Bone mineralization entails two mineralization phases: primary and secondary mineralization. Primary mineralization is achieved when matrix vesicles are secreted by osteoblasts, and thereafter, bone mineral density gradually increases during secondary mineralization. Nearby extracellular phosphate ions (PO43−) flow into the vesicles via membrane transporters and enzymes located on the vesicles’ membranes, while calcium ions (Ca2+), abundant in the tissue fluid, are also transported into the vesicles. The accumulation of Ca2+ and PO43− in the matrix vesicles induces crystal nucleation and growth. The calcium phosphate crystals grow radially within the vesicle, penetrate the vesicle’s membrane, and continue to grow outside the vesicle, ultimately forming mineralized nodules. The mineralized nodules then attach to collagen fibrils, mineralizing them from the contact sites (i.e., collagen mineralization). Afterward, the bone mineral density gradually increases during the secondary mineralization process.
- bone mineralization
- osteoblast
- osteocyte
- matrix vesicle
1. Introduction
2. Matrix Vesicle-Meditated Mineralization
2.1. Nucleation of Calcium Phosphates in Matrix Vesicles
Bone mineralization initiates inside matrix vesicles, which are small extracellular vesicles secreted by osteoblasts [18,19,35,36,37][18][19][35][36][37]. Matrix vesicles contain several membrane transporters and enzymes related to mineralization on their plasma membranes and in their interior, thus providing an adequate microenvironment for calcium phosphate nucleation and subsequent growth to eventually form hydroxyapatite crystals [Ca10(PO4)6(OH)2]. Regarding calcium phosphate crystal nucleation, it is generally known that Ca2+ strongly binds to the negatively charged inner leaflet of the plasma membrane [38]. Plasma membranes consisting of phosphatidylcholine and phosphatidylserine have a substantial capacity for Ca2+ binding, with several types of binding sites proposed [39].2.2. Distribution of Ca and P in the Vicinity of Matrix Vesicles in the Osteoid
2.3. Local Synthesis of PO
43−
by the Activities of TNAP and ENPP1
One of the most important enzymes enabling mineralization is TNAP, a glycosylphosphatidylinositol anchor enzyme associated with the cell membranes of matrix vesicles and osteoblastic cells. TNAP can hydrolyze various phosphate esters, especially pyrophosphates (PPi), and is broadly recognized as a hallmark of osteoblastic cells. However, the method of PPi supplementation is important. Currently, it is believed that ENPP1 mainly supplies PPi. ENPP1 is composed of two N-terminal somatomedin B-like domains, a catalytic domain, and a nuclease-like domain. Crystalline structure analysis of ENPP1 demonstrated that the nucleotides are accommodated in a pocket formed by an insertion loop in the catalytic domain of ENPP1, implying a preference for an ATP substrate [7]. Therefore, in bone mineralization, the catalytic activity of ENPP1 may generate PPi, presumably using ATPs in the extracellular fluid. The resultant PPi is then hydrolyzed by TNAP into PO43−. However, PPi is also known to inhibit mineralization by binding to nascent hydroxyapatite crystals, thereby preventing crystal overgrowth [50,51,52][44][45][46]. Hence, a balance between PPi and PO43− is important for normal bone mineralization. TNAP is not uniformly distributed on the cell membranes of osteoblasts; it was distinctly observed on the basolateral sides rather than the secretory (osteoidal) domains [37,53][37][47].2.4. Transport of PPi and PO
43−
via ANK and Pit1/Pit2
ANK, encoded by the progressive ankylosis gene (Ank), can serve as a non-enzymatic PPi channel, allowing PPi to pass through the plasma membrane to the outside of the cell [16,17][16][17]. As shown in ourthe recent reports, the immunoreactivity of ENPP1 was detected not only in the cell membranes but also in the cytoplasmic region of osteoblasts and osteocytes, indicating the presence of both extracellular and intracellular PPi in these cells [54][48]. It is therefore likely that the ANK-mediated outflow of intracellular PPi may be involved in the dynamic equilibrium between intra- and extracellular levels of PPi. After the outflow of PPi to the extracellular region, TNAP hydrolyzes PPi into PO43−. Extracellular PO43− may pass through the plasma membrane of the matrix vesicles by Pit1 and Pit2 mediation. Pit1 and Pit2 are type III sodium-inorganic phosphate (Pi) co-transporters encoded by Slc20a1 and Slc20a2 [8,9,10,11,12][8][9][10][11][12]. Recently, it has been reported that Pit1 and Pit2 form heterodimers, sense extracellular PO43− concentrations, and increase the expression of matrix Gla protein (MGP) and osteopontin via the extracellular signal-regulated kinase (ERK) pathway [55,56,57][49][50][51].2.5. PHOSPHO1 for PO
43−
Production inside Matrix Vesicles
Alternative to the biological function of ENPP1/TNAP, PHOSPHO1 is an enzyme highly expressed in mineralizing osteoblasts and hypertrophic chondrocytes [60][52]. This enzyme has been implicated in bone and cartilage formation and is thought to function inside cells and matrix vesicles to generate PO43− using phosphocholine and phosphoethanolamine, which are components of the lipid bilayers of matrix vesicles [13,14,15][13][14][15].3. Development of Mineralized Nodules and Collagen Mineralization
3.1. Growth of Mineralized Nodules
The calcium phosphate crystals that are nucleated inside the matrix vesicles grow in all directions and then penetrate the plasma membrane to exit the vesicles, eventually forming mineralized nodules, which are also referred to as calcifying globules [1,3,4][1][3][4]. Under TEM observation, mineralized nodules appear as globular structures composed of radially assembled hydroxyapatite crystals [44,68][53][54]. It seems likely that the growth of mineralized nodules is regulated by non-collagenous proteins in the osteoid. Among these materials, osteopontin is especially suited to regulating mineralization because it is a negatively charged and highly phosphorylated molecule that can effectively inhibit hydroxyapatite formation and growth [6,69][6][55]. Osteocalcin is another important bone matrix protein subjected to vitamin K-dependent carboxylation at its glutamate residues.
3.2. Collagen Mineralization
Collagen mineralization begins at the point of contact with mineralized nodules. TEM observations demonstrated that mineralization spreads from the contact point of the mineralized nodules toward the periphery of the collagen fibrils [2]. This finding suggests that collagen mineralization orderly progresses from the contact points with mineralized nodules, presumably allowing the regular deposition of calcium phosphate crystals onto the collagen fibrils. At a higher magnification, the spicules of calcium phosphate crystals can be seen on the fibrillar structures identical to the superhelix (tropocollagen) of collagen fibrils, thus indicating that mineral crystals are deposited on the superhelix, which serves as a scaffold for collagen mineralization. After contact with the mineralized nodules, the collagen fibrils eventually become completely mineralized. Proteoglycans such as decorin and biglycan, which directly bind the collagen surface through GAG chains, inhibit the growth of mineral crystals [74,75,76][56][57][58]. Collagen mineralization in the osteoid increases proportionally based on the distance from the osteoblasts, whereas the amount of decorin in the osteoid decreases further away from the bone surface [77][59]. In the osteoid close to the osteoblasts, proteoglycans combined with the surface of newly formed collagen fibrils are localized to the large space between collagen fibrils.4. Osteocyte Network and the Biological Function of Regulating Bone Mineralization
4.1. Distribution of the Osteocyte Network
Osteoblasts secrete bone matrix proteins and can become embedded in the bone matrix, where they differentiate into osteocytes. Immediately before becoming embedded into the bone matrix, osteoblasts rearrange the actin filament assembly along the cell membranes and the cytoplasmic processes, which resemble that of embedded osteocytes [24]. This implies that the osteoblasts approaching osteocytic differentiation and the newly-differentiated osteocytes decide the geometrical structure of the cellular network of their cytoplasmic processes.4.2. Osteocyte-Derived Molecules Involved in Peripheral Mineralization
Osteocytes physiologically synthesize several important molecules, e.g., dentin matrix protein (DMP) 1, matrix extracellular phosphoglycoprotein (MEPE), osteopontin, and Phex, for regulating surrounding bone mineralization. DMP1 has a high Ca2+-binding capacity and, therefore, is postulated to play a role in bone mineralization in the vicinity of osteocytes [33]. DMP1 belongs to the small integrin-binding ligand N-linked glycoprotein (SIBLING) family, which also includes MEPE, osteopontin, bone sialoprotein, and dentin sialo-phosphoprotein, and is encoded by a gene located on human chromosome 4q21 and mouse chromosome 5q21 [92,93][60][61]. Since osteocytes express abundant MEPE [96][62], DMP1 [33], and osteopontin, especially in Hyp mice fed a high-phosphate diet [97][63], it can be easily assumed that osteocyte-derived SIBLINGs would regulate peripheral bone mineralization by the osteocytes. This postulation is evidenced by the report that a DMP1 absence results in rickets or osteomalacia in mice [98][64] and by autosomal recessive hypophosphatemic rickets/osteomalacia (ARHR) in human patients [99][65]. Hence, osteocytes seem to be involved in the regulation of the surrounding mineralization. However, Phex/SIBLINGs are usually associated with the congenital deformities rickets and osteomalacia. Therefore, it is necessary to elucidate whether SIBLINGs play an important role in the physiological regulation of bone mineralization in a normal state (Figure 41).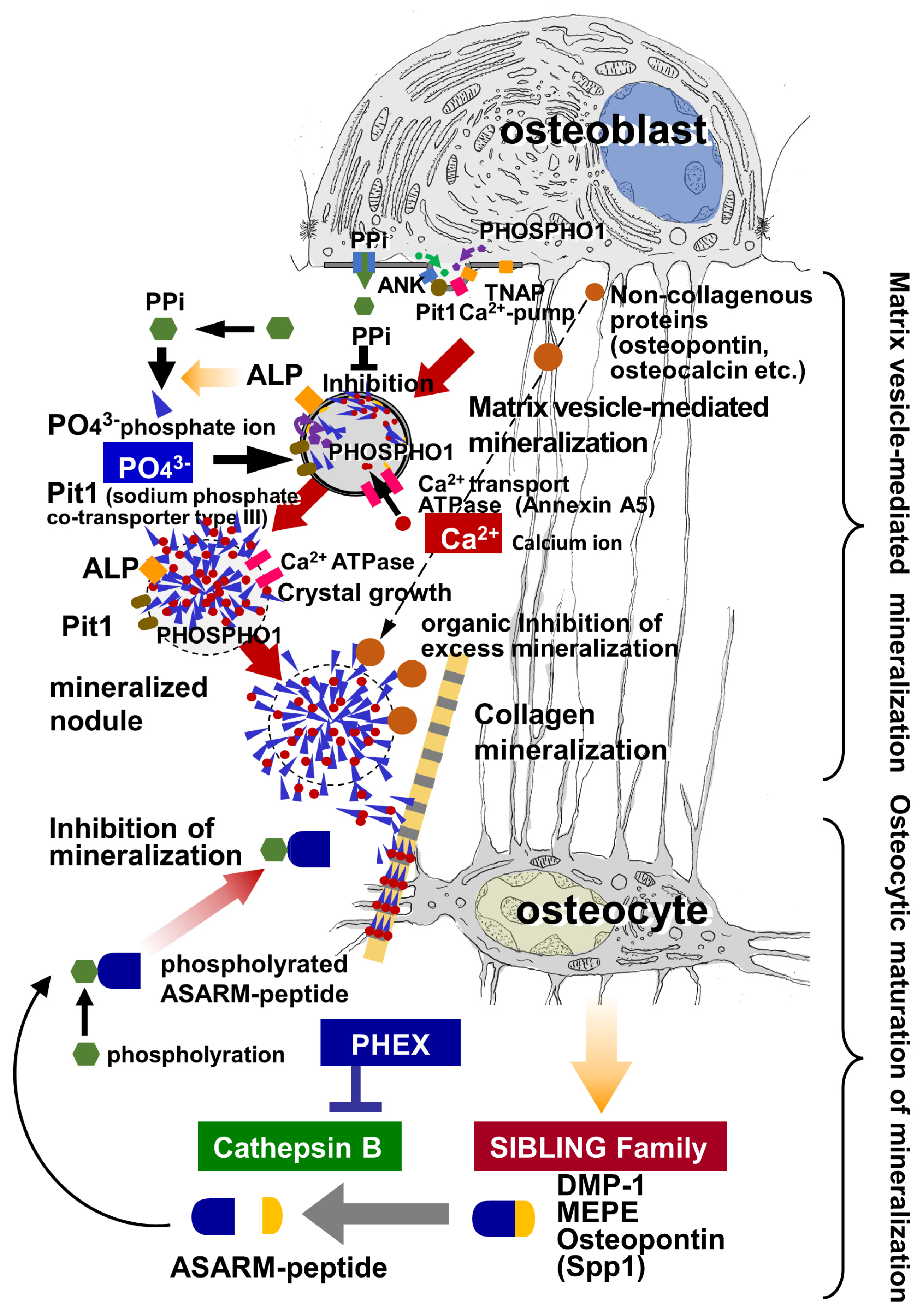
5. Cellular Interplay between Osteoblasts and Osteocytes in Bone Mineralization
Osteoblasts secrete matrix vesicles, which provide initiation sites for mineralization during primary mineralization, while osteocytes appear to regulate bone mineralization through Phex/SIBLINGs. Taking these findings into account, the interplay between osteoblasts and osteocytes in the regulation of bone mineralization seems likely. Matrix vesicles secreted by osteoblasts grow into globular assemblies of needle-like calcium phosphate crystals, called mineralized nodules, which then induce collagen mineralization. During nucleation and subsequent growth inside the vesicles, the influx of Ca2+ and PO43− is promoted by many enzymes and membrane transporters located on the matrix vesicles and mineralized nodules (particularly, they are located on the ruptured membranes of the vesicles). However, the growth of large, terminal mineralized nodules that are distant from osteoblasts, as well as collagen mineralization, may be regulated by a mechanism other than enzymes associated with matrix vesicles secreted by the osteoblasts. In the osteoid, there seem to be two possible pathways that supply Ca2+ and PO43− to terminal mineralized nodules and collagen mineralization: one is from the osteoblast-covered bone surface, and the other is from osteocytic canaliculi, which are opened to the osteoid.6. Conclusions
Primary mineralization in bone is achieved by matrix vesicle-mediated mineralization; matrix vesicles contain a variety of membrane transporters and enzymes involved in the nucleation and subsequent growth of crystalline calcium phosphates inside the vesicles. For proper mineralization, the biological accumulation of Ca2+ and PO43− in the vesicles is necessary. Of particular importance is the influx of PO43− into matrix vesicles, which involves a complex interplay among ENPP1, ANK, TNAP, and Pit1. Crystalline calcium phosphates grow radially, penetrate the vesicle membranes, and then exit the vesicles to form mineralized nodules, which are globular assemblies of needle-shaped mineral crystals. In contrast to primary mineralization, secondary mineralization increases bone mineral density, presumably due to osteocytic functions. Osteocytes appear to regulate bone mineralization, which is mediated by Phex/SIBLINGs. Thus, bone mineralization is biologically regulated by osteoblasts and osteocytes.References
- Ozawa, H.; Yamada, M.; Yamamoto, T. Ultrastructural Observations on the Location of Lead and Calcium in the Mineralizing Dentine of Rat Incisor. In Matrix Vesicles; Ascenzi, A., Bonucci, E., de Bernard, B., Eds.; Wiching Editore Srl: Milano, Italy, 1981; pp. 179–187.
- Hasegawa, T.; Li, M.; Hara, K.; Sasaki, M.; Tabata, C.; de Freitas, P.H.L.; Hongo, H.; Suzuki, R.; Kobayashi, M.; Inoue, K.; et al. Morphological Assessment of Bone Mineralization in Tibial Metaphyses of Ascorbic Acid-Deficient ODS rats. Biomed. Res. 2011, 32, 259–269.
- Boonrungsiman, S.; Gentleman, E.; Carzaniga, R.; Evans, N.D.; McComb, D.W.; Porter, A.E.; Stevens, M.M. The role of intra-cellular calcium phosphate in osteoblast-mediated bone apatite formation. Proc. Natl. Acad. Sci. USA 2012, 109, 14170–14175.
- Ansari, S.; de Wildt, B.W.; Vis, M.A.; de Korte, C.E.; Ito, K.; Hofmann, S.; Yuana, Y. Matrix Vesicles: Role in Bone Mineral-ization and Potential Use as Therapeutics. Pharmaceuticals 2021, 14, 289.
- Matsuzawa, T.; Anderson, H.C. Phosphatases of epiphyseal carti-lage studied by electron microscopic cytochemical methods. J. Histochem. Cytochem. 1971, 19, 801–808.
- De Bruyn, J.R.; Goiko, M.; Mozaffari, M.; Bator, D.; Dauphinee, R.L.; Liao, Y.; Flemming, R.L.; Bramble, M.S.; Hunter, G.K.; Goldberg, H.A. Dynamic light scattering study of inhibition of nucleation and growth of hydroxyapatite crystals by osteopon-tin. PLoS ONE 2013, 8, e56764.
- Kato, K.; Nishimasu, H.; Okudaira, S.; Mihara, E.; Ishitani, R.; Takagi, J.; Aoki, J.; Nureki, O. Crystal structure of Enpp1, an extracellular glycoprotein involved in bone mineralization and insulin signaling. Proc. Natl. Acad. Sci. USA 2012, 109, 16876–168781.
- Bai, L.; Collins, J.F.; Ghishan, F.K. Cloning and characterization of a type III Na-dependent phosphate cotransporter from mouse intestine. Am. J. Physiol. Cell. Physiol. 2000, 279, C1135–C1143.
- Bai, L.; Collins, J.F.; Xu, H.; Xu, L.; Ghishan, F.K. Molecular cloning of a murine type III sodium-dependent phosphate co-transporter (Pit-2) gene promoter. Biochim. Biophys. Acta 2001, 1522, 42–45.
- Collins, J.F.; Bai, L.; Ghishan, F.K. The SLC20 family of proteins: Dual functions as sodium-phosphate cotransporters and viral receptors. Pflügers Arch. 2004, 447, 647–652.
- Zoidis, E.; Ghirlanda-Keller, C.; Gosteli-Peter, M.; Zapf, J.; Schmid, C. Regulation of phosphate (Pi) transport and NaPi-III transporter (Pit-1) mRNA in rat osteoblasts. J. Endocrinol. 2004, 181, 531–540.
- Forster, I.C.; Hernando, N.; Biber, J.; Murer, H. Phosphate transporters of the SLC20 and SLC34 families. Mol. Asp. Med. 2013, 34, 386–395.
- Roberts, S.J.; Stewart, A.J.; Sadler, P.J.; Farquharson, C. Human PHOSPHO1 exhibits high specific phosphoethanolamine and phosphocholine phosphatase activities. Biochem. J. 2004, 382, 59–65.
- Roberts, S.; Narisawa, S.; Harmey, D.; Millán, J.L.; Farquharson, C. Functional Involvement of PHOSPHO1 in Matrix Vesicle-Mediated Skeletal Mineralization. J. Bone Miner. Res. 2007, 22, 617–627.
- Ciancaglini, P.; Yadav, M.C.; Simão, A.M.S.; Narisawa, S.; Pizauro, J.M.; Farquharson, C.; Hoylaerts, M.F.; Millán, J.L. Kinetic Analysis of Substrate Utilization by Native and TNAP-, NPP1- or PHOSPHO1-Deficient Matrix Vesicles. J. Bone Miner. Res. 2010, 25, 716–723.
- Ho, A.M.; Johnson, M.D.; Kingsley, D.M. Role of the mouse ank gene in tissue calcification and arthritis. Science 2000, 15, 265–270.
- Gurley, K.A.; Reimer, R.J.; Kingsley, D.M. Biochemical and Genetic Analysis of ANK in Arthritis and Bone Disease. Am. J. Hum. Genet. 2006, 79, 1017–1029.
- Hasegawa, T.; Yamamoto, T.; Tsuchiya, E.; Hongo, H.; Tsuboi, K.; Kudo, A.; Abe, M.; Yoshida, T.; Nagai, T.; Khadiza, N.; et al. Ultrastructural and biochemical aspects of matrix vesicle-mediated mineralization. Jpn. Dent. Sci. Rev. 2016, 53, 34–45.
- Hasegawa, T. Ultrastructure and biological function of matrix vesicles in bone mineralization. Histochem. Cell Biol. 2018, 149, 289–304.
- Xu, T.; Bianco, P.; Fisher, L.W.; Longenecker, G.; Smith, E.; Goldstein, S.; Bonadio, J.; Boskey, A.; Heegaard, A.-M.; Sommer, B.; et al. Targeted disruption of the biglycan gene leads to an osteoporosis-like phenotype in mice. Nat. Genet. 1998, 20, 78–82.
- Corsi, A.; Xu, T.; Chen, X.; Boyde, A.; Liang, J.; Mankani, M.; Sommer, B.; Iozzo, R.V.; Eichstetter, I.; Robey, P.G.; et al. Phenotypic Effects of Biglycan Deficiency Are Linked to Collagen Fibril Abnormalities, Are Synergized by Decorin Deficiency, and Mimic Ehlers-Danlos-Like Changes in Bone and Other Connective Tissues. J. Bone Miner. Res. 2002, 17, 1180–1189.
- Kemp, L.P.; Morris, J.A.; Medina-Gomez, C.; Forgetta, V.; Warrington, N.M.; Youlten, S.E.; Zheng, J.; Gregson, C.L.; Grundberg, E.; Trajanoska, K.; et al. Identification of 153 new loci associated with heel bone mineral density and functional in-volvement of GPC6 in osteoporosis. Nat. Genetics. 2017, 49, 1468–1475.
- Hao, J.-X.; Shen, M.-J.; Wang, C.-Y.; Wei, J.-H.; Wan, Q.-Q.; Zhu, Y.-F.; Ye, T.; Luo, M.-L.; Qin, W.-P.; Li, Y.-T.; et al. Regulation of biomineralization by proteoglycans: From mechanisms to application. Carbohydr. Polym. 2022, 294, 119773.
- Nagai, T.; Hasegawa, T.; Yimin; Yamamoto, T.; Hongo, H.; Abe, M.; Yoshida, T.; Yokoyama, A.; de Freitas, P.H.L.; Li, M.; et al. Immunocytochemical assessment of cell differentiation of podoplanin-positive osteoblasts into os-teocytes in murine bone. Histochem. Cell. Biol. 2021, 155, 369–380.
- Plotkin, L.I.; Bellido, T. Osteocytic signalling pathways as therapeutic targets for bone fragility. Nat. Rev. Endocrinol. 2016, 12, 593–605.
- Gould, N.R.; Torre, O.M.; Leser, J.M.; Stains, J.P. The cytoskeleton and connected elements in bone cell mechano-transduction. Bone 2021, 149, 115971.
- Moriishi, T.; Komori, T. Osteocytes: Their Lacunocanalicular Structure and Mechanoresponses. Int. J. Mol. Sci. 2022, 23, 4373.
- Wang, H.; Du, T.; Li, R.; Main, R.P.; Yang, H. Interactive effects of various loading parameters on the fluid dynamics within the lacunar-canalicular system for a single osteocyte. Bone 2022, 158, 116367.
- Sato, T.; Verma, S.; Andrade, C.D.C.; Omeara, M.; Campbell, N.; Wang, J.S.; Cetinbas, M.; Lang, A.; Ausk, B.J.; Brooks, D.J.; et al. A FAK/HDAC5 signaling axis controls osteocyte mechanotransduction. Nat. Commun. 2020, 11, 3282.
- Ubaidus, S.; Li, M.; Sultana, S.; de Freitas, P.H.L.; Oda, K.; Maeda, T.; Takagi, R.; Amizuka, N. FGF23 ismainly synthesized by osteocytes in the regularly distributed osteocytic lacunar canalicular system established after physiological bone remodeling. J. Electron. Microsc. 2009, 58, 381–392.
- Rowe, P.S.; Kumagai, Y.; Gutierrez, G.; Garrett, I.R.; Blacher, R.; Rosen, D.; Cundy, J.; Navvab, S.; Chen, D.; Drezner, M.K.; et al. MEPE has the properties of an osteoblastic phosphatonin and minhibin. Bone 2004, 34, 303–319.
- Rowe, P.S.; Garrett, I.R.; Schwarz, P.M.; Carnes, D.L.; Lafer, E.; Mundy, G.R.; Gutierrez, G.E. Surface plasmon resonance (SPR) confirms that MEPE binds to PHEX via the MEPE–ASARM motif: A model for impaired mineralization in X-linked rickets (HYP). Bone 2005, 36, 33–46.
- Sasaki, M.; Hasegawa, T.; Yamada, T.; Hongo, H.; de Freitas, P.H.; Suzuki, R.; Yamamoto, T.; Tabata, C.; Toyosawa, S.; Yamamoto, T.; et al. Altered distribution of bone matrix proteins and defective bone min-eralization in klotho-deficient mice. Bone 2013, 57, 206–219.
- Oya, K.; Ishida, K.; Nishida, T.; Sato, S.; Kishino, M.; Hirose, K.; Ogawa, Y.; Ikebe, K.; Takeshige, F.; Yasuda, H.; et al. Immunohistochemical analysis of dentin matrix protein 1 (Dmp1) phosphorylation by Fam20C in bone: Impli-cations for the induction of biomineralization. Histochem. Cell. Biol. 2017, 147, 341–351.
- Anderson, H.C. Vesicles Associated with Calcification in the Matrix of Epiphyseal Cartilage. J. Cell Biol. 1969, 41, 59–72.
- Bonucci, E. Fine Structure and Histochemistry of "Calcifying Globules" in Epiphyseal Cartilage. Cell Tissue Res. 1970, 103, 192–217.
- Amizuka, N.; Hasegawa, T.; Oda, K.; Luiz de Freitas, P.H.; Hoshi, K.; Li, M.; Ozawa, H. Histology of Epiphyseal Cartilage Cal-cification and Endochondral Ossification. Front. Biosci. 2012, 4, 2085–2100.
- Tadross, M.R.; Tsien, R.W.; Yue, D.T. Ca2+ channel nanodomains boost local Ca2+ amplitude. Proc. Natl. Acad. Sci. USA 2013, 110, 15794–15799.
- Melcrova, A.; Pokorna, S.; Pullanchery, S.; Kohagen, M.; Jurkiewicz, P.; Hof, M.; Jungwirth, P.; Cremer, P.S.; Cwiklik, L. The complex nature of calcium cation interactions with phospholipid bilayers. Sci. Rep. 2016, 6, 38035.
- Hoshi, K.; Ejiri, S.; Ozawa, H. Localizational Alterations of Calcium, Phosphorus, and Calcification-Related Organics Such as Proteoglycans and Alkaline Phosphatase During Bone Calcification. J. Bone Miner. Res. 2001, 16, 289–298.
- Genge, B.R.; Wu, L.N.; Wuthier, R.E. Identification of phospholipid-dependent calcium-binding proteins as constituents of ma-trix vesicles. J. Biol. Chem. 1989, 264, 10917–10921.
- Genge, B.R.; Cao, X.; Wu, L.N.; Buzzi, W.R.; Showman, R.W.; Arsenault, A.L.; Ishikawa, Y.; Wuthier, R.E. Establishment of the primary structure of the major lipid dependent Ca2+ binding proteins of chicken growth plate cartilage matrix vesicles: Iden-tity with anchorin CII (annexin V) and annexin II. J. Bone. Miner. Res. 1992, 7, 807–819.
- Balcerzak, M.; Malinowska, A.; Thouverey, C.; Sekrecka, A.; Dadlez, M.; Buchet, R.; Pikula, S. Proteome analysis of matrix vesicles isolated from femurs of chicken embryo. Proteomics 2007, 8, 192–205.
- Terkeltaub, R.; Rosenbach, M.; Fong, F.; Goding, J. Causal link between nucleotide pyrophosphohydrolase overactivity and increased intracellular inorganic pyrophosphate generation demonstrated by transfection of cultured fibroblasts and osteo-blasts with plasma cell membrane glycoprotein-1. Arthritis. Rheum. 1994, 37, 934–941.
- Johnson, K.; Vaingankar, S.; Chen, Y.; Moffa, A.; Goldring, M.; Sano, K.; Jin-Hua, P.; Sali, A.; Goding, J.; Terkeltaub, R. Dif-ferential mechanisms of inorganic pyrophosphate production by plasma cell membrane glycoprotein-1 and B10 in chondro-cytes. Arthritis. Rheum. 1999, 42, 1986–1997.
- Johnson, K.; Moffa, A.; Chen, Y.; Pritzker, K.; Goding, J.; Terkeltaub, R. Matrix vesicle plasma membrane glycoprotein-1 reg-ulates mineralization by murine osteoblastic MC3T3 cells. J. Bone. Miner. Res. 1999, 14, 883–892.
- Nakano, Y.; Beertsen, W.; van den Bos, T.; Kawamoto, T.; Oda, K.; Takano, Y. Site-specific localization of two distinct phos-phatasesalong the osteoblast plasma membrane: Tissue non-specificalkaline phosphatase and plasma membrane calcium ATPase. Bone 2004, 35, 1077–1085.
- Yamamoto, T.; Hasegawa, T.; Mae, T.; Hongo, H.; Yamamoto, T.; Abe, M.; Nasoori, A.; Morimoto, Y.; Maruoka, H.; Kubota, K.; et al. Comparative immunolocalization of tissue nonspecific alkaline phosphatase and ectonucleotide pyrophosphatase/phosphodiesterase 1 in murine bone. J. Oral Biosci. 2021, 63, 259–264.
- Beck, L.; Leroy, C.; Salaün, C.; Margall-Ducos, G.; Desdouets, C.; Friedlander, G. Identification of a Novel Function of PiT1 Critical for Cell Proliferation and Independent of Its Phosphate Transport Activity. J. Biol. Chem. 2009, 284, 31363–31374.
- Salaün, C.; Leroy, C.; Rousseau, A.; Boitez, V.; Beck, L.; Friedlander, G. Identification of a novel transport-independent function of PiT1/SLC20A1 in the regulation of TNF-induced apoptosis. J. Biol. Chem. 2010, 285, 34408–34418.
- Forand, A.; Koumakis, E.; Rousseau, A.; Sassier, Y.; Journe, C.; Merlin, J.F.; Leroy, C.; Boitez, V.; Codogno, P.; Friedlander, G.; et al. Disruption of the Phosphate Transporter Pit1 in Hepatocytes Improves Glucose Metabolism and Insulin Signaling by Modulating the USP7/IRS1 Interaction. Cell. Rep. 2016, 16, 2736–2748.
- Houston, B.; Stewart, A.J.; Farquharson, C. PHOSPHO1—A novel phosphatase specifically expressed at sites of mineralisation in bone and cartilage. Bone 2004, 34, 629–637.
- Ozawa, H. Ultrastructural concepts on biological calcification; Focused on matrix vesicles. Jpn. J. Oral Biol. 1985, 27, 751–774.
- Weiner, S. Organization of extracellularly mineralized tissues: A comparative study of biological crystal growth. CRC Crit. Rev. Biochem. 1986, 20, 365–408.
- Boskey, A.L.; Christensen, B.; Taleb, H.; Sørensen, E.S. Post-translational modification of osteopontin: Effects on in vitro hy-droxyapatite formation and growth. Biochem. Biophys. Res. Commun. 2012, 419, 333–338.
- Boskey, A.L.; Spevak, L.; Doty, S.B.; Rosenberg, L. Effects of bone CS-proteoglycans, DS-decorin, and DS-biglycan on hydrox-yapatite formation in a gelatin gel. Calcif. Tissue. Int. 1997, 61, 298–305.
- Tavafoghi, M.; Cerruti, M. The role of amino acids in hydroxyapatite mineralization. J. R. Soc. Interface 2016, 13, 20160462.
- Wang, K.; Ren, Y.; Lin, S.; Jing, Y.; Ma, C.; Wang, J.; Yuan, X.B.; Han, X.; Zhao, H.; Wang, Z.; et al. Osteocytes but not osteoblasts directly build mineralized bone structures. Int. J. Biol. Sci. 2021, 17, 2430–2448.
- Hoshi, K.; Kemmotsu, S.; Takeuchi, Y.; Amizuka, N.; Ozawa, H. The Primary Calcification in Bones Follows Removal of Decorin and Fusion of Collagen Fibrils. J. Bone Miner. Res. 1999, 14, 273–280.
- Liu, S.; Rowe, P.S.N.; Vierthaler, L.; Zhou, J.; Quarles, L.D. Phosphorylated acidic serine–aspartate-rich MEPE-associated motif peptide from matrix extracellular phosphoglycoprotein inhibits phosphate regulating gene with homologies to endopeptidases on the X-chromosome enzyme activity. J. Endocrinol. 2007, 192, 261–267.
- Staines, K.A.; MacRae, V.E.; Farquharson, C. The importance of the SIBLING family of proteins on skeletal mineralisation and bone remodelling. J. Endocrinol. 2012, 214, 241–255.
- Yamada, T.; Matsukawa, N.; Matsumoto, M.; Morimoto, S.; Ogihara, T.; Ochi, T.; Yoshikawa, H.; Nampei, A.; Hashimoto, J.; Hayashida, K.; et al. Matrix extracellular phosphoglycoprotein (MEPE) is highly expressed in osteocytes in human bone. J. Bone Miner. Metab. 2004, 22, 176–184.
- Hoac, B.; Østergaard, M.; Wittig, N.K.; Boukpessi, T.; Buss, D.J.; Chaussain, C.; Birkedal, H.; Murshed, M.; McKee, M.D. Genetic Ablation of Osteopontin in Osteomalacic Hyp Mice Partially Rescues the Deficient Mineralization Without Correcting Hypo-phosphatemia. J. Bone. Miner. Res. 2020, 35, 2032–2048.
- Feng, J.Q.; Ward, L.M.; Liu, S.; Lu, Y.; Xie, Y.; Yuan, B.; Yu, X.; Rauch, F.; Davis, S.I.; Zhang, S.; et al. Loss of DMP1 causes rickets and osteomalacia and identifies a role for osteocytes in mineral metabolism. Nat. Genet. 2006, 38, 1310–1315.
- Mäkitie, O.; Pereira, R.C.; Kaitila, I.; Turan, S.; Bastepe, M.; Laine, T.; Kröger, H.; Cole, W.G.; Jüppner, H. Long-term clinical outcome and carrier phenotype in autosomal recessive hypophosphatemia caused by a novel DMP1 mutation. J. Bone Miner. Res. 2010, 25, 2165–2174.
