Precision medicine requires highly sensitive and specific diagnostic strategies with high spatiotemporal resolution. Accurate detection and monitoring of endogenously generated biomarkers at the very early disease stage is of extensive importance for precise diagnosis and treatment. Aggregation-induced emission luminogens (AIEgens) have emerged as a new type of excellent optical agents, which show great promise for numerous biomedical applications. In this review, we highlight the recent aAdvances of AIE-based probes for detecting reactive species (including reactive oxygen species (ROS), reactive nitrogen species (RNS), reactive sulfur species (RSS), and reactive carbonyl species (RCS)) and related biomedical applications are introduced. The molecular design strategies for increasing the sensitivity, tuning the response wavelength, and realizing afterglow imaging are summarized, and theranostic applications in reactive species-related major diseases such as cancer, inflammation, and vascular diseases are reviewed.
- aggregation-induced emission
- reactive oxygen nitrogen species
- activatable probe
- theranostics
- fluorescence
- photoacoustic
- afterglow
- bioimaging
1. Introduction
2. Detection of Reactive Oxygen Nitrogen Species
When designing a specific chemical/biological probe, a usually requisite is to synthesize molecules with specific recognition groups or moieties.The boronate subunit is a popularly used building block for H2O2 sensors, as the boronate cage is nonfluorescent and the conversion of arylboronates to phenols results in turn-on emission [95,96,97][95][96][97]. The deprotonated H2O2 is a potent nucleophile, which can attack the boron center to generate a labile borate species that hydrolyses to the corresponding phenol [98]. For O2•− detection, the diphenyl phosphinyl group can be introduced into an organic compound, in which the fluorescence is strongly quenched at first, and obvious turn-on fluorescent signal is realized in the presence of O2•− [99,100][99][100]. The oxidative properties of ClO− can be utilized to destroy C=C or C=N bonds rapidly, therefor, the conjugation of fluorescence quencher through C=C or C=N bonds has turned out to be an efficient strategy to construct ClO− probes [101,102][101][102]. Some arylboronate groups, diphenylphosphinate groups, and nitrophenyloxoacetamide moieties have been employed as the response substitutes for ONOO− detection [103,104,105][103][104][105]. The tunability of molecular structure will alter the photophysical properties and biomedical applications as well. H2O2 is an overexpressed molecule in many serious diseases, and thus, it is regarded as a pivotal biomarker for some biological processes and disease diagnoses [106,107,108][106][107][108]. A variety of H2O2-activatable probes have been exploited based on AIEgens, which exhibit excellent performance for both in vitro and in vivo applications [109,110,111,112][109][110][111][112]. Xia and Lou et al. developed a H2O2-responsive AIEgen for peroxidase-mediated selective imaging and inhibition of inflammatory cells [113].The probe consisted of a TPE core and two tyrosine (Tyr) moieties, which could undergo enzyme-catalyzed dityrosine formation in the presence of peroxidase and H2O2. By conjugating two hydrophilic Tyr groups, the hydrophobic TPE molecule became hydrophilic TT, which showed weak fluorescence in aqueous solution due to the excited-state energy consumption via intense molecular motion. As a result, the H2O2-responsive and myeloperoxidase (MPO)-mediated TT self-assembly enabled turn-on fluorescence, which could be used for selectively imaging and inhibiting inflammatory cells containing overexpressed H2O2 and MPO. The AIE process could be activated through dityrosine linkage-induced hydrophobic aggregates formation, which helped to distinguish between inflammatory and normal cells. Additionally, the in situ formation of TT aggregates could inhibit RAW264.7 cell growth through inducing mitochondria damage and cell apoptosis. Wang and Li et al. reported a ROS-responsive theranostic nanoplatform for accurate diagnosis and therapy of inflammation diseases [114]. A two-photon AIEgen (TP) was conjugated with the widely used anti-inflammatory glucocorticoid, prednisolone (Pred) with the ROS-sensitive linkage to afford the compound TPP. Then, the TPP was encapsulated with an amphiphilic block copolymer PMPC−PMEMA (PMM) to give polymeric micelles (TPP@PMM). Noteworthy, the PMEMA part served as the hydrophobic block in the NPs formation, which could be oxidized in response to ROS to yield the hydrophilic sulphone product. The ROS-triggered hydrophobic-to-hydrophilic conversion was able to realize ROS-mediated drug delivery at an inflammatory site. This shell-core dual ROS-responsive nanoplatform was used in three different inflammatory murine models including acute lung injury, atherosclerosis, and arthritis. The deep-penetration two-photon fluorescence diagnosis and efficient serial ROS sensitive anti-inflammation could be used for both acute and chronic inflammation theranostics. Two-photon imaging with the AIEgen helped to provide unambiguous delineation of inflammatory tissue with minimum autofluorescence interference. Moreover, TPP@PMM also possessed excellent anti-inflammatory effect that reduced the inflammatory response and decreased inflammatory cytokines expression.3. Detection of Gasotransmitters
Small gaseous molecules including NO, CO, and H2S, function as important signal transmitters in living systems as they are associated with many biological functions and major diseases [148,149,150,151][115][116][117][118]. NO is a neutral diatomic free radical that is produced from L-arginine by NO synthase (NOSs) isoforms such as neuronal NOS (nNOS), inducible NOS (iNOS), and endothelial NOS (eNOS) [152,153][119][120]. CO is the second gasotransmitter that is generated as a byproduct of haem cleavage by two distinct haem oxygenases [154][121]. H2S is predominantly formed from Cys or its derivatives by the enzymes cystathionine β-synthase and cystathionine γ-lyase [155][122]. All these gasotransmitters play vital roles in vasorelaxation and inflammatory responses, thus, numerous molecular probes have been developed for precise monitoring of related diseases [156,157,158][123][124][125]. For example, the o-diamino aromatic moiety is a recognition group for NO, and the cyclization reaction of o-diamine with NO produces a triazole moiety, which alters the electronic property and conjugation nature [159,160,161][126][127][128]. For H2S detection, the popularly used approaches include reduction of azides into amines and nucleophilic addition of H2S to the electrophilic group [162,163][129][130]. Some representative AIEgens for sensing gasotransmitters are listed Figure 13, which show great potential for applications in biological imaging and disease diagnosis.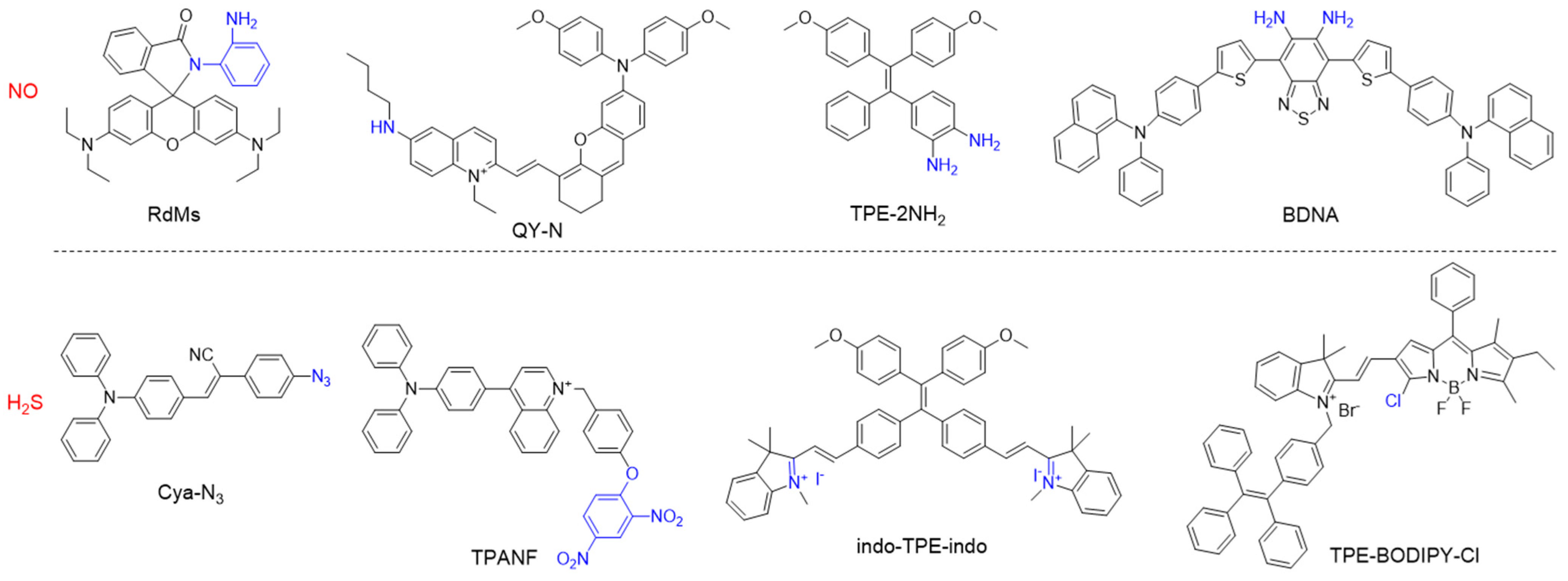
References
- Park, S.M.; Aalipour, A.; Vermesh, O.; Yu, J.H.; Gambhir, S.S. Towards clinically translatable in vivo nanodiagnostics. Nat. Rev. Mater. 2017, 2, 17014.
- Letai, A. Functional precision cancer medicine–moving beyond pure genomics. Nat. Med. 2016, 23, 1028–1035.
- Ashley, E.A. Towards precision medicine. Nat. Rev. Genet. 2016, 17, 507–522.
- Weissleder, R. Scaling down imaging: Molecular mapping of cancer in mice. Nat. Rev. Cancer 2012, 2, 11–18.
- Willmann, J.K.; Bruggen, N.V.; Dinkelborg, L.M.; Gambhir, S.S. Molecular imaging in drug development. Nat. Rev. Drug Discov. 2008, 7, 591–607.
- Nicolson, F.; Kircher, M.F.; Stone, N.; Matousek, P. Spatially offset Raman spectroscopy for biomedical applications. Chem. Soc. Rev. 2021, 50, 556–568.
- Naumova, A.V.; Modo, M.; Moore, A.; Murry, C.E.; Frank, J.A. Clinical imaging in regenerative medicine. Nat. Biotechnol. 2014, 32, 804–818.
- Mustafa, D.A.; Al-Shimmari, H.A.T.; Radhi, M.M. Use of MgCl2 Nanoparticles as Alternative Contrast Media in Magnatic Resonance Imaging Molecular Imaging and Analyzed by Voltammetric Technique. Nano Biomed. Eng. 2020, 12, 148–152.
- Wang, L.V.; Yao, J. A practical guide to photoacoustic tomography in the life sciences. Nat. Methods 2016, 13, 627–638.
- Gottschalk, S.; Degtyaruk, O.; Mc Larney, B.; Rebling, J.; Hutter, M.A.; Deán-Ben, X.L.; Shoham, S.; Razansky, D. Rapid volumetric optoacoustic imaging of neural dynamics across the mouse brain. Nat. Biomed. Eng. 2019, 3, 392–401.
- Li, W.; Yan, Z.; Ren, J.; Qu, X. Manipulating cell fate: Dynamic control of cell behaviors on functional platforms. Chem. Soc. Rev. 2018, 47, 8639–8684.
- Ni, J.-S.; Min, T.; Li, Y.; Zha, M.; Zhang, P.; Ho, L.; Li, K. Planar AIEgens with enhanced solid-state luminescence and ROS generation for multidrug-resistant bacteria treatment. Angew. Chem. Int. Ed. 2020, 59, 10179–10185.
- Li, C.; Chen, G.; Zhang, Y.; Wu, F.; Wang, Q. Advanced Fluorescence Imaging Technology in the Near-Infrared-II Window for Biomedical Applications. J. Am. Chem. Soc. 2020, 142, 14789–14804.
- Zhang, Y.; Zhou, J.; Peng, S.; Yu, W.; Fan, X.; Liu, W.; Ye, Z.; Qi, J.; Feng, Z.; Qian, J. Hot-Band-Absorption-Induced Anti-Stokes Fluorescence of Aggregation-Induced Emission Dots and the Influence on the Nonlinear Optical Effect. Biosensors 2021, 11, 468.
- Liu, R.; Xu, Y.; Xu, K.; Dai, Z. Current trends and key considerations in the clinical translation of targeted fluorescent probes for intraoperative navigation. Aggregate 2021, 2, e23.
- Zhou, J.; del Rosal, B.; Jaque, D.; Uchiyama, S.; Jin, D. Advances and challenges for fluorescence nanothermometry. Nat. Methods 2020, 17, 967–980.
- Fan, Y.; Wang, P.; Lu, Y.; Wang, R.; Zhou, L.; Zheng, X.; Li, X.; Piper, J.A.; Zhang, F. Lifetime-engineered NIR-II nanoparticles unlock multiplexed in vivo imaging. Nat. Nanotechnol. 2018, 13, 941–946.
- Wu, Y.; Zeng, F.; Zhao, Y.; Wu, S. Emerging contrast agents for multispectral optoacoustic imaging and their biomedical applications. Chem. Soc. Rev. 2021, 50, 7924–7940.
- Ji, X.; Ge, L.; Liu, C.; Tang, Z.; Xiao, Y.; Chen, W.; Lei, Z.; Gao, W.; Blake, S.; De, D.; et al. Capturing functional two-dimensional nanosheets from sandwich-structure vermiculite for cancer theranostics. Nat. Commun. 2021, 12, 1124.
- Kang, M.; Zhang, Z.; Song, N.; Li, M.; Sun, P.; Chen, X.; Wang, D.; Tang, B.Z. Aggregation-enhanced theranostics: AIE sparkles in biomedical field. Aggregate 2020, 1, 80–106.
- Liu, Y.; Li, Y.; Koo, S.; Sun, Y.; Liu, Y.; Liu, X.; Pan, Y.; Zhang, Z.; Du, M.; Lu, S.; et al. Versatile types of inorganic/organic NIR-IIa/IIb fluorophores: From strategic design toward molecular imaging and theranostics. Chem. Rev. 2022, 122, 209–268.
- Qi, J.; Fang, Y.; Kwok, R.T.K.; Zhang, X.; Hu, X.; Lam, J.W.Y.; Ding, D.; Tang, B.Z. Highly Stable Organic Small Molecular Nanoparticles as an Advanced and Biocompatible Phototheranostic Agent of Tumor in Living Mice. ACS Nano 2017, 11, 7177–7188.
- Cai, Y.; Chen, X.; Si, J.; Mou, X.; Dong, X. All-in-One Nanomedicine: Multifunctional Single-Component Nanoparticles for Cancer Theranostics. Small 2021, 17, 2103072.
- Liu, Y.; Bhattarai, P.; Dai, Z.; Chen, X. Photothermal therapy and photoacoustic imaging via nanotheranostics in fighting cancer. Chem. Soc. Rev. 2019, 48, 2053–2108.
- Wu, L.; Huang, J.; Pu, K.; James, T.D. Dual-locked spectroscopic probes for sensing and therapy. Nat. Rev. Chem. 2021, 5, 406–421.
- Qi, J.; Feng, L.; Zhang, X.; Zhang, H.; Huang, L.; Zhou, Y.; Zhao, Z.; Duan, X.; Xu, F.; Kwok, R.T.K.; et al. Facilitation of molecular motion to develop turn-on photoacoustic bioprobe for detecting nitric oxide in encephalitis. Nat. Commun. 2021, 12, 960.
- Antaris, A.L.; Chen, H.; Cheng, K.; Sun, Y.; Hong, G.; Qu, C.; Diao, S.; Deng, Z.; Hu, X.; Zhang, B.; et al. A small-molecule dye for NIR-II imaging. Nat. Mater. 2016, 15, 235–242.
- Yang, J.; Zhang, Y.; Wu, X.; Dai, W.; Chen, D.; Shi, J.; Tong, B.; Peng, Q.; Xie, H.; Cai, Z.; et al. Rational design of pyrrole derivatives with aggregation-induced phosphorescence characteristics for time-resolved and two-photon luminescence imaging. Nat. Commun. 2021, 12, 4883.
- Ding, Z.; Gu, Y.; Zheng, C.; Gu, Y.; Yang, J.; Li, D.; Xu, Y.; Wang, P. Organic small molecule-based photothermal agents for cancer therapy: Design strategies from single-molecule optimization to synergistic enhancement. Coordin. Chem. Rev. 2022, 464, 214564.
- Ji, C.; Cheng, W.; Yuan, Q.; Müllen, K.; Yin, M. From Dyestuff Chemistry to Cancer Theranostics: The Rise of Rylenecarboximides. Acc. Chem. Res. 2019, 52, 2266–2277.
- Yang, J.; Fang, M.; Li, Z. Organic luminescent materials: The concentration on aggregates from aggregation-induced emission. Aggregate 2020, 1, 6–18.
- Luo, J.; Xie, Z.; Lam, J.W.Y.; Cheng, L.; Chen, H.; Qiu, C.; Kwok, H.S.; Zhan, X.; Liu, Y.; Zhu, D.; et al. Aggregation-induced emission of 1-methyl-1, 2, 3, 4, 5-pentaphenylsilole. Chem. Commun. 2001, 1740–1741.
- Chen, Y.; Lam, J.W.Y.; Kwok, R.T.K.; Liu, B.; Tang, B.Z. Aggregation-induced emission: Fundamental understanding and future developments. Mater. Horiz. 2019, 6, 428–433.
- Gao, M.; Tang, B.Z. AIE-based cancer theranostics. Coordin. Chem. Rev. 2020, 402, 213076.
- Zha, M.; Yang, G.; Li, Y.; Zhang, C.; Li, B.; Li, K. Recent Advances in AIEgen-Based Photodynamic Therapy and Immunotherapy. Adv. Healthc. Mater. 2021, 10, 2101066.
- Zang, T.; Xie, Y.; Su, S.; Liu, F.; Chen, Q.; Jing, J.; Zhang, R.; Niu, G.; Zhang, X. In Vitro Light-Up Visualization of a Subunit-Specific Enzyme by an AIE Probe via Restriction of Single Molecular Motion. Angew. Chem. Int. Ed. 2020, 59, 10003–10007.
- Zhao, Z.; Zhang, H.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission: New vistas at the aggregate level. Angew. Chem. Int. Ed. 2020, 59, 9888–9907.
- Hong, Y.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission. Chem. Soc. Rev. 2011, 40, 5361–5388.
- Mei, J.; Hong, Y.; Lam, J.W.Y.; Qin, A.; Tang, Y.; Tang, B.Z. Aggregation-Induced Emission: The Whole Is More Brilliant than the Parts. Adv. Mater. 2014, 26, 5429–5479.
- Feng, G.; Liu, B. Aggregation-Induced Emission (AIE) Dots: Emerging Theranostic Nanolights. Acc. Chem. Res. 2018, 51, 1404–1414.
- Wang, Z.; Zhou, Y.; Xu, R.; Xu, Y.; Dang, D.; Shen, Q.; Meng, L.; Tang, B.Z. Seeing the unseen: AIE luminogens for super-resolution imaging. Coordin. Chem. Rev. 2022, 451, 214279.
- Yang, G.; Ni, J.-S.; Li, Y.; Zha, M.; Tu, Y.; Li, K. Acceptor Engineering for Optimized ROS Generation Facilitates Reprogramming Macrophages to M1 Phenotype in Photodynamic Immunotherapy. Angew. Chem. Int. Ed. 2021, 60, 5386–5393.
- Qi, J.; Sun, C.; Li, D.; Zhang, H.; Yu, W.; Zebibula, A.; Lam, J.W.Y.; Xi, W.; Zhu, L.; Cai, F.; et al. Aggregation-Induced Emission Luminogen with Near-Infrared-II Excitation and Near-Infrared-I Emission for Ultradeep Intravital Two-Photon Microscopy. ACS Nano 2018, 12, 7936–7945.
- Li, Q.; Li, Y.; Min, T.; Gong, J.; Du, L.; Phillips, D.L.; Liu, J.; Lam, J.W.Y.; Sung, H.H.Y.; Williams, I.D.; et al. Time-Dependent Photodynamic Therapy for Multiple Targets: A Highly Efficient AIE-Active Photosensitizer for Selective Bacterial Elimination and Cancer Cell Ablation. Angew. Chem. Int. Ed. 2020, 59, 9470–9477.
- He, Z.; Gao, Y.; Zhang, H.; Xue, Y.; Meng, F.; Luo, L. Mitochondrion-Anchored Photosensitizer with Near Infrared-I Aggregation-Induced Emission for Near Infrared-II Two-Photon Photodynamic Therapy. Adv. Healthc. Mater. 2021, 10, 2101056.
- Naghibi, S.; Chen, T.; Ghahfarokhi, A.J.; Tang, Y. AIEgen-enhanced protein imaging: Probe design and sensing mechanisms. Aggregate 2021, 2, e41.
- Huang, J.; Nie, H.; Zeng, J.; Zhuang, Z.; Gan, S.; Cai, Y.; Guo, J.; Su, S.-J.; Zhao, Z.; Tang, B.Z. Highly Efficient Nondoped OLEDs with Negligible Efficiency Roll-Off Fabricated from Aggregation-Induced Delayed Fluorescence Luminogens. Angew. Chem. Int. Ed. 2017, 56, 12971–12976.
- Xu, Y.; Xu, R.; Wang, Z.; Zhou, Y.; Shen, Q.; Ji, W.; Dang, D.; Meng, L.; Tang, B.Z. Recent advances in luminescent materials for super-resolution imaging via stimulated emission depletion nanoscopy. Chem. Soc. Rev. 2021, 50, 667–690.
- Liu, C.; Wang, X.; Liu, J.; Yue, Q.; Chen, S.; Lam, J.W.Y.; Luo, L.; Tang, B.Z. Near-Infrared AIE Dots with Chemiluminescence for Deep-Tissue Imaging. Adv. Mater. 2020, 32, 2004685.
- Hu, Q.; Hu, H.; Zhang, X.; Fan, K.; Hong, Y.; Raston, C.L.; Tang, Y. In situ monitored vortex fluidic-mediated protein refolding/unfolding using an aggregation-induced emission bioprobe. Molecules 2021, 26, 4273.
- Cai, X.; Liu, B. Aggregation-Induced Emission: Recent Advances in Materials and Biomedical Applications. Angew. Chem. Int. Ed. 2020, 59, 9868–9886.
- Qi, J.; Ou, H.; Liu, Q.; Ding, D. Gathering brings strength: How organic aggregates boost disease phototheranostics. Aggregate 2021, 2, 95–113.
- Zhou, T.; Hu, R.; Wang, L.; Qiu, Y.; Zhang, G.; Deng, Q.; Zhang, H.; Yin, P.; Situ, B.; Zhan, C.; et al. An AIE-Active Conjugated Polymer with High ROS-Generation Ability and Biocompatibility for Efficient Photodynamic Therapy of Bacterial Infections. Angew. Chem. Int. Ed. 2020, 59, 9952–9956.
- Zhang, Z.; Fang, X.; Liu, Z.; Liu, H.; Chen, D.; He, S.; Zheng, J.; Yang, B.; Qin, W.; Zhang, X.; et al. Semiconducting Polymer Dots with Dual-Enhanced NIR-IIa Fluorescence for Through-Skull Mouse-Brain Imaging. Angew. Chem. Int. Ed. 2020, 59, 3691–3698.
- Liu, M.; Chen, Y.; Guo, Y.; Yuan, H.; Cui, T.; Yao, S.; Jin, S.; Fan, H.; Wang, C.; Xie, R.; et al. Golgi apparatus-targeted aggregation-induced emission luminogens for effective cancer photodynamic therapy. Nat. Commun. 2022, 13, 2179.
- Valko, M.; Rhodes, C.J.; Moncol, J.; Izakovic, M.; Mazur, M. Free radicals, metals and antioxidants in oxidative stress-induced cancer. Chem.-Biol. Interact. 2006, 160, 1–40.
- Schieber, M.; Chandel, N.S. ROS function in redox signaling and oxidative stress. Curr. Biol. 2014, 24, R453–R462.
- Rani, V.; Deep, G.; Singh, R.K.; Palle, K.; Yadav, U.C.S. Oxidative stress and metabolic disorders: Pathogenesis and therapeutic strategies. Life Sci. 2016, 148, 183–193.
- Wang, Z.; Zhang, Y.; Ju, E.; Liu, Z.; Cao, F.; Chen, Z.; Ren, J.; Qu, X. Biomimetic nanoflowers by self-assembly of nanozymes to induce intracellular oxidative damage against hypoxic tumors. Nat. Commun. 2018, 9, 3334.
- Tang, D.; Wang, Y.; Wijaya, A.; Liu, B.; Maruf, A.; Wang, J.; Xu, J.; Liao, X.; Wu, W.; Wang, G. ROS-responsive biomimetic nanoparticles for potential application in targeted anti-atherosclerosis. Regen. Biomater. 2021, 8, rbab033.
- Zhang, Y.; Yang, H.; Wei, D.; Zhang, X.; Wang, J.; Wu, X.; Chang, J. Mitochondria-targeted nanoparticles in treatment of neurodegenerative diseases. Exploration 2021, 1, 20210115.
- Duanghathaipornsuk, S.; Farrell, E.J.; Alba-Rubio, A.C.; Zelenay, P.; Kim, D.-S. Detection Technologies for Reactive Oxygen Species: Fluorescence and Electrochemical Methods and Their Applications. Biosensors 2021, 11, 30.
- Nguyen, K.T.; Zhao, Y. Engineered Hybrid Nanoparticles for On-Demand Diagnostics and Therapeutics. Acc. Chem. Res. 2015, 48, 3016–3025.
- Wang, C.; Ding, S.; Wang, S.; Shi, Z.; Pandey, N.K.; Chudal, L.; Wang, L.; Zhang, Z.; Wen, Y.; Yao, H.; et al. Endogenous tumor microenvironment-responsive multifunctional nanoplatforms for precision cancer theranostics. Coordin. Chem. Rev. 2021, 426, 213529.
- Barani, M.; Mukhtar, M.; Rahdar, A.; Sargaz, S.; Pandey, S.; Kang, M. Recent Advances in Nanotechnology-Based Diagnosis and Treatments of Human Osteosarcoma. Biosensors 2021, 11, 55.
- Paul, B.D.; Snyder, S.H. H2S signalling through protein sulfhydration and beyond. Nat. Rev. Mol. Cell Bio. 2012, 13, 499–507.
- Jiang, Y.; Pu, K. Molecular Probes for Autofluorescence-Free Optical Imaging. Chem. Rev. 2021, 121, 13086–13131.
- Yang, X.; Zhang, D.; Ye, Y.; Zhao, Y. Recent advances in multifunctional fluorescent probes for viscosity and analytes. Coordin. Chem. Rev. 2022, 453, 214336.
- Jiao, X.; Li, Y.; Niu, J.; Xie, X.; Wang, X.; Tang, B. Small-Molecule Fluorescent Probes for Imaging and Detection of Reactive Oxygen, Nitrogen, and Sulfur Species in Biological Systems. Anal. Chem. 2018, 90, 533–555.
- Zhan, Z.; Dai, Y.; Li, Q.; Lv, Y. Small molecule-based bioluminescence and chemiluminescence probes for sensing and imaging of reactive species. TrAC Trend. Anal. Chem. 2021, 134, 116129.
- Li, J.; Wang, T.; Jiang, F.; Hong, Z.; Su, X.; Li, S.; Han, S. Activatable Dual ROS-Producing Probe for Dual Organelle-Engaged Photodynamic Therapy. ACS Appl. Bio Mater. 2021, 4, 4618–4628.
- Hu, J.-J.; Jiang, W.; Yuan, L.; Duan, C.; Yuan, Q.; Long, Z.; Lou, X.; Xia, F. Recent advances in stimuli-responsive theranostic systems with aggregation-induced emission characteristics. Aggregate 2021, 2, 48–65.
- Tang, G.; He, J.; Liu, J.; Yan, X.; Fan, K. Nanozyme for tumor therapy: Surface modification matters. Exploration 2021, 1, 75–89.
- Reza, A.H.M.M.; Zhu, X.; Qin, J.; Tang, Y. Microalgae-Derived Health Supplements to Therapeutic Shifts: Redox-Based Study Opportunities with AIE-Based Technologies. Adv. Healthc. Mater. 2021, 10, 2101223.
- Mujika, J.I.; Uranga, J.; Matxain, J.M. Computational Study on the Attack of •OH Radicals on Aromatic Amino Acids. Chem.-Eur. J. 2013, 19, 6862–6873.
- Vicente-Gutierrez, C.; Bonora, N.; Bobo-Jimenez, V.; Jimenez-Blasco, D.; Lopez-Fabuel, I.; Fernandez, E.; Josephine, C.; Bonvento, G.; Enriquez, J.A.; Almeida, A.; et al. Astrocytic mitochondrial ROS modulate brain metabolism and mouse behaviour. Nat. Metab. 2019, 1, 201–211.
- Vaccaro, A.; Dor, Y.K.; Nambara, K.; Pollina, E.A.; Lin, C.; Greenberg, M.E.; Rogulj, D. Sleep Loss Can Cause Death through Accumulation of Reactive Oxygen Species in the Gut. Cell 2020, 181, 1307–1328.
- Yardeni, T.; Tanes, C.E.; Bittinger, K.; Mattei, L.M.; Schaefer, P.M.; Singh, L.N.; Wu, G.D.; Murdock, D.G.; Wallace, D.C. Host mitochondria influence gut microbiome diversity: A role for ROS. Sci. Signal. 2019, 12, eaaw3159.
- Cheng, D.; Xu, W.; Gong, X.; Yuan, L.; Zhang, X.-B. Design Strategy of Fluorescent Probes for Live Drug-Induced Acute Liver Injury Imaging. Acc. Chem. Res. 2021, 54, 403–415.
- Li, C.; Li, S.; Zhao, J.; Sun, M.; Wang, W.; Lu, M.; Qu, A.; Hao, C.; Chen, C.; Xu, C.; et al. Ultrasmall Magneto-chiral Cobalt Hydroxide Nanoparticles Enable Dynamic Detection of Reactive Oxygen Species in Vivo. J. Am. Chem. Soc. 2022, 144, 1580–1588.
- Zhang, X.; Chen, Y.; He, H.; Wang, S.; Lei, Z.; Zhang, F. ROS/RNS and Base Dual Activatable Merocyanine-Based NIR-II Fluorescent Molecular Probe for in vivo Biosensing. Angew. Chem. Int. Ed. 2021, 60, 26337–26341.
- Li, J.; Rao, J.; Pu, K. Recent progress on semiconducting polymer nanoparticles for molecular imaging and cancer phototherapy. Biomaterials 2018, 155, 217–235.
- Wang, H.; Wang, X.; Li, P.; Dong, M.; Yao, S.Q.; Tang, B. Fluorescent probes for visualizing ROS-associated proteins in disease. Chem. Sci. 2021, 12, 11620–11646.
- Ong, S.Y.; Zhang, C.; Dong, X.; Yao, S.Q. Recent advances in polymeric nanoparticles for enhanced fluorescence and photoacoustic imaging. Angew. Chem. Int. Ed. 2021, 60, 17797–17809.
- Wang, D.; Tang, B.Z. Aggregation-induced emission luminogens for activity-based sensing. Acc. Chem. Res. 2019, 52, 2559–2570.
- Niu, G.; Zhang, R.; Shi, X.; Park, H.; Xie, S.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. AIE luminogens as fluorescent bioprobes. TrAC Trend. Anal. Chem. 2020, 123, 115769.
- Li, H.; Kim, H.; Han, J.; Nguyen, V.-N.; Peng, X.; Yoon, J. Activity-based smart AIEgens for detection, bioimaging, and therapeutics: Recent progress and outlook. Aggregate 2021, 2, e51.
- Ouyang, J.; Sun, L.; Zeng, F.; Wu, S. Biomarker-activatable probes based on smart AIEgens for fluorescence and optoacoustic imaging. Coordin. Chem. Rev. 2022, 458, 214438.
- Mei, J.; Leung, N.L.C.; Kwok, R.T.K.; Lam, J.W.Y.; Tang, B.Z. Aggregation-induced emission: Together we shine, united we soar! Chem. Rev. 2015, 115, 11718–11940.
- Ding, D.; Li, K.; Liu, B.; Tang, B.Z. Bioprobes based on AIE fluorogens. Acc. Chem. Res. 2013, 46, 2441–2453.
- Qian, J.; Tang, B.Z. AIE luminogens for bioimaging and theranostics: From organelles to animals. Chem 2017, 3, 56–91.
- Qi, J.; Chen, C.; Ding, D.; Tang, B.Z. Aggregation-Induced Emission Luminogens: Union Is Strength, Gathering Illuminates Healthcare. Adv. Healthc. Mater. 2018, 7, 1800477.
- Zhang, H.; Zhao, Z.; Turley, A.T.; Wang, L.; McGonigal, P.R.; Tu, Y.; Li, Y.; Wang, Z.; Kwok, R.T.K.; Lam, J.W.Y.; et al. Aggregate science: From structures to properties. Adv. Mater. 2020, 32, 2001457.
- Li, J.; Wang, J.; Li, H.; Song, N.; Wang, D.; Tang, B.Z. Supramolecular materials based on AIE luminogens (AIEgens): Construction and applications. Chem. Soc. Rev. 2020, 49, 1144–1172.
- Chan, J.; Dodani, S.C.; Chang, C.J. Reaction-based small-molecule fluorescent probes for chemoselective bioimaging. Nat. Chem. 2012, 4, 973–984.
- Nguyen, V.-N.; Ha, J.; Cho, M.; Li, H.; Swamy, K.M.K.; Yoon, J. Recent developments of BODIPY-based colorimetric and fluorescent probes for the detection of reactive oxygen/nitrogen species and cancer diagnosis. Coordin. Chem. Rev. 2021, 439, 213936.
- Jiang, G.; Li, C.; Liu, X.; Chen, Q.; Li, X.; Gu, X.; Zhang, P.; Lai, Q.; Wang, J. Lipid droplet-targetable fluorescence guided photodynamic therapy of cancer cells with an activatable AIE-active fluorescent probe for hydrogen peroxide. Adv. Optical Mater. 2020, 8, 2001119.
- Miller, E.W.; Tulyathan, O.; Isacoff, E.Y.; Chang, C.J. Molecular imaging of hydrogen peroxide produced for cell signaling. Nat. Chem. Biol. 2007, 3, 349.
- Gao, X.; Feng, G.; Manghnani, P.N.; Hu, F.; Jiang, N.; Liu, J.; Liu, B.; Sun, J.Z.; Tang, B.Z. A two-channel responsive fluorescent probe with AIE characteristics and its application for selective imaging of superoxide anions in living cells. Chem. Commun. 2017, 53, 1653–1656.
- Xiao, H.; Zhang, W.; Li, P.; Zhang, W.; Wang, X.; Tang, B. Versatile Fluorescent Probes for Imaging the Superoxide Anion in Living Cells and In Vivo. Angew. Chem. Int. Ed. 2020, 59, 4216–4230.
- Duan, Q.; Zheng, G.; Li, Z.; Cheng, K.; Zhang, J.; Yang, L.; Jiang, Y.; Zhang, H.; He, J.; Sun, H. An ultra-sensitive ratiometric fluorescent probe for hypochlorous acid detection by the synergistic effect of AIE and TBET and its application of detecting exogenous/endogenous HOCl in living cells. J. Mater. Chem. B 2019, 7, 5125–5131.
- Qiao, W.; Ma, T.; Wang, S.; Li, L.; Liu, M.; Jiang, H.; Wu, Y.; Zhu, J.; Li, Z. Designing Squaraine Dyes with Bright Deep-Red Aggregation-Induced Emission for Specific and Ratiometric Fluorescent Detection of Hypochlorite. Adv. Funct. Mater. 2021, 31, 2105452.
- Wu, W.; Mao, D.; Cai, X.; Duan, Y.; Hu, F.; Kong, D.; Liu, B. ONOO− and ClO− Responsive Organic Nanoparticles for Specific in Vivo Image-Guided Photodynamic Bacterial Ablation. Chem. Mater. 2018, 30, 3867–3873.
- Jiang, G.; Li, C.; Lai, Q.; Liu, X.; Chen, Q.; Zhang, P.; Wang, J.; Tang, B.Z. An easily available ratiometric AIE probe for peroxynitrite in vitro and in vivo imaging. Sensor. Actuat. B Chem. 2021, 329, 129223.
- Han, X.; Yang, X.; Zhang, Y.; Li, Z.; Cao, W.; Zhang, D.; Ye, Y. A novel activatable AIEgen fluorescent probe for peroxynitrite detection and its application in EC1 cells. Sensor. Actuat. B Chem. 2020, 321, 128510.
- Zeng, Z.; Liew, S.S.; Wei, X.; Pu, K. Hemicyanine-Based Near-Infrared Activatable Probes for Imaging and Diagnosis of Diseases. Angew. Chem. Int. Ed. 2021, 60, 26454–26475.
- Vassalle, C.; Maltinti, M.; Sabatino, L. Targeting oxidative stress for disease prevention and therapy: Where do we stand, and where do we go from here. Molecules 2020, 25, 2653.
- Ma, B.; Xu, H.; Zhuang, W.; Wang, Y.; Li, G.; Wang, Y. ROS Responsive Nanoplatform with Two-Photon AIE Imaging for Atherosclerosis Diagnosis and “Two-Pronged” Therapy. Small 2020, 16, 2003253.
- Zhang, W.; Liu, W.; Li, P.; Huang, F.; Wang, H.; Tang, B. Rapid-Response Fluorescent Probe for Hydrogen Peroxide in Living Cells Based on Increased Polarity of C−B Bonds. Anal. Chem. 2015, 87, 9825–9828.
- Li, W.; Wang, L.; Tang, H.; Cao, D. An interface-targeting and H2O2-activatable probe liberating AIEgen: Enabling on-site imaging and dynamic movement tracking of lipid droplets. Chem. Commun. 2019, 55, 4491–4494.
- Mao, D.; Wu, W.; Ji, S.; Chen, C.; Hu, F.; Kong, D.; Ding, D.; Liu, B. Chemiluminescence-Guided Cancer Therapy Using a Chemiexcited Photosensitizer. Chem 2017, 3, 991–1007.
- Xu, L.; Sun, L.; Zeng, F.; Wu, S. Near-Infrared Fluorescent Nanoprobe for Detecting Hydrogen Peroxide in Inflammation and Ischemic Kidney Injury. Chin. J. Chem. 2020, 38, 1304–1310.
- Cheng, Y.; Dai, J.; Sun, C.; Liu, R.; Zhai, T.; Lou, X.; Xia, F. An Intracellular H2O2-Responsive AIEgen for the Peroxidase-Mediated Selective Imaging and Inhibition of Inflammatory Cells. Angew. Chem. Int. Ed. 2018, 57, 3123–3127.
- Ma, B.; Xu, H.; Zhuang, W.; Wang, Y.; Li, G.; Wang, Y. Reactive Oxygen Species Responsive Theranostic Nanoplatform for Two-Photon Aggregation-Induced Emission Imaging and Therapy of Acute and Chronic Inflammation. ACS Nano 2020, 14, 5862–5873.
- Shatalin, K.; Shatalina, E.; Mironov, A.; Nudler, E. H2S: A Universal Defense Against Antibiotics in Bacteria. Science 2011, 334, 986–990.
- Szabo, C. Gasotransmitters in cancer: From pathophysiology to experimental therapy. Nat. Rev. Drug Discov. 2016, 15, 185–203.
- Kumar, N.; Bhalla, V.; Kumar, M. Recent developments of fluorescent probes for the detection of gasotransmitters (NO, CO and H2S). Coordin. Chem. Rev. 2013, 257, 2335–2347.
- Opoku-Damoah, Y.; Zhang, R.; Ta, H.T.; Xu, Z.P. Therapeutic gas-releasing nanomedicines with controlled release: Advances and perspectives. Exploration 2022, 2, 20210181.
- Lundberg, J.O.; Gladwin, M.T.; Weitzberg, E. Strategies to increase nitric oxide signalling in cardiovascular disease. Nat. Rev. Drug Discov. 2015, 14, 623–641.
- Zhou, L.; Li, X.; Wang, K.; Shen, F.; Zhang, L.; Li, P.; Shang, T.; Wang, J.; Huang, N. Cu∥-loaded polydopamine coatings with in situ nitric oxide generation function for improved hemocompatibility. Regen. Biomater. 2020, 7, 153–160.
- Qian, Y.; Matson, J.B. Gasotransmitter delivery via self-assembling peptides: Treating diseases with natural signaling gases. Adv. Drug Delivery Rev. 2017, 110–111, 137–156.
- Li, Z.; Polhemus, D.J.; Lefer, D.J. Evolution of hydrogen sulfide therapeutics to treat cardiovascular disease. Circ. Res. 2018, 123, 590–600.
- Zhou, Y.; Zhang, X.; Yang, S.; Li, Y.; Qing, Z.; Zheng, J.; Li, J.; Yang, R. Ratiometric Visualization of NO/H2S Cross-Talk in Living Cells and Tissues Using a Nitroxyl-Responsive Two-Photon Fluorescence Probe. Anal. Chem. 2017, 89, 4587–4594.
- Yang, M.; Fan, J.; Du, J.; Peng, X. Small-molecule fluorescent probes for imaging gaseous signaling molecules: Current progress and future implications. Chem. Sci. 2020, 11, 5127–5141.
- Zhu, T.; Ren, N.; Liu, X.; Dong, Y.; Wang, R.; Gao, J.; Sun, J.; Zhu, Y.; Wang, L.; Fan, C.; et al. Probing the Intracellular Dynamics of Nitric Oxide and Hydrogen Sulfide Using an Activatable NIR II Fluorescence Reporter. Angew. Chem. Int. Ed. 2021, 60, 8450–8454.
- Liu, L.; Zhang, F.; Xu, B.; Tian, W. Silica nanoparticles based on an AIE-active molecule for ratiometric detection of RNS in vitro. J. Mater. Chem. B 2017, 5, 9197–9203.
- Hu, W.; Xie, M.; Zhao, H.; Tang, Y.; Yao, S.; He, T.; Ye, C.; Wang, Q.; Lu, X.; Huang, W.; et al. Nitric oxide activatable photosensitizer accompanying extremely elevated two-photon absorption for efficient fluorescence imaging and photodynamic therapy. Chem. Sci. 2018, 9, 999–1005.
- Lucero, M.Y.; East, A.K.; Reinhardt, C.J.; Sedgwick, A.C.; Su, S.; Lee, M.C.; Chan, J. Development of NIR-II photoacoustic probes tailored for deep-tissue sensing of nitric oxide. J. Am. Chem. Soc. 2021, 143, 7196–7202.
- Lin, V.S.; Chen, W.; Xian, M.; Chang, C.J. Chemical probes for molecular imaging and detection of hydrogen sulfide and reactive sulfur species in biological systems. Chem. Soc. Rev. 2015, 44, 4596–4618.
- Zhou, Y.; Mazur, F.; Fan, Q.; Chandrawati, R. Synthetic nanoprobes for biological hydrogen sulfide detection and imaging. View 2022, 3, 20210008.
- Sun, L.; Ouyang, J.; Ma, Y.; Zeng, Z.; Zeng, C.; Zeng, F.; Wu, S. An Activatable Probe with Aggregation-Induced Emission for Detecting and Imaging Herbal Medicine Induced Liver Injury with Optoacoustic Imaging and NIR-II Fluorescence Imaging. Adv. Healthc. Mater. 2021, 10, 2100867.
