2. Rapid, Non-Genomic Steroid Actions
The first non-genomic action of steroid molecules was described in 1942 when Hans Selye observed the anesthetic effect of progesterone immediately after its injection into peritoneum of rats and mice differently from what was observed with respect to its main effect that took place only within hours after its administration
[11][17]. Subsequently, Spach and Streeten demonstrated that Na
+ ion variation occurred within few minutes after aldosterone administration in dog erythrocytes, offering new compelling evidence on non-genomic effects of this hormone because these cells lack nuclei, and therefore the observed in vitro effects might be attributable exclusively to its non-genomic mechanism
[12][18]. However, these rapid hormone effects were not clarified until their recent recognition for several steroid hormones, including 1α,25(OH)
2D
3 [10][13].
Recently, 25(OH)D
3, for a considerable time deemed only a metabolic precursor of 1α,25-(OH)
2D
3, has been proved to be an agonist ligand of VDR and capable of initiating rapid, non-genomic actions
[13][14][16,19].
As opposed to their genomic counterpart, non-genomic rapid responses appear rapidly (within a range of seconds or minutes), are not susceptible to cycloheximide or actinomycin D, and also occur in response to steroids coupled to macromolecules which block their cell entering
[15][20]. Therefore, a major difference for discerning between genomic and non-genomic actions is the time course and the sensitivity of transcription and protein synthesis inhibitors.
3. Mechanisms of Membrane-Associated Proteins for 1α,25(OH)2D3-Mediated Rapid, Non-Genomic Actions
Concerning the existence of non-genomic calcitriol actions, t
o our knowledge, the earliest observations for these actions were described by Nemere et al. in 1984
[16][21], who observed that this hormone was able to induce a rapid increase of intracellular calcium (Ca
2+) concentrations both by promoting its release from intracellular stores and by stimulating its intestinal uptake in the vascularly perfused duodenum of normal, vitamin D-replete chicks. They observed that calcitriol significantly increased Ca
2+ transport within 14 min compared with controls in a mechanism independent of genome activation and de novo protein synthesis. This rapid effect on transepithelial Ca
2+ movement across the intestine has been termed transcaltachia.
It is established that the biologically active vitamin D
3 metabolite stimulates different signaling molecules, including phospholipase A2 (PLA2), phospholipase C (PLC), and phosphatidylinositol-3 kinase (PI3K), and promotes the generation of second messengers, such as Ca
2+ ions, phosphatidylinositol (3,4,5)-trisphosphate (PIP3), and cyclic AMP (cAMP), culminating in the activation of several downstream protein kinases (protein kinase C (PKC), calcium/calmodulin-dependent protein kinase II gamma (CaMKIIG), Src, and mitogen-activated protein (MAP) kinases)
[17][18][19][20][21][22,23,24,25,26]. In addition to the above-mentioned non-genomic actions of 1α,25(OH)
2D
3, this secosteroid hormone also mediates the opening of Ca
2+, Cl
−, and Pi channels.
Early studies performed by Norman and colleagues
[22][23][27,28] suggested that a distinct membrane VDR could mediate non-genomic actions in response to 1α,25(OH)
2D
3. The biologically active form of vitamin D
3 promotes the generation of non-genomic responses and, especially in the case of transcaltachia, in its 6-s-cis configuration. By contrast, binding of secosteroid in the 6-s-trans form could be responsible for genomic responses.
This rapid effect was also observed in primary muscle cell cultures isolated from a chicken embryonic heart
[24][43]. In particular, 1α,25(OH)
2D
3 induces a fast increase of both tissue Ca
2+ uptake and cAMP levels within 10 min in primary myocytes. Moreover,
scholarauthors found that this effect was inhibited by a specific protein kinase A (PKA) suppressor, suggesting that the regulation of Ca
2+ ion channel activity by the biologically active form of vitamin D
3 was mediated by the second messenger cAMP.
Baran et al.
[25][44] demonstrated that the secosteroid 1α,25(OH)
2D
3 not only evokes a rapid opening of Ca
2+ channels but also a rapid activation of phospholipase C (PLC) in ROS 24/1 osteoblastic cells lacking the VDR, implying that these effects occur independently of the VDR signaling mechanisms.
This could suggest that, in addition to membrane-associated VDR, the existence of other membrane receptors in conjunction with vitamin D might be essential for rapid, non-genomic effects of 1α,25(OH)
2D
3.
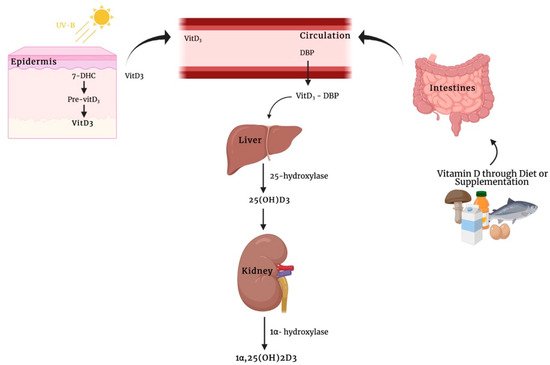
Given the highly liposoluble nature of vitamin D
3, 1α,25(OH)
2D
3, can penetrate biological membranes, and when inside the cells, may interact with heat shock proteins (HSPs) so as to be transferred to the nucleus and the mitochondria
[26][46]. Alternatively, vitamin D
3 metabolites bound to a specific vitamin D
3 binding protein (DBP) can undergo endocytosis through an LDL receptor-related protein 2 (LRP2)- and cubilin (CUBN)-mediated mechanism. Subsequently, 25(OH)D
3 bound to DBP is transported into the proximal tubule epithelium of the kidneys via megalin-mediated endocytosis, where it undergoes a hydroxylation step to transform into 1α,25(OH)
2D
3 [27][28][47,48]. In this light, additional studies should be performed to clarify the physiological significance and the potential mechanistic aspect for vitamin D
3 transporters.
One of best membrane-associated proteins able to bind vitamin D
3 compounds is the protein disulfide isomerase family A member 3 (Pdia3), also known as 1α,25(OH)
2D
3-membrane-associated rapid response to steroid (MAARS), which has been described as a crucial protein in 1α,25(OH)
2D
3-initiated rapid membrane non-genomic signaling pathways
[29][49].
This protein was first purified in the study of Nemere et al.
[30][50], describing the existence of a putative plasmalemmal receptor for the biologically active form of vitamin D
3 involved in the transcaltachia on the basal-lateral membranes (BLM) of chick intestinal epithelium. This conclusion was supported by the observation that an altered, but still present, specific binding for [
3H] 1α,25(OH)
2D
3 was discovered in membrane fractions purified from vitamin D-deficient chicks with respect to the corresponding fractions obtained from normal animals. In addition, the BLM-VDR exhibited down-regulation of specific [
3H] 1α,25(OH)
2D
3 binding upon exposure to nonradioactive 1α,25(OH)
2D
3.
Later studies showed that this candidate plasmalemmal receptor essential for the rapid responses by 1α,25(OH)
2D
3 is Pdia3
[31][32][51,52]. Its primary function is to catalyze the formation, reduction, and isomerization of disulfide bonds, interacting with lectin-like molecular chaperones, calnexin (CANX) and calreticulin (CALR), to ensure the correct folding of newly synthesized glycoproteins
[33][53].
4. 25(OH)D3-Mediated Rapid, Non-Genomic Actions
In the study conducted by Lou et al.
[14][19], it was assumed that calcifediol is an agonist ligand of VDR with anti-proliferative effects and gene regulatory functions, despite the fact it binds to VDR with a lower affinity compared to the biologically active metabolite of vitamin D
3.
Another study by Asano et al. showed an interesting non-genomic mechanism of 25(OH)D
3, where this compound could regulate lipogenesis, thereby reducing the risk of metabolic disease-associated complications by altering sterol regulatory element-binding proteins (SREBPs) activation via the ubiquitin-mediated proteasomal degradation of SREBP cleavage-activating protein (SCAP)
[34][68].
In recent years, 1α,25(OH)
2D
3, the biologically active metabolite of vitamin D
3, has attracted attention due to its involvement in several biological processes, including the regulation of the serum levels of calcium and phosphate, as well as its influence on bone and mineral metabolism.
There is now compelling evidence that 1α,25(OH)
2D
3 affects the target cells through genomic pathways and membrane receptor-mediated rapid, non-genomic responses. This latter mechanism has also been described for virtually all the steroid molecules, such as aldosterone, testosterone, estrogens, and cortisol
[13][29][35][16,49,69].
Recently, even the direct metabolic precursor of 1α,25(OH)
2D
3, named calcifediol, it has been revealed able to activate rapid, nontranscriptional actions, such as an acute and sustained rise in intracellular Ca
2+ levels, similarly to that observed with the biologically active form of vitamin D
3 [13][36][16,70].
Although growing evidence has led to a significant knowledge concerning calcifediol and calcitriol rapid, non-genomic activities, their impact on physiological processes needs to be clarified. In fact, it is difficult to identify vitamin D-deficiency-associated diseases which occur exclusively due to aberrations of its rapid actions.