Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Prakash Parthasarathy and Version 2 by Vivi Li.
Processing significant amounts of dye effluent discharges into receiving waters can supply major benefits to countries that are affected by the water crisis and anticipated future stress in many areas in the world. When compared to most conventional adsorbents, biochars can provide an economically attractive solution. In comparison to many other textile effluent treatment processes, adsorption technology provides an economical, easily managed, and highly effective treatment option.
- effluents
- dye removal
- biochar
- adsorption
- dye absorption capabilities
1. Introduction
Dyestuffs color and pollute receiving waters, streams, and rivers as a result of inadequate processing of the industrial effluents by a variety of industrial applications including the food and beverage companies, paper and pulp processing, paint manufacturing, pharmaceutical processing, printing, textiles, dyeing, and printing [1]. Many dyes pose a grave danger to the water environmental ecosystem due to their chemical properties, with serious consequences for human health, animal, and plant ecosystems [2][3][2,3]. Aside from a limited number of studies indicating that specific dyes are toxic, the presence of dyestuffs into receiving waters reduces the photosynthetic process by inhibiting light from passing through [4]. During the degradation process, dyes consume the dissolved oxygen concentrations of the receiving water, therefore decreasing the water quality standards for aquatic species. This has detrimental visual aesthetic impacts which may result in health reproductive issues in fishes [5]. Specific dyes have a negative impact on the skin, kidneys, liver, reproductive system, heart, brain, and nervous system, and some may be carcinogenic or mutagenic.
Data on dyestuff effluent discharge volumes and production quantities are not readily available or recorded around the world. According to available data, 700,100 tons of dyestuffs are produced every year for 10,000 dyes. According to industry figures, the global dyestuffs produced yearly is 1.8 to 1.9 × 106 tons with more than 11,000 dye pigments applied primarily in the food, textile, cosmetics, leather, paper, and plastics industries [6]. Depending on the type of dyestuff and the process technology used, 1–10% of dye is not used in the dyeing process, indicating that significant amounts of dye are discharged to the water bodies via various means [7].
The majority of dyestuffs have specific characteristics such as chemically stable and light fastness [8]. Furthermore, the dye color reduces light penetration in streams and rivers, therefore reducing photosynthesis and dissolved oxygen content. They prevent a variety of chemical functions based on the material to which they are applied and the color they impart (Figure 1). All of these properties are advantageous to the dye user and are enhanced by dyestuff manufacturers. However, the huge volumes of effluent makes the treatment of these dyes to comply with environmental effluent discharge standards very problematic. In addition, water can be colored in certain cases with dye concentrations as little as 1 ppm. The majority of dyeing applications employ copious amount of water during the dyeing, washing, and rinsing stages [9][10][9,10].

Figure 1.
(
A
) Dye house discharge. (
B
) River quality affected by dyestuffs.
2. Dye Classifications
Dyes are colored molecules or ions that can be applied to a wide range of materials including food, beverages, and textiles in solution or as a dispersion. Most dyes have a high-water solubility, and often contain a sulfonic acid group, usually in the form of a sodium salt, which is responsible for the solubility of many water-soluble dyestuffs [11]. Dye colors are created by chemical groups absorbing light of various wavelengths in the visible region of the spectrum. Different unsaturated chemical groups on chromophores promote this key distinguishing property. Figure 2 depicts the more common ones.
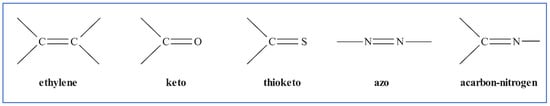
Figure 2.
Color-producing chromophores or groups.
Auxochromes are groups that can also enhance the water solubility and improve the dye absorption potential for adsorbing material; examples include substituted sulfonic, hydroxyl, carbonyl, or amino groups. Dyes can be classified based on their chemistry or their types of application. As a result, the chemical structure and type of dye must be a primary consideration in determining which dye wastewater process treatment technology should be applied for effluent removal, as well as determining what adsorbent properties are required for the adsorption of the specific dyestuff type.
2.1. Reactive Dyes
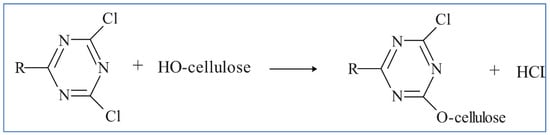
Reactive dyes are used extensively in the dyeing of cellulosic textile fibers, namely, flax and cotton. Due to their high adhesion to a substrate, they can also be used to dye linen, viscose, and silk [12][13][12,13]. These reactive compounds in the dye can form chemical bonds with textile uptake of fibers. The uptake of the dichlorotriazine type of reactive dye which becomes attached to the cellulose fiber by displacing the chloride grouping is depicted in the mechanistic schemes below. One or both chlorides may be present. Figure 3 and Figure 4 show the typical dye uptake mechanisms for dyeing cellulosic materials.

Figure 3.
Typical mechanisms for dyeing cellulose.
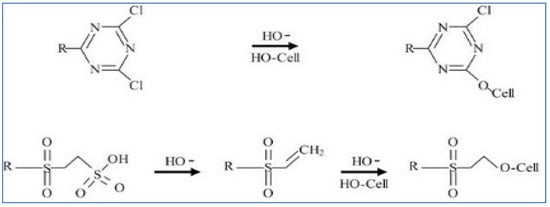

Figure 4.
Reactive dyeing mechanism on cellulose.
Due to reactive dyes having a strong bonding affinity for cellulose, consequently, the hydroxyl group containing biosorbents have demonstrated a very strong dye uptake capacity to remove reactive dye compounds from textile dyehouse effluents [14].
2.2. Disperse Dyes
Disperse dyes are non-ionic substances that are commonly applied to polyesters but can be used in acetate or nylon fabrics. These dyes are water soluble and can be used for these fibers by diffusion into the fibers at increased temperatures. As there are no basic chemical groups, there are no attractive sites for acid dye groups, despite a weak attraction for basic dyes. The dye attachment mechanism is based on weak Van der Waals forces and dipole-dipole interactions, implying that like mechanisms may occur during the removal of disperse dyes onto biochar adsorbents [15]. Figure 5 depicts disperse blue 6 as an example of this class.

Figure 5.
Disperse Blue 6 dye compound.
The dispersed dyestuff occurs typically as a fine suspension that can be filtered from the effluent discharge by biochar.
2.3. Vat Dyes
The majority of vat dyes have a ketonic style chromophore which may be applied to color cellulosic fibers and materials such as viscose, cotton, and linen. This is a broad category of dyes that includes indanthrones, anthraquinones, carbazoles, benzanthrones, polycyclic quinones, and acridones.

Figure 6.
Structure of the vat red 13 dye.
The large anthraquinone groups suggest that the removal process may involve electron clouds of the anthraquinone dye by adsorption onto the positively charged surface groups and adequately sized pore diameters of biochars.
2.4. Direct Dyes
Direct dyes or substantive dyes, as there is no fixation phase necessary, may be applied to color cotton yarn, viscose, and loose cotton of fabrics [17]. Mordant chemicals, such as chromium compounds that can undergo complexation by attaching substrate to chromophore to form an insoluble color, are used in some direct dyes, but not all, to fix the dye and improve color fastness. In the case of dark color shades such as black or navy blue dyes, this technique has proven to be cost-effective in achieving high color fastness. These dyes are now being reviewed due to environment and safety concerns which have limited their use. The mechanism for the application of direct dyes involves establishing non-ionic forces to attach the dyestuff to the textile fiber material [18]. The structure of direct yellow 24 dye is depicted in Figure 7.

Figure 7.
Structure of the direct yellow 24 dye.
Direct dyestuffs produce negatively charged ions in solution and can be adsorbed onto positive sites on biochars.
2.5. Basic Dye
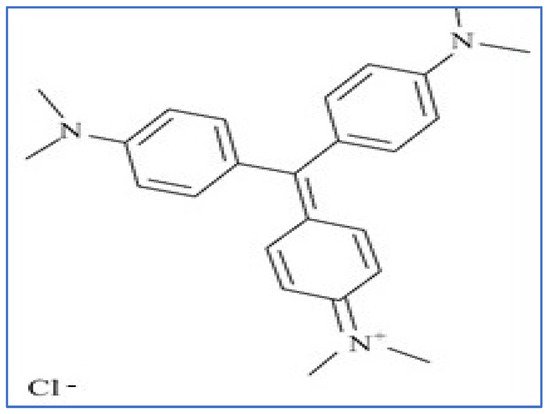
Congo red (CR) is a member of the very large group of basic dyes that are characterized by the color, high tinctorial strength, and brilliance. These basic dyes are most commonly used on acrylic fibers, but they can also be used on other fibrous textiles when mordants are used. Furthermore, basic dyes are soluble media but not soluble in alkaline solutions. These dyes are primarily made up of imino or amino groups that are linked to triarylmethane or xanthene; they are also used in typewriter ribbon, carbon paper, and inks [19]. Monoazo, methane, and oxazine are the three main subclasses. Figure 8 depicts the structure of a basic CR dye.

Figure 8.
Structure of basic crystal violet (CV) dye.
40][103], two capacities for the removal of CV are listed: 175 mg/g and a quite high value of 1220 mg/g. The capacity values reported in the literature range from 600 mg/g for a bentonite-alginate composite [56][119] to 75 mg/g for chitosan hydrogel beads [57][120]. Table 1 shows the final values for the adsorption of BR9 onto biochar materials from animal residue [48][111] after 1 and 3 h of heat treatment. At one (90 m2/g) and three hours (95 m2/g), the BR9 capacities were 50 and 52 mg/g, respectively. The general trend in surface areas and pyrolysis times was followed by these capacities. Literature values are slightly lower but of relative magnitude, for example, 29 and 15 mg/g for sepiolite [58][121] and fish bone [59][122], respectively.
Table 2 shows the anionic dye adsorption properties on unmodified biochars. Acid orange 7 (AO 7) [41][104], CR [33][34][38][39][40][42][45][60][61][62][63][96,97,101,102,103,105,108,123,124,125,126], reactive red RR 120 [64][127], Remazol violet 5R (RV5R), Remazol orange 3R (RO 3R), Remazol blue R (RBR) [65][128], orange G (OG) [45][108], and methyl orange (MO) [66][129] are some of them.
Table 2 shows only the dye removal composition (%) for the adsorption of AO7 using biochar derived from groundnut shell. Only a few instances of AO7 adsorption capacity potential are documented in articles, and they range from 50 to 180 mg/g on fly ash [67][130], oxihumolite [68][131], and chemically reactivated sawdust [69][132].
72][135]. Microwave treatment was used following phosphoric acid activation to synthesize mesoporous activated carbon. The carbon showed a higher surface area and a 350 mg/g adsorption capability.
At temperatures between 513 to 553 K and reaction durations varying from 0.5 to 6.0 h, many bamboo biochars were generated [73][136]. Their CR absorption capabilities ranged from 30–100 mg/g, with the maximum values seen in biochars generated at high temperatures of 513 K and 553 K, as well as the longer treatment times of 5 and 6 h. After only 4 h at 523 K, an activated carbon obtained from apricot seeds [71][134] had a low CR capacity of 33 mg/g. This was ascribed to the small pyrolysis temperature and specific surface area. The latter two investigations’ adsorption capacity potential was similar to that of date seed carbon. More research into the production of biochars through microwave and plasma pyrolysis techniques should be conducted.
Anionic reactive red (RR) 120 was adsorbed at a high (330 mg/g) capacity on biochar synthesized from Eucheuma spinosum [64][127]. Fe3O4-activated magnetic nanoparticles [74][137] and activated carbon [75][138] have high capacity values in the literature, exhibiting adsorption capacity potentials of 165 and 255 mg/g, respectively. Biochar made from green sea algae [65][128] has been reported to remove brilliant violet 5R, Remazol, and brilliant orange 3R dyes with removal percentages of more than 70%. On coffee shell activated carbon [76][139] and calcined eggshell [77][140], the reported figures for Remazol dyes are relatively low, at 65 and 15 mg/g for brilliant orange 3R and brilliant violet 5R, respectively.
Table 2 demonstrates that biochar derived from switchgrass [45][108] generated at 873 K displayed a poor orange G adsorption capacity of 8 mg/g. The poor surface area (255 m2/g) of the char could have contributed to this low capacity. Other published values include 9 mg/g for activated carbon derived from Thespesia populnea [78][141] and 19 mg/g for nanoporous activated carbon [79][142]. All of these values indicate that orange G dye is one to be treated. When it came to adsorbing MO, corn cob char [66][129] had an adsorption capacity potential of 85 mg/g, while amidoxime char [80][143] had a potential of 140 mg/g.
MB adsorption capacities (Table 1) are 110, 150, 38, and 195 mg/g for biochar materials derived from microalgae [42][105], Wodyetia [44][107], and switchgrass at 873 and 1173 K [23][45][45,108], respectively. Pyrolysis temperature significantly influences the adsorption potential of switchgrass biochar. MB adsorption capacity values in the literature for sugarcane bagasse [46][109], phosphoric acid-treated olive seed carbon [49][112], and bamboo cane active carbon [50][113] are 110, 135, and 455 mg/g, respectively. Biochar adsorption capacities for MG dye are remarkably decent, with values of 12,500 mg/g on crab shell [38][101], 1740 mg/g on wheat/bran straw-fed larvae biochar [39][102], and 5300 mg/g on biochar synthesized from Ulothrix algae [40][103].
The adsorbent potential values reported in the literature for activated carbon made from grape processing residue [51][114], shrimp shell [52][115], and plastic waste [53][116] are 665, 320, and 1430 mg/g, respectively. The amount of Rh B adsorbed on the surface of biochar sourced from tapioca shell is 33 mg/g [36][99] compared to reported values of 77 using Acacia mangium wood-derived carbon [54][117] and 30–40 mg/g on activated carbons from carnauba, macauba, and pine nut wastes activated using calcium chloride and phosphoric acid [55][118].
On biochar from mango leaves [39][102] and Ulothrix zonata algae [
As CR is one of the most researched anionic dyestuffs, the citations in Table 2 are merely illustrative. Adsorption capacity potential of biochars derived from chlorella microalgae species [42][105], phoenix dactylifera [62][125], cotton stalk [63][126], orange skin [60][123], carapace (crab shell) [38][101], activated carbon [61][124], spirulina algae species [33][96], wheat bran larvae [39][102], switchgrass (charred at 873 K and 1173 K) [45][108], and Ulothrix algae species [40][103] are 160, 25, 250, 90, 20,315, 230, 85, 8, 23, and 345 mg/g. The study on switchgrass indicated that at elevated pyrolysis temperatures, a high-quality biochar is generated.
Most investigations have found that the CR dye adsorption capacity potential is below 100 mg/g. The maximum value was observed from pyrolyzed crab shell with 80 m2/g surface area. This result was achieved at a pH of 4 and volume to mass ratio of 2; nonetheless, at a CR concentration above 20 g/L. The activated carbon obtained from date stone exhibited a low adsorption capacity potential of 35 mg/g. The huge dye molecular size (695 g/mol) and the total pore volume of 0.086 are responsible for the poor capacities. CR dye, unlike other anionic acid dyes, is a direct dye with no anionic bonding characteristics [70][71][133,134]. The significance of examining adsorbent pore size distribution is shown by this phenomenon [
On Fe/Mn impregnated fronds pyrolyzed at 973 K [84][147] and 1073 K thermally treated fronds [81][144], the biochar from date palm frond [41][104] exhibited an MB adsorption capacity of 205 mg/g, whereas the biochars obtained from modified date frond demonstrated capacities of 300 and 210 mg/g. Biochar made from tapioca skin has a 30% MG removal capacity and a 65% Rh B elimination potential [36][99]. The removal capacity of biochar made from sulfur-doped tapioca peel was 75 and 90%, respectively [36][99]. 30 and 33 mg/g were reported as the greatest adsorption potential.
The sulfur-doped tapioca skin was coated with chitosan at 873 K, which raised the dye absorption capabilities to 50 mg/g for MG and 40 mg/g for Rh B, respectively [82][145]. Unaltered areca plant biochar showed an MG capacity of 190 mg/g [43][106]. After activating with K2FeO4, the adsorption potential of biochar prepared from wakame increased to 250 mg/g. The biochar sourced from chlorella microalgae displayed an MG adsorption capacity of 110 mg/g and is one among many seaweed/algae species biochars with potentials lying between 25 and 130 mg/g. The adsorption capability of MG on biochar obtained from chlorella was close to 80 mg/g [89][151].
Only 10 mg/g capacity of Rh B was accepted by seaweed biochar. Biochar made from calcined wakame seaweed, on the other hand, has extraordinarily high adsorption capabilities for MG, Rh B, and MB, with 4065, 840, and 530 mg/g, respectively [85][148]. Biochar generated from rice husk had an MG adsorption capacity of 65 mg/g [37][100]; after alteration with Cu + Al [87][149], the potential elevated to 470 mg/g. Biswas et al. [46][109] found that biochar made from sugarcane bagasse had a potential of 70 mg/g, while biochars made from steam-activated bagasse and ZnCl2-modified bagasse had capacities of 5220 and 90 mg/g, respectively [46][109].
Since most basic dyes ionize in water, they have a positively charged colored ion or cationic species. These dyes are commonly classified as cationic dyes and adsorb most effectively onto negatively charged functional groups on the biochars [20].
3. Adsorption of Dye onto Biochar Materials for Dye Removal
3.1. Biochar as a Dye Removal Adsorbent
Activated carbons are amorphous carbon-based substances with decent porosity and large internal surface area. Its feedstock can be almost any organic substances with a relatively decent carbon composition, covering traditional materials such as hard and softwood, coconut hull, peat and lignite coal, natural and artificial polymers. Surface areas for marketable carbons typically fall between the 500–1500 m2/g range [21][86] and can even reach 3000 m2/g. Carbon materials are again classified into two sub-categories based on whether they are used to remove pollutant particles from fluids. The first ones are typically microporous with a pore size of 2 nm diameter and are usually granular, whilst the latter are mesoporous materials with a pore size ranging between 2–50 nm diameter and are usually in powder form [22][71]. Both types are useful in wastewater treatment, where they aid in decolorization, odor removal, metal recovery, and organics adsorption. The pore volume, internal surface area, and size distribution are all proportional to its adsorption capacity. Organics have been reported to adsorb in pores just fitting the adsorbate molecule [23][45]. Humic acids and dyes with size ranging between 1.5 and 3.0 nm that support adsorption phenomenon in mesopores are examples [24][87]. As a result, the biochar’s pore size dissemination influences its adsorption potential for ions of varying size and shape. The electric strength between the adsorbate and the adsorbent (carbon surface) has proven to improve dye removal efficacy greatly. The dissociation equilibria and functionality of specific functional sites on biochar material surfaces, such as carboxylic-lactonic groups, phenolic-alcolohic hydroxyl groups, aromatic-heterocyclic carbons, ketone-carbonyl groups, pyridinic-N, pyrollic-N, and quaternary-N nitrogen species, can influence adsorption. These potential biochar surface sites are influenced by several factors, including biomass source, pyrolysis parameters such as heating rate, temperature, residence time, nature of pyrolysis, etc. Despite that activated and modified carbons are broadly used as adsorbent materials, they are relatively expensive due to the high costs of raw materials, energy, and chemical production. As a result, many researchers have focused on developing novel, high-capacity, low-cost adsorbents obtained from biomass residues. Metal organic frameworks and nano-adsorbent substances have lately been used to create highly efficient adsorbents [25][88]; however, the cost of treatment renders these materials prohibitively expensive. As a result, in recent years, several low-cost adsorbents known as biochars have been produced by biomass pyrolysis and used in polluted water treatment applications. The technique is affordable and cost-effective only when the adsorbent is inexpensive and copious [26][89]. Pyrolysis of biomass leftovers into value-added biochar materials is a cost-effective process that produces high-value-added products: syngas and bio-oil. The pyrolysis process requires energy to run, but the process is driven by the by-products of the side reactions, and biochars possess a larger surface area and pore volume, as well as chemically functional moiety content, making them a much more potent adsorbent material than the biomass feedstock [27][28][29][30][90,91,92,93]. There are over a thousand papers on color removal in the literature, with over a hundred of these based on dye elimination employing biochar substances, including biochar products derived from vermicompost, cabbage residues, algae, and animal litters [31][32][33][34][35][94,95,96,97,98]. There have also been numerous publications on the synthesis and usage of altered biochar materials for dye color elimination. The dye potentials of unmodified/unaltered biochars and others are shown in Table 1 and Table 2. Table 1 shows the adsorption properties for cationic dye uptake onto unmodified biochars. MG [36][37][38][39][40][99,100,101,102,103], MB [41][42][43][44][45][46][104,105,106,107,108,109], rhodamine B (Rh B) [36][99], basic red 9 (BR 9), and CV [40][47][103,110] values are included in the data. Many citations only present the quantity of dye removed (in %) [36][37][41][43][47][48][99,100,104,106,110,111], which is valuable, however, this value varies with adsorbent quantity, dye concentration, and adsorbate volume.Table 1.
Basic dyes (cationic) and their adsorption onto biochars.
| Dye | Biochar Feedstock | Pyrolysis Conditions | Pore Volume (cm3/g) | BET Surface Area (m2/g) | Adsorption Capacity (mg/g) or Dye Removal (%) | Isotherm Type | Kinetic Model | Parameters | Mechanism | Reference | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Temperature (K) | Heating Rate (K/min) | Time (min) | pH | Equilibrium Time (min) | |||||||||
| MB | Date palm fronds | 973 | - | 240 | 0.134 | 430 | 205 | - | - | 6 | 36 | - | [41][104] |
| MG | Tapioca peel | 1073 | 10 | 180 | - | - | 32% | Langmuir, Freundlich | Pseudo I-order, Pseudo-II order | 2–10 | 0–180 | - | [36][99] |
| Rh B | Tapioca peel | 1073 | 10 | 180 | - | - | 66% | Langmuir, Freundlich | Pseudo I-order, Pseudo-II order | 2–10 | 0–180 | - | [36][99] |
| MB | Chlorella sp. microalgae | MW heating (2450 MHz, 800 W) | - | - | - | 3 | 110 | Freundlich, Temkin | Pseudo I-order, Pseudo-II order, Elovich | 2–10 | |||
Table 2.
Acid dyes (anionic) and their adsorption onto biochars.
| Dye | Biochar Feedstock | Pyrolysis Conditions | Pore Volume (cm3/g) | BET Surface Area (m2/g) | Adsorption Capacity (mg/g) or Dye Removal (%) | Isotherm Type | Kinetic Model | Adsorbent Parameters | Mechanism | Reference | |||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Temperature (K) | Heating Rate (K/min) | Time (min) | pH | Equilibrium Time (min) | |||||||||
| CR | Chlorella sp. microalgal | MW heating (2450 MHz, 800 W) | - | - | - | 3 | 160 | Langmuir, Freundlich, Temkin | Pseudo I-order, Pseudo II-order, Elovich | 2–10 | 240 | Boyd, Intraparticle diffusion | [42][105] |
| CR | Rice husk | 773 | 5 | 180 | - | - | 66–97% | Langmuir, Freundlich | - | 2, 4, 6, 7, 9, 11 | 5760 | - | [34][97] |
| RR 120 | Eucheuma spinosum | 573–873 | 10 | 120 | - | - | 330 | Langmuir, Freundlich, Temkin | Pseudo I-order, Pseudo II-order, Elovich | 3–9 | 20 | Electrostatic interaction, Ion exchange, Metal complexation, Hydrogen bonding | [ |
3.2. Dye Removal Using Adsorption onto Modified Biochars
Several investigations are now being conducted to improve the adsorption efficacy of biochars. Many papers on various biochar modification techniques are available. Treatment or activation of biochar with bases and acids, chemical impregnation, size alteration, and encapsulation are some of the modification techniques. The cationic dye adsorption characteristics and performance properties are shown in Table 3, whereas the anionic dye adsorption characteristics and performance properties are shown in Table 4.Table 3.
Basic dyes (cationic) and their adsorption onto modified (altered) biochars.
| Dye | Modified Biochar Feedstock | Pyrolysis Conditions | Pore Volume (cm3/g) | BET Surface Area (m2/g) | Adsorption Capacity (mg/g) or Dye Removal (%) | Isotherm Type | Kinetic Model | Adsorbent Parameters | Mechanism | Reference | |||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Temperature (K) | Heating Rate (K/min) | Time | pH | Equilibrium Time (min) | |||||||||||||||||||||||||||||||
| MB | Date palm fronds | 1073 | 20 | 240 | - | 70 | 210 | - | - | 7 | 180 | - | [81][144] | ||||||||||||||||||||||
| MG | Tapioca peel + S- doped | 1073 | 10 | 180 | - | 145 | 30 | Langmuir, Freundlich | Pseudo I-order, Pseudo II-order | 2–10 | 1080 | - | [36][99] | ||||||||||||||||||||||
| 64 | ] | [ | 127 | ] | |||||||||||||||||||||||||||||||
| Rh B | Tapioca peel + S- doped | 1073 | 10 | 180 | - | 145 | 30 | Langmuir, Freundlich | Pseudo I-order, Pseudo II-order | 2–10 | 1080 | - | [36][99] | 7200 | Boyd, Intraparticle diffusion | [42 | CR | Phoenix dactylifera leaves | 673] | - | -[ | - | 1105 | 25 | Langmuir, Freundlich | Pseudo I-order, Pseudo II-order | 5.8 | 120 | -] | ||||||
| [ | 62 | ] | [ | 125 | ] | ||||||||||||||||||||||||||||||
| MB | Areca leaf + K2FeO4− | 473 | 5 | 60 | - | 20 | 250 | Langmuir, Freundlich | Pseudo I-order, Pseudo II-order | 7 | 720 | Electrostatic attraction | [43][106] | MG | Rice husk | CR673–873 | - | 60 | - | - | 65 | Langmuir, Freundlich | Pseudo I-order, Pseudo-II order, Elovich | 2, 4, 6, 8 | 1440 | - | [37][100] | ||||||||
| Cotton stalks | 873 | - | 120 | - | 120 | 50 | Langmuir, Freundlich | Pseudo I-order, Pseudo II-order | 2–12 | 160 | Electrostatic attraction, Hydrogen bonding | [ | 82][145] | MG | Crab shell | 1073 | - | 120 | 0.086 | 82 | 12,500 | Langmuir | Pseudo-II order | 7 | 2 | Electrostatic attraction, Hydrogen bonding, π-π interactions | [38][101] | ||||||||
| MB | Areca leaf | 473 | 5 | 60 | - | 21 | 120 | Langmuir, Freundlich | Pseudo I-order, Pseudo-II order | 7 | 720 | Electrostatic attraction | [43][106] | ||||||||||||||||||||||
| 673 | 8 | 90 | - | - | 250 | Langmuir, Freundlich, Temkin, Dubinin-Radushkevich | |||||||||||||||||||||||||||||
| MG | Pseudo I-order, Pseudo II-order | 2–10 | 180 | Electrostatic attraction | [ | 63 | ] | [ | 126 | ] | |||||||||||||||||||||||||
| Chitosan-tapioca peel + S-doped | CR | Orange peel | 1073 | 15 | 15 | - | 20 | - | - | - | - | ||||||||||||||||||||||||
| Rh B | Chitosan-tapioca peel + S-doped | [ | 873 | - | 120 | - | 120 | 40 | 60 | ] | [ | 123 | ] | ||||||||||||||||||||||
| Langmuir, Freundlich | Pseudo I-order, Pseudo II-order | 2–12 | 160 | Electrostatic attraction, Hydrogen bonding, π-π interactions | [ | 82 | ] | [ | 145 | ] | Remazol BV 5R | Green marine algae (Caulerpa scalpelliformis) | 573–773 | 5 | 120 | - | - | 70% | Langmuir, Freundlich, Sips, T | Pseudo I-order, Pseudo II-order | 2–5 | - | - | ||||||||||||
| MB | Sugarcane bagasse + steam | [ | 1073 | 65 | 10 | ] | 120 | 0.356 | 570 | 5220 | [ | Langmuir, Freundlich | 128] | - | 7.4 | MB | Wodyetia Bifurcate | 973 | 10 | 30 | - | - | 150 | Sips | Pseudo I-order, Pseudo-II order | - | 30 | - | [44][107] | ||||||
| 180 | - | [ | 83 | ] | [ | 146 | ] | Remazol BO 3R | Green marine algae (Caulerpa scalpelliformis) | 573–773 | 5 | 120 | - | - | 77% | Langmuir, Freundlich, Sips, Temkin | Pseudo I-order, Pseudo II-order | 2–5 | - | - | [65][128] | MG | Waste wheat straw/wheat bran | 1073 | 15 | 90 | - | - | 1740 | Langmuir | Pseudo-II order | 2, 4, 6, 8, 10 | - | Electrostatic interaction, Chemisorption | [39][ |
| Remazol BO 3R | Green marine algae (Caulerpa scalpelliformis) | 102 | 573–773 | 5 | 120 | - | - | 75% | Langmuir, Freundlich, Sips, Temkin | Pseudo I-order, Pseudo II-order | 2–5 | - | -] | ||||||||||||||||||||||
| [ | 65 | ] | [ | 128 | ] | CV | Waste wheat straw/wheat bran | 1073 | 15 | 90 | - | - | 175 | Langmuir | Pseudo-II order | 2, 4, 6, 8, 10 | - | Electrostatic interaction, Chemisorption |
[39][102] | ||||||||||||||||
| Remazol BO 3R | Crab shell | 1073 | - | 120 | 0.086 | 82 | 20,315 | Langmuir | Pseudo I-order, Pseudo II-order | 4 | 2 | Electrostatic attraction, Hydrogen bonding, π-π interactions | [38][101] | MB | Switchgrass | 873 | - | 60 | 0.029 | 255 | 40 | Langmuir | Pseudo-II order | 6 | - | Intraparticle diffusion | [45][108] | ||||||||
| MB | |||||||||||||||||||||||||||||||||||
| CR | Activated Carbon | 723 | 20 | ||||||||||||||||||||||||||||||||
| MB | Date palm fronds with Fe/Mn | 973 | 3 | 240 | - | 430 | 300 | Langmuir, Freundlich | Pseudo I-order, Pseudo II-order, Intraparticle diffusion, Elovich | 4–10 | 240 | Surface adsorption, π-π interactions, Ion exchange, Pore-filling | [84][147] | 120 | - | - | 230 | Freundlich | - | ||||||||||||||||
| MB | Wakame Undaria pinnatifida leaves with calcination | 1073 | 10 | 120 | - | 1160 | 840 | Langmuir, Freundlich | Pseudo I-order, Pseudo II-order | 2–12 | 300 | Surface adsorption, Hydrogen bonding, π-π interactions, Pore-filling | [85][148] | 2–10 | 120 | - | [61][124 | ||||||||||||||||||
| Rh B | Wakame Undaria pinnatifida leaves with calcination | 1073 | 10 | 120 | - | 1160 | 530 | Langmuir, Freundlich | Pseudo I-order, Pseudo II-order | 2–12 | 300 | Surface adsorption, Hydrogen bonding, π-π interactions, Pore-filling | [85][148] | ] | |||||||||||||||||||||
| MG | Wakame Undaria pinnatifida leaves with calcination | 1073 | 10 | 120 | - | 1160 | 4065 | Langmuir, Freundlich | Pseudo I-order, Pseudo II-order | 2–12 | 300 | Surface adsorption, Hydrogen bonding, π-π interactions, Pore-filling | [85][148] | Switchgrass- | 1173 | - | 60 | 0.058 | 640 | 200 | Langmuir | Pseudo-II order | 6 | - | Intraparticle diffusion | [45][108] | |||||||||
| CR | Spirulina platensis algae | 723 | 20 | 120 | - | - | Freundlich | - | 2–10 | 120 | - | [33][96] | |||||||||||||||||||||||
| MG | Corn straw | 773 | - | 180 | - | 35 | 520 | Langmuir, Freundlich, Temkin | Pseudo I-order, Pseudo II-order, Intra diffusion | 2–9 | 20 | [86][76] | CV | Mango leaves | 1073 | - | 60 | - | 170 | 180 | - | 8 | 48 | - | [47 | ||||||||||
| CR | Waste wheat straw/wheat bran | ] | 1073 | 15 | 90 | - | - | 90 | Langmuir | Pseudo II-order | 2, 4, 6, 8, 10 | - | Chemisorption, Electrostatic interaction[110] | ||||||||||||||||||||||
| [ | 39 | ||||||||||||||||||||||||||||||||||
| MG | Rice husk + Cu + Al | 353 | - | ] | 60 | [ | 0.350 | 102 | ] | 200 | 470 | Langmuir, Freundlich | 9 | 200 | Pore-filling, π- π interactions | [87][149] | MG | Ulothrix zonata algae | 1073 | 15 | 90 | - | 130 | 5300 | Freundlich | Pseudo-II order | 2, 4, 6, 10 | 840 | Chemisorption | [40][103] | |||||
| OG | Switchgrass | 873 | - | 60 | 0.029 | 255 | 8 | Langmuir | Pseudo II-order | 6 | - | Outer boundary | [45][108] | CV | Ulothrix zonata algae | 1073 | 15 | 90 | - | 130 | 1220 | Freundlich | Pseudo-II order | 2, 4, 6, 10 | 840 | Chemisorption | |||||||||
| CR | Switchgrass | [ | 873 | 40 | ] | - | [ | 103 | ] | ||||||||||||||||||||||||||
| 60 | 0.029 | 255 | 8 | Langmuir | Pseudo II-order | 6 | - | Outer boundary | [ | 45 | ] | [ | 108] | BR 9 | Bovine bones | 1073 | 10 | 60 | 0.271 | 90 | 50 | Langmuir, Freundlich | Pseudo-II order | 7 | 180 | - | |||||||||
| CR | [ | 48 | Switchgrass | ] | [ | 111 | ] | ||||||||||||||||||||||||||||
| 1173 | 60 | 0.058 | 640 | 20 | Langmuir | Pseudo II-order | 6 | - | Outer boundary | [ | 45 | ][108] | BR 9 | Bovine bones | 1073 | 10 | 180 | 0.193 | 95 | 50 | |||||||||||||||
| CR | Ulothrix zonata algae | 1073 | Pseudo I-order | 7 | 15 | 90 | 180 | - | - | 130 | 345 | [48][111] | |||||||||||||||||||||||
| Freundlich | MB | Sugarcane bagasse | 773 | 10 | 90 | - | 260 | 70 | Langmuir, Freundlich | Pseudo I-order, Pseudo-II order | 7.4 | 180 | Intraparticle diffusion | [46][109] | |||||||||||||||||||||
