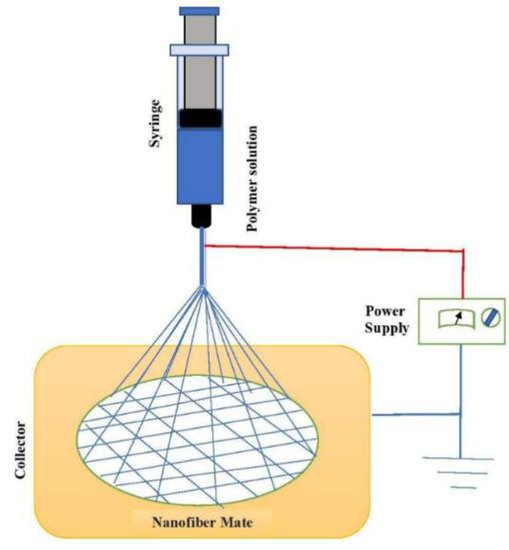
3.1.2. Nerve Regeneration
Peripheral nerve injuries (PNI), caused by traffic accidents, natural disasters, and unsatisfactory treatments
[32][85], are intractable clinical problems that create heavy burdens for patients. Therefore, various electrospinning nanofiber materials play a considerable role in nerve regeneration. So, an aligned fibrin/functionalized self-assembling peptide (AFG/fSAP) interpenetrating hydrogel was fabricated by electrospinning and molecular self-assembly, where the prepared hydrogels show synergistic topographical and biochemical cues
[32][85]. The scaffolds with aligned structures illustrated considerable impact on the regeneration of peripheral nerves
[32][85]. Also, they upregulated regeneration-associated gene expression and activated PI3K/Akt and MAPK signaling pathways in regenerated nerves.
Aligned chitosan nanofiber hydrogel (AChiG) incorporated with a bioactive peptide mixture composed of Ac-RGIDKRHWNSQGG (RGI) and Ac-KLTWQELYQLKYKGIGG (KLT), named AChiG-RGI/KLT, was applied as nerve conduit filler to remodel sciatic nerve defects in rats. AChiG-RGI/KLT oriented the Schwann cells, and supported the proliferation and excretion of neurotrophic factors by Schwann cells. At an early infection stage, AChiG-RGI/KLT promoted nerve regeneration and enhanced vascular infiltration. After twelve weeks, AChiG-RGI/KLT facilitated nerve repair and functional restoration in rats
[33][87].
3.1.3. Vascular Tissue
Recently, electrospinning has been utilized to synthesize nanofiber-based scaffolds. Several works have investigated and described the applications of nanofiber-based scaffolds for vascular scaffolds of small diameter and vascular grafts
[34][35][36][37][38][39][40][88,89,90,91,92,93,94]. Though the vascular scaffolds fabricated by present electrospinning techniques can mimic the compositions of human blood vessels, these technologies have many difficulties in fabrication of vascular scaffolds of small-diameters (<1.5 mm)
[34][88]. Therefore, a biodegradable poly
l-lactide-co-caprolactone (PLCL) with biomimetic mechanical characteristics was employed to fabricate small diameter <1.5 mm PLCL/tussah silk fibroin (TSF) nanofiber vascular scaffolds for grafting. The biological behaviors of PLCL/TSF nanofiber vascular scaffolds were tested by in vitro culture of vascular endothelial cells (ECs). The scaffolds efficiently allowed vascular endothelial cell adhesion and proliferation
[34][88]. Also, to mimic and fabricate the specific structures of natural blood vessels, a new approach has been developed
[35][89]. In this process dual-oriented/bilayered small-diameter tubular nanofiber scaffolds were prepared by a mixture of PCL, poly
d,l-lactide-co-glycolide (PLGA) and gelatin. The two bilayered nanofibers were orientated perpendicular to each other, aiming at guiding cell-specific orientation of smooth muscle cells (SMCs) and endothelial cells (ECs) in vitro, respectively. The findings revealed that the presence of gelatin highly induced the hydrophilicity of the scaffold as well as its mechanical property. The in vitro degradation indicated that by blending of three biodegradable polymers, the degradation rate of the scaffold accelerated. Moreover, electrospun scaffolds could enhance proliferation of both SMCs and ECs. Furthermore, topographic cues signed by oriented nanofibers could direct the growth and orientation of smooth muscle cells and endothelial cells
[35][89].
3.1.4. Skin Tissue Engineering
Skin is the outermost and the most important barrier organ in the body and plays a vital role in protection of internal tissues from external damage
[41][42][43][96,97,98]. Therefore, the protection of this barrier against damage is vital to prevent microorganisms from penetrating and forming infections in wounds and other dangerous side effects. Also, the protection and healing of wounds are very urgent factors in keeping patients safe and healthy
[42][43][97,98]. Recently, the development of novel nanomaterials for wound dressing and antibiotic agents is one of the more important challenges facing current medical technological innovations
[44][99]. Thus, many researchers have been motivated to fabricate an ideal wound dressing and antibiotics with appropriate properties using nanomaterials, such as different types of fabricated electrospun nanofibers to enhance wound healing. Electrospun nanofibers have opened new ways to synthesizing and fabricating of novel materials to be used in skin tissue engineering through nano/microscale polymeric fibers, inorganic/organic compositions, biomaterials, elastomers, and other types of materials.
Polymeric nanofibers can be incorporated with nanofillers, such as nanoparticles and nanotubes, to improve their superior properties and enhance applications in skin tissue engineering. Electrospun nanofibers of glucose (G)-reduced graphene oxide (rGO) (0–1.0 wt%) blended with PVA to form PVA/GrGO scaffolds, referred as (PG) scaffolds, and crosslinked chemically with acidic glutaraldehyde (GA) in acetone medium to mimic the (ECM) to apply in skin tissue engineering. Further increase in concentrations of G-rGO in PG scaffolds cause a decrease in tensile strengths and elongations, increasing the thermal properties. The biological properties of PG scaffolds were examined using in vitro hemolysis, using CCD-986Sk (a human skin fibroblast cell line). Results indicated G-rGO incorporation in PVA nanofibers induced a small shift from hydrophilic to hydrophobic. Moreover, the PVA/G-rGO scaffolds did not possess hemolysis of red blood cells, even at a G-rGO immobilizing of 1.0 wt%, and PG-1.0 scaffold (with a GRGO loading of 1.0 wt%) there was good compatibility with fibroblasts and highly increased metabolic activity after seed for twenty-one days, as compared with PG-0 controls
[41][96].
Another nanofiber scaffold was synthesized from PU and cellulose acetate via electrospinning. RGO/Ag nanocomposite and/or curcumin was incorporated with the materials attributed to the antibacterial activity of rGO/Ag-nanocomposite. The scaffolds could prevent both of Gram-negative and Gram-positive bacteria via direct contact with them. In vivo histopathological experiments demonstrated that the scaffolds of the blended rGO/Ag nanocomposites and curcumin showed excellent wound healing and could promote the healing rate of artificial wounds, which provides considerable biomedical potential for nanomaterials in wound healing
[42][97].
4. Conclusions
Nowadays, electrospun nanofibers with superior performance have illustrated considerable and promising results for biomedical applications. Their unique characteristics makes them convenient to apply in various fields. However, fabrication of nanofibers with distinct morphological and superior properties and considerable yield is still challenging. Such fabricated nanofibers can be functionalized through various techniques to widespread application in different strategic fields. Hence, considerable developments have been achieved even though there are still various challenges. Much effort has been put into improving desired performance of fabricated electrospun nanofiber and functionalized nanofiber application in specific areas. However, further work still needs to be done to obtain more promising results and eventual practical electrospun nanofibrous materials by advanced electrospinning techniques and scaling up the fabrication of nanofibers from the laboratory to commercial scales, especially for biomedical applications. The development of nanomaterials/biomaterials from biodegradable, biobased electrospun nanofibers is imperative to improve the applications of electrospun nanofibers in diverse fields, such as biomedical applications.
Also, electrospun nanofiber materials can play a vital role to overcome many challenges facing tissue engineering technologies in regeneration of tissues and organs where the scaffolds fabricated by electrospun nanofibers materials can mimic 3D with other physiological properties of tissues and organs in vitro to then transplant in vivo. Also, they are promising materials to regenerate injured nerve through tissue engineering technologies. Another serious challenge facing tissue engineering technologies to mimic and fabricate nerve grafts is losing the structural integrity, or biological functionality, of some natural polymer nanofibers used in a media simulating that in the human body. Therefore, further intensive research is required to explore and fabricate new materials to overcome such challenges. Using nanomaterials has many disadvantages, such as aggregation, uncontrollable release, costs, and potential cellular toxicity. Therefore, further research is needed to avoid such disadvantages.
On the other hand, current utilization of electrospun nanofibers in biomedical applications, such as bone cell proliferation, nerve regeneration, vascular tissue, and skin tissue engineering has been demonstrated. Many of these results were obtained for small rodent models which may be accompanied by restrictions regarding their immunological responses compared with humans. Therefore, these studies must be confirmed with suitable, and larger, animal models.