Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Anatoly Antipov and Version 2 by Rita Xu.
Among the most effective energy systems for stationary applications, a special place is occupied by redox flow battery (RFB) technology, encompassing easy scalability with independent scaling of power density and energy capacity, no detrimental effects of a deep discharge, very low self-discharge, low cost for a large system compared to other types of batteries, and long cycle life.
- hybrid flow batteries
- zinc-bromine flow batteries
- hydrogen-bromate flow batteries
1. Introduction
The well-known 17 Sustainable Development Goals (SDGs) are at the hearing today to form the 2030 ecological agenda majors [1]. Among them, the reliable technologies to provide energy in a sustainable way are limited by insufficient usage of renewable energy sources. Due to the intermittent availability of the latter, the demand for effective energy storage systems (ESS) today is of crucial importance.
The distributed energy resources (DER) model is another ace in the hole that offers more flexible and integrative energy storage mechanisms within the local power grid close to the end user, thus minimizing the demand for power transmission lines usage [2]. An essential solution to the problem of energy accumulation and storage in distributed networks is the use of chemical power sources (CPS).
However, the wide distribution and commercialization of flow batteries are currently hindered by their insufficient energy capacity and power, primarily due to the low energy density of reagents used [3][4]. To achieve significant capacity, the electrolyte tanks have to be large enough, along with the aqueous electrolyte, which often makes the battery very heavy and suitable only for stationary applications. Hybrid flow batteries (e.g., zinc-bromine, zinc-cerium, zinc-iron, iron-iron), which have a liquid-solid electrochemical reaction, are prone to additional degradation due to dendrite formation and increased resistance, while the common all-liquid systems such as vanadium and polysulfide bromide often require electrolyte conditioning, a process of balancing the electrolyte chemistry using additional hardware or maintenance steps, which is required daily.
The high application potential of RFB ensures continued interest of the scientific community both in (i) studying the properties and principles of their operation to improve commonly proposed systems, and in (ii) the development of energy storage devices with new reagent types or RFB concepts. These modern studies of RFB are primarily aimed at overcoming two fundamental barriers that significantly hinder the development of the direction: (a) insufficient specific power due to small values of exchange currents for heterogeneous redox reactions used in RFB and (b) low energy density compared to the competing CPS, due to insufficient energy capacity of the reagents per unit volume or mass. To solve these issues scientific community proposed a number of concepts, such as the use of hybrid systems, in which one of the half-reactions in the battery is either replaced by a hydrogen oxidation reaction [4][5][6][7][5,6,7,8] or an electroactive component in a solid state [8][9][10][9,10,11]. In novel fundamental concepts, high-capacity electrolytes are proposed either in specific states, e.g., [7][11][12][13][8,12,13,14] or in specific forms, e.g., various salts of halates as highly soluble multielectron redox-active components [14][15].
2. Halogen Hybrid Flow Batteries Perspective Concepts Analysis
2.1. Hybrid Principle Basis
The hybrid device may lose performance in some way, but this change can push a device from completely non-functional to viable in terms of functionality. So, in the case of lithium batteries, the lithium-metal electrode was sacrificed and replaced by lithium-intercalated graphite, which led to a 30% decrease in theoretical energy density, nevertheless making it possible to create a battery with a long-life cycle [15][88]. In general, a natural consequence of this approach is the free combination of design and chemistry for the cathode and anode half-cells of the electrochemical system in a way to balance the system as much as possible in terms of performance. When doing this optimization, there is no need to stay within the same design or operation principle for the power source: the ability to combine different technologies is the key to obtaining new energy storage devices that simultaneously combine the advantages of capacitors, electrochemical or fuel cells, flow batteries and other technologies. At present, this approach to the development of new energy sources is used everywhere. Thus, an increase in the energy capacity of a conventional double-layer capacitor with the reversible redox electrochemical processes on the electrode near-surface layer leads to a new type of electrochemical capacitor–supercapacitor [16][17][18][19][20][21][89,90,91,92,93,94]. The combination of various chemistries of galvanic cells has led to the appearance of lithium-sulfur batteries with outstanding performance in terms of specific energy capacity—a value of more than 500 W h has been experimentally achieved [22][23][24][25][26][27][28][95,96,97,98,99,100,101]. Even higher energy intensity (comparable to the energy intensity of gasoline fuel) is demonstrated by a hybrid of a fuel cell and a lithium-ion battery, a lithium-air battery, where electric current is generated due to oxidation of lithium at the anode and oxygen reduction from the air at the cathode [29][30][31][32][33][34][35][36][37][102,103,104,105,106,107,108,109,110]. Despite a number of difficulties associated with the use of metal anodes, the choice of catalysts and electrolytes, metal-air electrochemical sources are the prominent result of combining the idea of electrolyte circulating through the system and the design of a galvanic cell, for example, for transport applications [38][39][40][41][42][43][44][45][46][47][111,112,113,114,115,116,117,118,119,120]. The principle of combining various electrochemical approaches can also play a decisive role in solving the problems of traditional fuel cells, where you can sacrifice air oxygen by replacing it with another reagent (whereas the hydrogen reaction is stored at an anode) because such a replacement immediately eliminates the crucial problems of conventional fuel cells: the high cost of the system due to utilization of platinum and other catalysts, and dissolution of cathode catalyst [48][49][50][121,122,123]. However, an adequate choice of new oxidizing agent for a hybrid flow battery is an extremely difficult task, since several requirements must be met simultaneously: electrolyte must have a high energy density and its reduction reaction must be kinetically fast to ensure the high energy efficiency of the device. Additionally, all reagents and products of the cathodic reaction must be nontoxic, stable and non-flammable, preferably liquids or gases, since, e.g., solids, pastes and suspensions are often commercially less preferable in flow battery design.2.2. Halogen Hybrid Flow Batteries
Considering the above-mentioned requisites one can recall the hydrogen-halogen hybrid flow batteries, primarily due to rapid, reversible kinetics, which leads to excellent system performance and the use of inexpensive reagents [10][51][52][53][54][11,75,124,125,126]. In such a system a halogen oxidizer X2 (for example, Br2, Cl2, or I2) is used in the form of an aqueous solution and hydrogen (H2) is used as a reducing agent. The potential difference between the halogen and hydrogen electrodes V is equal to the EMF (from 0.54 V for I2 to 1.4 V for Cl2). It should be noted that F2, which is not considered here, has the largest potential difference relative to the standard hydrogen electrode (3.05 V), however, due to technological difficulties in the separation of gaseous fluorine from a mixture of gases, its use is unreasonable due to energy considerations and the problem of materials’ compatibility with this reagent. In this battery the following reactions undergo on electrodes (from left to right—charge mode, from right to left—discharge):2.3. Halate Hybrid Flow Batteries
The solution to the problem of the insufficient energy capacity of hydrogen-halogen flow batteries was proposed in 2015 by Yu.V. Tolmachev in [14][15]—to abandon the use of dissolved halogens in favor of a new family of aqueous multielectron oxidants—solutions of halogen oxoacids. Lithium bromate LiBrO3, which should be converted at the cathode to lithium bromide LiBr, serves as a promising object of study in [14][15]. Due to the very high solubility of both substances in water (according to [14][15] even at room temperature, the concentration of a saturated solution of lithium bromate is about 9 mol l−1, and bromide—more than 10 mol l−1) and the transfer of 6 electrons for bromate-bromide transition results in very high charge densities, more than 1000 A h kg−1, which are more than an order of magnitude higher than similar indicators for other redox flow batteries systems proposed to date. Other advantages of this oxidizing agent, mentioned in [14][15], and demonstrated experimentally in [75][144], are its long-term chemical stability (at moderately low pH), low toxicity of the reagent and product, the utilization of commodity chemicals, low cost per unit of generated electrical energy (due to both a relatively inexpensive reagent and the absence of expensive catalysts for the cathodic reaction, in particular, noble metals), low self-discharge rate and the absence of fire and explosion hazards. Additionally, in addition to lithium bromate, the use of commercially available sodium bromates with water solubility up to 2 mol l−1, seems very promising [14][15]. The reason why the bromate anion (as well as other oxohalogenate ions) did not attract attention for applications in electrochemical energy earlier is its low electrochemical activity: on all the studied electrodes, including noble metals, its direct electroreduction reaction at high-rate proceeds at high overvoltages. For the first time, it has been experimentally confirmed that despite the direct electroreduction of bromate anion via heterogeneous reactions cannot be performed without the huge cathode overvoltage, nevertheless, it can be carried out via redox mediator autocatalysis (EC″ mechanism) through a combination of the following bromine reduction reactions:-
The high reversibility of the electrochemical step (5) of the EC″ mechanism for the bromine/bromide redox couple used in the system, even on carbon electrodes. That is a prerequisite for high current density, sufficient voltage for under load conditions and the specific power of the system;
-
Irreversibility of the chemical step (5) of the EC″ mechanism due to sufficiently high solution acidity (acid volume concentration approximately in the molar range);
-
The first order of the comproportionation reaction (5) with respect to the anions of bromate and bromide, the second order—with respect to the activity of protons:
-
where aH = 10−pH proton activity (in the form of hydroxonium ions), fBrO3 and fBr are activity coefficients of the bromate and the bromide anions, correspondingly.
-
The six-electron nature of the electrochemical process (the Br atom changes its oxidation state from +5, which corresponds to BrO3−, to −1, which corresponds to Br−) during the cycle is a prerequisite for ensuring a high specific energy capacity of the system.

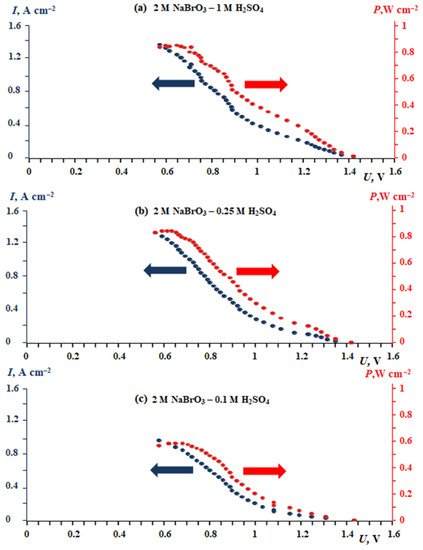
Figure 2. Dependence of current density and specific power vs. voltage for a laboratory membrane electrode assembly prototype (see Figure 13a) with an active surface area of 50 cm2 at different acidic concentrations of the bulk solution: (a) 1 mol L−1 H2SO4, (b) 0.25 mol L−1 H2SO4, (c) 0.1 mol L−1 H2SO4.
