Inflammatory Bowel Diseases, including ulcerative colitis (UC) and Crohn’s disease (CD), are chronic, relapsing inflammatory conditions of the gastrointestinal (GI) tract. Interactions between the environmental factors and commensal intestinal microflora in genetically predisposed individuals are considered the leading cause of an inappropriate immune response and as a result, the development of inflammatory disease. Iron is an indispensable nutrient for life. A lack of it leads to iron deficiency anaemia (IDA), which currently affects about 1.2 billion people worldwide. The primary means of IDA treatment is oral or parenteral iron supplementation. This can be burdened with numerous side effects such as oxidative stress, systemic and local-intestinal inflammation, dysbiosis, carcinogenic processes and gastrointestinal adverse events.
1. Anaemia as a Complication of Inflammatory Bowel Diseases
1. Anaemia as a Complication of IBD
Anaemia is the most prevalent extraintestinal complication of IBD, with an estimated 70% of inpatients and 20% of outpatients with IBD developing anaemia, which is believed to affect one-third of the IBD patients at any one time
[1][75]. Traditionally, anaemia was classified as IDA and anaemia of chronic disease (ACD). In IBD patients, both the aforementioned types of anaemia may occur because the local inflammation in the intestines contributes to the onset of systemic inflammation
[2][76].
IDA is a consequence of chronic haemorrhages from the ulcerated mucosa, impaired dietary iron absorption, as well as self-imposed dietary restrictions relating to gastrointestinal symptoms. Moreover, appetite loss during an exacerbation of the disease and a range of other factors such as medicines used for IBD treatment (e.g., proton pump inhibitors, methotrexate, thiopurines and sulfasalazine) also negatively impact iron absorption and erythropoiesis
[1][75]. In turn, ACD is connected with the systemic immune response that accompanies inflammatory diseases such as IBD
[3][77]. Immune cells release pro-inflammatory cytokines, mainly interleukin-6 (IL-6), which upregulates the expression of liver hepcidin, the main regulator of iron homeostasis, thereby decreasing iron uptake from the enterocytes and a reduced ability to take advantage of sufficient iron for effective erythropoiesis
[4][78]. This issue is described in more detail in the section about iron metabolism.
2. Iron Replacement Therapy in Inflammatory Bowel Diseases
2. Iron Replacement Therapy in IBD
As described in the previous paragraph, the high incidence of anaemia in patients with IBD requires therapeutic intervention, either intravenous or oral iron administration. The European Crohn’s and Colitis Organization
[5][79] guidelines recommend intravenous iron supply as a mainstay treatment for IBD patients and front-line therapy for haemoglobin (Hb) levels < 10 g/dL (e.g., iron sucrose, ferric gluconate, ferric carboxymaltose, iron isomaltoside). Another indication for intravenous iron is the active phase of the disease because inflammation impairs iron absorption in the intestines
[6][80]. An intravenous iron supply is also recommended in patients with a poor iron tolerance and a previous unsuccessful attempt at oral iron treatment
[7][8][81,82]. If oral iron supplementation must be used, it should be limited to IBD patients with mild anaemia (Hb ≥ 11.0 g/dL), with an inactive disease and no prior intolerance to oral iron
[9][83].
3. Negative Consequences of Oral Iron
Oral iron administration can cause side effects due to a large amount of non-absorbed iron (about 90%) remaining in the intestines, including gastroduodenitis, nausea, bloating, vomiting, dyspepsia, constipation, diarrhoea, abdominal pain or darkening of the stools
[10][47]. Thus, oral iron therapy is not optimal with as many as 50% of patients discontinuing the therapy
[11][8]. The aforementioned gastrointestinal symptoms most likely result from a combination of a high concentration of free iron radicals induced by redox cycling in the gut lumen and at the mucosal surface which can promote inflammation and alterations in the gut microbiota composition
[12][13][9,45].
Moreover, non-absorbed iron can be toxic and exacerbates disease activity in IBD. A study conducted in rats with dextran sulphate sodium (DSS)-induced colitis indicated that dietary iron administration aggravates colitis and is associated with oxidative stress, neutrophil infiltration and NF-kappaB pathway activation which increases the expression of pro-inflammatory cytokines such as interferon-γ (IFN-γ), tumour necrosis factor α (TNF-α), and inducible nitric oxide synthase (iNOS). These negative effects can be ameliorated by vitamin E
[14][84]. Another animal study showed that a diet without iron sulphate combined with intravenous iron administration prevents the development of chronic ileitis in a mouse model of CD, suggesting that oral iron sulphate replacement therapy may trigger the inflammatory processes linked to the progression of CD-like ileitis
[15][85]. There is an imbalance between pro-oxidative and antioxidant mechanisms in CD, with patients having increased levels of reactive oxygen intermediates (ROI) and DNA oxidation products, as well as markedly raised iron levels in combination with reduced copper and activity of zinc superoxide dismutase (Cu/Zn SOD)
[16][86]. Oral iron due to its pro-oxidative capacity may promote a pro-inflammatory effect.
4. Impact of Iron on the Intestine
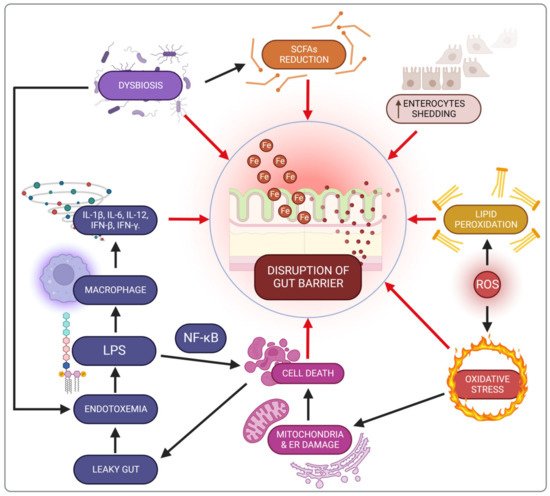
Iron has a significant impact on intestinal functioning, potentially via various mechanisms. Experimental evidence suggests that excessive iron in the lumen may be also harmful to the intestinal mucous membrane. Despite its key role in cellular processes, free iron in the colon can generate toxic free radicals that may directly impair the integrity of the intestinal epithelium via oxidative stress. The relationship between iron and redox stress is discussed in more detail in the section about iron and redox stress.
A key element of the physical intestinal barrier is a single layer of epithelial cells, mainly consisting of enterocytes, which in addition to absorbing nutrients, play an important role in immune activity, mediating the release of cytokines and the expression of receptors engaged in the immune response
[17][87]. The epithelial layer also includes the goblet cells secreting mucus, Paneth cells synthesising defensins, enterochromatophilic cells releasing hormones and neuropeptides, and M cells that capture antigens from the intestinal lumen. The epithelial integrity and as well as selective permeability are dependent on appropriate connections between cells, mainly tight junctions, including claudins, occludins, protein junctional adhesion molecules, and tricellulins
[18][88].
Oral iron supplementation contributes to lipid peroxidation of cellular membranes and disrupts energy processes in the cell due to mitochondrial damage, as well as promoting endoplasmic reticulum dysfunction
[19][89]. Consequently, these cellular disorders trigger death pathways, thereby destroying the mechanical intestinal barrier and increasing permeability, known as a leaky gut
[10][47]. There are also changes in the enterocyte shedding-proliferation axis
[20][90]. This impairment of the intestinal barrier increases the risk of exposure to bacterial endotoxins (e.g., LPS), leading to metabolic endotoxemia and microinflammation (gut-derived inflammation). LPS binding to its receptor complex on macrophages results in markedly increased production of pro-inflammatory cytokines such as IL-1β, IL-6, IL-12, IFN-β or IFN-γ, which may worsen intestinal inflammation in IBD patients. This impaired integrity has been demonstrated in an in vitro study with Caco-2 cells exposed to iron
[21][91].
The proper functioning of the intestinal barrier also involves short-chain fatty acids (SCFAs) including butyrate, acetate and propionate, produced by the microbiota in the colon through the anaerobic fermentation of indigestible polysaccharides such as dietary fibre and resistant starch. There are two main pathways for the conversion of butyrate–CoA into butyrate. The first pathway includes a two-step reaction using butyrate kinase and phosphate butyryltransferase. The second pathway is a single-step reaction conducted by butyryl–CoA: acetate Co-A transferase
[22][92]. SCFAs are the main source of energy to colonocytes and modulate the immune response through inhibition of the LPS-induced NF-κB pathway, and reduced production of pro-inflammatory cytokines and chemokine by the epithelial intestinal cells (EIC). Besides, SCFAs increase the secretion of antimicrobial peptides (LL-37 and CAP-18) and IL-18, a cytokine that maintains homeostasis in EIC, thereby protecting against colitis
[23][24][93,94].
The influence of oral iron supply on SCFAs production remains still unclear. Some research indicates that iron can contribute to increases in gut SCFAs production, thus positively affecting gut health
[25][95]. Some in vitro studies compared the impact on SCFAs production between the normal iron condition and iron deficiency, which does not fully reflect increased iron content in the gut lumen on SCFAs production
[26][96]. Another study investigated the influence of oral iron supplementation on colitis exacerbation and the composition of the gut microbiome. A decreased abundance of SCFA-producing genera was shown without assessing the levels of faecal SCFAs
[27][97]. However, the literature also provides evidence for the adverse impact of oral heme iron on murine colitis model, reducing the level of butyrate production and expression of butyrate kinase, phosphate butyryltransferase and the α subunit of butyryl–CoA: acetate Co-A transferase
[22][92].
In conclusion, the loss of intestinal barrier integrity is an early event which contributes to chronic inflammation (Figure 1).
Figure 1. Iron-related gut barrier disruption mechanisms. SCFAs—short-chain fatty acids; IFN-β—interferon β; IFN-γ—interferon-γ; IL-1β—interleukin 1β; IL-6—interleukin 6; IL-12—interleukin 12; LPS—lipopolysaccharide; NF-kβ—nuclear factor kappa-light-chain-enhancer of activated B cells; ROS—reactive oxygen species.
5. Impact of Iron on the Microbiota in Inflammatory Bowel Diseases Patients
5. Impact of Iron on the Microbiota in IBD Patients
Besides the unfavourable impact of iron on the intestinal epithelial barrier via oxidative stress, iron may also affect the gut microbiota
[28][98]. As mentioned above, the altered gut microbiota is a crucial factor in driving inflammation in IBD but it is unclear if dysbiosis is the cause or the outcome of mucosal inflammation. Numerous studies have indicated differences in the composition and diversity of the intestinal microbiota among IBD patients in comparison to healthy individuals. Characteristic changes observed in patients with IBD include increased bacteria such as Proteobacteria, Fusobacterium species, and
Ruminococcus gnavus and in turn, decreased protective groups such as Lachnospiraceae, Bifidobacterium species,
Roseburia, and
Sutterella [29][30][99,100]. Iron leads to a shift in the microbiota composition and exacerbation of dysbiosis in IBD. Low iron bioavailability results in a high concentration in the intestinal lumen and accessibility to the gut microflora. Lee et al.
[31][101] designed an open-labelled clinical trial with iron-deficient participants with or without IBD and performed pre-iron therapy and post-iron therapy measurements whereby the individuals served as the controls. This study compared the effects of
per oral (p.o) versus
intravenous (i.v) iron replacement therapy (IRT), indicating that oral iron supplementation decreased the diversity of intestinal microflora in patients with IBD and ID, especially
Faecalibacterium prausnitzii,
Ruminococcus bromii,
Dorea sp. and
Collinsella aerofaciens. It is worth noting that dysbiosis aggravated by iron can also be a causative factor disrupting the integrity of the EIC and promoting an oxidative pro-inflammatory microenvironment.
In summary, the need for iron supplementation in IBD patients who additionally suffer from anaemia exacerbates the dysbiosis caused by the disease, thus worsening their clinical condition.