2. Composition of the YAP and Hippo Signaling Pathways
Yes-associated protein (YAP) is a key member of the Hippo signaling pathway. It was originally identified in
Drosophila, with a molecular weight of 65 kDa, and is also known as YAP65
[12][13]. YAP was confirmed to be encoded by the
yap gene on human chromosome 11q22. It is a proline-rich protein that plays a role in controlling the cell number and organ size in
Drosophila development by coordinating cell proliferation, the cell cycle, and apoptosis
[13][14][14,15]. The Hippo signaling pathway includes three main components: upstream kinase cascade initiation, kinase cascade core, and transcription-related activity. The core components of the mammalian kinase cascade include Mst1/2, Sav1, LATS1/2, Mob1, YAP, and TAZ (transcriptional coactivator with the PDZ-binding motif). Dong et al.
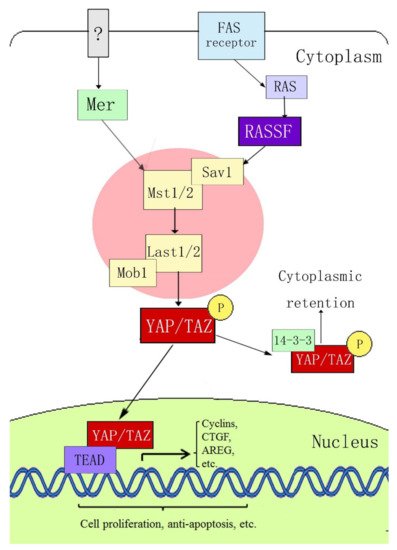
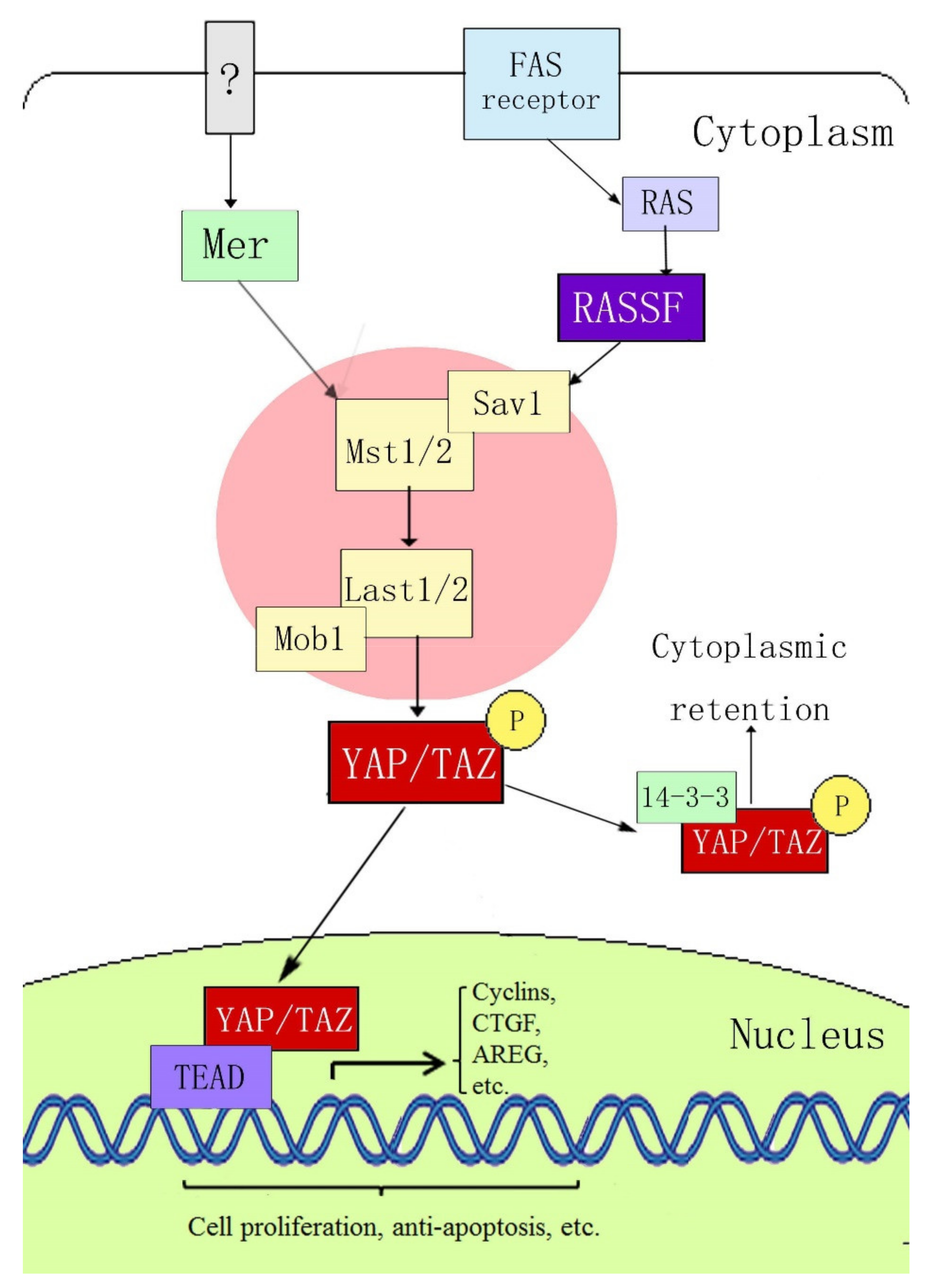
[15][16] first determined the signal transmission sequence of the Hippo–YAP pathway in mammals; when an external stimulation signal activates the Hippo pathway, Mst1/2 and Sav form a complex, which binds to LATS1/2 through the adaptor protein WW45, resulting in activation of LATS1/2, which subsequently phosphorylates YAP at the ser127 site. YAP transcriptional activity depends on its subcellular localization: YAP is active in its dephosphorylated state and can thus enter the nucleus and activate gene transcription by interacting with transcription factors TEAD (TEA domain family members) 1–4, Smads, p63/p73, Runx, and ErbB4. When phosphorylated through the upstream kinase cascade, YAP binds to the cytoplasmic 14-3-3 protein, is inactivated, and remains in the cytoplasm, where it cannot act as a transcription factor (
Figure 1).
Figure 1. Hippo–YAP signaling pathway.
3. Possible Mechanisms of YAP in Psoriasis and Skin Tumor Pathogenesis
YAP and its upstream pathway (the Hippo signaling pathway) play important roles in regulating cell proliferation and apoptosis to regulate the growth, development, and size of tissues and organs; epithelial mesenchymal transformation (EMT); intercellular contact inhibition; and stem cell self-renewal. Tumor research accounts for the vast majority of studies on YAP
[16][17][17,18]. YAP is highly expressed in lung cancer
[18][19], breast cancer
[19][20], ovarian cancer
[20][21][21,22], colon cancer
[22][23], liver cancer
[23][24], and other cancers. In some tumors, YAP expression is positively correlated with the survival rate and degree of tumor malignancy. Psoriasis and tumors are characterized by excessive cell proliferation, abnormal differentiation, vasodilation, and hyperplasia. Therefore, anything that can affect these pathophysiological mechanisms may be a key link in their pathogenesis.
3.1. YAP Regulates Apoptosis
The importance of the Hippo–YAP signaling pathway in regulating apoptosis has been confirmed by a number of studies. In
Drosophila, the Yorkie (Yki) protein, a homologue of YAP, can inhibit the activity of the
Drosophila apoptosis-related gene
reaper (RPR) by promoting the binding of p53 to ASPP1 (the apoptosis-stimulating protein of p53). Yki can also inhibit the activation of defective head involvement (HID), both of which inhibit
Drosophila apoptosis
[24][25]. The Hippo–YAP signaling pathway can also increase the expression of
Drosophila inhibitor of apoptosis protein 1 (DIP1)
[25][26]. YAP also inhibits apoptosis in human periodontal ligament stem cells
[26][27], human liver cancer cells
[27][28], endometrial stromal cells
[28][29], and meningioma cells
[29][30].
YAP can also promote apoptosis in some cases. During DNA damage, the promyelocytic leukemia gene (PML) recruits YAP and p73 into the nucleus. YAP acts as a transcriptional activator to enhance binding with p73 and activates the transcription of apoptosis-related genes
[30][31]. YAP accelerates amyloid-β-peptide (Aβ)-induced apoptosis through nuclear translocation, induces p73-mediated Bax expression and activation, and promotes the activation of the apoptosis-related protein caspase-3
[31][32]. In addition, YAP mediates c-Jun-dependent apoptosis
[32][33].
3.2. YAP Regulates Cell Proliferation and Differentiation and Maintains the Three-Dimensional Structure of Skin
In mice and
Drosophila, YAP overexpression induces the proliferation of undifferentiated intestinal progenitor cells. Reduced YAP expression induces the differentiation of intestinal progenitor cells. YAP activation or inhibition of upstream negative regulatory proteins can induce the activation or enhancement of the Notch and Wnt signaling pathways, which are related to the inhibition of stem cell differentiation and promotion of stem cell proliferation
[33][34][34,35]. Schlegelmilch et al.
[35][36] confirmed that YAP regulates epidermal stem cell proliferation and maintains the three-dimensional structure of the skin by interacting with the transcription factor TEAD; the epidermis of YAP-knockout mice became thinner, the stratum corneum decreased, and the arrangement of the epidermal structure became disordered. Zhang et al.
[36][37] also reported high expression of YAP in monolayer basal epidermal progenitor cells, and that the expression of nuclear YAP decreased gradually with age, which was related to the proliferation potential of epidermal progenitor cells. YAP promotes the proliferation of basal epidermal progenitor cells and inhibits terminal differentiation. In vitro studies have shown that, when YAP is activated, the proliferation rate of primary mouse keratinocytes (MKSs) increases, whereas the rates of differentiation and apoptosis decrease, and these characteristics are reversed after inhibiting YAP expression. YAP acts as a molecular switch for epidermal stem/progenitor cell activation. The C-terminus of YAP was found to regulate the balance between stem/progenitor cell proliferation and differentiation in the postnatal interfollicular epidermis
[37][38]. Overexpression of YAP can promote immortalized proliferation of human primary keratinocytes, hinder their normal differentiation process, increase the expression of the epithelial proliferation markers p63 and PCNA, and decrease the expression of the epidermal differentiation markers 14-3-3σ and LEKTI
[38][39]. YAP knockout resulted in a reduced level of expression of transforming growth factor (TGF)-β, a decrease in the proliferation rate of epidermal basal cells, and hindrance of skin wound healing. Interestingly, skin wound healing is dependent on YAP expression
[39][40]. YAP is mainly localized in the cytoplasm of differentiated cells
[40][41]; however, when epithelial tumor cells lose polarity and exhibit enhanced invasiveness, YAP is mainly located in the nucleus. Therefore, the regulation of YAP subcellular localization is crucial for the conversion between proliferation and terminal differentiation
[35][36].
3.3. YAP Regulates Cell Density and Intercellular Contact Inhibition
Contact inhibition refers to the biological characteristic in which cells stop growing because of mutual contact. It is an important regulatory mechanism that maintains the normal morphology of body tissues and prevents disordered cell proliferation in vivo. Contact inhibition allows the cells to stop proliferating when they accumulate in large numbers. Two notable characteristics of many tumor cell lines cultured in vitro are their lack of contact inhibition and the ability to grow without support
[41][42]. When the intercellular density increases, YAP localizes to the cytoplasm, and the activities of cell proliferation-related genes and apoptosis-inhibiting genes are inhibited. In contrast, at a low cell density, YAP localizes to the nucleus and its proliferation-inducing activity is increased; that is, contact inhibition between cells occurs and is mediated by YAP
[42][43]. The integrated membrane protein angiomotin (AMOT) and related proteins can inhibit YAP activity by chelating and binding with YAP in the cytoplasm, thereby inhibiting cell proliferation and intercellular contact and maintaining normal cell density
[43][44]. Zhang et al.
[44][45] found that at high cell densities, overexpression of 14-3-3ζ (an apoptosis inhibitor protein) in human umbilical cord mesenchymal stem cell-exosomes (hucMSC-EX) promoted the binding of LATS to YAP, enhanced the phosphorylation of YAP at ser127, and inhibited YAP activity. After transfecting YAP into the non-neoplastic breast epithelial cell line MCF10A by retrovirus, Overholtzer et al.
[45][46] found that overexpression of YAP could abolish the cell contact inhibition effect, promote growth-factor-independent proliferation, inhibit apoptosis, promote epithelial mesenchymal transition (EMT), and anchor independent growth in soft agar. Taken together, these changes are indicative of malignant transformation of cells.
3.4. YAP can Regulate Angiogenesis
The main pathological feature of psoriasis is the painful expansion of superficial dermal vessels with a clinical manifestation, such as the Auspitz sign
[46][47]. Tumor tissues require a nutrient supply, which is often accompanied by extensive angiogenesis
[47][48]. Choi et al.
[48][49] demonstrated that YAP is an important regulator of angiogenesis in mice; after phosphorylation, YAP is inactivated and then redistributed in a cell-contact-dependent manner through E-cadherin. In mice, YAP knockout is associated with a significant reduction in the number of endothelial buds in the common duct network and aortic ring. During angiogenesis, the vascular endothelial growth factor (VEGF)–VEGF receptor 2 (VEGFR2) signaling axis depends on YAP/TAZ activation
[49][50]. VEGFR regulates YAP/TAZ through the Rho GTPase, mitogen-activated protein kinase (MAPK), and phosphatidylinositol 3-kinase (PI3K) pathways to regulate angiogenesis
[50][51][52][53][51,52,53,54]. During postnatal development of human umbilical vein endothelial cells (HUVECs) and mouse retina, Ang2 is a key YAP target gene in endothelial cells that mediates the regulation of angiogenesis and vascular remodeling by YAP
[54][55].