Proteolysis-targeting chimeras (PROTACs) are molecules that selectively degrade a protein of interest (POI). The incorporation of ligands that recruit mouse double minute 2 (MDM2) into PROTACs, forming the so-called MDM2-based PROTACs, has shown promise in cancer treatment due to its dual mechanism of action: a PROTAC that recruits MDM2 prevents its binding to p53, resulting not only in the degradation of POI but also in the increase of intracellular levels of the p53 suppressor, with the activation of a whole set of biological processes, such as cell cycle arrest or apoptosis. In addition, these PROTACs, in certain cases, allow for the degradation of the target, with nanomolar potency, in a rapid and sustained manner over time, with less susceptibility to the development of resistance and tolerance, without causing changes in protein expression, and with selectivity to the target, including the respective isoforms or mutations, and to the cell type, overcoming some limitations associated with the use of inhibitors for the same therapeutic target.
- proteolysis-targeting chimera (PROTAC)
- mouse double minute 2 (MDM2)
- p53
- cancer
- targeted protein degradation
1. PROteolysis-TArgeting Chimeras—PROTACs
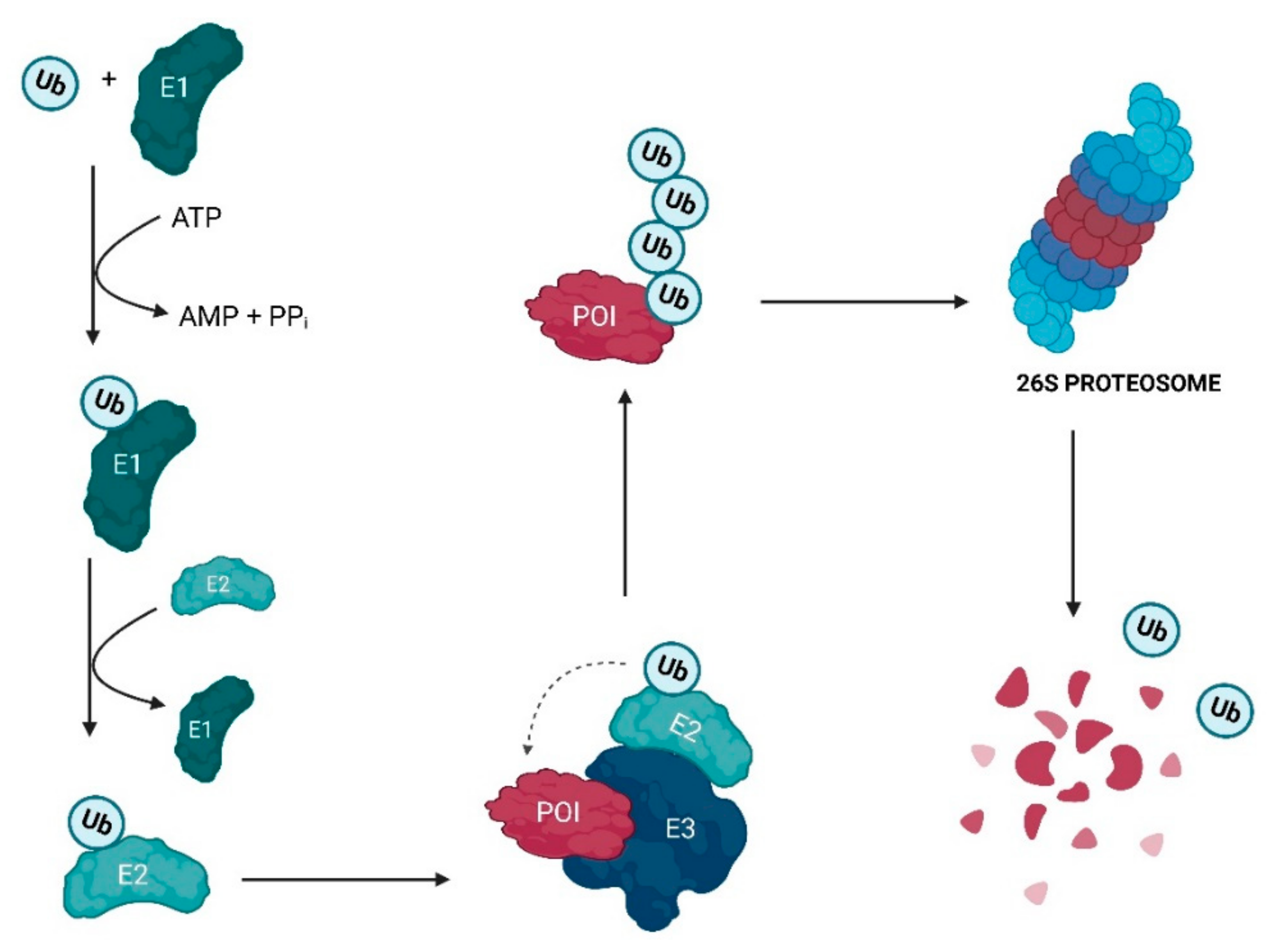
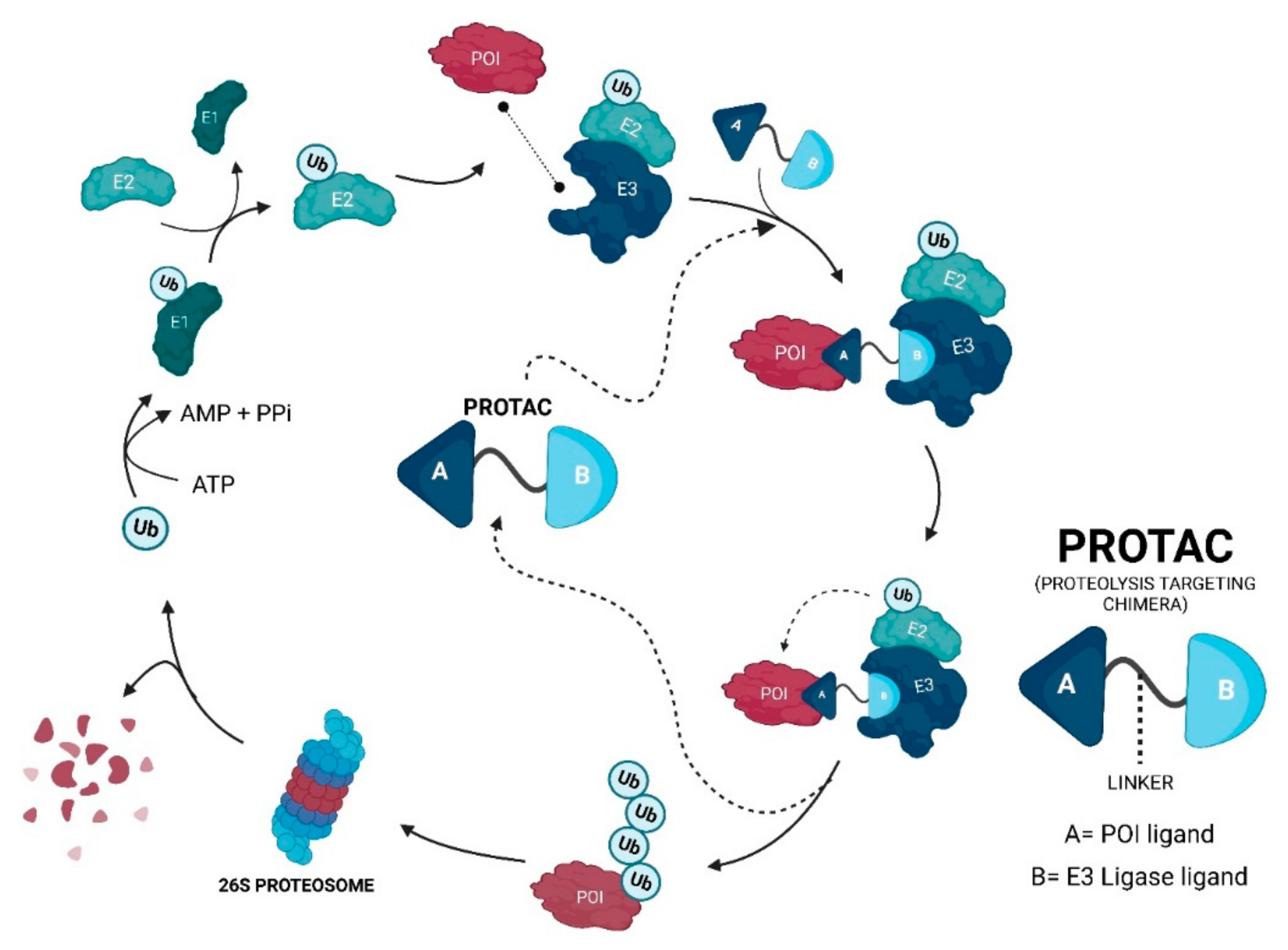
2. Evolutionary Perspective of PROTACs
2.1. First Generation—Peptide-Based PROTACs (2001–2008)
2.2. Second Generation—Small Molecule-Based PROTACs
2.3. Third Generation—Spatiotemporal Controllable PROTACs
3. PROTACs That Recruit MDM2 E3 Ligase
Although presenting very promising results, the first generation of PROTACs, regarding the degradation of key proteins in tumor development [32][20], has some disadvantages, mainly due to high molecular weight, which ends up compromising its stability and cellular permeability, such as hindering the entire process of synthesis and purification [6]. These aspects are extremely relevant when the aim is to develop a drug since a complex synthesis and purification process causes disadvantages for the pharmaceutical industry and, thus, compromises its entry into the market [33]. The creation of smaller PROTACs, using small chemical ligands, as well as linkers of a chemical origin, is a way to circumvent the problems presented by the previous generation, as was seen with the creation of the first small-based PROTAC in 2008, which, interestingly, was also the first MDM2-based PROTAC [6]. When this first MDM2-based PROTAC was created, the main advantage revealed at the time was that it only allowed for a reduction in its dimensions [6]. However, recruiting the MDM2 E3 ligase brings more benefits in terms of cancer treatment since it is responsible for the regulation of the tumor suppressor p53, which has a relevant role in the development of a high number of cancers [34][35]. Although the number of PROTACs recruiting MDM2 is still reduced, they have enormous potential in certain types of cancer, with a focus on those in which both the POI and E3 ligase are overexpressed [36]. This is because an MDM2-based PROTAC, in addition to having the ability to promote POI degradation by recruiting MDM2 with this binding ends up preventing its main function, that is, it binds to p53, leads to an increase in intracellular levels of the suppressor (Figure 3) [16].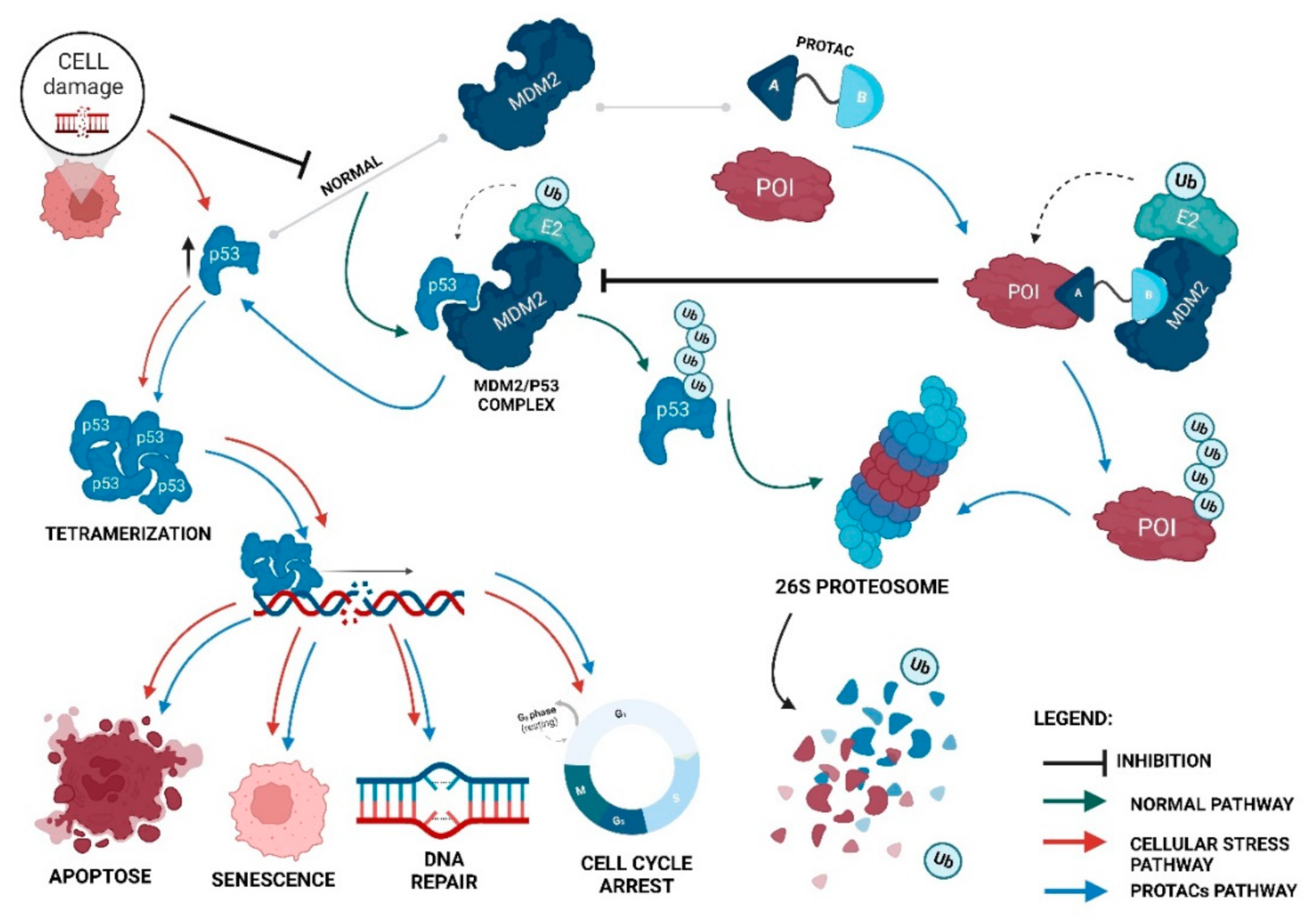 Figure 3. The impact of MDM2-based PROTACs on the MDM2/p53 cycle. Under normal conditions, p53 levels are kept low due to the presence of the MDM2 E3 ligase, which, when binding to the tumor suppressor, promotes its polyubiquitination, being subsequently recognized by the 26S proteasome, and degraded. When cell damage occurs, p53 is stabilized and forms a tetramer, and its ubiquitination by MDM2 is prevented. The tetramer is transported towards the nucleus, where it activates the transcription of those genes responsible for the processes of apoptosis, senescence, DNA repair, and cell-cycle arrest. However, in the presence of an MDM2-based PROTAC, when recruiting MDM2, this prevents it from binding to its natural substrate, p53, increasing the levels of the suppressor, which culminates not only in the degradation of POI, but also in the activation of processes that prevent the development of cancer cells [16].
Therefore, with MDM2-based PROTACs, it is possible not only to degrade the target oncogenic proteins, but also prevents MDM2 from binding to p53, activating its tumor suppressor action [16]. This duality of effect presented by this type of PROTAC makes their use in cancer therapy of great interest; they may become a great therapeutic weapon in the coming years.
MDM2-based PROTACs - Summary table of all MDM2-based PROTACs created to date for the treatment of several types of cancer: https://www.mdpi.com/1841390
Figure 3. The impact of MDM2-based PROTACs on the MDM2/p53 cycle. Under normal conditions, p53 levels are kept low due to the presence of the MDM2 E3 ligase, which, when binding to the tumor suppressor, promotes its polyubiquitination, being subsequently recognized by the 26S proteasome, and degraded. When cell damage occurs, p53 is stabilized and forms a tetramer, and its ubiquitination by MDM2 is prevented. The tetramer is transported towards the nucleus, where it activates the transcription of those genes responsible for the processes of apoptosis, senescence, DNA repair, and cell-cycle arrest. However, in the presence of an MDM2-based PROTAC, when recruiting MDM2, this prevents it from binding to its natural substrate, p53, increasing the levels of the suppressor, which culminates not only in the degradation of POI, but also in the activation of processes that prevent the development of cancer cells [16].
Therefore, with MDM2-based PROTACs, it is possible not only to degrade the target oncogenic proteins, but also prevents MDM2 from binding to p53, activating its tumor suppressor action [16]. This duality of effect presented by this type of PROTAC makes their use in cancer therapy of great interest; they may become a great therapeutic weapon in the coming years.
MDM2-based PROTACs - Summary table of all MDM2-based PROTACs created to date for the treatment of several types of cancer: https://www.mdpi.com/1841390
4. Advantages of MDM2-Based PROTACs
Nowadays, conventional therapies with mAbs, capable of blocking extracellular targets or SMIs which can inhibit extra and intracellular compounds (and that respect Lipinski’s Rule of 5 (Ro5) for oral administration), unfortunately have some limitations [37]. However, SMIs perform a stoichiometric inhibition [38], in which one molecule of inhibitor inhibits one molecule of the target, following an occupancy-driven model, in which high concentrations are required to maintain a certain level of occupancy of the target to exert the desired effect [38][39]. Consequently, the use of high concentrations promotes the occurrence of adverse effects associated with some disadvantages, such as the inability to inhibit “undruggable proteins”, protein accumulation, and increasing its expression or the development of resistance [39] [37]. The development of PROTACs, more specifically MDM2-based PROTACs, can promote therapeutic effects through target degradation, as seen above, and has some advantages over inhibition, such as the following.-
PROTACs can display cooperativity—binding to a second protein in PROTACs can lead to the formation of the ternary complex being favored (positive cooperativity), or it can lead to the formation of binary complexes (negative cooperativity) or simply remain not affected (neutral cooperativity). If positive cooperativity exists, a stable ternary complex can be formed even in the presence of low affinities with the POI [38][39];
-
PROTACs are less susceptible to the development of resistance—the resistance me-chanism will have to encompass both of the pathways of the PROTAC, given that, in the absence of the degradation capacity, the inhibition of the target or MDM2 by itself may still have anticancer action [42].
5. Disadvantages of MDM2-Based PROTACs
For a PROTAC to be able to exert its action, it must go inside the cell, and this can be a limiting step given that they are usually large molecules [41]. As seen above, the strategy of reducing the size of the ligands or adding peptides that promote membrane permeability (poly-arginine tail) can help to overcome these difficulties [41]. Furthermore, PROTACs, when in high concentrations, can develop the hook effect [42]. However, if this presents positive cooperativity, then this effect can be attenuated, favoring the formation of the ternary complex and reducing the required concentrations [44]. From a pharmacokinetic viewpoint, PROTACs are usually given by parenteral administration [37]. Although not following Lipinski’s Ro5, given that MDM2-based PROTACs have the highest molecular weight and most lipophilic type of PROTACs, with 3 aromatic rings in the MDM2 ligand, their oral administration will be a challenge, but, currently, there are examples of PROTACs that may have good bioavailability when administered orally, and MDM2-based PROTACs may become good candidates for oral anticancer therapies [45]. In addition to absorption issues, its metabolism is also an aspect to be taken into account since, in vivo, they will give rise to metabolites, which, even if they are not able to degrade the target, may perhaps inhibit POI or MDM2 [45]. Furthermore, the fact that these PROTACs have a high molecular weight may hinder their ability to enter the interior of liver cells and, thus, be metabolized; in this sense, studies to assess this ability and predict its impact are necessary [46]. Another relevant issue is related to the fact that genotoxic stress promotes the expression of MDM2 isoforms that lack the full N-terminal p53 binding domain and varying extensions of the central acid domain, resulting from alternative splicing, with the consequential loss of its ubiquitinating activity [47]. However, as far as we know, there are no studies that allow us to understand the impact that these isoforms may have on the performance of MDM2-based PROTACs [47]. Therefore, MDM2-based PROTACs offer a full set of aspects that can be improved, making it necessary to conduct more studies to clarify, create, and improve these promising molecules in therapy.6. Conclusions
The MDM2-based PROTACs, although still under-represented within the universe of PROTACs, have unique and very advantageous anti-tumor characteristics, given that, in addition to degrading proteins, such as the AR [6], BCR4 [42], PARP1 [48], or their own MDM2 [40], they have the ability to activate the p53 tumor suppressor pathway. This leads to the consequent activation of apoptosis processes in tumor cells, leading to their cell death, presenting, in the end, a synergistic effect—an effect much superior to the use of isolated inhibitors [42]. Therefore, with a dual mechanism of action, which requires the formation of a stable ternary complex, MDM2-based PROTACs, with some refinement, may well become candidates for the future anticancer therapeutic arsenal, as they have already proven in certain cases to be able to be effective in vitro and in vivo at concentrations in the nanomolar order, given their catalytic and synergistic mode of action, and with characteristics that overcome the current disadvantages of conventional therapies, highlighting the lack of selectivity for the target and cell type, the development of resistance, tolerance, accumulation of the target protein, and processes of up-regulation of the target [40][42][43][43]. Furthermore, the great advantage of PROTACs, and of those that recruit MDM2, lies in the possibility of degrading previously “undruggable proteins”, that is, they allow for the degradation of targets which do not have an active site, thus being able, with these new molecules, to interfere with new signaling pathways crucial for the development and proliferation of cancer cells, something that has never been explored before [7]. Therefore, the MDM2-based PROTACs need more studies to be improved, present new characteristics, as well as overcoming their current limitations. However, everything points to these molecules having an incredible potential to become an innovative therapeutic strategy for cancer treatment.References
- Mariell Pettersson; Craig M. Crews; PROteolysis TArgeting Chimeras (PROTACs) — Past, present and future. Drug Discovery Today: Technologies 2019, 31, 15-27, 10.1016/j.ddtec.2019.01.002.
- Kathleen M. Sakamoto; Kyung B. Kim; Akiko Kumagai; Frank Mercurio; Craig M. Crews; Raymond J. Deshaies; Protacs: Chimeric molecules that target proteins to the Skp1–Cullin–F box complex for ubiquitination and degradation. Proceedings of the National Academy of Sciences 2001, 98, 8554-8559, 10.1073/pnas.141230798.
- Yutian Zou; Danhui Ma; Yinyin Wang; The PROTAC technology in drug development. Cell Biochemistry and Function 2019, 37, 21-30, 10.1002/cbf.3369.
- Markella Konstantinidou; Jingyao Li; Bidong Zhang; Zefeng Wang; Shabnam Shaabani; Frans Ter Brake; Khaled Essa; Alexander Dömling; PROTACs– a game-changing technology. Expert Opinion on Drug Discovery 2019, 14, 1255-1268, 10.1080/17460441.2019.1659242.
- Ke Li; Craig M. Crews; PROTACs: past, present and future. Chemical Society Reviews 2022, 51, 5214-5236, 10.1039/d2cs00193d.
- Ashley R. Schneekloth; Mathieu Pucheault; Hyun Seop Tae; Craig M. Crews; Targeted intracellular protein degradation induced by a small molecule: En route to chemical proteomics. Bioorganic & Medicinal Chemistry Letters 2008, 18, 5904-5908, 10.1016/j.bmcl.2008.07.114.
- Brandon Dale; Meng Cheng; Kwang-Su Park; H. Ümit Kaniskan; Yue Xiong; Jian Jin; Advancing targeted protein degradation for cancer therapy. Nature Cancer 2021, 21, 638-654, 10.1038/s41568-021-00365-x.
- Gary Kleiger; Thibault Mayor; Perilous journey: a tour of the ubiquitin–proteasome system. Trends in Cell Biology 2014, 24, 352-359, 10.1016/j.tcb.2013.12.003.
- Markella Konstantinidou; Jingyao Li; Bidong Zhang; Zefeng Wang; Shabnam Shaabani; Frans Ter Brake; Khaled Essa; Alexander Dömling; PROTACs– a game-changing technology. Expert Opinion on Drug Discovery 2019, 14, 1255-1268, 10.1080/17460441.2019.1659242.
- Deepa Nath; Sadaf Shadan; The ubiquitin system. Nature Cell Biology 2009, 458, 421-421, 10.1038/458421a.
- Mohammed A. Mansour; Ubiquitination: Friend and foe in cancer. The International Journal of Biochemistry & Cell Biology 2018, 101, 80-93, 10.1016/j.biocel.2018.06.001.
- Markella Konstantinidou; Jingyao Li; Bidong Zhang; Zefeng Wang; Shabnam Shaabani; Frans Ter Brake; Khaled Essa; Alexander Dömling; PROTACs– a game-changing technology. Expert Opinion on Drug Discovery 2019, 14, 1255-1268, 10.1080/17460441.2019.1659242.
- Zhenyi Hu; Craig M. Crews; Recent Developments in PROTAC‐Mediated Protein Degradation: From Bench to Clinic. ChemBioChem 2021, 23, 1-23, 10.1002/cbic.202100270.
- Chaoguo Cao; Ming He; Liguo Wang; Yuna He; Yu Rao; Chemistries of bifunctional PROTAC degraders. Chemical Society Reviews 2022, 51, 7066–7114, 10.1039/d2cs00220e.
- Markella Konstantinidou; Jingyao Li; Bidong Zhang; Zefeng Wang; Shabnam Shaabani; Frans Ter Brake; Khaled Essa; Alexander Dömling; PROTACs– a game-changing technology. Expert Opinion on Drug Discovery 2019, 14, 1255-1268, 10.1080/17460441.2019.1659242.
- Jing Liu; Jia Ma; Yi Liu; Jun Xia; Yuyun Li; Z. Peter Wang; Wenyi Wei; PROTACs: A novel strategy for cancer therapy. Seminars in Cancer Biology 2020, 67, 171-179, 10.1016/j.semcancer.2020.02.006.
- Xin Zhou; Ru Dong; Jin-Yang Zhang; Xin Zheng; Li-Ping Sun; PROTAC: A promising technology for cancer treatment. European Journal of Medicinal Chemistry 2020, 203, 112539, 10.1016/j.ejmech.2020.112539.
- Kathleen M. Sakamoto; Kyung B. Kim; Akiko Kumagai; Frank Mercurio; Craig M. Crews; Raymond J. Deshaies; Protacs: Chimeric molecules that target proteins to the Skp1–Cullin–F box complex for ubiquitination and degradation. Proceedings of the National Academy of Sciences 2001, 98, 8554-8559, 10.1073/pnas.141230798.
- Kathleen M. Sakamoto; Kyung B. Kim; Rati Verma; Andy Ransick; Bernd Stein; Craig M. Crews; Raymond Deshaies; Development of Protacs to Target Cancer-promoting Proteins for Ubiquitination and Degradation. Molecular & Cellular Proteomics 2003, 2, 1350-1358, 10.1074/mcp.t300009-mcp200.
- John S. Schneekloth Jr.; Fabiana N. Fonseca; Michael Koldobskiy; Amit Mandal; Raymond Deshaies; Kathleen Sakamoto; Craig M. Crews; Chemical Genetic Control of Protein Levels: Selective in Vivo Targeted Degradation. Journal of the American Chemical Society 2004, 126, 3748-3754, 10.1021/ja039025z.
- A A Rodriguez-Gonzalez; K Cyrus; M Salcius; K Kim; C M Crews; R J Deshaies; K M Sakamoto; Targeting steroid hormone receptors for ubiquitination and degradation in breast and prostate cancer. Oncogene 2008, 27, 7201-7211, 10.1038/onc.2008.320.
- Yukihiro Itoh; Minoru Ishikawa; Mikihiko Naito; Yuichi Hashimoto; Protein Knockdown Using Methyl Bestatin−Ligand Hybrid Molecules: Design and Synthesis of Inducers of Ubiquitination-Mediated Degradation of Cellular Retinoic Acid-Binding Proteins. Journal of the American Chemical Society 2010, 132, 5820-5826, 10.1021/ja100691p.
- Georg E. Winter; Dennis L. Buckley; Joshiawa Paulk; Justin M. Roberts; Amanda Souza; Sirano Dhe-Paganon; James E. Bradner; Phthalimide conjugation as a strategy for in vivo target protein degradation. Science 2015, 348, 1376-1381, 10.1126/science.aab1433.
- Dennis L. Buckley; Jeffrey L. Gustafson; Inge Van Molle; Anke G. Roth; Hyun Seop Tae; Peter C. Gareiss; William L. Jorgensen; Alessio Ciulli; Craig M. Crews; Small-Molecule Inhibitors of the Interaction between the E3 Ligase VHL and HIF1α. Angewandte Chemie International Edition 2012, 51, 11463-11467, 10.1002/anie.201206231.
- Zhenyi Hu; Craig M. Crews; Recent Developments in PROTAC‐Mediated Protein Degradation: From Bench to Clinic. ChemBioChem 2021, 23, 1–23, 10.1002/cbic.202100270.
- John Hines; Jonathan D. Gough; Timothy W. Corson; Craig M. Crews; Posttranslational protein knockdown coupled to receptor tyrosine kinase activation with phosphoPROTACs. Proceedings of the National Academy of Sciences 2013, 110, 8942-8947, 10.1073/pnas.1217206110.
- Jing Liu; He Chen; Leina Ma; Zhixiang He; Dong Wang; Yi Liu; Qian Lin; Tinghu Zhang; Nathanael Gray; H. Ümit Kaniskan; et al.Jian JinWenyi Wei Light-induced control of protein destruction by opto-PROTAC. Science Advances 2020, 6, eaay5154, 10.1126/sciadv.aay5154.
- Martin Reynders; Bryan S. Matsuura; Marleen Bérouti; Daniele Simoneschi; Antonio Marzio; Michele Pagano; Dirk Trauner; PHOTACs enable optical control of protein degradation. Science Advances 2020, 6, 0, 10.1126/sciadv.aay5064.
- Patrick Pfaff; Kusal Samarasinghe; Craig M. Crews; Erick M. Carreira; Reversible Spatiotemporal Control of Induced Protein Degradation by Bistable PhotoPROTACs. ACS Central Science 2019, 5, 1682-1690, 10.1021/acscentsci.9b00713.
- Honorine Lebraud; David J. Wright; Christopher N. Johnson; Tom D. Heightman; Protein Degradation by In-Cell Self-Assembly of Proteolysis Targeting Chimeras. ACS Central Science 2016, 2, 927-934, 10.1021/acscentsci.6b00280.
- Marı́a Maneiro; Nafsika Forte; Maria M. Shchepinova; Cyrille S. Kounde; Vijay Chudasama; James Richard Baker; Edward W. Tate; Antibody–PROTAC Conjugates Enable HER2-Dependent Targeted Protein Degradation of BRD4. ACS Chemical Biology 2020, 15, 1306-1312, 10.1021/acschembio.0c00285.
- Kathleen M. Sakamoto; Kyung B. Kim; Akiko Kumagai; Frank Mercurio; Craig M. Crews; Raymond J. Deshaies; Protacs: Chimeric molecules that target proteins to the Skp1–Cullin–F box complex for ubiquitination and degradation. Proceedings of the National Academy of Sciences 2001, 98, 8554-8559, 10.1073/pnas.141230798.
- Scott D. Edmondson; Bin Yang; Charlene Fallan; Proteolysis targeting chimeras (PROTACs) in ‘beyond rule-of-five’ chemical space: Recent progress and future challenges. Bioorganic & Medicinal Chemistry Letters 2019, 29, 1555-1564, 10.1016/j.bmcl.2019.04.030.
- Abiodun Anifowose; Ayodeji A. Agbowuro; Xiaoxiao Yang; Binghe Wang; Anticancer strategies by upregulating p53 through inhibition of its ubiquitination by MDM2. Medicinal Chemistry Research 2020, 29, 1105-1121, 10.1007/s00044-020-02574-9.
- Shusuke Tomoshige; Minoru Ishikawa; In vivo synthetic chemistry of proteolysis targeting chimeras (PROTACs). Bioorganic & Medicinal Chemistry 2021, 41, 116221, 10.1016/j.bmc.2021.116221.
- Cristina Nieto-Jiménez; Esther Cabañas Morafraile; Carlos Alonso-Moreno; Alberto Ocaña; Clinical considerations for the design of PROTACs in cancer. Molecular Cancer 2022, 21, 1-9, 10.1186/s12943-022-01535-7.
- Sainan An; Liwu Fu; Small-molecule PROTACs: An emerging and promising approach for the development of targeted therapy drugs. eBioMedicine 2018, 36, 553-562, 10.1016/j.ebiom.2018.09.005.
- Gary Kleiger; Thibault Mayor; Perilous journey: a tour of the ubiquitin–proteasome system. Trends in Cell Biology 2014, 24, 352-359, 10.1016/j.tcb.2013.12.003.
- Markella Konstantinidou; Jingyao Li; Bidong Zhang; Zefeng Wang; Shabnam Shaabani; Frans Ter Brake; Khaled Essa; Alexander Dömling; PROTACs– a game-changing technology. Expert Opinion on Drug Discovery 2019, 14, 1255-1268, 10.1080/17460441.2019.1659242.
- Shipeng He; Junhui Ma; Yuxin Fang; Ying Liu; Shanchao Wu; Guoqiang Dong; Wei Wang; Chunquan Sheng; Homo-PROTAC mediated suicide of MDM2 to treat non-small cell lung cancer. Acta Pharmaceutica Sinica B 2020, 11, 1617-1628, 10.1016/j.apsb.2020.11.022.
- Xinyi Li; Wenchen Pu; Qingquan Zheng; Min Ai; Song Chen; Yong Peng; Proteolysis-targeting chimeras (PROTACs) in cancer therapy. Molecular Cancer 2022, 21, 1-30, 10.1186/s12943-021-01434-3.
- John Hines; Schan Lartigue; Hanqing Dong; Yimin Qian; Craig M. Crews; MDM2-Recruiting PROTAC Offers Superior, Synergistic Antiproliferative Activity via Simultaneous Degradation of BRD4 and Stabilization of p53. Cancer Research 2019, 79, 251-262, 10.1158/0008-5472.can-18-2918.
- Bohan Ma; Fan Niu; Xiaoyan Qu; Wangxiao He; Chao Feng; Simeng Wang; Zhenlin Ouyang; Jin Yan; Yurong Wen; Dan Xu; et al.Yongping ShaoPeter X. MaWuyuan Lu A tetrameric protein scaffold as a nano-carrier of antitumor peptides for cancer therapy. Biomaterials 2019, 204, 1-12, 10.1016/j.biomaterials.2019.03.004.
- Markella Konstantinidou; Jingyao Li; Bidong Zhang; Zefeng Wang; Shabnam Shaabani; Frans Ter Brake; Khaled Essa; Alexander Dömling; PROTACs– a game-changing technology. Expert Opinion on Drug Discovery 2019, 14, 1255-1268, 10.1080/17460441.2019.1659242.
- Scott D. Edmondson; Bin Yang; Charlene Fallan; Proteolysis targeting chimeras (PROTACs) in ‘beyond rule-of-five’ chemical space: Recent progress and future challenges. Bioorganic & Medicinal Chemistry Letters 2019, 29, 1555-1564, 10.1016/j.bmcl.2019.04.030.
- Yichao Wan; Chunxing Yan; Han Gao; Tingting Liu; Small-molecule PROTACs: novel agents for cancer therapy. Future Medicinal Chemistry 2020, 12, 915-938, 10.4155/fmc-2019-0340.
- Orit Karni-Schmidt; Maria Lokshin; Carol Prives; The Roles of MDM2 and MDMX in Cancer. Annual Review of Pathology: Mechanisms of Disease 2016, 11, 617-644, 10.1146/annurev-pathol-012414-040349.
- Qiuye Zhao; Tianlong Lan; Shang Su; Yu Rao; Induction of apoptosis in MDA-MB-231 breast cancer cells by a PARP1-targeting PROTAC small molecule. Chemical Communications 2018, 55, 369-372, 10.1039/c8cc07813k.
