Endoscopic ultrasound-guided hepaticogastrostomy (EUS-HGS) is widely performed worldwide for various benign and malignant biliary diseases in cases of difficult or unsuccessful endoscopic transpapillary cholangiopancreatography (ERCP). Furthermore, its applicability as primary drainage has also been reported. Although recent advances in EUS systems and equipment have made EUS-HGS easier and safer, the risk of serious adverse events such as bile leak and stent migration still exists. Physicians and assistants need not only sufficient skills and experience in ERCP-related procedures and basic EUS-related procedures such as fine needle aspiration and pancreatic fluid collection drainage, but also knowledge and techniques specific to EUS-HGS.
- hepaticogastrostomy
- endoscopic ultrasound
- endoscopic ultrasound-guided biliary drainage (EUS-BD)
- endoscopic ultrasound-guided hepaticogastrostomy (EUS-HGS)
1. Introduction
2. Step-by-Step Tutorial on EUS-HGS Procedure including Devise Selection
In EUS-HGS, the left lateral branch of the intrahepatic bile duct is first punctured from the stomach or jejunum (in the case of post-gastrectomy) with an fine needle aspiration (FNA) needle, followed by injection of contrast medium and insertion of a guidewire. After the needle is removed, a dilation device is inserted into the bile duct to dilate the tract. Next, an introducer of a SEMSself-expandable metal stent (SEMS) or plastic stent (PS) is inserted into the bile duct. Finally, a stent is deployed between the bile duct and the stomach or jejunum (Figure 1).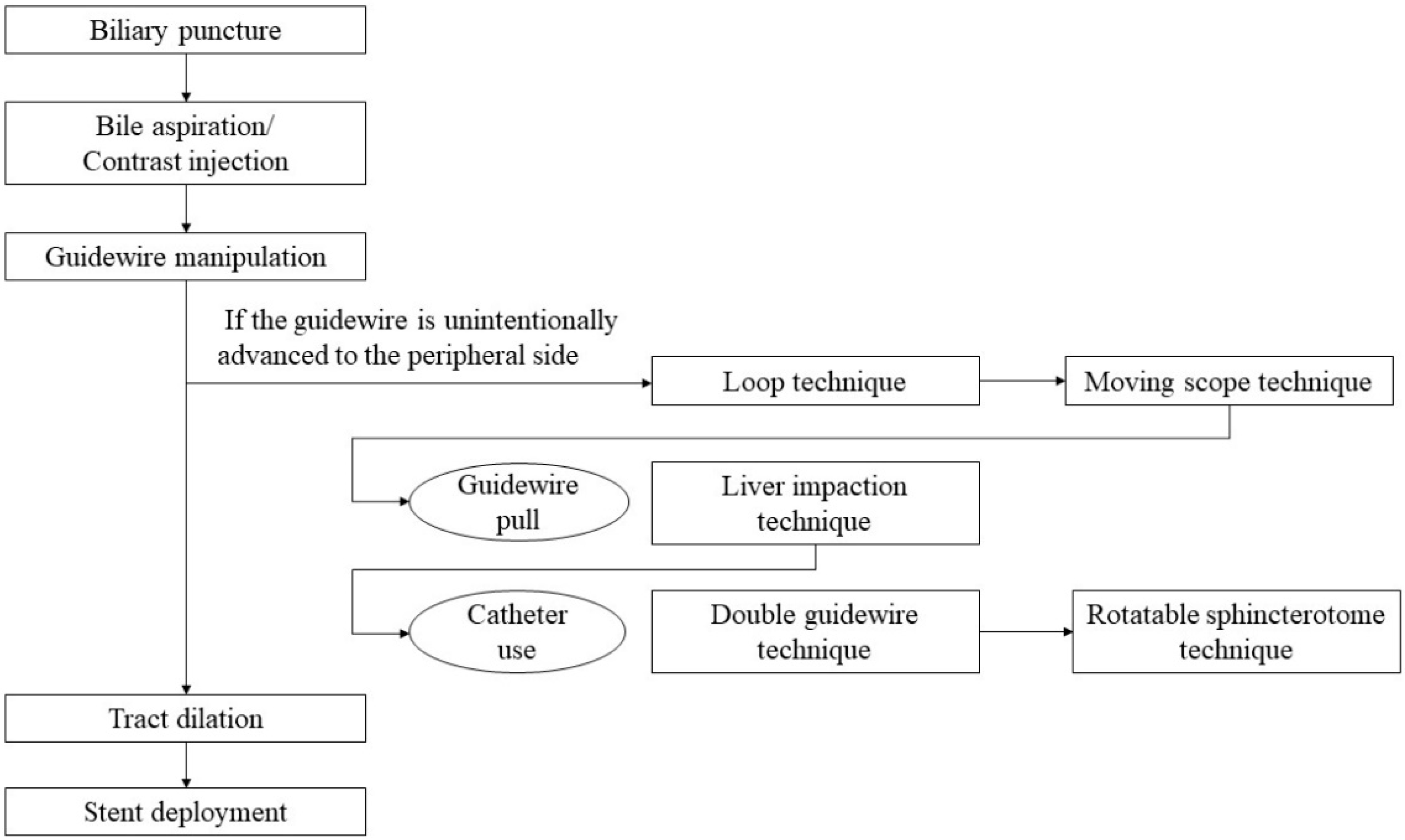
2.1. Selection of Bile Duct Puncture Site and Scope Position
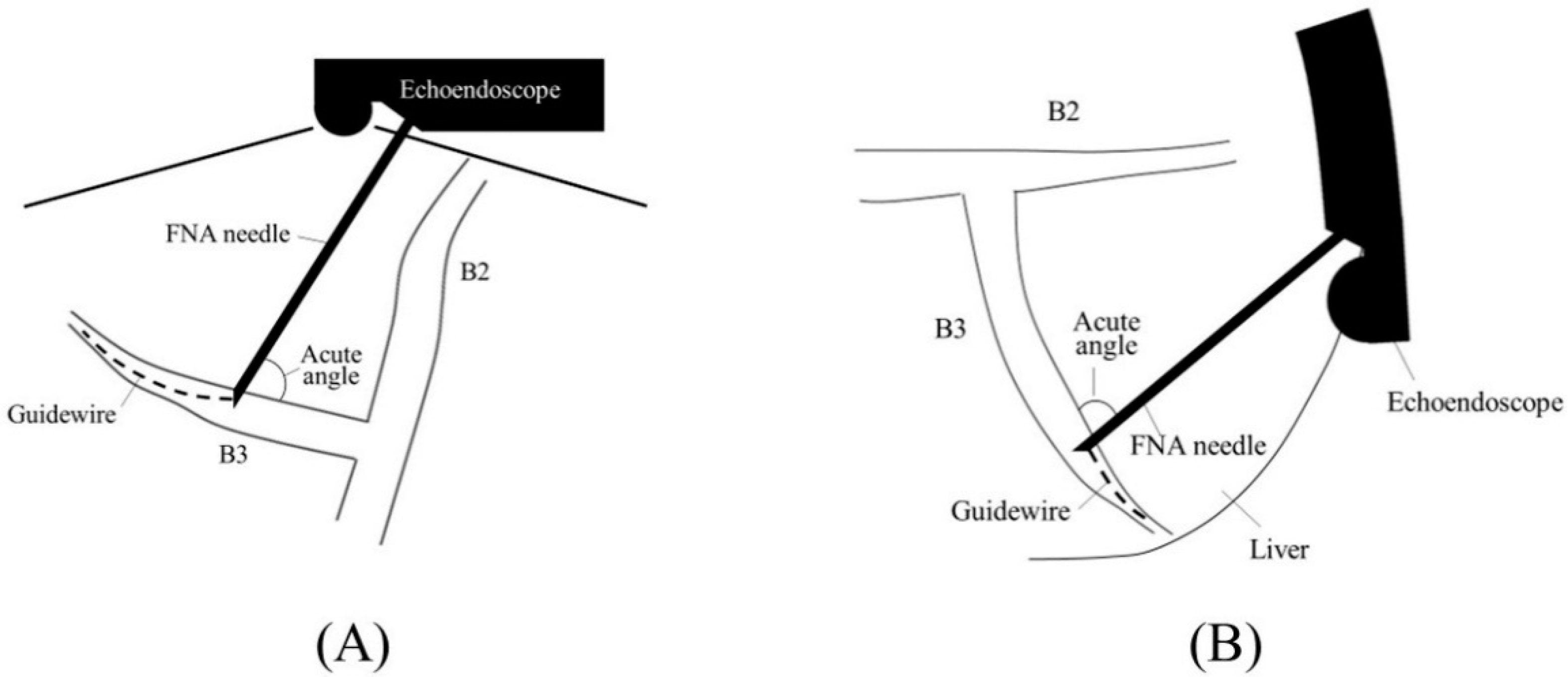
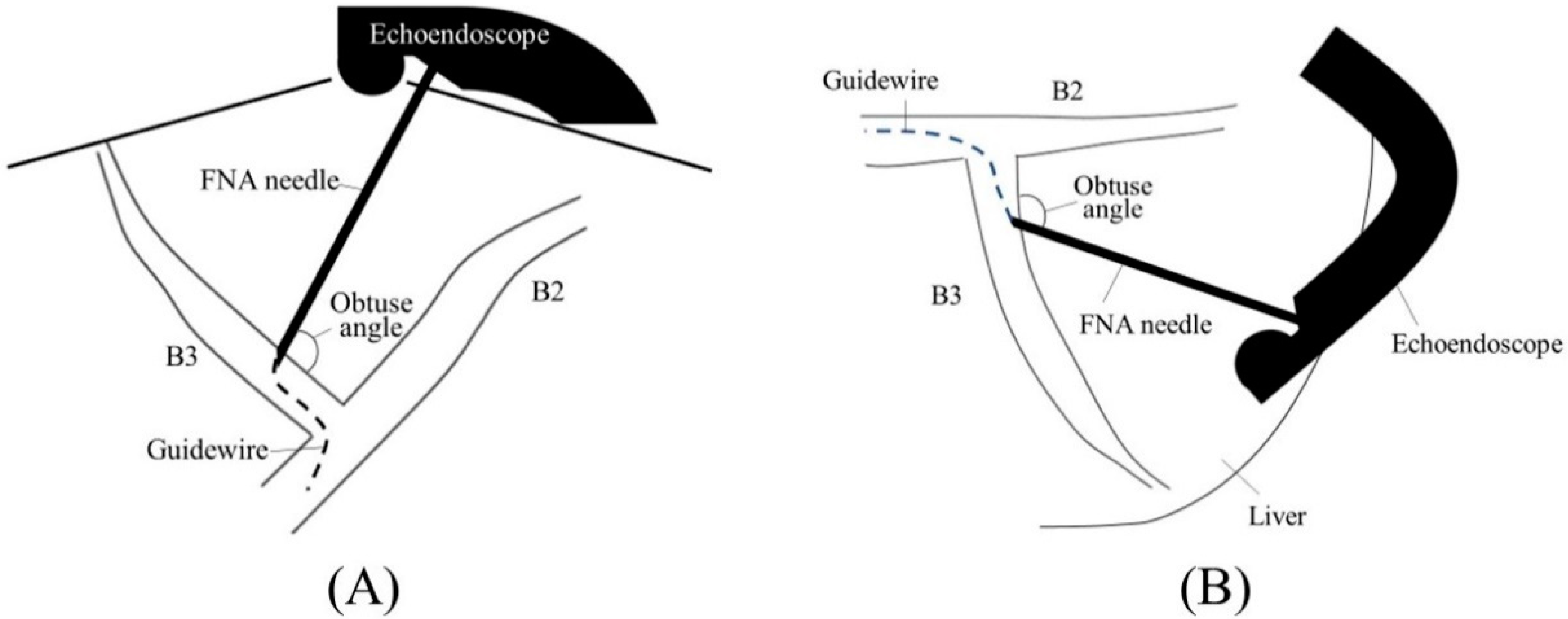

2.2. Biliary Puncture
There are various types of FNA needles, each with a different tip shape and different materials for the needle and sheath. Nitinol needles are more flexible and less prone to bending than steel needles. Additionally, the coil sheath has a higher lumen retention when bent than the plastic sheath. These properties are useful for performing EUS-HGS. The EZ-shot 3 plus (Olympus Medical Systems) (Figure 75A) is the only commercially available nitinol needle with a coil sheath. In EUS-HGS, one of the most difficult steps is the manipulation of the guidewire through the needle [31][22]. The main issue is guidewire shearing, which in turn created a risk of leaving a tip of the guidewire in the patient. The EchoTip Access Needle (Cook Medical, Winston Salem, NC, USA) is a dedicated needle for interventional EUS, which has a sharp stylet for puncture, and the needle tip becomes blunt when the stylet is removed, thus avoiding guidewire shearing [17,32][23][24] (Figure 75B).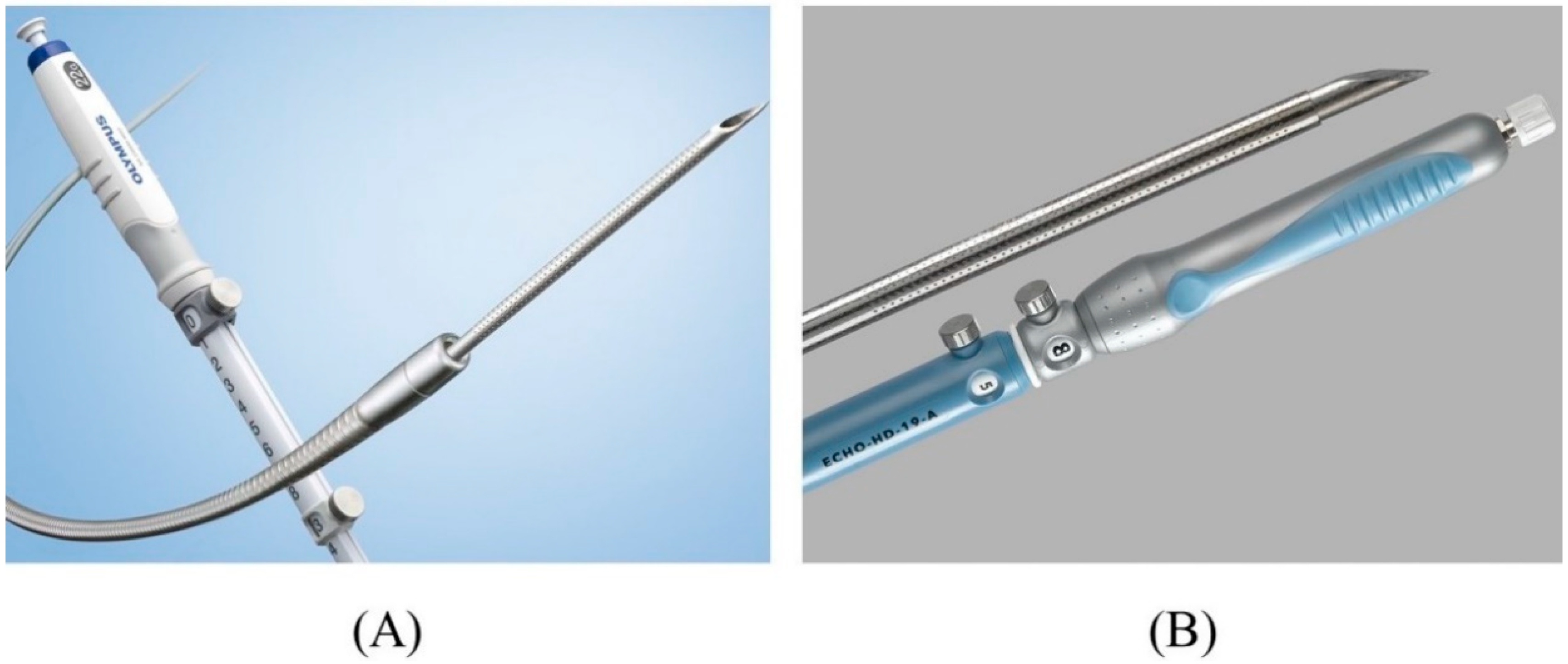
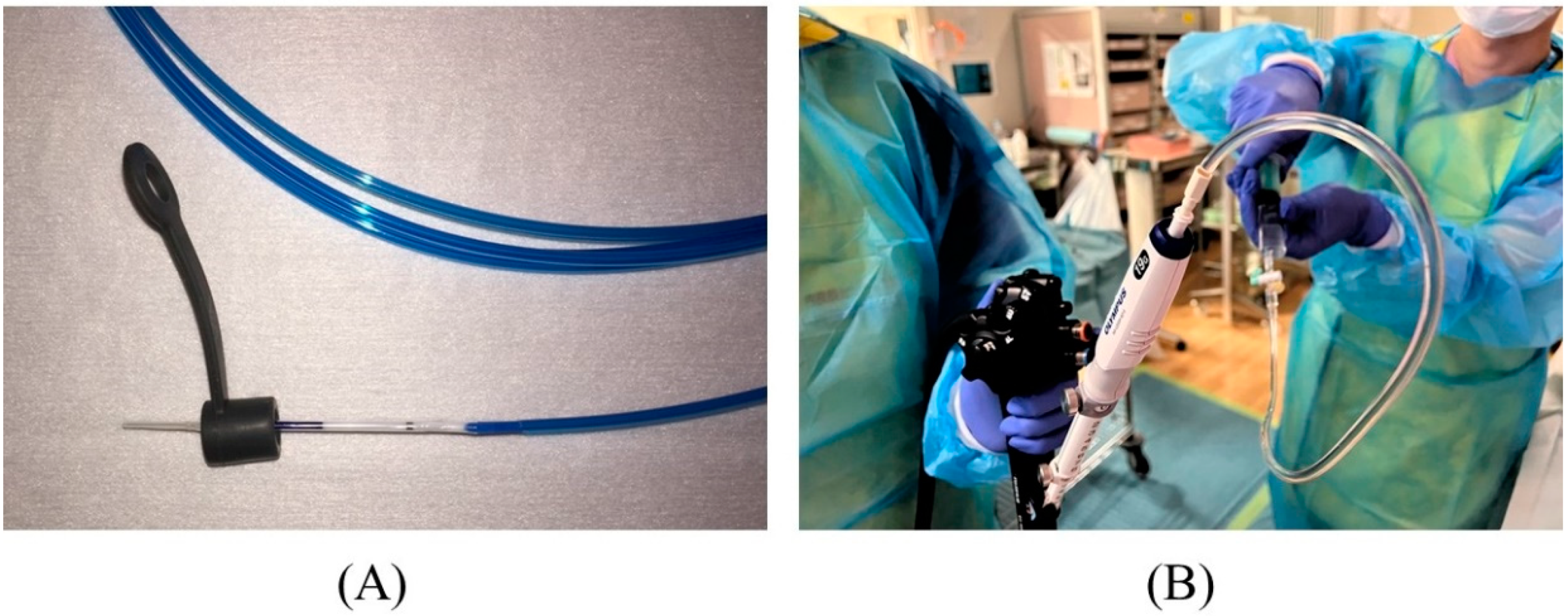 Figure 86. Preparation for puncture. The biopsy valve is attached to a dilation device (A). The needle stylet is removed, and a syringe filled with contrast medium is attached to the needle to pre-fill the lumen with contrast medium (B).
Figure 86. Preparation for puncture. The biopsy valve is attached to a dilation device (A). The needle stylet is removed, and a syringe filled with contrast medium is attached to the needle to pre-fill the lumen with contrast medium (B).
2.3. Contrast Injection
2.4. Guidewire Manipulation
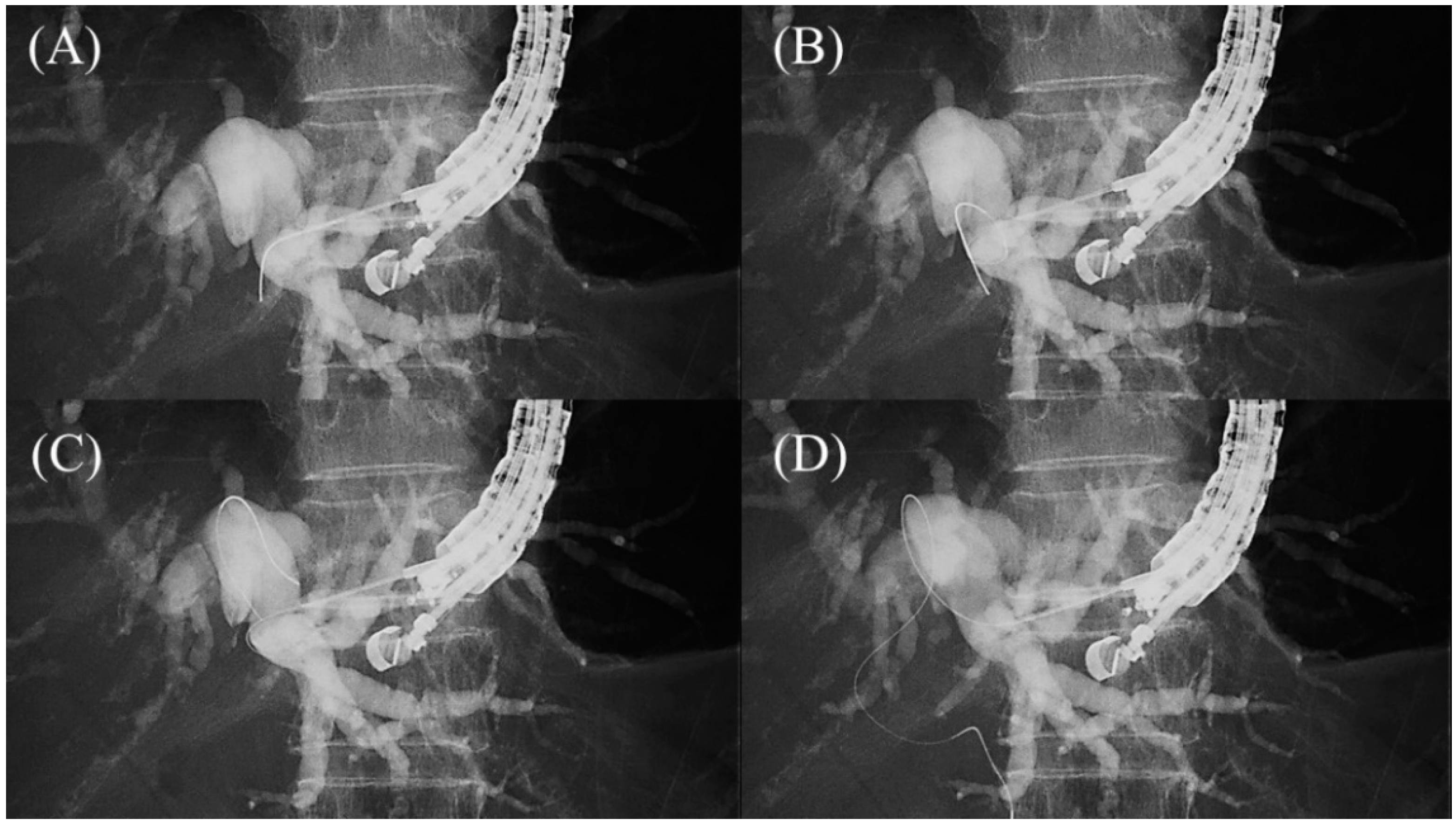 Figure 107. Loop technique for redirection of a guidewire. If a guidewire is unintentionally advanced to the peripheral side, push the guidewire with rotation. When the tip of the guidewire is caught on a lateral branch (A), the guidewire will bend and form a loop by pushing force (B). If the loop is facing the hilar region, the guidewire can be advanced to the hilum by pushing further (C,D).
Figure 107. Loop technique for redirection of a guidewire. If a guidewire is unintentionally advanced to the peripheral side, push the guidewire with rotation. When the tip of the guidewire is caught on a lateral branch (A), the guidewire will bend and form a loop by pushing force (B). If the loop is facing the hilar region, the guidewire can be advanced to the hilum by pushing further (C,D).
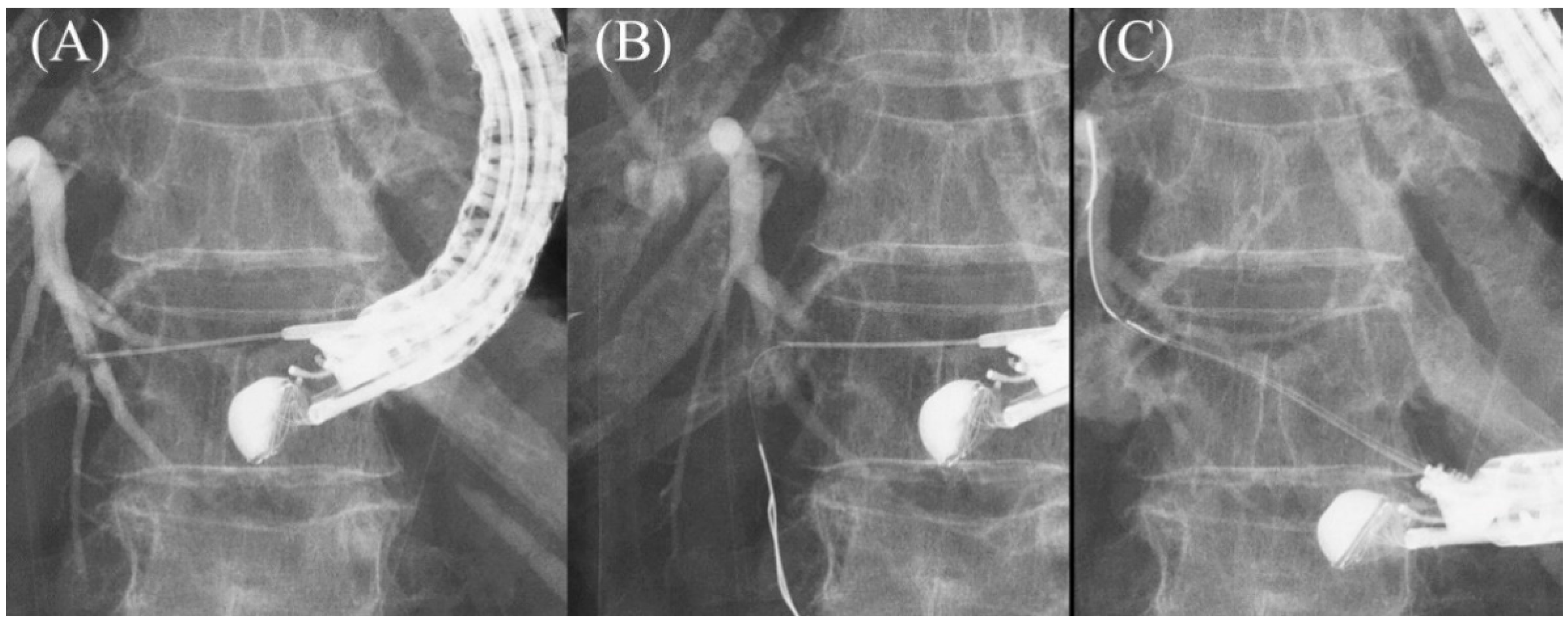 Figure 118. Moving scope technique for redirection of a guidewire. If a guidewire is unintentionally advanced to the peripheral side (A,B), push the scope while turning the large wheel upward to change the needle direction to the cranial side, allowing the guidewire to proceed toward the hilum (C).
Figure 118. Moving scope technique for redirection of a guidewire. If a guidewire is unintentionally advanced to the peripheral side (A,B), push the scope while turning the large wheel upward to change the needle direction to the cranial side, allowing the guidewire to proceed toward the hilum (C).
2.5. Tract Dilation
After a sufficient length of guidewire is placed, the needle is replaced with a dilatation device. In ERCP, the elevator is usually raised completely after device removal to prevent guidewire dislodgement. However, in EUS-HGS, the elevator should not be raised further after the needle is removed, because it is most critical to maintain ultrasound visualization of the puncture line to ensure subsequent device insertion. The more skilled the physician is in ERCP, the more likely it is that he or she will do this unconsciously, so care must be taken. The dilatation of the tract is carried out using a mechanical dilator such as a bougie dilator or balloon dilator, or a diathermic dilator. The bougie dilator is the safest, but insertion of an introducer of covered SEMS is often difficult because the size of the hole opened on the bile duct is the smallest, usually only 7 Fr. The balloon dilator can make the largest hole, but it is associated with the risk of bile leak. The diathermic dilator is the most reliable in penetrating the bile duct wall, but the burning effect can cause bleeding from the surrounding liver parenchyma and hepatic artery. Therefore, the bougie dilator is appropriate for stents with small caliber introducers (7 Fr or less), such as plastic stents and some kinds of covered SEMS, while the balloon dilator is suitable for conventional covered SEMS where the introducer is usually 8 Fr or more. The diathermic dilator had better be used as a rescue when the bile duct wall is too hard to be breached by other dilators [8]. In the initial era of EUS-HGS, mechanical dilation was accomplished gradually: the ERCP catheter was inserted first after the needle removal, followed by sequential dilatation with a bougie dilator or balloon dilator [45,46,47][30][31][32]. Recently, however, the properties of mechanical dilators have been improved so that they can be inserted directly without dilation by the ERCP catheter. Balloon dilators include Hurricane RX (Boston Scientific Corp), which has a rigid shaft with a stylet (Figure 149A) [46][31], and REN (Kaneka Medics), which has an ultra-thin tip of 3 Fr (Figure 149B) [48][33]. ES dilator (Zeon Medical, Tokyo, Japan) is a 7 Fr bougie dilator which has an ultra-thin tip of 2.5 Fr (Figure 149C) [49,50,51][34][35][36]. REN and ES dilator are dedicated dilation devices for EUS-HGS that are adapted to 0.025 inch guidewires, and the gap between the tip of these devices and the 0.025 inch guidewire is extremely small.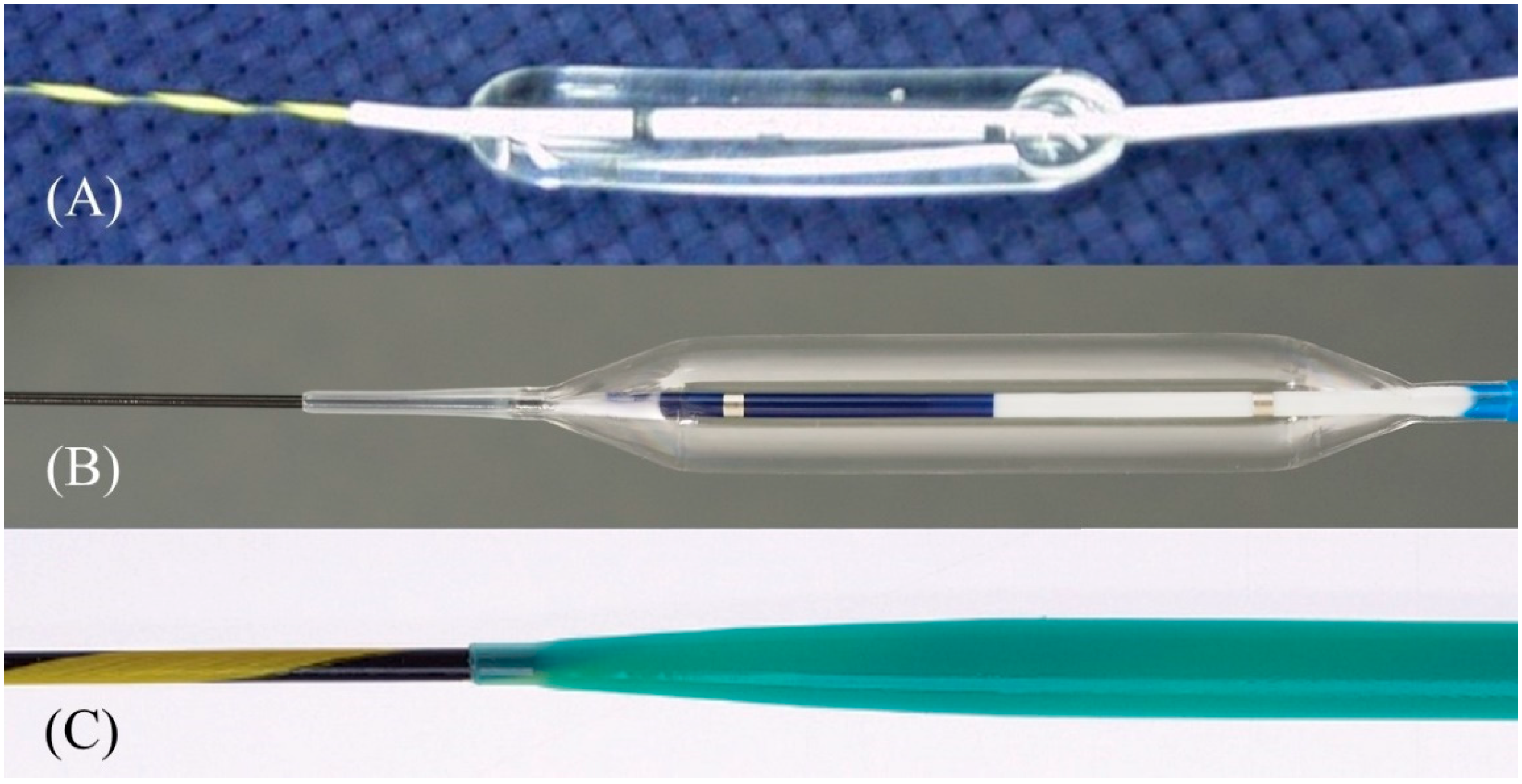 Figure 149. One-step mechanical dilation devices. Hurricane (Boston Scientific) is a balloon dilator with a rigid shaft and stylet (Courtesy of Boston Scientific) (A). REN (Kaneka Medics) is a balloon dilator with an ultra-tapered tip adapted to a 0.025 inch guidewire (Courtesy of Kaneka Medics) (B). ES dilator (Zeon Medical) is a bougie dilator with an ultra-tapered tip adapted to a 0.025 inch guidewire. (Courtesy of Zeon Medical) (C).
Figure 149. One-step mechanical dilation devices. Hurricane (Boston Scientific) is a balloon dilator with a rigid shaft and stylet (Courtesy of Boston Scientific) (A). REN (Kaneka Medics) is a balloon dilator with an ultra-tapered tip adapted to a 0.025 inch guidewire (Courtesy of Kaneka Medics) (B). ES dilator (Zeon Medical) is a bougie dilator with an ultra-tapered tip adapted to a 0.025 inch guidewire. (Courtesy of Zeon Medical) (C).
2.6. Stent Deployment
In the early days of EUS-HGS, plastic stents were predominantly used [55,56,57,58][37][38][39][40]. Although plastic stents are inexpensive and easy to place, they are prone to stent clogging due to their small caliber and bile leakage due to their lack of self-expandability. Therefore, conventional biliary-covered SEMS with a length of 6 cm or 8 cm have come into use in the expectation of preventing bile leaks by closing the fistula with self-expandability and prolonging the stent patency period with a large diameter [59,60][41][42]. In fact, the adverse events of EUS-HGS with a covered SEMS have been reported to be lower than with a plastic stent [53][43]. However, the migration of the gastric end of the stent into the abdominal cavity leading to fatal biliary peritonitis has been recognized as a major problem with a covered SEMS. For this reason, some experts initially recommended the use of a plastic stent for EUS-HGS and its replacement with a covered SEMS after fistula maturation [61,62][44][45]. However, recent advances in methodology and instrumentation have made it possible to prevent migration. Currently, various types of SEMS are available for EUS-HGS with respect to stent design (braided or laser-cut type), coverage (partial or full), presence or absence of anti-migration properties at the gastric end, and size of the introducer. As a dedicated device for EUS-HGS, several partially covered braided SEMSs with anti-migration properties have been released by Korean companies (Figure 160A–D) [17,73,74,75,76,77,78,79][23][46][47][48][49][50][51][52]. In Japan, the most common SEMS for EUS-HGS is Niti-S S-type stent (modified Giobor stent; Taewoong Medical, Seoul, Korea), which is a partially covered SEMS with a 1 cm uncovered portion at the hepatic end [71,80][53][54]. Since this stent is a braided SEMS with a cross-wire structure, it gradually expands in the stomach from the non-expanded part in the gastric wall to form a smooth and gently sloping stent surface. Therefore, the effect of holding down the gastric wall is weak. Furthermore, the shortening rate of the stent is large, which tends to cause delayed migration of the gastric end into the peritoneal cavity (Figure 17A–C). In order to prevent this, the stent length should be longer than 10 cm, but even a long stent cannot prevent it completely as mentioned above. For this reason, Niti-S Spring Stopper Stent (Taewoong Medical) was developed with a spring-type stopper at the gastric end to prevent migration (Figure 18). This stent can reliably prevent delayed migration of the gastric end. Meanwhile, pre-dilation of the tract is usually required for these SEMSs insertion because the diameter of the introducer is 8.5 Fr.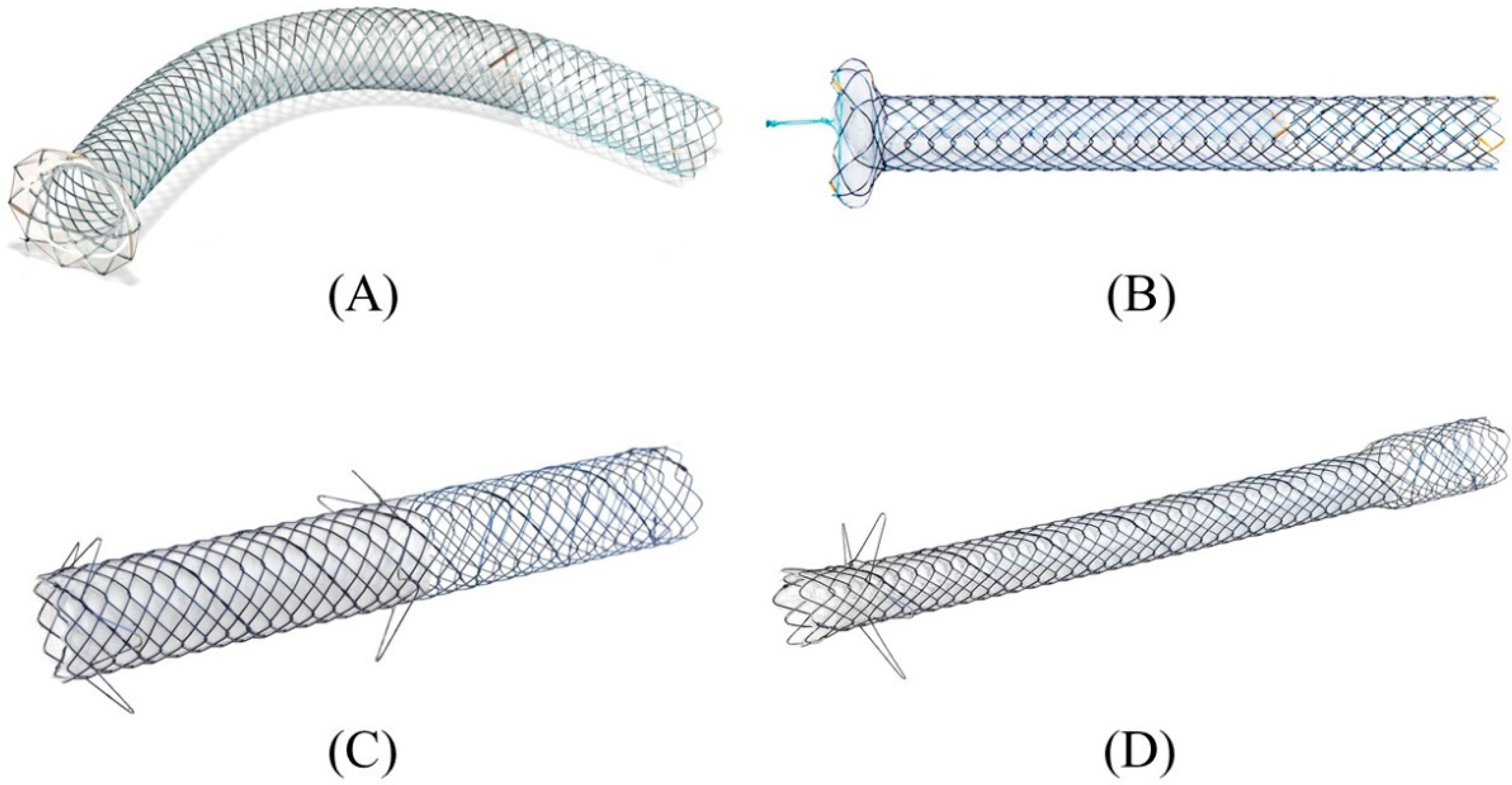 Figure 160. Partially covered SEMSs with anti-migration properties dedicated for EUS-HGS developed by Korean companies. GIOBOR stent (Taewoong medical) (A). HANARO stent BPD (M.I.Tech, Seoul, Korea) (B). Hybrid BONA stent (Standard Sci. Tech, Seoul, Korea) (C). DEUS (Standard Sci. Tech) (D). Courtesy of each company.
Figure 160. Partially covered SEMSs with anti-migration properties dedicated for EUS-HGS developed by Korean companies. GIOBOR stent (Taewoong medical) (A). HANARO stent BPD (M.I.Tech, Seoul, Korea) (B). Hybrid BONA stent (Standard Sci. Tech, Seoul, Korea) (C). DEUS (Standard Sci. Tech) (D). Courtesy of each company.
