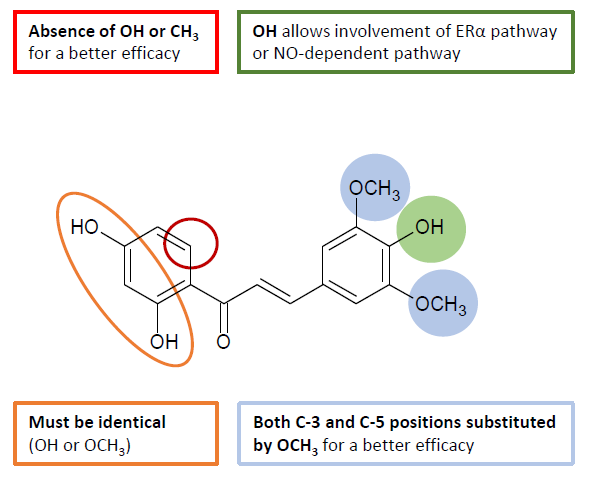
Figure 6. Structure-activity relationship of chalcones for vasodilation.
References
- Marta Perro Neves; Sara M. Cravo; Raquel T. Lima; M. Helena Vasconcelos; M. São José Nascimento; Artur Silva; Madalena Pinto; Honorina Cidade; Arlene Correa; Solid-phase synthesis of 2′-hydroxychalcones. Effects on cell growth inhibition, cell cycle and apoptosis of human tumor cell lines. Bioorganic & Medicinal Chemistry 2012, 20, 25-33, 10.1016/j.bmc.2011.11.042.
- Yesseny Vásquez-Martínez; Mauricio Osorio; Diego San Martín; Marcela Carvajal; Alejandra Vergara; Elizabeth Sanchez; Marcela Raimondi; Susana Zacchino; Carolina Mascayano; Claudia Torrent; Francisco Cabezas; Sophia Mejias; Margarita Montoya; Marcelo Cortez-San Martín; Antimicrobial, Anti-Inflammatory and Antioxidant Activities of Polyoxygenated Chalcones. Journal of the Brazilian Chemical Society 2018, null, , 10.21577/0103-5053.20180177.
- Ognyan Petrov; Yordanka Ivanova; Mariana Gerova; SOCl2/EtOH: Catalytic system for synthesis of chalcones. Catalysis Communications 2008, 9, 315-316, 10.1016/j.catcom.2007.06.013.
- Marina Roussaki; Belinda Hall; Sofia Costa Lima; Anabela Cordeiro Da Silva; Shane R. Wilkinson; Anastasia Detsi; ChemInform Abstract: Synthesis and anti-Parasitic Activity of a Novel Quinolinone-Chalcone Series.. ChemInform 2014, 45, , 10.1002/chin.201415168.
- Anupama Shirali; Madhavi Sriram; John J. Hall; Benson L. Nguyen; Rajsekhar Guddneppanavar; Mallinath B. Hadimani; J. Freeland Ackley; Rogelio Siles; Christopher J. Jelinek; Phyllis Arthasery; Rodney C. Brown; Victor Leon Murrell; Austin McMordie; Suman Sharma; David J. Chaplin; Kevin Pinney; Sanjay Sharma; Development of Synthetic Methodology Suitable for the Radiosynthesis of Combretastatin A-1 (CA1) and Its Corresponding Prodrug CA1P†. Journal of Natural Products 2009, 72, 414-421, 10.1021/np800661r.
