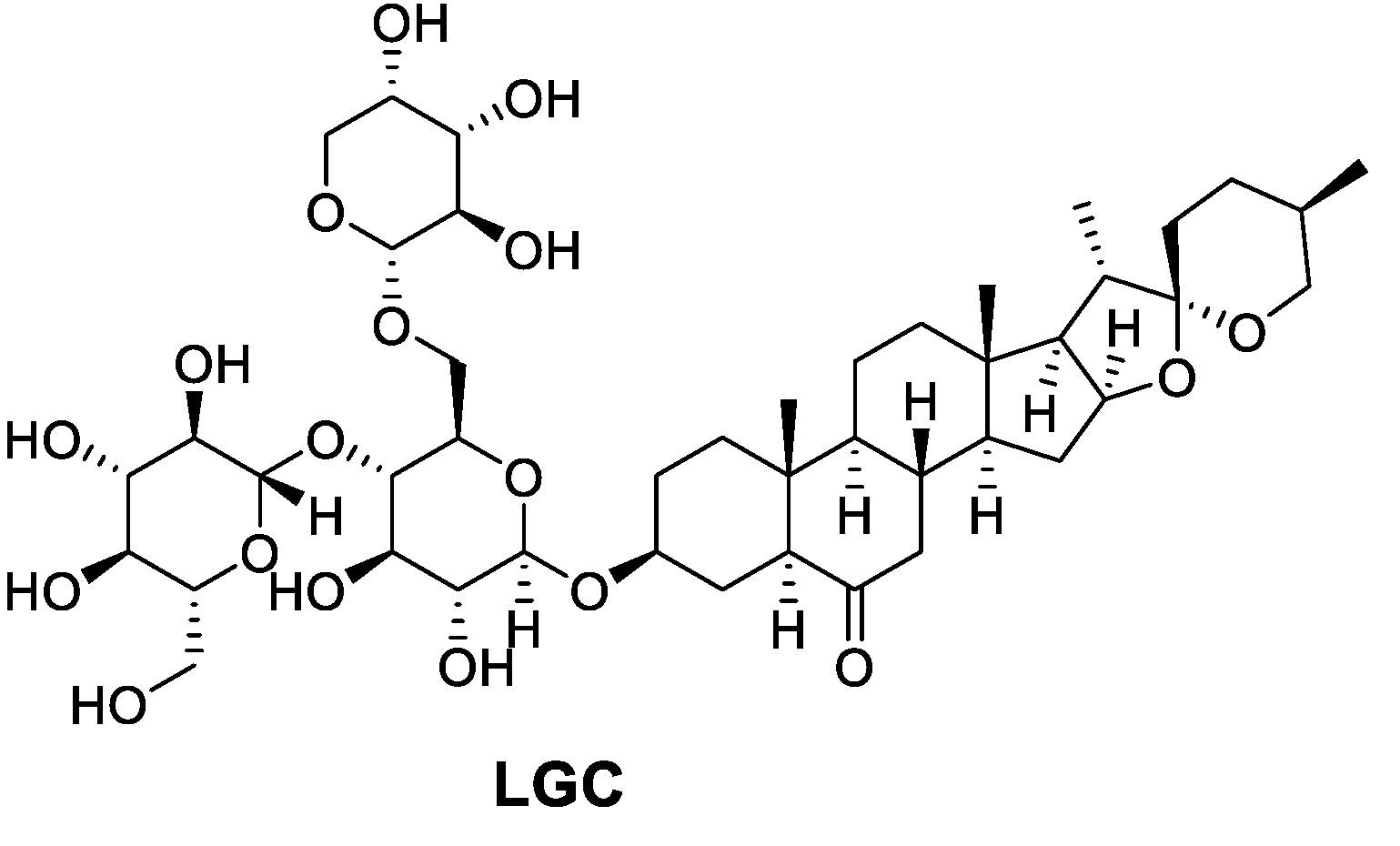
Laxogenin C (LGC) is a natural spirostanol deriving from plant hormone which has shown growanti-ing regulation similar to those of brassinosteroidflammatory, antibacterial, and growth-regulating activities.
- plant growth regulator
- LC-MS/MS
- metabolomics
- Laxogenin C
- Metabolism
1. Introduction
Laxogenin C is a class of natural product steroid glycosides, which is derived from laxogenin. It has the same active functional group as the plant hormones brassinosteroids and therefore shares similarities in their physiological activity with brassinosteroids. Its chemical name is laxogenin-3-O-β-D-glucopyranosyl- (1→4)-[α-L-arabinopyranosyl- (1→6) ]-β-D-glucopyranose. Its chemical formula is C44H70O18 and its structure is shown in Figure 1.
2. Tomato Growth Responding to LGC and SA Regulation
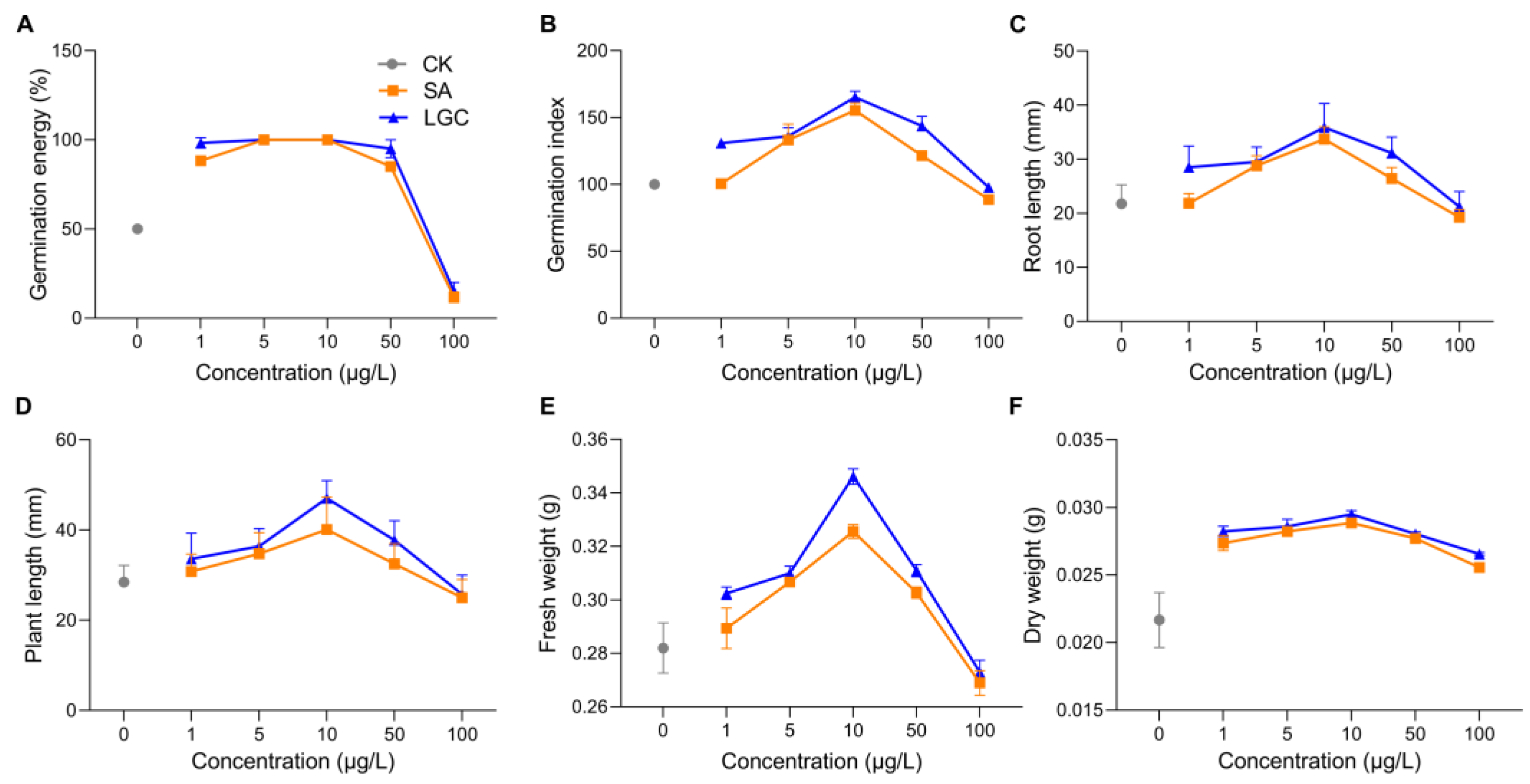
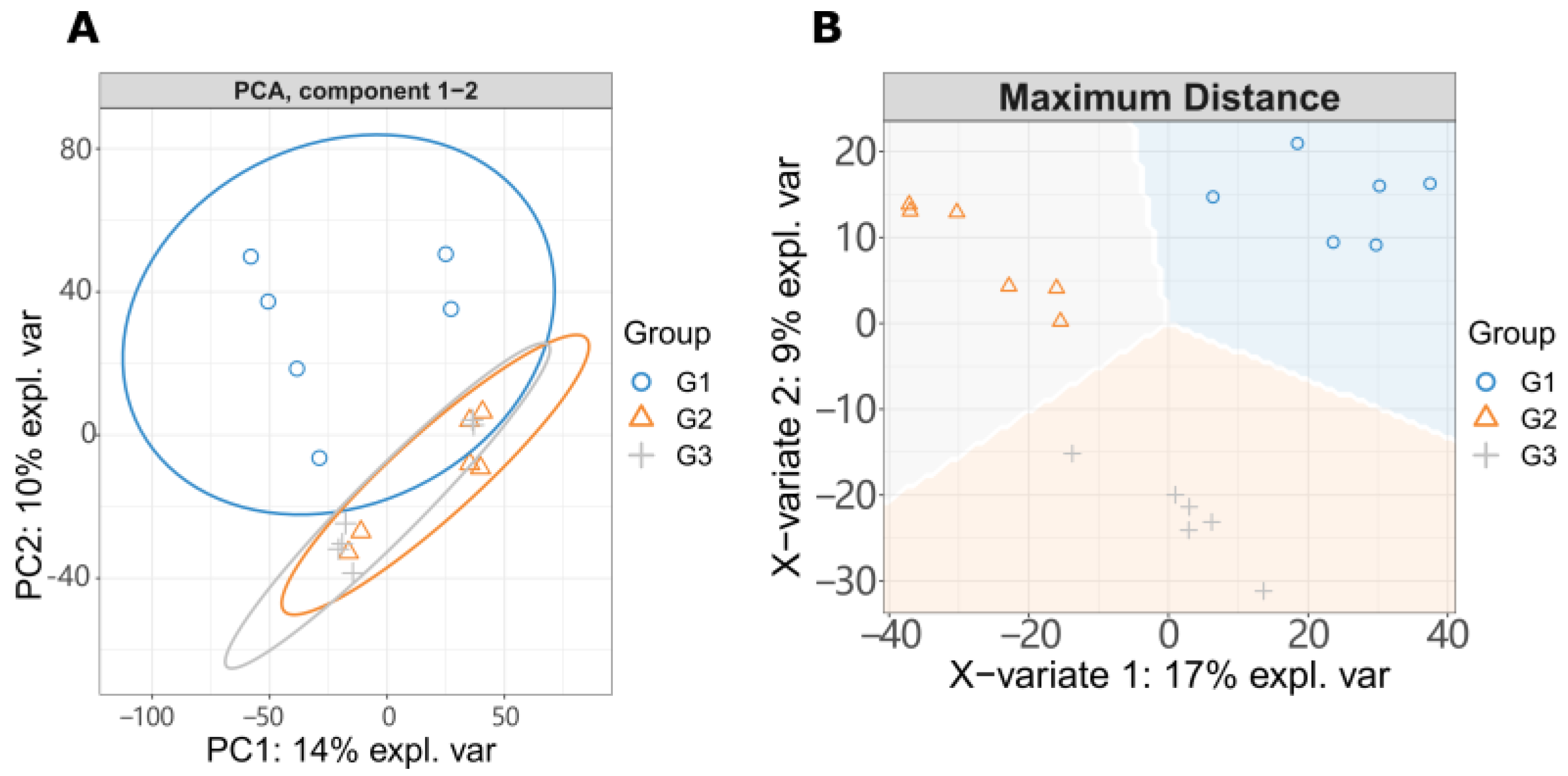
32. LC-MS/MS Metabolome AnalHistorysis
Laxogenin C was first isolated from the rhizomes of Smilax sieboldii by Kubo et al [1]. At the same time, it was also isolated from the rhizomes of Smilax lebrunii by Jia and Ju [2]. It was also isolated from the bulbs of Allium chinense by Jiang et al [3] and the stem of Smilax scobinicaulis by Zhang[4]. Studies have shown that it has some bioactivities on both animals and plants. It has anti-inflammatory activity and antinociceptive effect. It regulates plant growth in a dose-dependent manner, promoting seed germination, seedling growth and increasing crop and fruit yield at low concentrations and inhibiting them at high concentrations. Spraying laxogenin C also alleviates the harm of drought stress on seedlings.
Kim et al.[5] found that showed laxogenin C had anti-inflammatory activity to inhibit ear edema.
Suh et al.[6] found that GABAB, and non-NMDA receptors located at the supraspinal level may play important roles in the production of antinociception induced by laxogenin C administered supraspinally. Furthermore, laxogenin C administered supraspinally may produce its antinociception by activating descending noradrenergic- but not opioidergic- and serotonergic-neurons.
Guo et al. [7]found that laxogenin C significantly promoted seed germination and seedling growth of rape.
Wang et al. [8]found that laxogenin C promoted seed germination and seedling growth and increased biological yield in maize. Laxogenin C could increase plant height, leaf width, leaf length, and biological yield of maize seedlings to different degrees.
Ma et al. [9] found that laxogenin C improved seed germination and seedling growth of watermelon. The concentration of laxogenin C varies, resulting in different sites of action, the indicators of improvement are also different. For example, different concentrations of Laxogenin C improve the weight of the roots or overground parts, respectively.
Ma et al.[10] found that moderate concentrations of laxogenin C had the positive effect on seed germination and seedling growth of radish, black bean and etchling. Besides, laxogenin C also increased their overground biological yield and the content of Vitamin C effectively. It showed that the suitable concentration of laxogenin C had a certain application value in the production of sprout vegetable.
Zhou and Zhang [11] found that spraying laxogenin C at the elongation-flowering stage coordinated yield components of winter wheat and increased the photosynthetic rate, consequently improved the winter wheat grain yield and quality.
Ding et al. [12]found that spraying exogenous laxogenin C alleviated the harm of drought stress on seedling by accumulating osmotic substances, increasing activity of antioxidant enzymes, removing peroxide, and enhancing photosynthetic ability.
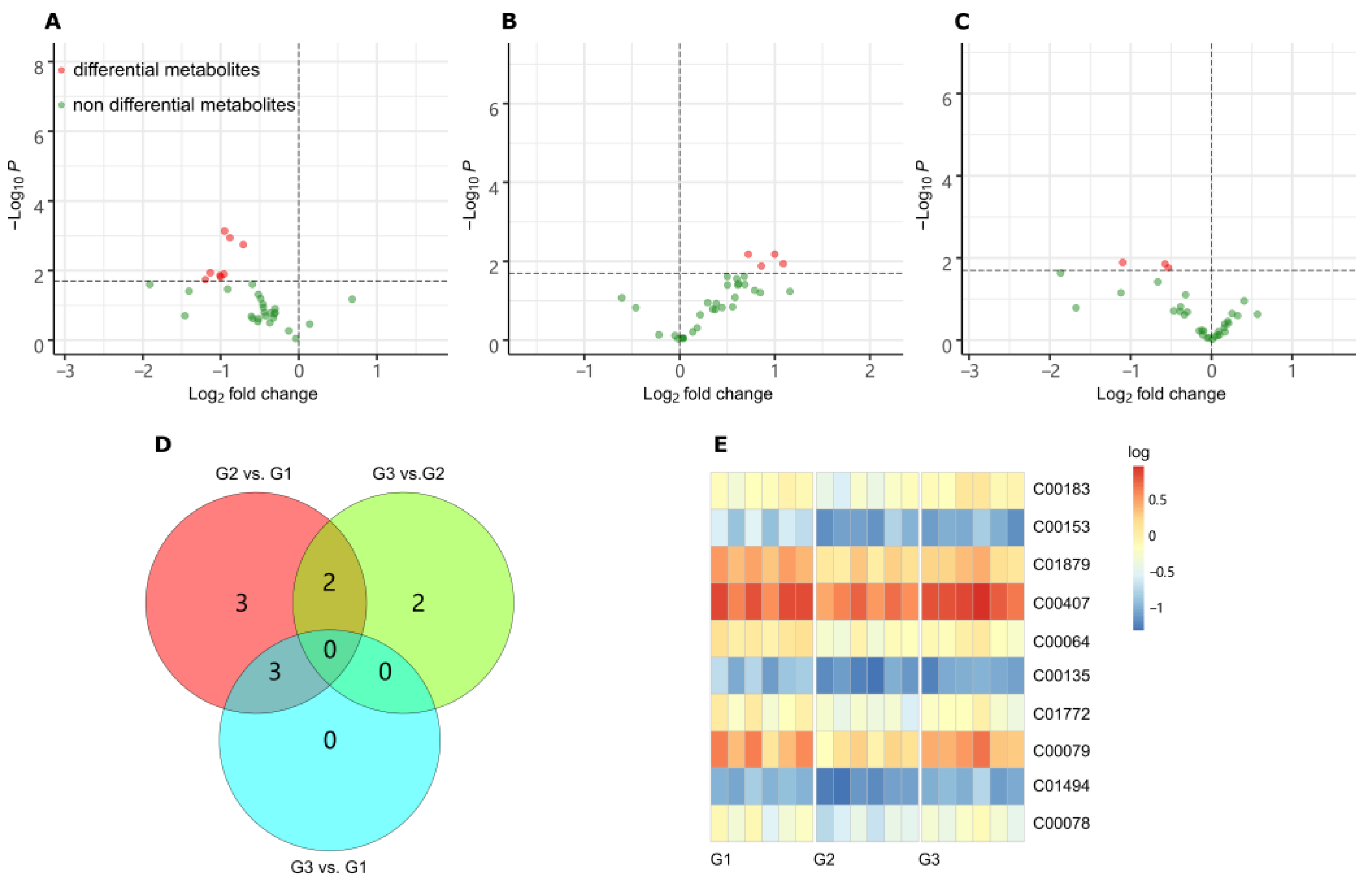
4
3. PathNeways Responding to LGC Regulation
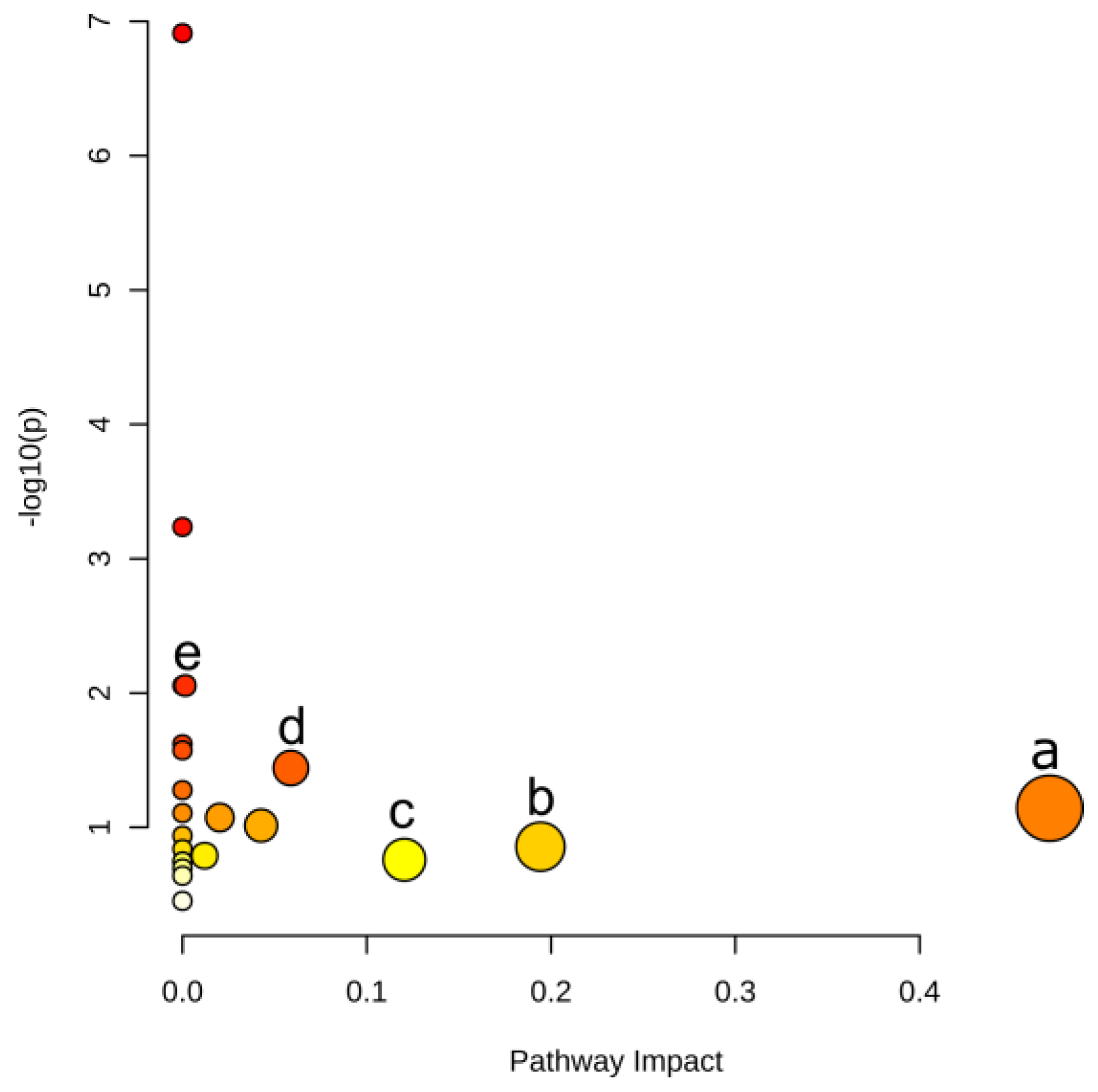
| Pathway | –logP | Impact | Matched Features |
|---|---|---|---|
| Phenylalanine metabolism | 1.14 | 0.470 | l-phenylalanine (C00079) |
| Alanine, aspartate and glutamate metabolism | 0.86 | 0.194 | l-glutamine (C00064) |
| Tryptophan metabolism | 0.76 | 0.120 | l-tryptophan (C00078) |
| Phenylpropanoid biosynthesis | 1.44 | 0.059 | l-phenylalanine (C00079); ferulate (C01494) |
| Phenylalanine, tyrosine and tryptophan biosynthesis | 2.05 | 0.002 | l-phenylalanine (C00079); l-tryptophan (C00078) |
5 discoveries
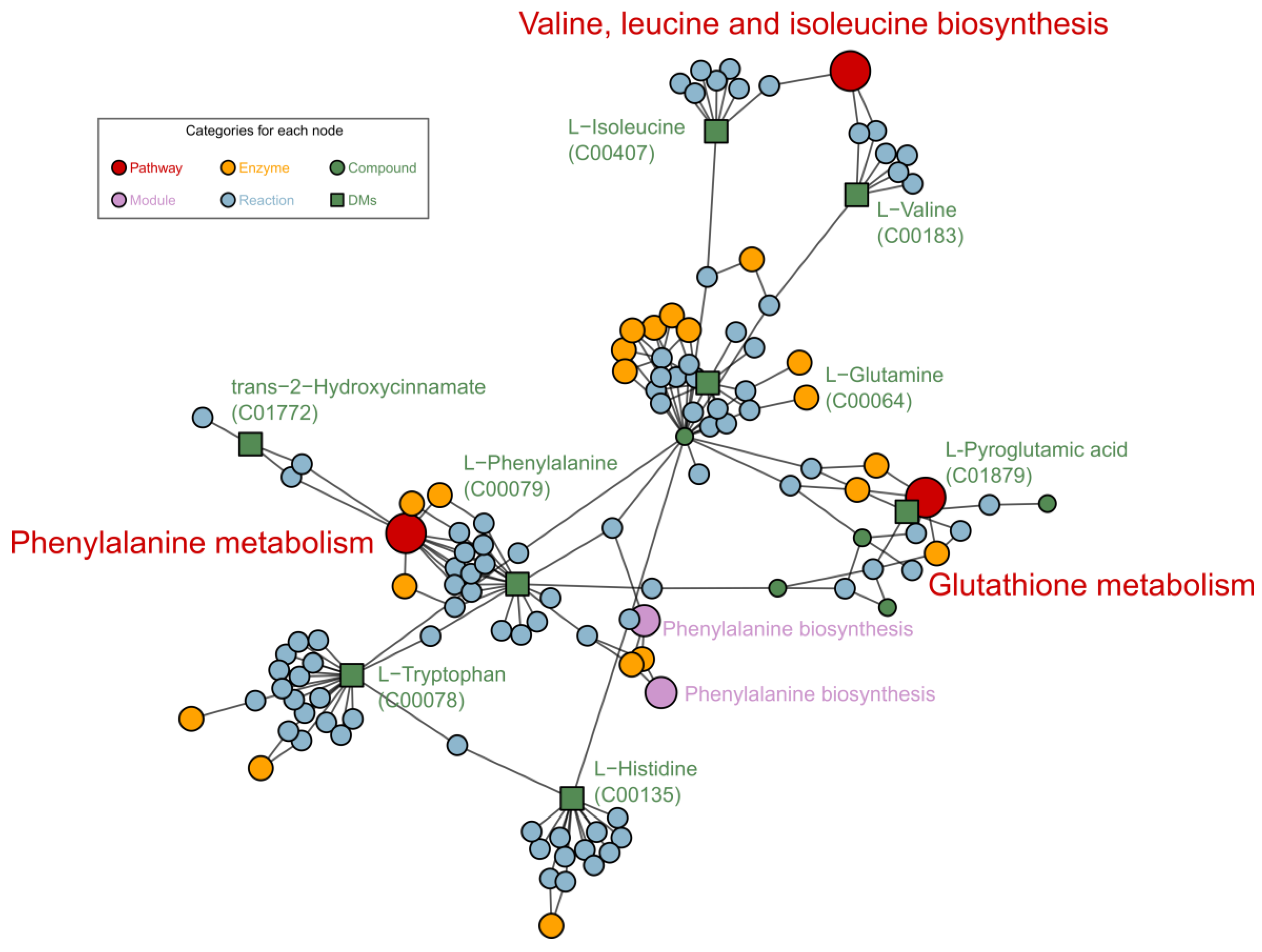
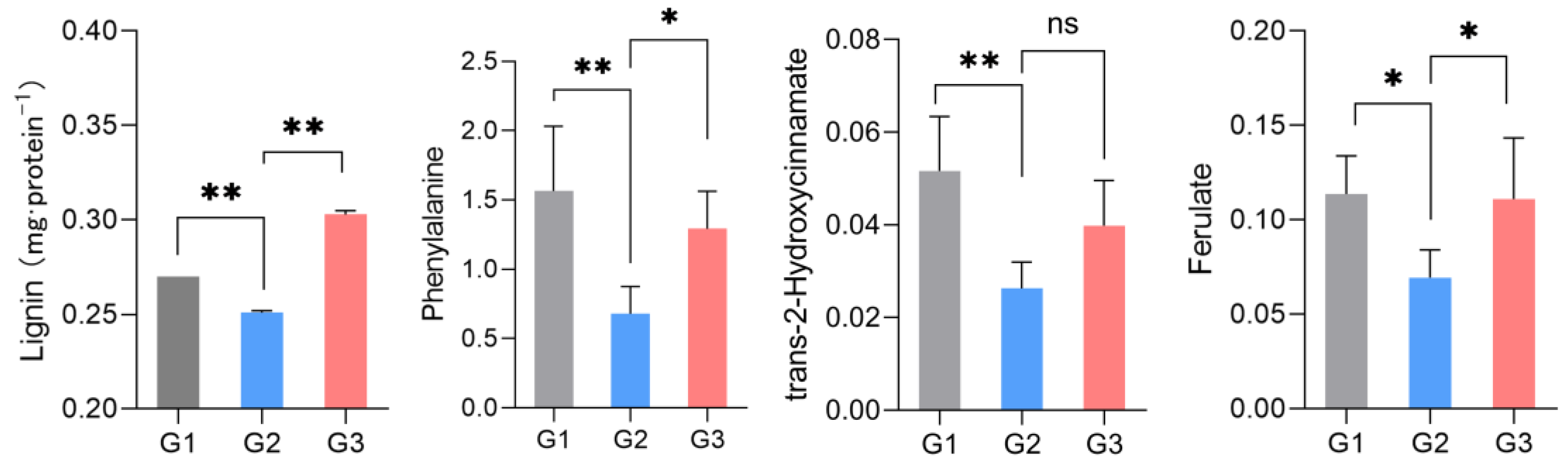
Metabolomic analysis revealed that the most significant metabolites regulated by laxogenin C in tomato were l-phenylalanine, nicotinamide and tryptophan. The most significant metabolic pathways regulated by laxogenin C were phenylalanine metabolism, alanine, aspartate and glutamate metabolism, tryptophan metabolism, phenylpropanoid biosynthesis and phenylalanine, tyrosine and tryptophan biosynthesis. Laxogenin C can affect the lignin content to a certain extent, thus regulating plant growth.
4. PhenotyAppe Veriflication
The invention of Zhang et al. discloses a patent for a plant growth regulator containing laxogenin and its derivatives that are pollution-free and environmentally friendly. Laxogenin and its derivatives come from medicinal plant resources and belong to the endogenous natural metabolites of plants, which can be absorbed and metabolized easily by plants, so it will not adversely affect the ecological environment or cause human health and safety problems, and is a non-toxic and non-polluting technology that meets the needs of green agriculture, low cost, high stability and wide range of applications.
5. Current status
The activity of brassinosteroids has been widely noted, such as promoting plant growth and enhancing plant resistance to abiotic stresses. More and more studies have proved the great potential of brassinosteroids in agricultural applications. Synthesis of brassinosteroids and their analogues has been started in the United States, Japan, China and other countries, which have been widely used in agricultural production. It was demonstrated that laxogenin C had biological activity on a variety of plants and was widely used in crops, vegetables and fruits. Studies have shown that laxogenin C promoted seed germination and seedling growth, increased biological yield of crops, enhanced photosynthesis, improved crop quality, increased nutrition and improve the taste of fruits and vegetables, and also enhanced plant resistance to abiotic stresses. The study of laxogenin and laxogenin provides a new way to extract analogues with brassinosteroids activity from plants.
References
- Rademacher, W. Plant Growth Regulators: Backgrounds and Uses in Plant Production. J. Plant Growth Regul. 2015, 34, 845–872. Tamotsu Nikaido; Taichi Ohmoto; Satoshi Kubo; Yoshihiro Mimaki; Yutaka Sashida; Steroidal saponins from the rhizomes of Smilax sieboldii. Phytochemistry 1992, 31, 2445-2450, 10.1016/0031-9422(92)83296-b.
- Steber, C.M.; McCourt, P. A Role for Brassinosteroids in Germination in Arabidopsis. Plant Physiol. 2001, 125, 763–769. Jia Zhong-Jian; Ju Yong; Steroidal saponins from Smilax lebrunii. Phytochemistry 1992, 31, 3173-3175, 10.1016/0031-9422(92)83469-f.
- Li, T.; Lei, W.; He, R.; Tang, X.; Han, J.; Zou, L.; Yin, Y.; Lin, H.; Zhang, D. Brassinosteroids Regulate Root Meristem Development by Mediating BIN2-UPB1 Module in Arabidopsis. PLoS Genet. 2020, 16, e1008883. Yong Jiang; Nai-Li Wang; Xin-Sheng Yao; Susumu Kitanaka; Steroidal saponins from the bulbs of Allium chinense. Advances in Plant Glycosides, Chemistry and Biology, Proceedings of the International Symposium in Plant Glycosides 1999, 6, 212-219, 10.1016/s0928-3420(99)80029-9.
- Huang, G.; Han, M.; Yao, W.; Wang, Y. Transcriptome Analysis Reveals the Regulation of Brassinosteroids on Petal Growth Gerbera hybrida. PeerJ 2017, 5, e3382. Zhang Cunli; Wang Dongmei; Zhu wei; Zhang Hongli; Study on chemical constituents from stem of Smilax scobinicaulis. Chinese Pharmaceutical Journal 2006, 41, 1540-1542.
- Ye, H.; Liu, S.; Tang, B.; Chen, J.; Xie, Z.; Nolan, T.M.; Jiang, H.; Guo, H.; Lin, H.-Y.; Li, L.; et al. RD26 Mediates Crosstalk between Drought and Brassinosteroid Signalling Pathways. Nat. Commun. 2017, 8, 14573. Sung Yong Kim; Kun Ho Son; Hyeun Wook Chang; Sam Sik Kang; Hyun Pyo Kim; Inhibition of mouse ear edema by steroidal and triterpenoid saponins. Archives of Pharmacal Research 1999, 22, 313-316, 10.1007/bf02976370.
- Hou, J.; Zhang, Q.; Zhou, Y.; Ahammed, G.J.; Zhou, Y.; Yu, J.; Fang, H.; Xia, X. Glutaredoxin GRXS16 Mediates Brassinosteroid-Induced Apoplastic H2O2 Production to Promote Pesticide Metabolism in Tomato. Environ. Pollut. 2018, 240, 227–234. Hong Suh; Dong Song; Keun Son; Mi Woo; Jae Do; Young Choi; Keun Lee; Yung Kim; Antinociceptive Effect of Smilaxin B Administered Intracerebroventricularly in the Mouse. Planta Medica 1996, 62, 141-145, 10.1055/s-2006-957837.
- De Bock, P.; Van Bockstaele, F.; Muylle, H.; Quataert, P.; Vermeir, P.; Eeckhout, M.; Cnops, G. Yield and Nutritional Characterization of Thirteen Quinoa (Chenopodium Quinoa Willd.) Varieties Grown in North-West Europe—Part I. Plants 2021, 10, 2689. Guo Jie; Guan Hua; Zhang Cunli; Gong Yuehua; Effects of Laxogenin and Smilscobinoside C on Seed Germination and Seedlings Growth of Rape. Northern Horticulture 2010, 24, 12~14, 10.11937/bfyy.201024004.
- Baik, I.-H.; Kim, K.-H.; Lee, K.-A. Antioxidant, Anti-Inflammatory and Antithrombotic Effects of Ginsenoside Compound K Enriched Extract Derived from Ginseng Sprouts. Molecules 2021, 26, 4102. Wang weiyan; Zhang cunli; Effects of Laxogenin and Smilscobinoside C on Seed Germination and Seedlings Growth of Maize. Seed 2011, 30, 15-17, :10.16590/j.cnki.1001-4705.2011.12.059.
- Le, N.T.M.; Cuong, D.X.; Thinh, P.V.; Minh, T.N.; Manh, T.D.; Duong, T.-H.; Minh, T.T.L.; Oanh, V.T.T. Phytochemical Screening and Evaluation of Antioxidant Properties and Antimicrobial Activity against Xanthomonas Axonopodis of Euphorbia Tirucalli Extracts in Binh Thuan Province, Vietnam. Molecules 2021, 26, 941. Ma li; Luo Yongjun; Wang weiyan; Zhang cunli; Effects of Laxogenin C on Seed Germination and Seedlings Growth of Watermelon. Northern Horticulture 2011, 35, 22-24, 10.11937/bfyy.2011024006.
- Shvets, S.A.; Gutsu, O.N.; Kintia, P.K. Steroidal Glycosides from Nicotiana Tabacum L. Seeds and Their Biological Activity. In Saponins Used in Food and Agriculture; Advances in Experimental Medicine and Biology; Waller, G.R., Yamasaki, K., Eds.; Springer: Boston, MA, USA, 1996; pp. 247–257. ISBN 978-1-4613-0413-5. Ma Huiling; Ye Zhanyang; Shang Xiaojiao; Shen Wei; Li Weini; Effect of Laxogenin C on Seed Germination and Seedlings Growth of Radish,Black Bean and Etchling. Northern Horticulture 2014, 05, 15~20.
- Santos Wagner, A.L.; Araniti, F.; Bruno, L.; Ishii-Iwamoto, E.L.; Abenavoli, M.R. The Steroid Saponin Protodioscin Modulates Arabidopsis Thaliana Root Morphology Altering Auxin Homeostasis, Transport and Distribution. Plants 2021, 10, 1600. Zhou Meng; Zhang Cunli; The effects of Laxogenin-C on photosynthetic characteristics of the flag leaf and yield and quality of winter wheat grain. Agricultural Science Journal of Yanbian University 2017, 39, 53-59, 10.13478/j.cnki.jasyu.2017.02.009.
- Nikaido, T.; Ohmoto, T.; Kubo, S.; Mimaki, Y.; Sashida, Y. Steroidal Saponins from the Rhizomes of Smilax sieboldii. Phytochemistry 1992, 31, 2445–2450. Ding Danyang; Zhang Yijie; Wang Kaiyue; Liu Zhihong; Lu Yutong; Shao Huifang; Physiological Achine of Laxogenin C on Increasing the Ressistance to Drought Stress of Tobacco Seedlings. Journal of Agricultural Science and Technology 2018, 20, 36-44, 10.13304/j.nykjdb.2017.0843.
- Iglesias-Arteaga, M.A.; Gil, R.P.; Martínez, C.S.P.; Manchado, F.C. Spirostanic Analogues of Teasterone. Synthesis, Characterisation and Biological Activity of Laxogenin, (23S)-Hydroxylaxogenin and 23-Ketolaxogenin (23-Oxolaxogenin). J. Chem. Soc. Perkin Trans. 1 2001, 261–266.
- Wang, Q.; Xu, J.; Liu, X.; Gong, W.; Zhang, C. Synthesis of Brassinosteroids Analogues from Laxogenin and Their Plant Growth Promotion. Nat. Prod. Res. 2015, 29, 149–157.
- Zhang, C.L.; Wang, D.M.; Zhu, W.; Zhang, H.L. Study on Chemical Constituents from Stem of Smilax scobinicaulis. Chin. Pharm. J. 2006, 41, 1540–1542.
- Joshi, V.; Joung, J.-G.; Fei, Z.; Jander, G. Interdependence of Threonine, Methionine and Isoleucine Metabolism in Plants: Accumulation and Transcriptional Regulation under Abiotic Stress. Amino Acids 2010, 39, 933–947.
- Thangamani, S.; Eldesouky, H.E.; Mohammad, H.; Pascuzzi, P.E.; Avramova, L.; Hazbun, T.R.; Seleem, M.N. Ebselen Exerts Antifungal Activity by Regulating Glutathione (GSH) and Reactive Oxygen Species (ROS) Production in Fungal Cells. Biochimica et Biophysica Acta (BBA)—General Subjects 2017, 1861, 3002–3010.
- Zhang, Z.; Lv, Z.; Wei, Z.; Li, C.; Shao, Y.; Zhang, W.; Zhao, X.; Xiong, J. Microsomal Glutathione Transferase 2 Modulates LTC4 Synthesis and ROS Production in Apostichopus Japonicus. Mol Immunol 2017, 91, 114–122.
