Intracytoplasmic sperm injection (ICSI) has been used for severe male factor infertility and non-male factors, such as unexplained infertility or advanced maternal age, without robust scientific evidence. However, applying ICSI blindly is not free of potential detrimental consequences since novel studies report possible health consequences to offspring. DNA methylation and epigenetic alterations in sperm cells of infertile men might help explain some of the adverse effects reported in ICSI studies on reproductive health in future generations.
- assisted reproductive technology
- human in vitro fertilization
- embryo development
- male infertility
- intracytoplasmic sperm injection
1. Background
2. Imprinting Alteration following ART
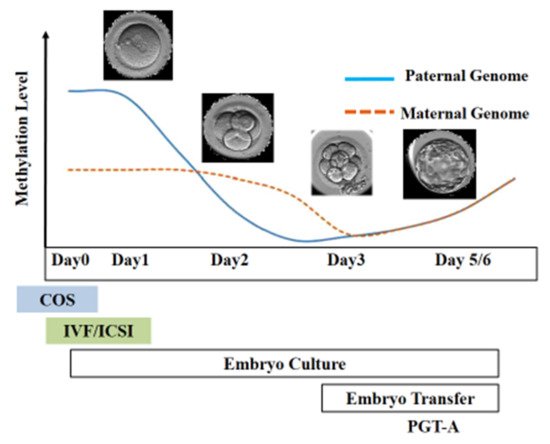
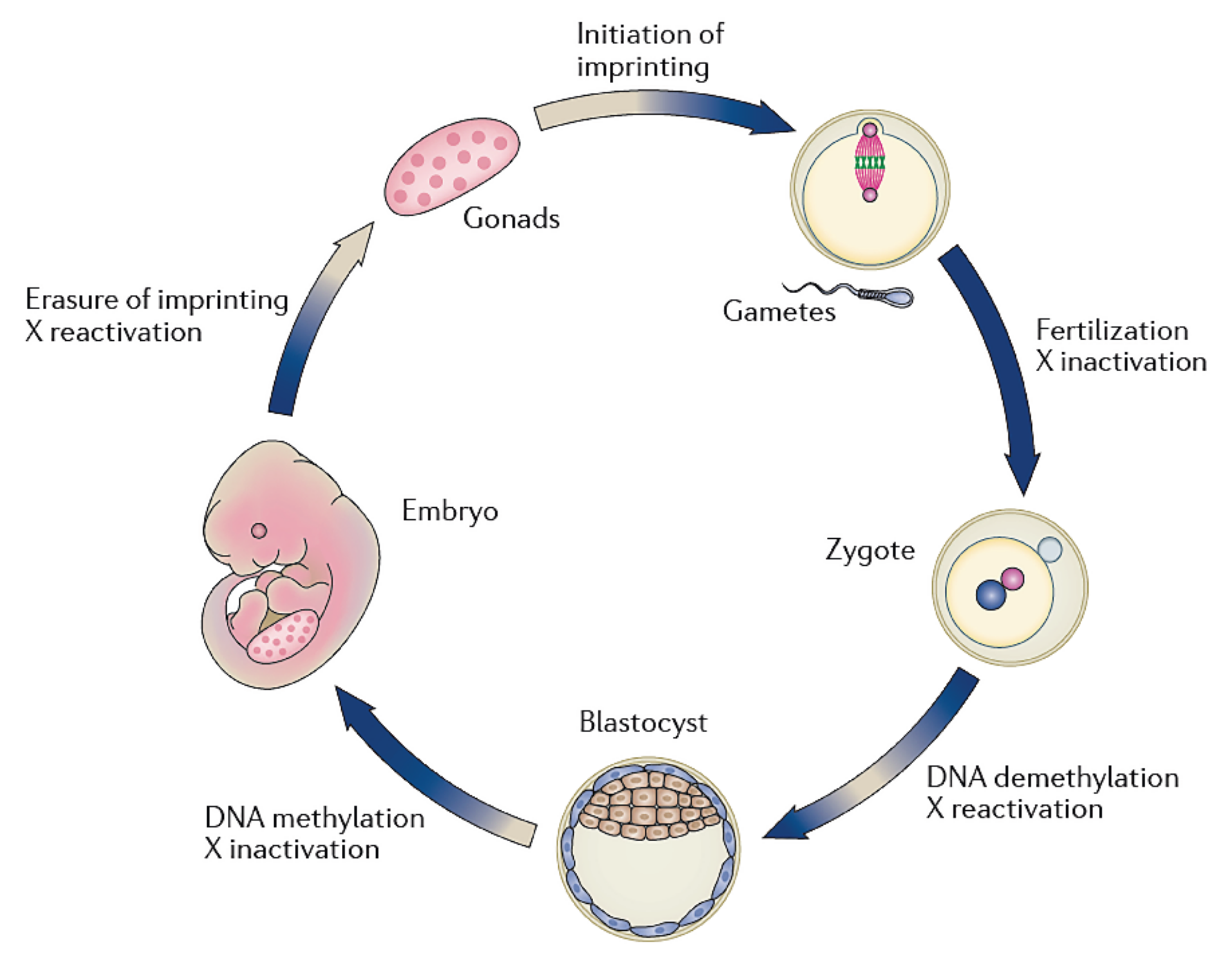
After fertilization, the zygote develops into a structure called the “blastocyst”, which includes about 200 cells, already differentiated into two types: the trophectoderm (TE) and the inner cell mass (ICM). The latter comprises a group of cells attached to the inside of the trophectoderm, which will eventually give rise to the fetus. TE cells are the blastocyst’s external layer, promoting the implantation process into the uterine lining and forming other extraembryonic tissues, including the placenta. Embryonic cells are guided toward their future lineages during early development through epigenetic reprogramming and subsequent re-establishment of cell-type-specific epigenetic signatures. This coincides with the period when gametes and embryos are cultivated inside the embryology laboratory. Therefore, during this critical time window, any artificial perturbations might lead to epigenetic modifications in the resultant offspring (Figure 1 and Figure 2). Studies reported imprinted loci to be vulnerable to external environmental cues during in vitro embryo culture. For example, KvDMR1 has been abnormally methylated in ART-related BWS in humans [42,60,61][16][17][18] and hypomethylated in ART-produced bovine progeny with large offspring syndrome (LOS) [62][19]. Additionally, several reports indicate that ART-related procedures, including OS, ICSI, and extended culture to the blastocyst stage, might promote epigenetic aberrations [41,42,55][16][20][21].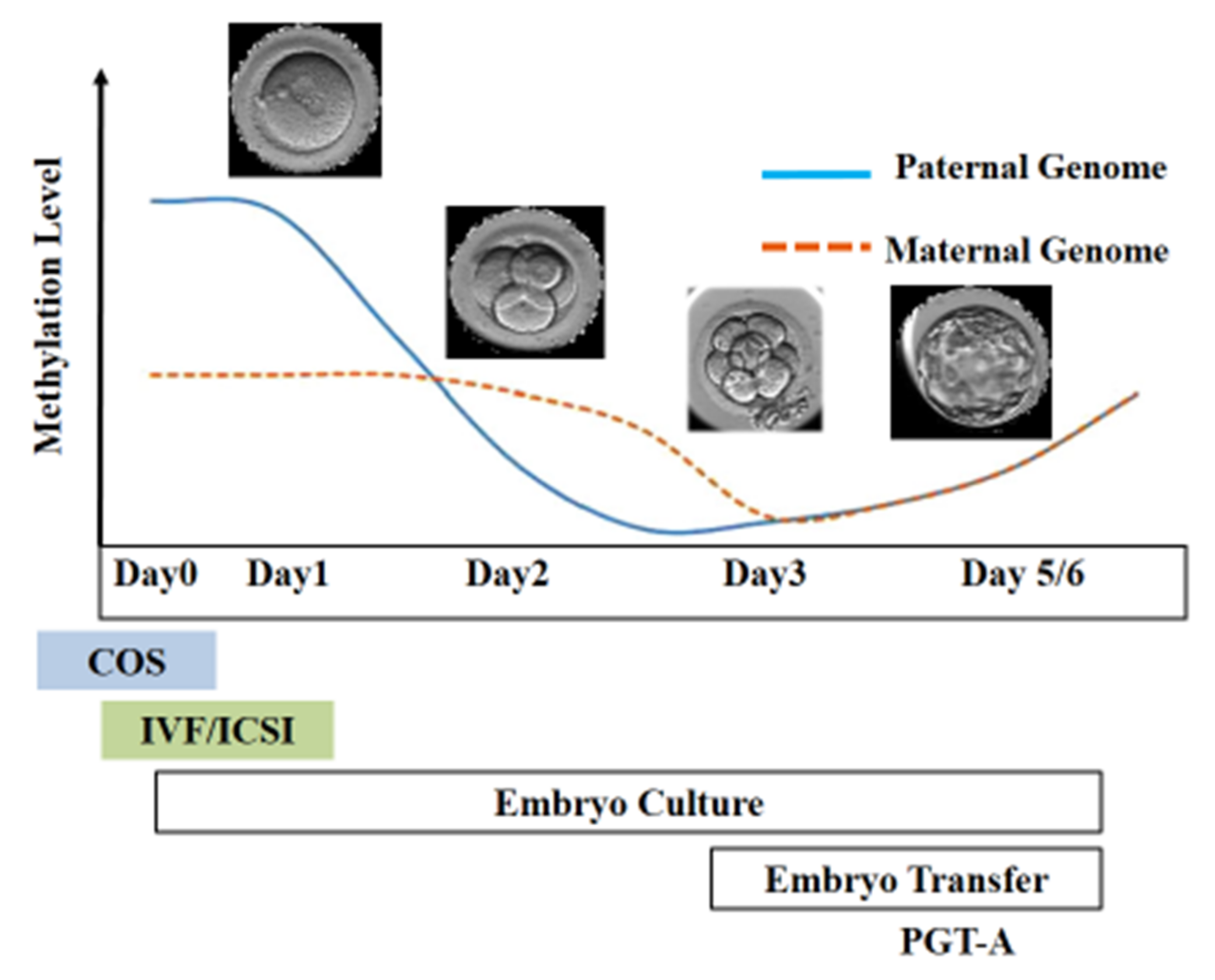
3. Spermatogenesis, Epigenetics, and Infertility
54. ICSI for Male Factor Infertility
5.1. Oligoasthenoteratozoospermia
4.1. Oligoasthenoteratozoospermia
Although ICSI should be encouraged mainly in severe male infertility, it can be challenging to establish when a male factor is compulsory for the ICSI technique. Standard semen assessment is performed to confirm the severity of male infertility and advise ICSI, but it is well reported that sperm analysis has limitations; for example, it does not assess the function and physiology of the sperm, and genetic or epigenetic assessment [83][34]. Sperm number, morphology, and motility are typically evaluated to decide on the ICSI procedure rather than standard IVF insemination [84][35]. It is worth mentioning that high-quality studies investigating pregnancy outcomes and live birth rate (LBR) between ICSI and IVF in couples with oligoasthenoteratozoospermia are still missing. However, a study published in 2005 by Shuai and collaborators explored these concerns. The authorscholars observed no differences between the two insemination procedures (IVF and ICSI) in fertilization, implantation, and pregnancy rates in couples undergoing ART with men diagnosed with moderate oligoasthenoteratozoospermia [85][36]. Sperm morphology is another parameter broadly used to choose for ICSI. In 1986, Kruger and colleagues suggested using strict criteria for sperm abnormalities and advising ICSI when the proportion of normal sperm in the ejaculate was <4% [86][37]. Additional studies confirmed this evidence and proposed that at least 5% of sperm is needed to be morphologically normal to obtain an acceptable fertilization rate using standard IVF [87,88][38][39]. Therefore, ICSI rather than IVF has been routinely recommended in patients with reduced sperm morphology (<5%) [89][40].5.2. Azoospermia
4.2. Azoospermia
The term azoospermia indicates the absence of sperm cells in the ejaculate. It affects around 1% of the general male population and about 15% of infertile men [93][41]. There are two different types of azoospermia: obstructive and non-obstructive. In obstructive azoospermia, normal and complete spermatogenesis is typically found, and sperm can be surgically collected from the testis [94][42]. By contrast, non-obstructive azoospermia is associated with the testicular alterations that result in the failure of sperm production. Typical testicular histopathological features in males with non-obstructive azoospermia include germ cell aplasia, maturation arrest, or hypospermatogenesis. The procedures mostly applied to collect sperm from azoospermic patients are percutaneous acquisition and open surgery [95][43]. Following sperm retrieval, ICSI can be applied to achieve oocyte fertilization [96][44].5.3. Antisperm Antibodies
4.3. Antisperm Antibodies
The presence of seminal antisperm antibodies (ASAs) is typically associated with a gap or rupture of the blood–testis barrier in the reproductive tract, which can be linked with several conditions [97][45]. However, elevated levels of ASAs in semen samples are observed in about 5–12% of men undergoing ART, and might negatively affect fertility, reducing sperm motility, capacitation, acrosome reaction, and oocyte sperm bounding [98][46].5.4. ICSI and Sperm DNA Fragmentation (SDF)
4.4. ICSI and Sperm DNA Fragmentation (SDF)
DNA fragmentation test is applied to assess the breakage of DNA strands inside the sperm head. This diagnostic test can predict fertility and normal embryo development and pregnancy outcomes than routine semen analysis parameters [99,100][47][48]. With the use of probes, sperm DNA breaks can be deeply scrutinized and quantified with the aid of fluorescence/optical microscopy or flow cytometry [100][48]. Sperm DNA fragmentation (SDF) is generally induced by oxidative stress resulting from environmental and lifestyle factors such as smoking, genital tract infections, obesity, and nutrition [101][49]. Moreover, SDF is frequently detected in men with infertility issues (e.g., varicocele), and it is more prevalent in those individuals than in fertile counterparts [102,103][50][51]. Scientific evidence indicates that a high level of SDF impairs the probabilities of success following ART [104,105][52][53].5.5. Globozoospermia
4.5. Globozoospermia
This condition is described by the entire lack of the acrosomal vesicle in the sperm head, with alteration of the nuclear membrane, and midpiece defects, resulting in a round-shaped sperm head. It is an uncommon condition involving a small percentage of infertile men (about 0.1%) [111][54]. Despite having normal sperm count and motility, globozoospermic sperm cannot fertilize the oocyte: therefore, ICSI remains the favorable option available.65. Use of ICSI for Couples with Partners Having Semen Analysis within Reference Ranges
One of the first Cochrane review papers was published in 2004 by van Rumste and collaborators to investigate whether ICSI improves LBR compared to IVF in couples whose male partners had semen analysis within reference ranges. The authorscholars showed a significantly higher fertilization rate in the IVF group but no difference in pregnancy, miscarriage, or LBR than ICSI insemination [113][55]. Subsequently, Bhattacharya and co-workers performed a multicenter randomized controlled study comparing clinical outcomes after ICSI or traditional IVF in couples with male partners having semen assessment within references ranges. The study randomly assigned 415 couples and was performed in four UK IVF units. Their results showed that the fertilization rate was higher with IVF than with ICSI (58% versus 47%; p = 0.0001). Standard IVF insemination provided an implantation rate of 30% compared to 22% for ICSI (p = 0.03). No significant difference was observed regarding the clinical pregnancy rate between IVF and ICSI (33% and 26%, respectively). Moreover, the overall laboratory time used was significantly shorter with IVF than with ICSI (22.9 min versus 38.1) [114][56]. Dang and co-workers reported similar results. They randomized 1064 patients undergoing ART to ICSI technique (n = 532) or standard IVF insemination (n = 532). After the first embryo transfer, LBR was 35% in the ICSI group versus 31% for couples assigned to conventional IVF (p = 0.27). They found higher TFF with IVF (6%) than with ICSI (5%). The study concluded that in couples undergoing ART with a male partner having so-called normal semen parameters, ICSI did not increase LBR compared with conventional IVF [115][57].
76. Contemporary Use (and Overuse) of ICSI
Since its first use almost 30 years ago, the application of ICSI as a fertilization method has raised steadily, even though the percentage of infertile couples with severe male factors has not increased [125][58]. Thus, it seems evident that currently, ICSI is applied broadly, even though there is no clear evidence of its benefit in couples without male factor infertility [113,114,123,125,126][55][56][58][59][60]. Boulet and collaborators analyzed data on ART between 1996 and 2012 and reported increased use of ICSI from 36.4% in 1996 to 76.2% in 2012, even though male-factor infertility remained unchanged at about 36% of cycles [118][61]. Another trial published by Dyer and colleagues analyzing the worldwide data on ART performed between 2008 and 2010 found that ICSI was used as a fertilization method in about 67% of about 4.5 million cycles completed [123][59]. However, there is considerable variation according to countries; in Asia, ICSI is applied in about 55% of the treatments, 65% of cases in Europe, 85% of patients in Latin America, and almost 100% of patients in the Middle East [123][59]. Moreover, in a large retrospective study performed in Australia between 2002 and 2013, analyzing about 585 thousand ART cycles, the authorscholars did not report any improvement when ICSI was used rather than standard IVF insemination for couples without obvious male infertility. They observed an LBR of about 10% lower with ICSI than IVF [122][62]. On this basis, one should ask why is ICSI preferred to standard IVF in routine practice for cases without a clear male factor? Possible factors to justify the broad ICSI application related to a general notion that ICSI reduces the risk of TFF. Naturally, fertilization failure is problematic to any couple undergoing ART; especially when counseling is not available and the physician is the person involved in delivering this bad news. In addition, in private settings, where the couple needs to pay for the treatment, the failed fertilization also represents a remarkable burden for the couple who will need to bear the costs of another cycle. The debate is ongoing [115][57] and the Practice Committee of the American Society for Reproductive Medicine (ASRM) has recently produced a committee opinion paper recommending against the extensive use of ICSI in couples undergoing MAR cycles without confirmed male factor infertility [124][63].References
- De Geyter, C.; Wyns, C.; Calhaz-Jorge, C.; De Mouzon, J.; Ferraretti, A.P.; Kupka, M.; Andersen, A.N.; Nygren, K.G.; Goossens, V. 20 years of the European IVF-monitoring Consortium registry: What have we learned? A comparison with registries from two other regions. Hum. Reprod. 2020, 35, 2832–2849.
- Steptoe, P.C.; Edwards, R.G. Birth after the reimplantation of a human embryo. Lancet 1978, 2, 366.
- Hiura, H.; Okae, H.; Chiba, H.; Miyauchi, N.; Sato, F.; Sato, A.; Arima, T. Imprinting methylation errors in ART. Reprod. Med. Biol. 2014, 13, 193–202.
- Ventura-Juncá, P.; Irarrázaval, I.; Rolle, A.J.; Gutiérrez, J.I.; Moreno, R.D.; Santos, M.J. In vitro fertilization (IVF) in mammals: Epigenetic and developmental alterations. Scientific and bioethical implications for IVF in humans. Biol. Res. 2015, 48, 68.
- Qin, J.; Sheng, X.; Wang, H.; Liang, D.; Tan, H.; Xia, J. Assisted reproductive technology and risk of congenital malformations: A metaanalysis based on cohort studies. Arch. Gynecol. Obstet. 2015, 292, 777–798.
- Hart, R.; Norman, R.J. The longer-term health outcomes for children born as a result of IVF treatment: Part I—General health outcomes. Hum. Reprod. Update 2013, 19, 232–243.
- Kessler, N.J.; Waterland, R.A.; Prentice, A.M.; Silver, M.J. Establishment of environmentally sensitive DNA methylation states in the very early human embryo. Sci. Adv. 2018, 4, 1–9.
- Palermo, G.; Joris, H.; Devroey, P.; Van Steirteghem, A.C. Pregnancies after intracytoplasmic injection of single spermatozoon into an oocyte. Lancet 1992, 340, 17–18.
- Palermo, G.D.; Neri, Q.V.; Rosenwaks, Z. To ICSI or not to ICSI. Semin. Reprod. Med. 2015, 33, 92–102.
- El Hajj, N.; Haertle, L.; Dittrich, M.; Denk, S.; Lehnen, H.; Hahn, T.; Schorsch, M.; Haaf, T. DNA methylation signatures in cord blood of ICSI children. Hum. Reprod. 2017, 32, 1761–1769.
- Vrooman, L.A.; Bartolomei, M.S. Can assisted reproductive technologies cause adult-onset disease? Evidence from human and mouse. Reprod. Toxicol. 2017, 68, 72–84.
- Gianotten, J.; Lombardi, M.P.; Zwinderman, A.H.; Lilford, R.J.; van der Veen, F. Idiopathic impaired spermatogenesis: Genetic epidemiology is unlikely to provide a short-cut to better understanding Hum. Reprod. Update 2004, 10, 533–539.
- Gunes, S.; Arslan, M.A.; Hekim, G.N.T.; Asci, R. The role of epigenetics in idiopathic male infertility. J. Assist. Reprod. Genet. 2016, 33, 553–569.
- Zhao, Y.; Garcia, B.A. Comprehensive catalog of currently documented histone modifications. Cold Spring Harb. Perspect. Biol. 2015, 7, a025064.
- Kouzarides, T. Chromatin Modifications and Their Function. Cell 2007, 128, 693–705.
- Eggermann, T.; Perez de Nanclares, G.; Maher, E.R.; Temple, I.K.; Tümer, Z.; Monk, D.; Mackay, D.J.; Grønskov, K.; Riccio, A.; Linglart, A.; et al. Imprinting disorders: A group of congenital disorders with overlapping patterns of molecular changes affecting imprinted loci. Clin. Epigenet. 2015, 7, 123.
- White, C.R.; Denomme, M.M.; Tekpetey, F.R.; Feyles, V.; Power, S.G.; Mann, M.R. High frequency of imprinted methylation errors in human preimplantation embryos. Sci. Rep. 2015, 5, 17311.
- Huntriss, J.D.; Hemmings, K.E.; Hinkins, M.; Rutherford, A.J.; Sturmey, R.G.; Elder, K.; Picton, H.M. Variable imprinting of the MEST gene in human preimplantation embryos. Eur. J. Hum. Genet. 2013, 21, 40–47.
- Chen, Z.; Robbins, K.M.; Wells, K.D.; Rivera, R.M. Large offspring syndrome: A bovine model for the human loss-of-imprinting overgrowth syndrome Beckwith-Wiedemann. Epigenetics 2013, 8, 591–601.
- Tunster, S.J.; Jensen, A.B.; John, R.M. Imprinted genes in mouse placental development and the regulation of fetal energy stores. Reproduction 2013, 145, R117–R137.
- Hiura, H.; Okae, H.; Miyauchi, N.; Sato, F.; Sato, A.; Van De Pette, M.; John, R.M.; Kagami, M.; Nakai, K.; Soejima, H.; et al. Characterization of DNA methylation errors in patients with imprinting disorders conceived by assisted reproduction technologies. Hum. Reprod. 2012, 27, 2541–2548.
- Esteves, S.C.; Roque, M.; Bedoschi, G.; Haahr, T.; Humaidan, P. Intracytoplasmic sperm injection for male infertility and consequences for offspring. Nat. Rev. Urol. 2018, 15, 535–562.
- Kretser, D.M.; Loveland, K.L.; Meinhardt, A.; Simorangkir, D.; Wreford, N. Spermatogenesis. Hum. Reprod. 1998, 13 (Suppl. 1), 1–8.
- Anway, M.D.; Cupp, A.S.; Uzumcu, M.; Skinner, M.K. Epigenetic transgenerational actions of endocrine disruptors and male fertility. Science 2005, 308, 1466–1469.
- Leslie, M. Epigenetics. Sperm RNA fragments modify offspring metabolism. Science 2016, 351, 13.
- Ben Maamar, M.; Beck, D.; Nilsson, E.; McCarrey, J.R.; Skinner, M.K. Affiliations expand Developmental alterations in DNA methylation during gametogenesis from primordial germ cells to sperm. iScience 2022, 25, 103786.
- Cornwall, G.A. Role of posttranslational protein modifications in epididymal sperm maturation and extracellular quality control. Adv. Exp. Med. Biol. 2014, 759, 159–180.
- Chang, M.C. Fertilizing capacity of spermatozoa deposited into the fallopian tubes. Nature 1951, 168, 697–698.
- Sullivan, R.; Legare, C.; Lamontagne-Proulx, J.; Breton, S.; Soulet, D. Revisiting structure/functions of the human epididymis. Andrology 2019, 7, 748–757.
- James, E.R.; Carrell, D.T.; Aston, K.I.; Jenkins, T.G.; Yeste, M.; Salas-Huetos, A. The Role of the Epididymis and the Contribution of Epididymosomes to Mammalian Reproduction. Int. J. Mol. Sci. 2020, 21, 5377.
- Sharma, A. Transgenerational epigenetics: Integrating soma to germline communication with gametic inheritance. Mech. Ageing Dev. 2017, 163, 15–22.
- Scott, I.M.; Rubinstein, G.M.; Poole, F.L., II; Lipscomb, G.L.; Schut, G.J.; Williams-Rhaesa, A.M.; Stevenson, D.M.; Amador-Noguez, D.; Kelly, R.M.; Adams, M.W.W. The thermophilic biomass-degrading bacterium Caldicellulosiruptor bescii utilizes two enzymes to oxidize glyceraldehyde 3-phosphate during glycolysis. J. Biol. Chem. 2019, 294, 9995–10005.
- Ben Maamar, M.; Nilsson, E.; Sadler-Riggleman, I.; Beck, D.; McCarrey, J.R.; Skinner, M.K. Developmental origins of transgenerational sperm DNA methylation epimutations following ancestral DDT exposure. Dev. Biol. 2019, 445, 280–293.
- Esteves, S.C.; Zini, A.; Aziz, N.; Alvarez, J.G.; Sabanegh, E.S., Jr.; Agarwal, A. Critical appraisal of World Health Organization’s new reference values for human semen characteristics and effect on diagnosis and treatment of subfertile men. Urology 2012, 79, 16–22.
- Babayev, S.N.; Park, C.W.; Bukulmez, O. Intracytoplasmic sperm injection indications: How rigorous? Semin. Reprod. Med. 2014, 32, 283–290.
- Shuai, H.L.; Ye, Q.; Huang, Y.H.; Xie, B.G. Comparison of conventional in vitro fertilisation and intracytoplasmic sperm injection outcomes in patients with moderate oligoasthenozoospermia. Andrologia 2015, 47, 499–504.
- Kruger, T.F.; Menkveld, R.; Stander, F.S.; Lombard, C.J.; Van der Merwe, J.P.; van Zyl, J.A.; Smith, K. Sperm morphologic features as a prognostic factor in in vitro fertilization. Fertil. Steril. 1986, 46, 1118–1123.
- Gunalp, S.; Onculoglu, C.; Gurgan, T.; Kruger, T.F.; Lombard, C.J. A study of semen parameters with emphasis on sperm morphology in a fertile population: An attempt to develop clinical thresholds. Hum. Reprod. 2001, 16, 110–114.
- Menkveld, R.; Wong, W.Y.; Lombard, C.J.; Wetzels, A.M.; Thomas, C.M.; Merkus, H.M.; Steegers-Theunissen, R.P. Semen parameters, including WHO and strict criteria morphology, in a fertile and subfertile population: An attempt to develop clinical thresholds. Hum. Reprod. 2001, 16, 1165–1171.
- Plachot, M.; Belaisch-Allart, J.; Mayenga, J.M.; Chouraqui, A.; Tesquier, L.; Serkine, A.M. Outcome of conventional IVF and ICSI on sibling oocytes in mild male fator infertility. Hum. Reprod. 2002, 17, 362–369.
- Devroey, P.; Van Steirteghem, A. A review of ten years experience of ICSI. Hum. Reprod. Update 2004, 10, 19–28.
- Esteves, S.C.; Miyaoka, R.; Agarwal, A. An update on the clinical assessment of the infertile male . Clinics 2011, 66, 691–700.
- Esteves, S.C.; Miyaoka, R.; Orosz, J.E.; Agarwal, A. An update on sperm retrieval techniques for azoospermic males. Clinics 2013, 68 (Suppl. 1), 99–110.
- Esteves, S.C. Novel concepts in male factor infertility: Clinical and laboratory perspectives. J. Assist. Reprod. Genet. 2016, 33, 1319–1335.
- Zini, A.; Fahmy, N.; Belzile, E.; Ciampi, A.; Al-Hathal, N.; Kotb, A. Antisperm antibodies are not associated with pregnancy rates after IVF and ICSI: Systematic review and meta- analysis. Hum. Reprod. 2011, 26, 1288–1295.
- Esteves, S.C.; Schneider, D.T.; Verza, S., Jr. Influence of antisperm antibodies in the semen on intracytoplasmic sperm injection outcome. Int. Braz. J. Urol. 2007, 33, 795–802.
- Roque, M.; Esteves, S.C. Effect of varicocele repair on sperm DNA fragmentation: A review. Int. Urol. Nephrol. 2018, 50, 583–603.
- Esteves, S.C.; Sharma, R.K.; Gosálvez, J.; Agarwal, A. A translational medicine appraisal of specialized andrology testing in unexplained male infertility. Int. Urol. Nephrol. 2014, 46, 1037–1052.
- Greco, E.; Scarselli, F.; Iacobelli, M.; Rienzi, L.; Ubaldi, F.; Ferrero, S.; Franco, G.; Anniballo, N.; Mendoza, C.; Tesarik, J. Efficient treatment of infertility due to sperm DNA damage by ICSI with testicular spermatozoa. Hum. Reprod. 2005, 20, 226–230.
- Majzoub, A.; Esteves, S.C.; Gosálvez, J.; Agarwal, A. Specialized sperm function tests in varicocele and the future of andrology laboratory. Asian J. Androl. 2016, 18, 205–212.
- Minhas, S.; Bettocchi, C.; Boeri, L.; Capogrosso, P.; Carvalho, J.; Cilesiz, N.C.; Cocci, A.; Corona, G.; Dimitropoulos, K.; Gül, M.; et al. EAU Working Group on Male Sexual and Reproductive Health. European Association of Urology Guidelines on Male Sexual and Reproductive Health: 2021 Update on Male Infertility. Eur. Urol. 2021, 80, 603–620.
- Esteves, S.C.; Zini, A.; Coward, R.M.; Evenson, D.P.; Gosálvez, J.; Lewis, S.E.M.; Sharma, R.; Humaidan, P. Sperm DNA fragmentation testing: Summary evidence and clinical practice recommendations. Andrologia 2021, 53, e13874.
- Practice Committees of American Society for Reproductive Medicine. The clinical utility of sperm DNA integrity testing: A guideline. Fertil. Steril. 2013, 99, 673–677.
- Dam, A.; Feenstra, I.; Westphal, J.; Ramos, L.; van Golde, R.; Kremer, J. Globozoospermia revisited. Hum. Reprod. Update 2007, 13, 63–75.
- Van Rumste, M.M.; Evers, J.L.; Farquhar, C.M. ICSI versus conventional techniques for oocyte insemination during IVF in patients with non- male factor subfertility a Cochrane review. Hum. Reprod. 2004, 19, 223–227.
- Bhattacharya, S.; Hamilton, M.; Shaaban, M.; Khalaf, Y.; Seddler, M.; Ghobara, T.; Braude, P.; Kennedy, R.; Rutherford, A.; Hartshorne, G.; et al. Conventional in- vitro fertilisation versus intracytoplasmic sperm injection for the treatment of non- male-factor infertility: A randomised controlled trial. Lancet 2001, 357, 2075–2079.
- Dang, V.Q.; Vuong, L.N.; Luu, T.M.; Pham, T.D.; Ho, T.M.; Ha, A.N.; Truong, B.T.; Phan, A.K.; Nguyen, D.P.; Pham, T.N.; et al. Intracytoplasmic sperm injection versus conventional in-vitro fertilisation in couples with infertility in whom the male partner has normal total sperm count and motility: An open-label, randomised controlled trial. Lancet 2021, 397, 1554–1563.
- Jain, T.; Gupta, R. Trends in the use of intracytoplasmic sperm injection in the United States. N. Engl. J. Med. 2007, 357, 251–257.
- Dyer, S.; Chambers, G.M.; de Mouzon, J.; Nygren, K.G.; Zegers-Hochschild, F.; Mansour, R.; Ishihara, O.; Banker, M.; Adamson, G.D. International Committee for Monitoring Assisted Reproductive Technologies world report: Assisted reproductive technology 2008, 2009 and 2010. Hum. Reprod. 2016, 31, 1588–1609.
- Cissen, M.; Bensdorp, A.; Cohlen, B.J.; Repping, S.; de Bruin, J.P.; van Wely, M. Assisted reproduction technologies for male subfertility. Cochrane Database Syst. Rev. 2016, 2, CD000360.
- Boulet, S.L.; Mehta, A.; Kissin, D.M.; Warner, L.; Kawwass, J.F.; Jamieson, J.D. Trends in use of and reproductive outcomes associated with intracytoplasmic sperm injection. JAMA 2015, 313, 255–263.
- Chambers, G.M.; Wand, H.; Macaldowie, A.; Chapman, M.G.; Farquhar, C.M.; Bowman, M.; Molloy, D.; Ledger, W. Population trends and live birth rates associated with common ART treatment strategies. Hum. Reprod. 2016, 31, 2632–2641.
- Practice Committees of American Society for Reproductive Medicine and Society for Assisted Reproduction Technology. Intracytoplasmic sperm injection (ICSI) for non- male factor indications: A committee opinion. Fertil. Steril. 2020, 114, 239–245.
