Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Aref Zayed and Version 4 by Conner Chen.
Steroids are compounds widely available in nature and synthesized for therapeutic and medical purposes. Although several analytical techniques are available for the quantification of steroids in clinical samples, their analysis is challenging due to their low levels and complex matrices. The efficiency and quick separation of the high performance liquid chromatography (HPLC) combined with the sensitivity, selectivity, simplicity, and cost-efficiency of fluorescence, make HPLC coupled to fluorescence detection (HPLC-FLD) an ideal tool for routine measurement and detection of steroids for clinical and medical applications.
- high performance liquid chromatography
- clinical
- steroids
- fluorescence
- quantification
1. Clinical Applications of HPLC-FLD Techniques
Quantification of steroids plays an important role in the diagnosis and treatment of endocrine disorders. Diseases, such as cancer, metabolic syndromes, and neurodegenerative diseases, are associated with abnormalities in the endocrine system. Steroids also play an important role in biochemical processes, such as aging, reproduction, and metabolic pathways [1][76]. Furthermore, disturbances in the endocrine system due to EDC from environment and food are becoming a major clinical concern. HPLC-FLD has been employed in many research studies, both in humans and animal models, to quantify the various types of steroids and investigate their role in diseases and clinical conditions.
2. Detection of Glucocorticosteroids
Glucocorticosteroids are widely recognized as markers for adrenal activity. Cortisol can reflect the short-term changes in the activity of the hypothalamic–pituitary–adrenocortical axis (HPA axis), making it a valuable surrogate marker for stress and glucose metabolism [2][56].
Many methods have been developed to detect unconjugated cortisol levels in various biological samples. However, the use of certain samples, such as urine, may not be recommended as these samples may contain many interfering substances thus complicating steroids extraction procedure [3][55]. In addition, the concentrations of corticosteroids in urine are low compared with those found in plasma, which makes the latter a better medium for measurement. Steroids have been measured by HPLC-FLD methods for various clinical purposes, such as investigating disease courses, pathogenesis, and metabolic processes [4][15].
The detection of corticosteroids in biological samples can be done directly depending on fluorescence signal enhancement upon the treatment of samples with the deproteinizing agent (ethanol) and sulfuric acid (Figure 1). A study to quantify corticosteroids in serum samples was performed using C18 analytical column after treatment with ethanol and sulfuric acid [5][53]. Cortisol, testosterone and corticosterone emitted fluorescence, with LOD for cortisol = 0.3 pg/dL (S/N = 3). In another study, the same group measured cortisol in urine samples producing a LOD = 0.26 pg/dL (S/N = 3) [6][54]. Sudo et al. used post-column derivatization with sulfuric acid to measure cortisol and corticosterone in rat urine (LOD was 0.5 pmol for corticosterone at (S/N = 5)) [3][55]. They analyzed other corticoids, such as prednisolone, 6α-methylprednisolone, dexamethasone, and betamethasone, but with lower sensitivity. Moreover, ethyl acetate was used for extraction after sulfuric acid hydrolysis to prevent the acid from entering the HPLC system. It was noticed that the fluorescence intensities were dependent on the reactor temperature and sulfuric acid [3][7][55,77].
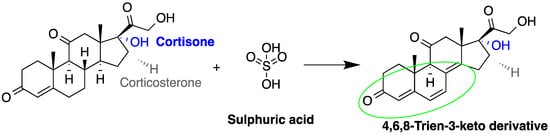
Figure 1. Fluorescence emission of steroids after treatment with sulfuric acid.
Since cortisol levels in hair might be used as a biomarker of chronic stress, a study developed a HPLC-FLD method to measure cortisol distribution in human hair samples. The method has achieved equal precision to mass spectrometry. Samples were prepared by pulverization and incubation in 0.1 M HCl, and extraction with ethyl acetate followed by derivatization with sulfuric acid. A detection limit of 1 pg/mg was achieved [2][56].
Derivatization with fluorophore-containing reagents was employed in many studies to detect and quantify corticosteroids in serum and urine samples. The derivatization process depends on functionalization of specific groups in corticosteroids structure (Figure 21). The primary alcohol functionality can be esterified through its reaction with either 2-(4-carboxyphenyl)-5,6-dimethylbenzimidazole (CDB) (Figure 21), 9-anthroyl nitrile (9-AN) (Figure 32), or 1-anthroyl nitrile (1-AN) (Figure 32). These derivatization processes proved to be selective as neither secondary alcohol, nor tertiary alcohol can react with fluorophore-containing reagents (Figure 32).
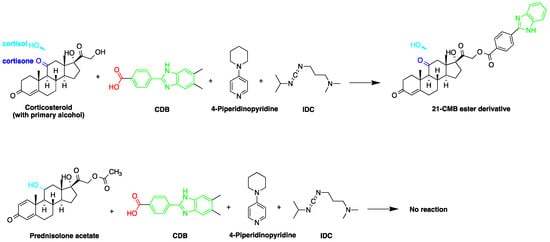
Figure 2. Esterification of primary alcohol functionality of cortisol and cortisone by 2-(4-carboxyphenyl)-5,6-dimethylbenzimidazole (CDB) in the presence of 4-piperidinopyridine and 1-isopropyl-3-(3-dimethylaminopropyl) carbodiimide (IDC) perchlorate. Prednisolone secondary alcohol shows no reaction.
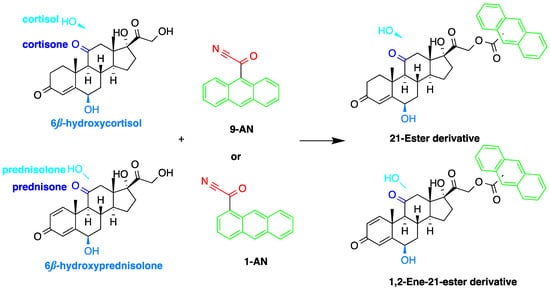
Figure 35.
Esterification of primary alcohol functionality of cortisol and cortisone by 9-anthroyl nitrile (9-AN) or 1-anthroyl nitrile (1-AN).
The derivatization through esterification of corticosteroid primary alcohol functionality with CDB was studied by Katayama et al. [8][78]. The results showed that the detector response to secondary alcohols was less than one fiftieth that of the primary alcohol. However, certain secondary alcohols in steroids, such as prednisolone acetate and testosterone, and tertiary alcohols, showed no reaction. Later, Katayama et al. used human plasma to detect eight corticosteroids by derivatization with CDB to their esters in acetonitrile. The esters were separated on a reversed-phase column (Zorbax ODS) with water:methanol (25:75, v/v) containing 5 mmol/L tetramethylammonium hydrogen sulfate as a mobile phase. The LOD ranged between 0.06 and 0.3 pg per 100 µL of plasma (S/N = 3) [9][58].
The use of 9-AN as a derivatizing agent for corticosteroids was reported in several studies. Neufled et al. used 9-AN to derivatize the primary alcohol in C21 corticosteroids. The reaction was carried out at 45 °C for 2 h and the fluorescent derivatives were separated on silica stationary phase with a mixture of 2-propanol and hexane as a mobile phase in the gradient mode. The low temperature in derivatization prevented the thermal degradation of corticosteroids and avoided reaction with secondary hydroxyl groups [10][11][46,50].
Measuring endogenous glucocorticoids and their metabolites is important as disturbances in the enzyme responsible for their metabolism can cause hypertension. One study described an HPLC-FLD method using 9-AN derivatization for the determination of cortisol, cortisone, and their tetrahydro- and allo-tetrahydro-metabolites in plasma and urine [12][45]. Following extraction with dichloromethane and SPE, 9-AN was used to derivatize the steroids in the samples to their fluorescent products. LOD (S/N = 3:1) achieved with this method was 3.0 ng/mL for all analytes. Similarly, Shibata et al. employed 9-AN for fluorescent derivatization and developed a method to investigate cortisone levels in plasma and urine samples of renal transplant patients who received prednisolone [13][47]. Cortisol, cortisone, prednisolone, prednisone, 6β-hydroxycortisol, and 6β-hydroxyprednisolone were derivatized to their fluorescent esters after extraction with ethyl acetate (Figure 42). The 6β-hydroxycortisone was used as an internal standard. LODs achieved for cortisol, cortisone, prednisolone and prednisone in plasma or urine were 0.1 ng/mL, while those for 6β-hydroxycortisol and 6β-hydroxyprednisolone in plasma or urine were 0.5 ng/mL.
Similar to 9-AN, fluorescent derivatization can be achieved using 1-AN. A study described the use of 1-AN to derivatize 18-oxygenated corticosteroids: 18-hydroxycortisol, 18-hydroxycortisone and 18-oxocortisol in human urine into their fluorescent 21-anthroyl esters [14][43] (Figure 43). A mixture of ether and dichloromethane was used to extract the steroids, and the anthroyl derivatives were enriched by SPE using a CN cartridge column. The LOD was 0.1 pmol (S/N = 5).
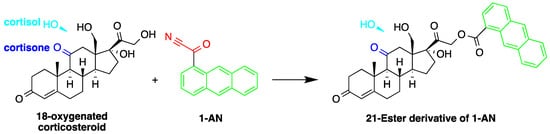
Figure 43. Fluorescent derivatization of 18-oxygenated corticosteroids by or 1-anthroyl nitrile (1-AN).
Fluorescent derivatization of corticosteroids can also be carried out using dansyl hydrazine targeting the carbonyl groups (Figure 57). In a study to determine corticosteroids in human plasma and urine samples were derivatized by dansyl hydrazine and quantified by HPLC-FLD, following extraction with methylene chloride. The linearity range of the method was between 0.5 and 60 ng of cortisol, proving it to be suitable for the routine analysis of cortisol in plasma and urine [15][71].
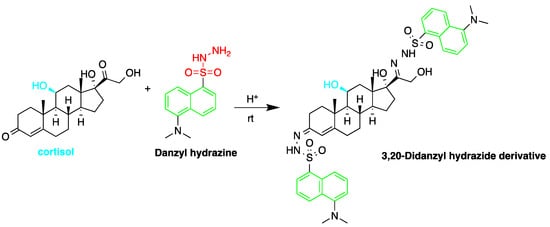
Figure 54. Fluorescent derivatization of carbonyl functionality of cortisol by dansyl hydrazine.
The detection of corticosteroids with ketolic groups in urine is key part in diagnostic procedure of Cushing’s syndrome. Ketol-containing corticosteroids, such as 17-hydroxycorticosteroid (breakdown product of cortisol), are usually excreted in urine as tetrahydro form making their detection using UV absorption at 240 nm not possible. To detect these steroids in urine, derivatization with amidine-containing compounds was used yielding fluorescent compounds [16][60] (Figure 65). Using this approach, the best sensitivity was achieved for cortisol.
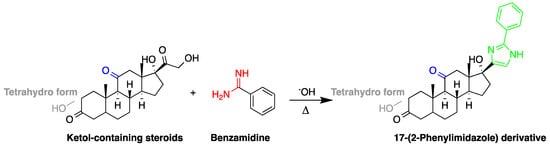
Figure 65. Fluorescent derivatization of ketolic-containing corticosteroids by benzamidine.
The utilization of ketolic groups was also performed by Yamaguchi et al. to detect and quantify corticosteroids in urine after their conversion into fluorescent quinoxalines (Figure 76) [17][67]. They detected nineteen 21-hydroxycorticosteroids in human urine samples achieving LOD = 0.14–29.4 pmol/50 µL injection volume (S/N = 3). In another study by the same group, prednisolone and prednisone were quantified in plasma samples following similar derivatization procedure and analyzed by reversed-phase liquid chromatography with isocratic elution. The LOD of prednisolone and prednisone was 3 ng/mL in plasma (S/N = 3) [18][65].
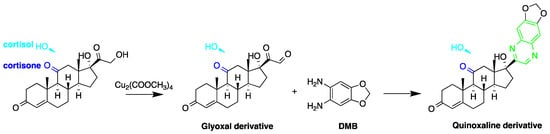
Figure 76. Fluorescent derivatization of ketol-containing corticosteroids to quinoxaline derivative.
Cholesterol quantification is commonly required in the analysis of biosamples. A study described a simple and sensitive method for the determination of cholesterol and phytosterols, such as β-sitosterol in biosamples (e.g., saliva and urine matrices) and food samples (cow and soybean milk) after derivatization with naproxen acyl chloride in toluene (Figure 87). The method employed a C8 column with a mixture of methanol, isopropanol, and water, achieving a LOD of about 25 nM (S/N = 3). The analysis of cholesterol and sitosterol is usually time-consuming, but using the method, a relatively short period of time was required since solvent concentration, evaporation, and replacement steps were not necessary [19][79].
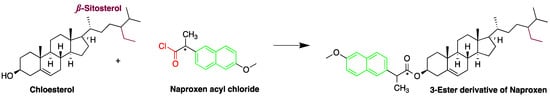
Figure 87. Derivatization of cholesterol by naproxen acyl chloride.
Free 7α-hydroxycholesterol (7-HC) levels in human serum were reported to be a good indicator for bile acid synthesis. An HPLC-FLD method was developed by Saisho et al. to quantify 7-HC levels in dog plasma, with the purpose of studying the effect of cholestyramine on plasma levels of 7-HC. 7-HC was converted to its fluorescence derivative, by two procedures, after being extracted and then purified [20][57]. The two derivatization reagents used were 1-AN and 7-methoxycoumarin-3-carbonyl azide (MC-CON3). The MC-CON3 derivatization resulted in higher fluorescence intensity compared to 1-AN route. 1-AN produced only a C-3 fluorescent derivative due to the bulky anthracene group and steric hindrance around the C-7 position. In comparison, MC-CON3 yielded a double coumarin derivative at the C-3 and C-7 positions (Figure 98) producing a LOD of 4 pg (S/N = 5).
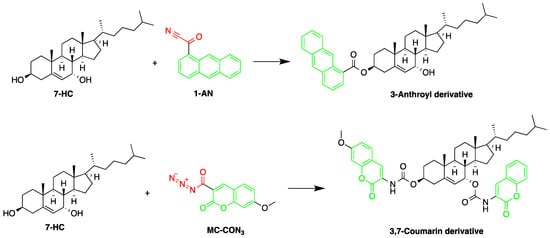
Figure 98.
Derivatization of 7α-hydroxycholesterol (7-HC) by 1-anthroyl nitrile (1-AN) or 7-methoxycoumarin-3-carbonyl azide (MC-CON
3
).
3. Detection of Steroid Hormones
Detection of steroid hormones and their metabolites are important in diagnosis of metabolic diseases. Several HPLC-FLD methods were described for the detection of steroid hormones. A study developed a method for monitoring progesterone and 17-hydroxyprogesterone in the serum from pregnant women. The quantification employed fluorescent derivatization using 4,4-difluoro-5,7-dimethyl-4-bora-3a,4adiaza-s-indacene-3-propionohydrazide (BODIPYTM FL hydrazide) [15][71] (Figure 109). The derivatization was carried out in ethanol at room temperature (about 22 °C) for 15 h. This derivatization method was reported to be 50 times faster than that using dansyl hydrazine. The LODs for progesterone, 17-hydroxyprogesterone, dehydroepiandrosterone, androstenedione, testosterone and 17-methyltestosterone were in the range of 550–3700 fmol per 10 µL injection (S/N = 5).
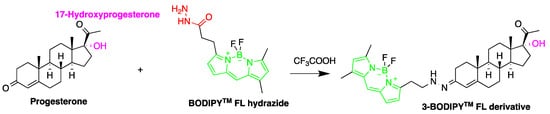
Figure 109.
Derivatization of progesterone by 4,4-difluoro-5,7-dimethyl-4-bora-3a,4adiaza-s-indacene-3-propionohydrazide (BOD-IPYTM FL hydrazide).
The derivatization reagent 1-AN was used by Shimada et al. to determine the endogenous steroid pregnenolone in Wistar and Sprague–Dawley rat brain samples [10][46]. The samples were homogenized in isotonic saline then deproteinized with methanol, before subjected to sequential steps of extraction and derivatization. The samples were derivatized with 1-AN (Figure 110) and the excess reagent was removed by purification, carried out on two successive silica gel columns. The 3β-hydroxy-16-methylpregna-5,16-dien-20-one was used as an internal standard.
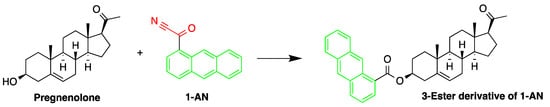
Figure 110. Derivatization of pregnenolone by 1-anthroyl nitrile (1-AN).
Several steroids in animal tissue were detected in another study using ultraviolet, fluorometric, and electrochemical detectors [21][80]. The study demonstrated the selectivity of FLD in determination of estradiol by exploiting its fluorescence emission, although it was eluted with the same fraction containing other steroids and their respective metabolites. Extraction with methanol allowed the separation of acidic corticosteroids (diethylstilbestrol, estradiol, zeranol/zearalenone, and their metabolites) from their neutral anabolic counterparts (testosterone, trenbolone and progesterone).
Urine is the best source for the estimation of estrogen concentrations, since they are primarily excreted renally as glucuronides and sulfates. Previous studies used urine samples for the determination of estrogen levels in their studies, albeit with different extraction methods [22][23][42,81]. Mao et al. used chemical hydrolysis in methanol and concentrated hydrochloric acid at 70 °C for 1 h to release the conjugation before SPE sample preparation, whereas Kumar et al. used a fabric phase sorptive extraction (FBSE) procedure, which offered shorter sample preparation times and 400 times higher sorbent loading. This method achieved a lower detection limit and analysis time, proving to be greener and more economical. The LODs for β-estradiol, 17α-ethinylestradiol, and bisphenol A were 20 pg/mL, 36 pg/mL, and 42 pg/mL, respectively.
In another study, Mao et al. used p-nitrobenzoyl chloride at 25 °C as a derivatization reagent, which can easily react with the hydroxyl and phenolic hydroxyl groups of organic chemicals (Figure 112) without the need for a catalyst [22][42]. Moreover, 4-nonylphenol, bisphenol A (BPA), 17α-ethinylestradiol, and three endogenic estrogens, including 17α-estradiol, 17β-estradiol, and estriol, were determined in urine samples collected from 20 healthy volunteers. Samples were hydrolyzed with HCl and subjected to SPE purification. Separation was performed on a C18 column with gradient elution using acetonitrile and water as a mobile phase. The LODs of the method were 2.7 µg/L for BPA and 17β-estradiol, 2.9 µg/L for 4-nonylphenol, 4.6 µg/L for 17α-estradiol and 17α-ethinylestradiol, and 8.3 µg/L for estriol.
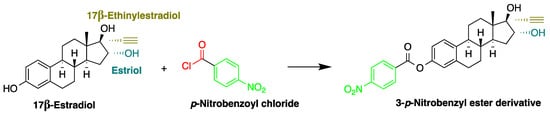
Figure 112. Derivatization of 17β-estradiol by p-nitrobenzoyl chloride.
A recent study examined the determination of estrone (E1), 17β-estradiol (E2), and estriol (E3) and their conjugated metabolites levels in cow and river buffalo meat [24][32]. Samples were extracted with methanol, enzymatically deconjugated and purified by C18 SPE before they are analyzed by HPLC-FLD. The effect of temperature on the studied steroid concentrations was also investigated and it was shown that heating processes was not able to significantly affect the level of phenolic estrogens in meat.
Estrogens in human urine samples were determined by HPLC-FLD following vortex-assisted dispersive liquid–liquid microextraction (VA-DLLME). Fine droplets of nonanoic acid floating on the top of sample solution were used to extract the estrogens using vortex-mix to assist the dispersion of the extraction solvent into the aqueous sample. Derivatization was not required in this work as the high extraction efficiency of DLLME improved the sensitivity and shortened the analysis time. LOD values were 0.01 ng/ml for E3, 0.01 ng/mL for βE2, and 0.06 ng/mL for E1, respectively [25][82].
The abuse of anabolic steroids has gained worldwide concerns, as frequent high doses can irreversibly affect the endocrine system, mineral metabolism, and may result in hepatic carcinomas [26]. Therefore, several committees have prohibited or imposed strict rules on their use for illegal purposes, such as increasing performance activity for athletes and stimulating meat production in cattle and poultry. Amin et al. used micellar chromatography with the detergent solution as the mobile phase to detect testosterone and bolasterone in human urine samples [26]. The method made use of fluorescence by means of energy transfer from the aromatic carbonyls in the anabolic steroids to the terbium ion in micellar media. No sample preparation was required and urine samples were injected directly onto the HPLC column. Excitation of terbium by means of energy transfer from steroids resulted in 183-fold fluorescence enhancement. The detection limits were 10 ng/200 µL injection volume for testosterone and 2 ng/200 µL for bolasterone.
4. Detection of Endocrine Disruptive Chemicals (EDCs)
The analysis and detection of steroid blood levels is vital for the investigation of food safety and effect on health [22][27][42,83]. Many studies have reported the presence of synthetic steroids in our daily food, both from plant and animal sources [28][29][30][31][61,84,85,86]. BPA, an industrial chemical, was found in biological fluids because of its ability to leach into food or liquids or through dental sealants into patient’s saliva [24][22][28][32][33][32,42,61,62,63]. One of the reasons behind their existence is that farmers increase their profit by using endocrine disruptive chemicals (EDCs) to support the feed conversion and growth rate in animals. Substances with hormonal actions are prohibited in the European community for use in animals intended for meat production due to their possible toxic effects on public health [27][83].
When bound to human estrogen receptors, they can stimulate the transcriptional activity of various estrogen receptor subtypes. The increase in levels of estrogen may be linked to the possibility of cancer occurrence among meat consumers [30][85].
In addition, studies have proved the ability of BPA to cross the placenta and blood brain barrier, in turn affecting the endocrine organs in animals and humans. This includes increasing prostate size, decreasing the number of produced sperms, and causing early puberty in females, in addition to effects on sexual differentiation [32][34][62,87]. Therefore, several HPLC-FLD methods were developed and used for quantification of EDC’s.
A sensitive HPLC method was developed for quantification of BPA and eight compounds of alkylphenol [35][59]. The alkylphenols assessed were; 4-sec-butylphenol, 2-tert-butylphenol, 3-tert-butylphenol, 4-tert-butylphenol, 4-n-pentylphenol, 4-tert-pentylphenol, 4-n-hexylphenol, and 4-n-heptylphenol. Derivatization was done using 2-(4-carboxyphenyl)-5,6-dimethylbenzimidazole (CDB) at 40 °C for 60 min. A C18 stationary phase was used for separation. The reported detection limits were in the range of 0.1–10.0 pg/mL. The method was applied to determination of bisphenol A in mother and infant rat serum samples.
The fluorescent reagent 4-(4,5-diphenyl-1H-imidazol-2-yl) benzoyl chloride (DIB-Cl) (Figure 132) was used by Sun et al. to detect EDC’s. To the evaporated sample residue, DIB-Cl suspension in acetonitrile and triethylamine in acetonitrile were added and reacted at room temperature [33][63].
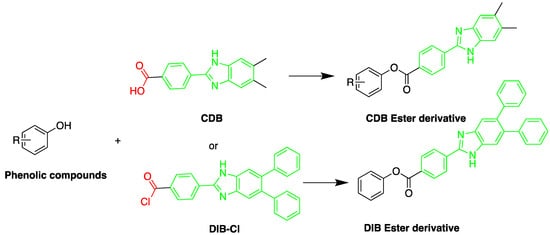
Figure 132. Derivatization of alkyl phenols by 2-(4-carboxyphenyl)-5,6-dimethylbenzimidazole (CDB) or 4-(4,5-diphenyl-1H-imidazol-2-yl) benzoyl chloride (DIB-Cl).
The study determined BPA in rat brain samples using HPLC-FLD coupled with a microdialysis [33][63]. A microdialysis probe was inserted into the hypothalamus of rat brains and artificial cerebrospinal fluid was used for perfusion. After the administration of a single intravenous or oral dose of BPA, concentrations were monitored in brain and plasma for 8 h. The obtained data proved that BPA could penetrate the blood brain barrier. The LOD of BPA was 0.3 ppb in 60 µL brain microdialysate at (S/N = 3).
Kuroda et al. used a similar method for determination of BPA in human blood serum and ascitic fluid samples. Samples were extracted by LLE using chloroform [32][62]. The LOD of BPA for both samples was 0.04 ppb at (S/N = 3). Human breast milk was also used to investigate the presence of BPA employing DIB-Cl as a fluorescent derivatizing agent [28][61]. Two steps of LLE were applied, and two C18 columns were used to separate DIB-BPA from the endogenous material in breast milk. The detection limit in 23 samples of healthy lactating women was 0.11 ng/mL at (S/N = 3).
The chromatographic conditions, sample preparation and detection limits of the HPLC-FLD methods that were employed in the steroid studies reviewed in this article are summarized and presented in Table 1.
Table 1.
HPLC-FLD method conditions used for steroid detection.
| Detected Steroids | Sample Type | Column Chemistry | Mobile Phase (v/v) | Derivatization Agent | Excitation (λex) and Emission Wavelengths (λem) (nm) | Extraction Method | LOD | References |
|---|---|---|---|---|---|---|---|---|
| PL and PN |
Human plasma | C18 | MeOH:ACN:1.0 M ammonium acetate (38:25:45) | DMB | 350, 390 | LLE | 3 ng/mL | Yamaguchi et al. 1991 [18][65] |
| F and E | Biological samples | Keystone Hypersil | H2O:MeOH:ACN (50:33.3:16.7) | 9-AN | 305–395, 430–470 | SPE | F: 50 pg E: 70 pg |
Haegele et al. 1991 [36][48] |
| F | Human serum | C18 | 1: 10 mM potassium biphthalate 2: ACN Tetrahydrofuran: 19 mM potassium biphthalate (40:6:54) Both adjusted to pH 1.85 with trifluoroacetic acid |
Sulfuric acid-ethanol | 365, 520 | - | 0.30 pg/dL | Nozaki et al. 1991 [5][53] |
| F | Human urine | C18 | Gradient of ACN: 36.4 mmol/L phosphate (45:55; pH 1.85 with trifluoroacetic acid) |
Sulfuric acid-ethanol | 365, 520 | SPE | 0.26 pg/dL | Nozaki et al. 1992 [6][54] |
| 7α-Hydroxycholesterol | Dog plasma | Develosil Ph-5 | Acetonitrile: Water (5:2) |
1-AN | 338, 411 | LLE | 4 pg | Saisho et al. 1998 [20][57] |
| Corticosteroids | Urine | Silica | 2-Propanol–hexane | 9-AN | 370, 470 | Enzyme hydrolysis, extraction with 0.5 M NaOH | NR | Neufeld et al. 1998 [11][50] |
| F and E | Human plasma | C18 | ACN: 0.3 mM ortho-phosphoric acid (470:530) | 9-AN | 360, 460 | SPE | 3.0 ng/mL | Glowka et al. 2009 [12][45] |
| Corticosterone | Rat urine | CN | ACN: H2O (24.5:75.5) | post-column reaction with sulfuric acid |
460, 510 | LLE | 0.5 pmol | Sudo et al. 1990 [3][55] |
| F | Human hair | C18 | MeOH:H2O 60:40 |
sulfuric acid | 360, 480 | LLE | 1 pg/mg | Gao et al. 2010 [2][56] |
| EE2, E2, and BPA | Human urine and aqueous samples | C18 | ACN:MeOH:H2O (30:15:55) | - | 280, 310 | FPSE | E2: 20 pg/mL EE2: 36 pg/mL BPA: 42 pg/mL |
Kumar et al. 2014 [23][81] |
| BPA, NP, E2, EE2, and E3 | Human urine | C18 | Gradient elution of ACN and H2O | p-nitrobenzoyl chloride | E2, E3: 282, 315 BPA, NP, EE2: 228, 316 |
SPE | BPA and E2: 2.7 μg/L NP: 2.9 μg/L E2 and EE2: 4.6 μg/L E3: 8.3 μg/L |
Mao et al. 2004 [22][42] |
| E, testosterone, methyltestosterone, bolasterone, testosterone acetate, progesterone | Urine | C18 | 0.01 M Tb(NO3)3, 0.1 M sodium dodecyl sulfate (SDS), and 20% acetonitrile |
- | 245, 547 | SPE | Down to 100 pg/mL | Amin et al. 1993 [26] |
| BPA and 8 alkylphenols (4-sec-Butylphenol, 2-tert-Butylphenol, 3-tert-butylphenol, 4-tert-butylphenol, 4-n-Pentylphenol, 4-tert-pentylphenol, 4-n-hexylphenol, and 4-n-heptylphenol) |
Rat plasma and blood | C18 | MeOH:Water (10:90) | 2-(4-carboxyphenyl)-5,6- dimethylbenzimidazole |
Derivatized: 336, 440 Native: 275, 315 |
LLE | BPA: 0.1 pg/mL Alkylphenol: 0.7–10 pg/mL |
Katayama et al. 2001 [35][59] |
| progesterone and 17-hydroxyprogesterone dehydroepiandrosterone, androstenedione, testosterone and 17-methyltestosterone |
Serum from pregnant and non-pregnant women | Wakosil 5C4 | Acetonitrile:Water (7:3) | BODIPY FL hydrazide | 495, 516 | LLE | 550–3700 fmol per 10 μL | Katayama et al. 1998 [37][68] |
| aldosterone, corticosterone, F, E, dexamethasone, fluocinolone acetonide, triamcinolone and triamcinolone acetonide |
Human plasma | C18 | Water:MeOH (25:75) containing 5 mmol/L tetramethylammonium hydrogen sulphate | CDB | 334, 418 | LLE | 0.06–0.3 pg per 100 μL | Katayama et al. 1992 [9][58] |
| BPA | Breast Milk | C18 | 1: ACN:H2O:MeOH (72:13:15) 2: ACN:0.1 M acetate buffer (pH 5.5):MeOH (55:12:33) |
DIB-Cl | 350, 475 | SPE then LLE | 0.11 ng/mL | Sun et al. 2004 [28][61] |
| BPA | Human blood serum and ascitic fluid samples | C18 | 1: ACN: H2O:MeOH (72:13:15) 2: ACN:0.1 M Acetate buffer (pH 5.5):MeOH (55:12:33) |
DIB-Cl | 350, 475 | LLE | 0.04 ppb | Kuroda et al. 2003 [32][62] |
| BPA | Rat brain rat plasma |
C18 | 1: ACN: H2O:MeOH:Tetrahydrofuran (55:10:35:2.5) 2: ACN:0.1 M Acetate buffer (pH 3.0):MeOH (35:10:55) |
DIB-Cl | 350, 475 | LLE | 0.3 ppb in 60 μL rat brain 4.6 ppb in 50 μL rat plasma |
Sun et al. 2002 [33][63] |
| F, E, PL, PN, 6β-OHF, 6β-OHP and 6β-OHE |
Human plasma and urine | Cosmosil 5SL | diethylene dioxide: ethyl acetate:chloroform:n-hexane:pyridine (500:100:100:1400:21) | 9-AN | 360, 460 | LLE | F, E, PL and PN: 0.1 ng/mL 6β-OHF and 6β-OHP: 0.5 ng/mL |
Shibata et al. 1997 [13][47] |
| Corticosterone | Rat serum | C18 | 60% MeOH: 40% 5 mM triethylamine, pH 3.3 |
- | 375, 485 | LLE | 0.1 ng | Mason et al. 1992 [7][77] |
| 18-Oxygenated corticosteroids, 18-hydroxycortisol, 18- hydroxycortisone and 18-oxocortisol |
Human urine | μBondasphere phenyl | A: 10 mM ammonium acetate: MeOH (50:50) B: ACN |
1-AN | 370, 470 | LLE and SPE | 0.1 pmol | Kurosawa et al. 1995 [14][43] |
| Cholesterol and sitosterol | Saliva and urine biosamples, Cow milk, and Soybean milk | C8 | MeOH:isopropanol:H2O (90:5:5) |
naproxen acyl chloride | 231, 352 | LLE | 25 nM per 10 μL injected volume | Lin et al. 2007 [19][79] |
| Pregnenolone | Rat brain | C18 | MeOH:H2O (9:1) | 1-AN | 370, 470 | SPE | NR | Shimada et al. 1996 [38][27] |
| C21 steroids; corticoids | Steroid standards | C18 | MeOH:H2O:cyclodextrin | 1-AN | 360, 460 | NR | NR | Shimada et al. 1991 [39][44] |
| Triamcinolone | Human plasma | C18 | ACN and 0.3 mM ortho-phosphoric acid |
9-AN | 360, 460 | SPE | 1 ng/mL | Glowka et al. 2006 [40][88] |
| Butane acid-(5-androsten-17-one-3beta-ol)-diester (A1998) | Rat plasma | C18 | 25 mM acetate buffer (pH 3.7):ACN Alfaxalone: (45:55) Pregnanolone: (40:60) |
Dansyl hydrazine | 332, 516 | LLE | 10 ng/mL | Visser et al. 2000 [41][69] |
| Alfaxalone and pregnanolone | Rat plasma | C18 | Gradient mixture of ACN and H2O |
Dansyl Hydrazine | 350, 520 | - | 0.025 μg/mL | Peng et al. 2007 [42][70] |
| EED | Oral contraceptive tablets | STAR RP-18e | ACN:H2O (47:53) | - | EED: 285, 310 | - | EED: 0.0538 μg/ml | Sarafinovska et al. 2006 [43][12] |
| EED and drospirenone | Oral contraceptive tablets | STAR RP-18e RP | ACN:H2O (47:53) | - | 285, 310 | - | EED: 0.00065 μg/mL DROSP: 0.0774 μg/mL |
Sarafinovska et al. 2009 [44][13] |
| EED | Coated tablets | LiChroCART 100RP | ACN:H2O (50:50) | - | 280, 310 | - | EED: 0.02 μg/mL | Silva et al. 2013 [45][89] |
| Sodium E1 sulphate, sodium equilin sulphate, E1 and equilin |
Raw materials and Pharmaceuticals |
5 ODS2 | TEA phosphate buffer (pH 4.0; 0.05 M):ACN 1— (70:30, v:v) 2—for unconjugated estrogens: (66:34) |
Postcolumn on line photochemical derivatization | 280, 410 or 312 | - | 0.01–1.38 pmol | Gatti et al. 1998 [46][90] |
| E1, 17β-Estradiol, E3, BPA, NP, OP |
Fish, chicken, aquaculture pond water sample | C18 | Gradient program of 1: H2O: 5% ACN 2: H2O: ACN |
BCEC-Cl | 279, 380 | DLLME | 0.02–0.07 µg/L | Wu et al. 2015 [47][29] |
| E1, E2, and E3 |
Cow and river Buffalo | C18 | 1: ACN:H2O:Formic Acid (40:60:0.4) 2: ACN:H2O:Formic Acid (90:10:0.4) |
- | 280, 310 | LLE and SPE | 5–10 ng/kg | Shahbazi et al. 2016 [48][31] |
| α- and β-Trenbolone | Bovine muscle and liver | C18 | MeOH:H2O (60:40) | - | 364, 460 | LLE then SPE | bovine muscle: 0.2 ng/g liver: 1.0 ng/g |
Yoshioka et al. 2000 [49][33] |
| NP, 4-nonylphenol mono-(NP1EO), diethoxylates (NP2EO), BPA, TBP, and OP |
Fish and shellfish | Inertsil PH | Gradient program of A: H2O B: MeOH |
- | 275, 300 | LLE | NP NP1EO and NP2EO : 2 ng/g BPA, BP and OP: 1 ng/g |
Tsuda et al. 2000 [24][32] |
| Nonylphenol and its ethoxylates | Fish tissue | Hypersil APS | Hexane:ethanol (98:2) | - | 230, 300 | Pressurized fluid extraction | 4–15 ng/mL | Datta et al. 2002 [50][28] |
| E2 and EE2 | Poultry litter | C18 | Gradient program of A: H2O B: ACN |
- | 280, 312 | LLE | E2: 4.0 μg/kg EE2: 2.6 μg/kg |
Lu et al. 2014 [51][91] |
| E2 and EE2 | Waste Water | C18 | Gradient program of A: H2O B: ACN |
- | 282, 306 | SPE | 2.5 ng/L | Liz et al. 2017 [52][17] |
| E3, E2, EE2, HEX, mestranol |
Water, sediment | Poroshell 120 EC | H2O:ACN 50:50 | - | 275, 310 | SPE | Water: 6–24 ng/L Sediment: 0.1–0.9 ng/g |
Perez et al. 2015 [53][92] |
| E2, 17α-EE2, and E1 | Water | C18 | Gradient program of A: ACN B: H2O acidified at pH 3.6 with glacial acetic acid |
- | 230, 302 | SPE | 10 to 1100 ng/L |
Patrolecco et al. 2013 [54][93] |
| OP, NP, BPA, diethylstilbestrol, E1, EE2, E2, and E3 |
Wastewater samples |
C18 | Gradient program of A: 5% ACN B: ACN |
EASC | 262, 430 | SPE | 0.3–0.7 ng/L | Zhang et al. 2012 [55][52] |
| E2 and EE2 |
Tap, surface and waste water | C18 | Water:acetonitrile mixture (50:50) | - | 280, 310 | DLLME | E2: 2.0 ng/L EE2: 6.5 ng/L |
Lima et al. 2013 [56][37] |
| BPA, 17β-estradiol, and 17α-ethynyl estradiol | Drinking water | LiChro-sorbs RP18 | 10 mM H3PO4:55% MeOH (45:55) | - | 280, 310 | PAC | BPA: 201 ng/L E2: 313 ng/L EE2: 284.5 ng/L |
Yoon et al. 2003 [57][94] |
| 17α- and 17β-Trenbolone | River water | C18 | Gradient program of MeOH:H2O |
- | 359, 458 | SPE | 4 ng/L | Durhan et al. 2005 [58][95] |
| NP, OP, NP polyethoxylates |
municipal wastewater treatment plants | C18 | Gradient program of H2O:ACN |
- | 229, 310 | LLE | OP: 2 ng/L NP: 11 ng/L NPE: 52 ng/L |
Snyder et al. 1999 [59][5] |
| E2, E3, BPA, and 17β-ethinylestradiol |
environmental waters |
C18 | ACN:0.02 mol/L phosphate solution (45:55) | - | 227, 315 | Synthesized in-tube SPME device | 0.006–0.10 ng/mL | Wen et al. 2006 [60][96] |
| E2, E3, EE2, 3-methyl ether EE2, NP, OP, POE(1-2) nonyl phenol and BPA |
River water | C18 | Gradient program of Milli-Q H2O and ACN |
- | 230, 290 | on-line SPE | 20–50 ng/L | Ying et al. 2002 [61][97] |
| Endocrine disruptors; BPA and EE2 | Environmental water samples | C18 | MeOH:0.025 mol/L Na2HPO4 buffer (70:30) | - | 220, 315 | A poly(acrylamide-vinylpyridine) monolithic capillary column |
BPA: 0.064 ng/mL 17α-ethinylestradiol: 0.12 ng/mL |
Fan et al. 2005 [62][98] |
| Brassinosteroids | Plant: (Vicia faba L.) |
C18 | ACN: H2O (90:10) | 9-Phenanthreneboronic acid | 305, 375 | LLE | 50 pg | Gamoh et al. 1989 [63][51] |
| Brassinolide | Arabidopsis thaliana, Daucus carota and Brassica campestris L. leaves’ samples |
C18 | Gradient program of A: H2O and ACN B: ACN and 0.1% Formic Acid |
- | 305, 375 | UA-DLLME | 8.0 ng/L | Lv et al. 2014 [64][99] |
| Brassinolide and castasterone | Pollen of orange (Citrus sinensis Osbeck) | C18 | ACN:H2O (80:20) | Dansylaminophenylboronic acid |
345, 515 | LLE | NR | Motegi et al. 1994 [65][72] |
| Norgestrel, norethindrone, EE2, gestodene, and norethisterone acetate | Meat samples | Hypersil GOLD | Gradient program of (A) H2O containing 5% ACN (B)ACN |
Fmoc-Cl | 250, 395 | MSPE | 1.4 × 103–8.7 × 103 | Qianyu Li et al. 2018 [66][73] |
| F and E | Human urine samples | C18 | ACN:0.3 mM orthophosphoric acid (470: 530) | 9-AN | 360, 460 | LLE | LLOQ: F: 27.6 nmol/L E: 27.7 nmol/L |
Kosicka et al. 2018 [67][49] |
| FFA | Edible oils and foodstuff | C8 | Gradient system: A: H2O B: ACN/DMF (1:1) C: ACN (100%) |
BCETS | 279, 380 | Supercritical CO2 and organic solvent extraction | 0.22–1.06 ng/mL | Li et al. 2011 [68][74] |
| OP, NP, TBP, BPA, E1, E2, E3 | Milk samples | C18 | Gradient program of A:5% ACN in H2O B: ACN |
BQEIC | 302, 401 | LLE | 10.5–13.8 ng/L | Liu et al. 2018 [69][75] |
| E3, 2-OHE2, 17β-E2, 17α-E2, EE2, HEX | Dairy products | C18 | Gradient Flow A: 1 mM formic acid in ACN B: 1 mM formic acid at pH 3.50 |
- | 280, 310/320 | HF-LPME | 0.23–14.8 μg/kg | Bárbara Socas-Rodríguez et al. 2014 [70][36] |
| Zearalenone | Edible oil | C18 | Gradient program of A: H2O, B: ACN |
274, 456 | SPE | 10 μg/kg | Drzymala et al. 2015 [71][100] | |
| EE2, E1, E2, E3, and progesterone | Drinking water and wastewater samples | C18 | Gradient program of A: H2O/CH3CN 90/10 v/v B: CH3CN |
200, 315 | SPE | Drinking water: 1–3.8 ng/L Sewage water: 3.8–7.5 ng/L |
Kozłowska-Tylingo et al. 2015 [72][20] | |
| E3, E2, E1 | Human urine | C18 | Gradient program of A: H2O, B: ACN |
280, 310 | VA-DLLME-FOA | E3: 0.01 ng/mL β-E2: 0.01 ng/mL E1: 0.06 ng/mL |
Wang et al. 2015 [25][82] | |
| 17-α-E2, 17-β-E2 benzoate and quinestrol |
Environmental water samples | Zorbax Eclipse SB-C18 | Gradient program of A: ACN, B: H2O |
265, 311 | IF-IHLME | 17-α-Estradiol: 0.04 ng/mL E2 and Quinestrol: 0.05 ng/mL |
Zhang et al. 2017 [73][101] | |
| E2 and EE2 | Tap water samples | Pursuit 5 C18 column | ACN:H2O (50:50), with 200 μL of H3PO4 | 230, 306 | Nanoparticles of graphene oxide/γ-Fe2O3 as a sorbent for SPE |
E2: 2.7 ng/L EE2: 0.8 ng/L |
Fernanda Nunes Ferreira et al. 2020 [74][102] | |
| 17-E2 and E3 | Water samples | C18 | H2O:MeOH:ACN (50:30:20) |
280, 310 | ultrasonication assisted DLLME | DLLME-HPLC/FLD: 7.16–69.22 ng/L | Zhang et al. 2020 [75][14] | |
| E2, 1,3,5(10)-Estratriene-3,17β-diol | Fish and prawn tissue samples | ODS C18 | H2O:MeOH (30:70) | 280,310 | MISPE | 0.023 mg/L | Jiang et al. 2009 [76][40] | |
| BPA, EE2, 4-t- OP, 4-OP, and 4-NP |
River water | C18 | Gradient program of A: ACN B: H2O |
277, 307 | Disposable pipette extraction (DPX) | BPA, EE2, 4-OP and 4-NP: 0.30 μg/L 4-t-OP: 0.60 μg/L |
Gabriela Corazza et al. 2017 [77][103] | |
| E1 and EE2 | Digested sludge | C18-PFP | A: H2O B: ACN E1: (50:50) EE2 (55:45) |
280, 310 | ultrasonic liquid extraction | E1: 0.305 μg/g EE2: 0.052 μg/g |
Vitória L. Louros et al. 2019 [78][104] | |
| E2 | Milk sample | XDB-C18 | MeOH:H2O (70:30) |
280, 310 | SPE | 0.7 ng/mL | Yanan Yuan et al. 2019 [79][105] | |
| Nine BPs | milk samples | C18 | Gradient program of 0.1% formic acid: ACN |
230, 305 | ultrasonically with acetonitrile and cleaned using the QuEChERS technique. |
1.0–3.1 µg/kg | Xiong et al. 2017 [80][38] | |
| EE | river water samples | 5C18 MS-II | ACN: 5.0 mM Tris-HNO3 buffer, pH 7.4 (60:40) | 310, 400 | C18 SPE disk | 7.4 ng/L | Ali et al. 2020 [81][64] | |
| E3, 17β-estradiol glucuronide, 17β-E2, 17α-E2, 17β-E2-3-methyl ether |
wastewater | UPLC C18 | Gradient program of water with 0.1% of ammonia: ACN |
280, 310 | Molecularly Imprinted SPE | 1.4 to 2.5 ng/mL | Rayco Guedes-Alonso et al. 2015 [82][41] |
Abbreviation: solid phase extraction (SPE), liquid–liquid extraction (LLE), dispersive-liquid–liquid extraction (DLLME), ultrasonic-assisted dispersive liquid–liquid microextraction (UA-DLLME), vortex-assisted dispersive liquid–liquid microextraction method based on floating organic acid droplet (VA-DLLME-FOA), ionic liquid foam floatation coupled with an ionic liquid-based homogeneous liquid–liquid microextraction (IF-IHLME), magnetic solid phase extraction (MSPE), fabric phase sorptive extraction (FPSE), molecularly imprinted solid-phase extraction (MISPE), hollow fiber liquid-phase microextraction (HF-LPME), 4-octylphenol (OP), 4-tert-octylphenol (4-t-OP), 4-nonylphenol (NP), 4-tert-butylphenol (TBP), bisphenol A (BPA), estrone (E1), 17β-estradiol (17β-E2), 17α-estradiol (17α-E2), estriol (E3), free fatty acids (FFA), 17α-ethinylestradiol (EE2), ethinylestradiol (EED), cortisol (F), cortisone (E), 4,40-(1,2-diethylethylene)diphenol (HEX), prednisolone (PL), prednisone (PN), 6β-hydroxycortisol (6β-OHF), 6β-hydroxyprednisolone (6β-OHP) and 6β-hydroxycortisone (6β-OHE), room temperature (RT).
Abbreviation: solid phase extraction (SPE), liquid–liquid extraction (LLE), dispersive-liquid–liquid extraction (DLLME), ultrasonic-assisted dispersive liquid–liquid microextraction (UA-DLLME), vortex-assisted dispersive liquid–liquid microextraction method based on floating organic acid droplet (VA-DLLME-FOA), ionic liquid foam floatation coupled with an ionic liquid-based homogeneous liquid–liquid microextraction (IF-IHLME), magnetic solid phase extraction (MSPE), fabric phase sorptive extraction (FPSE), molecularly imprinted solid-phase extraction (MISPE), hollow fiber liquid-phase microextraction (HF-LPME), 4-octylphenol (OP), 4-tert-octylphenol (4-t-OP), 4-nonylphenol (NP), 4-tert-butylphenol (TBP), bisphenol A (BPA), estrone (E1), 17β-estradiol (17β-E2), 17α-estradiol (17α-E2), estriol (E3), free fatty acids (FFA), 17α-ethinylestradiol (EE2), ethinylestradiol (EED), cortisol (F), cortisone (E), 4,40-(1,2-diethylethylene)diphenol (HEX), prednisolone (PL), prednisone (PN), 6β-hydroxycortisol (6β-OHF), 6β-hydroxyprednisolone (6β-OHP) and 6β-hydroxycortisone (6β-OHE), room temperature (RT).
