Dactylospongia elegans Thiele (Thorectidae) is a wealth pool of various classes of sesquiterpenes, including hydroquinones, quinones, and tetronic acid derivatives. These metabolites possessed a wide array of potent bioactivities such as antitumor, cytotoxicity, antibacterial, and anti-inflammatory.
1. Introduction
The marine environment is extraordinarily rich in diverse species of organisms that represent an enormous source of biometabolites, many of which have unique chemical entities not present in terrestrial sources
[1,2,3][1][2][3]. The molecular diversity and exceptional complexity of marine metabolites have been highlighted in many studies
[4,5][4][5]. The marine environment investigations have greatly been limited to subtropical and tropical regions, in addition, the exploration has been recently extended to colder regions. However, it is fact that many of these unique marine resources have barely been investigated.
Sponges (filter feeders, phylum Porifera) are evolutionarily ancient metazoans that occur in marine benthic, quasi-terrestrial, deep-sea, and fresh-water ecosystems
[6,7][6][7]. They represent one of the important members of marine communities that have potential biotechnological and ecological roles
[8,9][8][9]. They are sessile multicellular invertebrates, having an enormous amount of tiny pores on their surfaces that allow entrance and circulation of water through canals where organic particles and microorganisms are filtered out and eaten
[10]. Calcarea, Demospongiae, Homoscleromorpha, and Hexactinellida are the four main classes of sponges
[11]. It is noteworthy that the secondary metabolites distribution among these four different classes of sponges is greatly varied as shown in
Table 1.
Table 1.
Reported secondary metabolites from the four main class of sponges [12].
|
Sponge Class
|
Compounds Classes
|
|
Calcarea
|
C27 to C29 ∆5,7,9(11),22 and C27 to C29 ∆5,7,22 sterols
Amino alcohols
|
|
Hexactinellida
|
5α(H)-Cholestan-3β-ol/cholest-5-en-3β-ol
Ceramide glycosides
|
|
Homoscleromorpha
|
Steroidal alkaloids
Peroxy-polyketides
|
|
Demospongiae
|
Pyrroloquinoline, azetidine, pyrrole-2-aminoimidazole, and pentacyclic guanidine alkaloids
Norditerpene and norsesterterpene peroxides
Tetramic acids
Steroidal saponins and glycosides
Isomalabaricane triterpenoids
Bengamide and bengazoles
Hydroxyimino- and 3β-hydroxymethyl-A-nor-sterols
3-Alkylpyridines/3-alkylpiperidines
Renieramycins and polyacetylenes
Pentacyclic hydroquinones/polyprenylated benzoquinones
Adenine- and cyanthiwigin diterpenes
Hypotaurocyamine (Sesquiterpene derivatives)
Diterpene thio/iso/cyanides and formamides
Sesquiterpene thio/iso/cyanides and formamides
Aaptamines and bromotyrosines
Suberitane-derived sesterterpenes
Diterpene, sesquiterpene, and sesterterpenefurans/lactones
Scalarane sesterterpenes/sesterterpene hydroquinones
Thiazole polyketides
Polybrominated diphenyl ethers
|
∆: Double bond.
Sponges are devoid of any physical capacity for defense; therefore, they need to develop specific means and adaptive responses for self-protection
[13]. They produce diverse secondary metabolites as defense ways against pathogenic fungi, algae, bacteria, and other predators, also to modulate and/or enable cellular communication
[14]. Sponges have been known as a fertile field for the discovery of bioactive metabolites with diverse structural features that have been proven to have a beneficial potential for humans as agricultural medicines, drugs, health foods, and cosmetics
[14,15][14][15].
2. Secondary Metabolites of D. elegans
Phytochemical studies of
D. elegans from 1992 until 2022, revealed that 101 metabolites have been separated and characterized from
D. elegans. These metabolites are grouped according to their chemical classes into sesquiterpenes, sesterterpenes, diterpenes, sterols, pregnanes, and other metabolites which are consequently discussed
(Table 2).
Table 2. List of reported metabolites from Dactylospongia elegans.
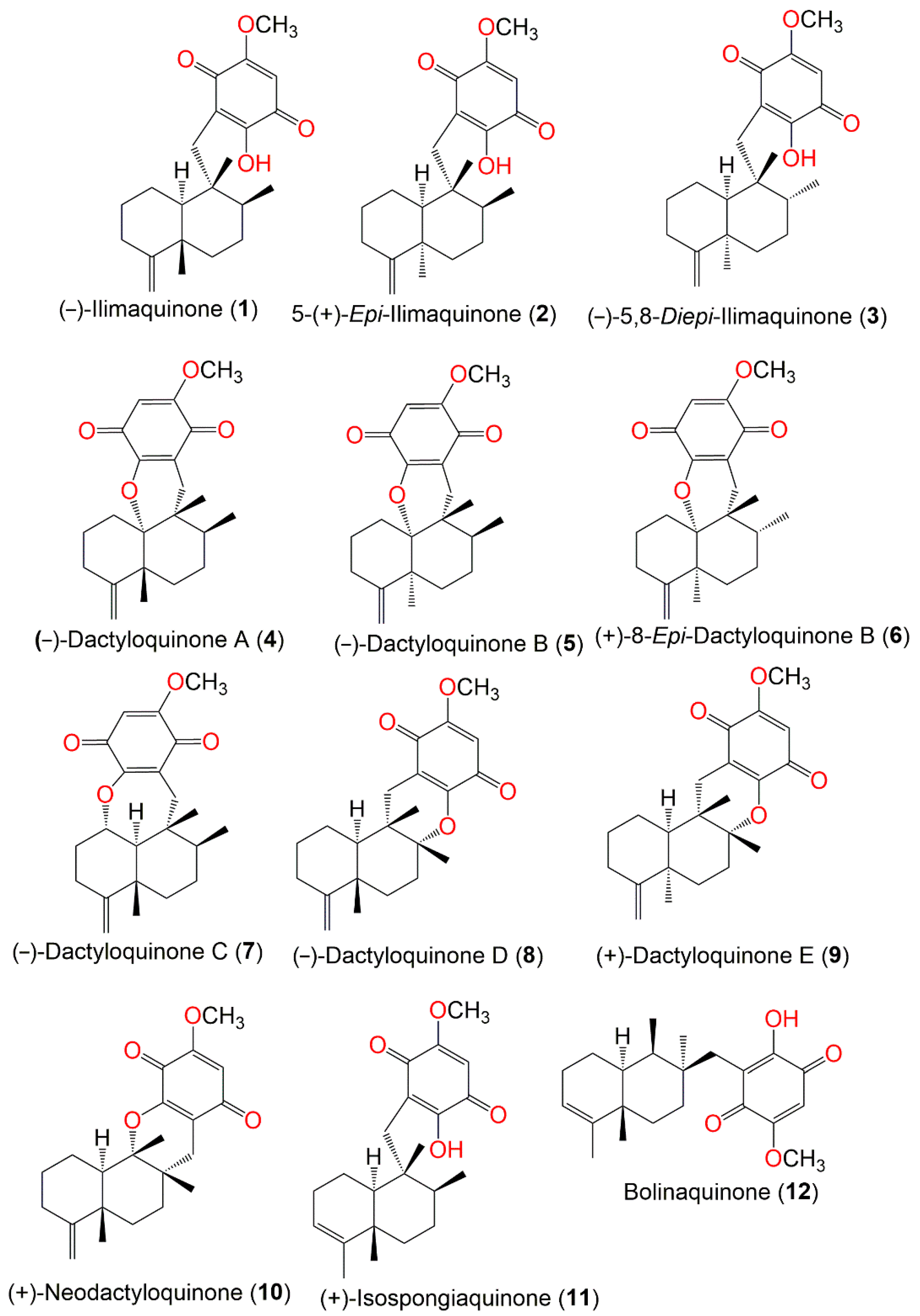
Figure 1.
Structures of compounds
1
–
12
.
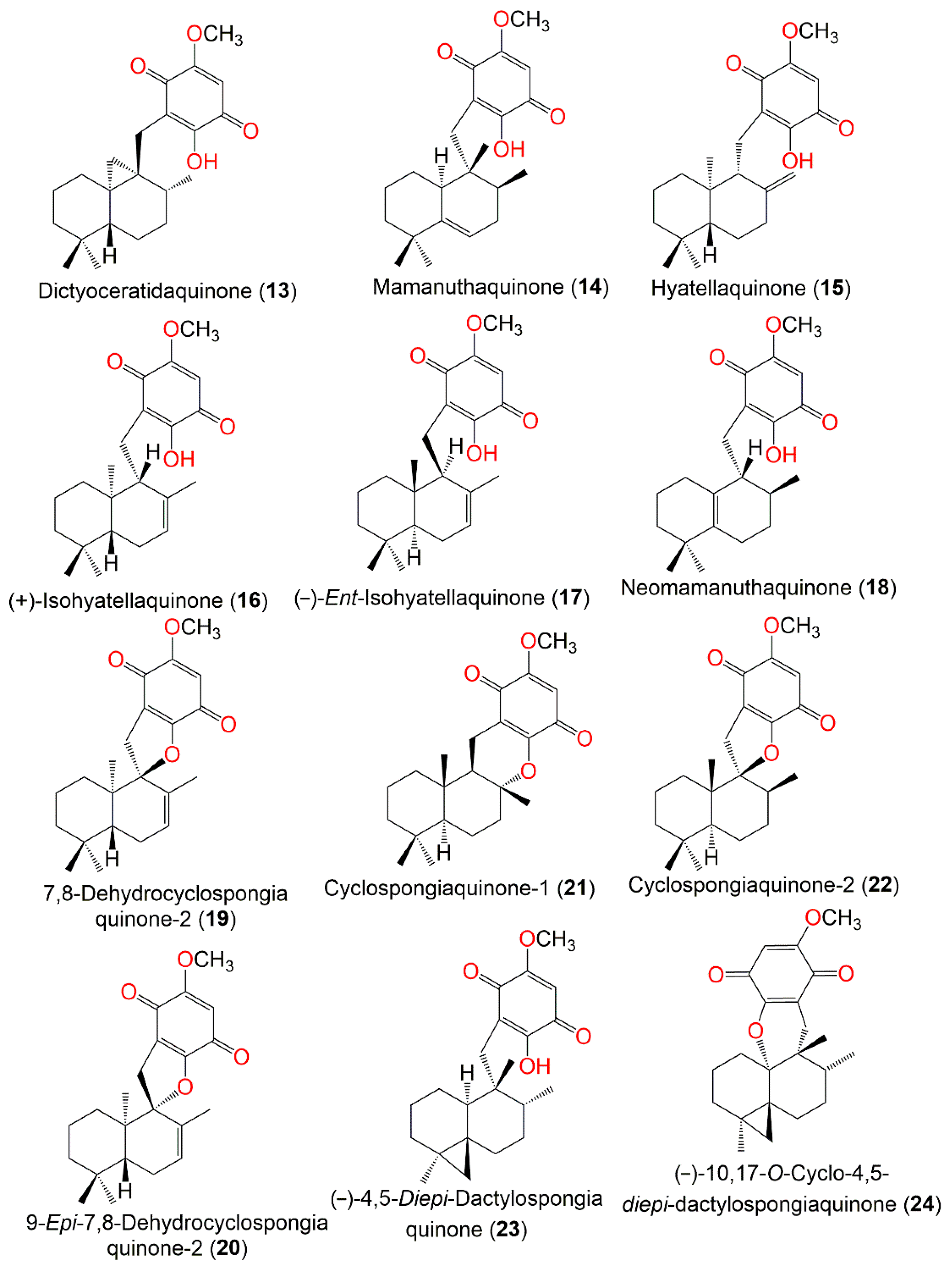
Figure 2.
Structures of compounds
13
–
24
.
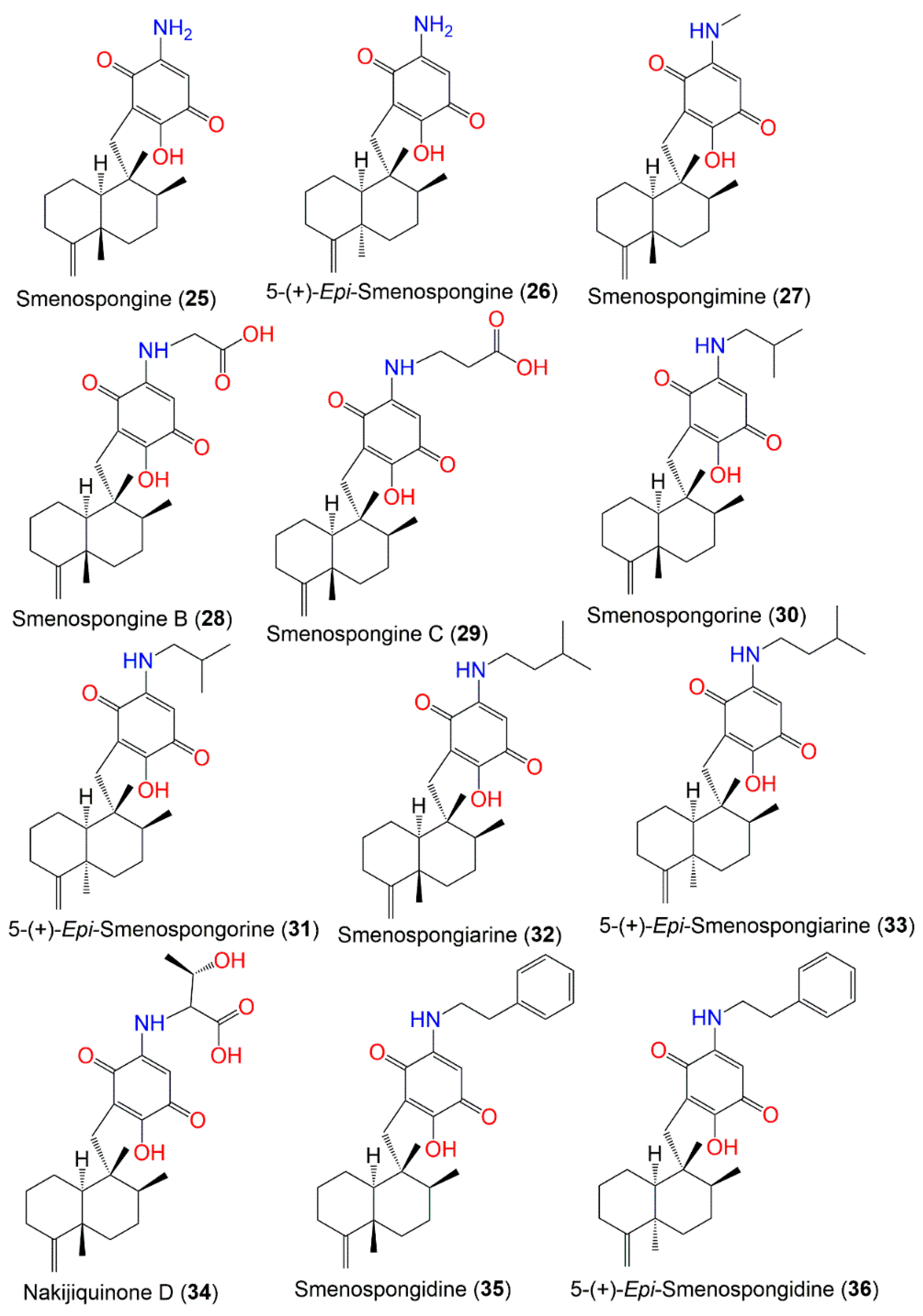
Figure 3.
Structures of compounds
25
–
36
.
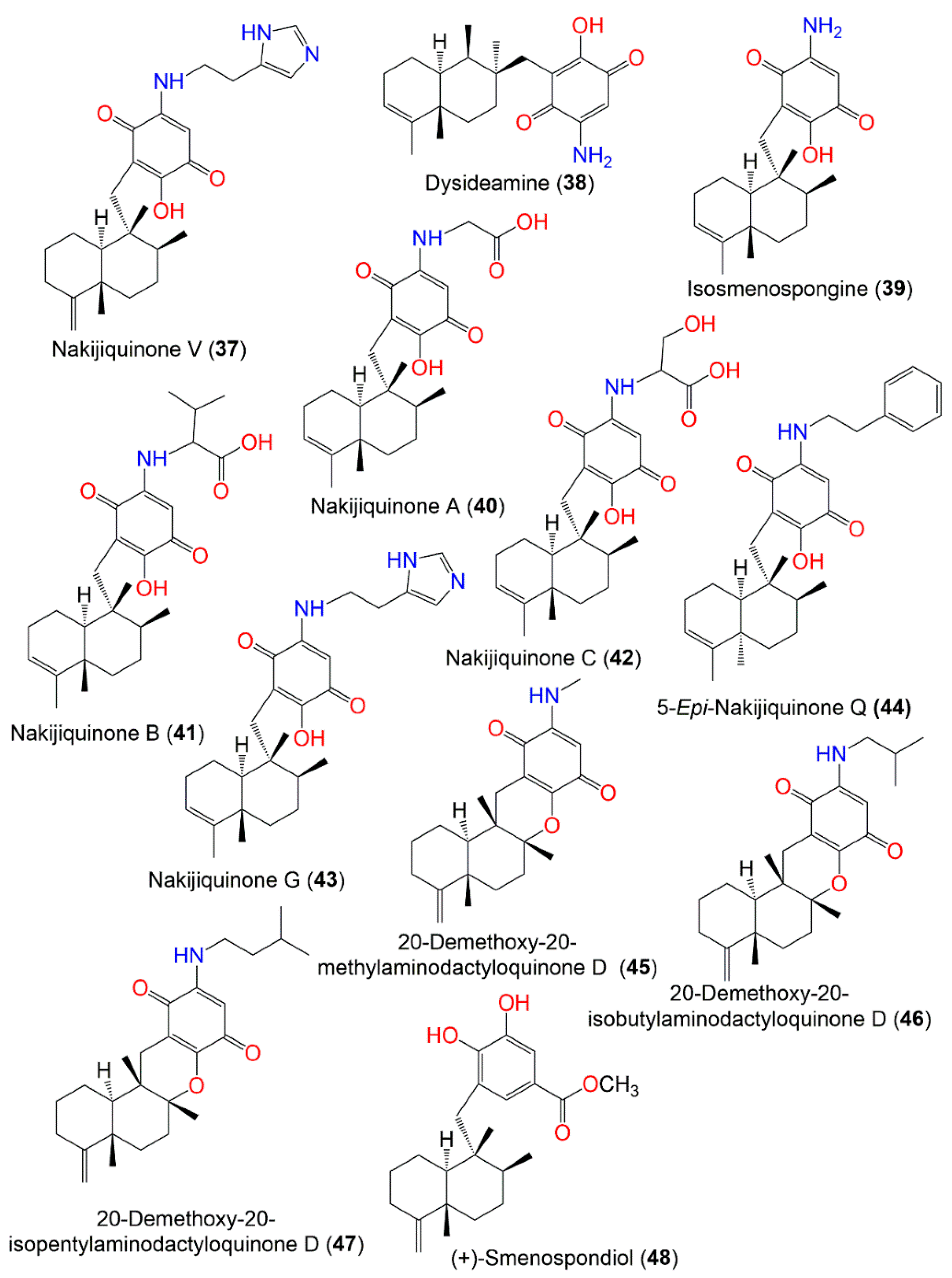
Figure 4.
Structures of compounds
37
–
48
.
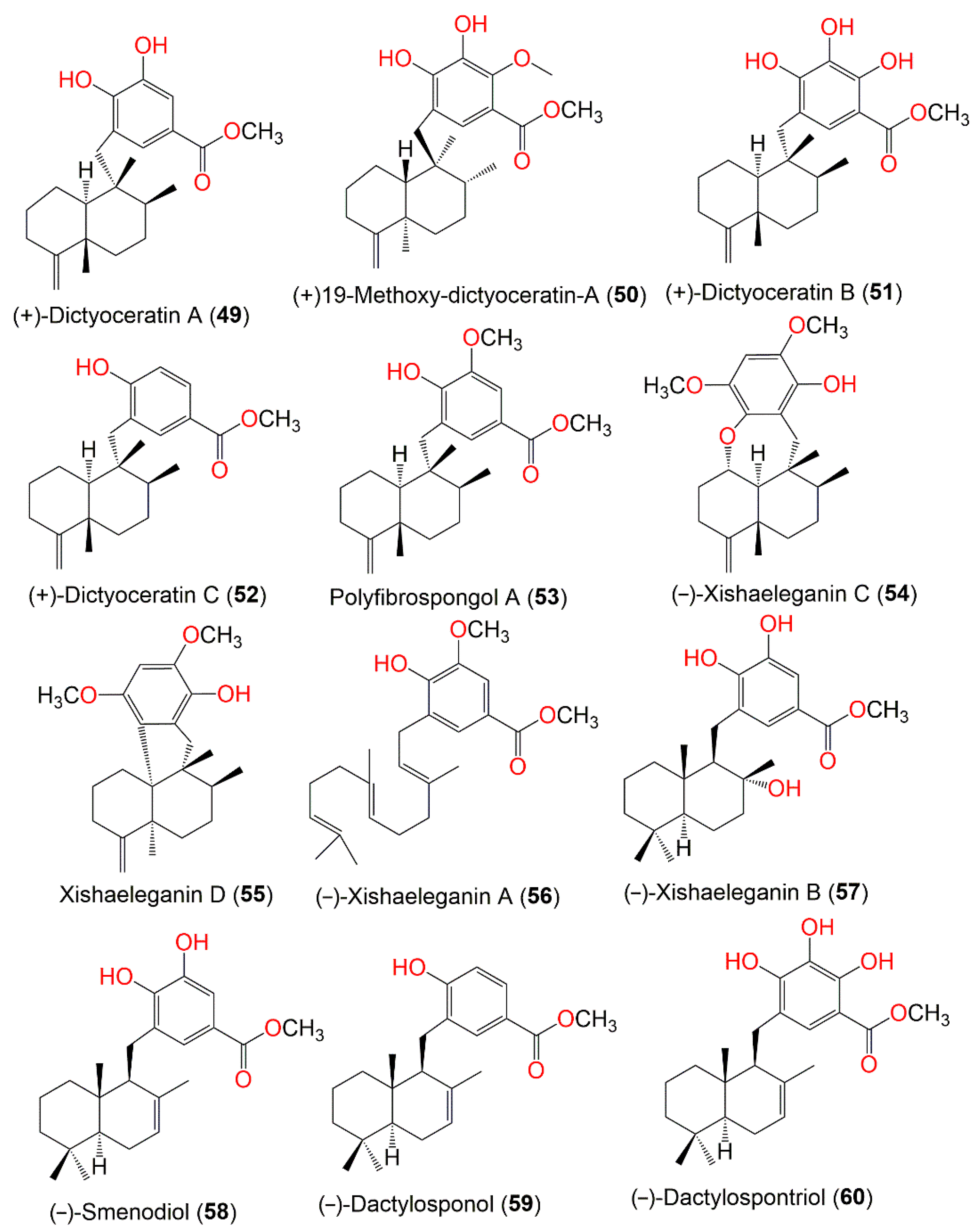
Figure 5.
Structures of compounds
49
–
60
.
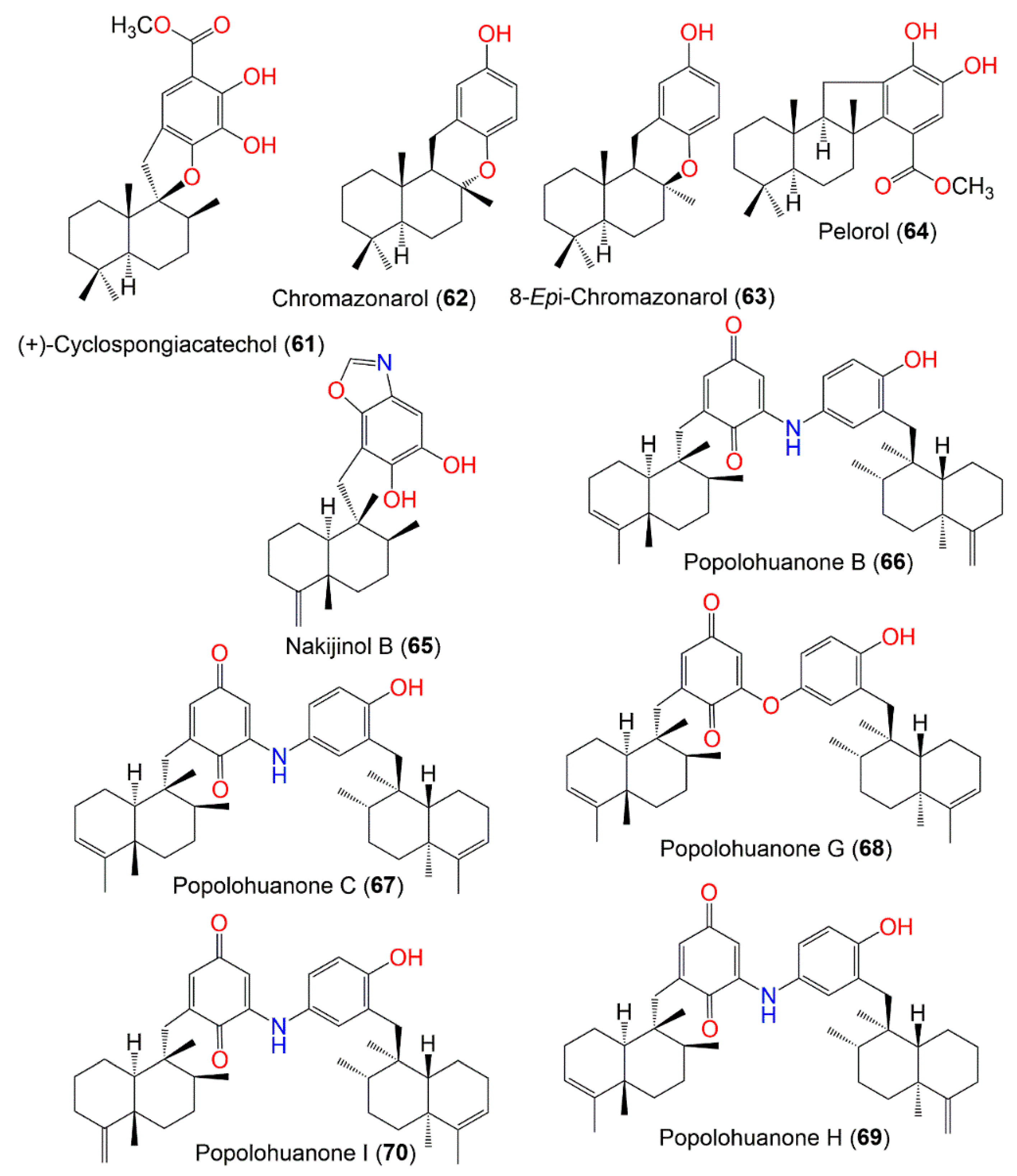
Figure 6.
Structures of compounds
61
–
70
.
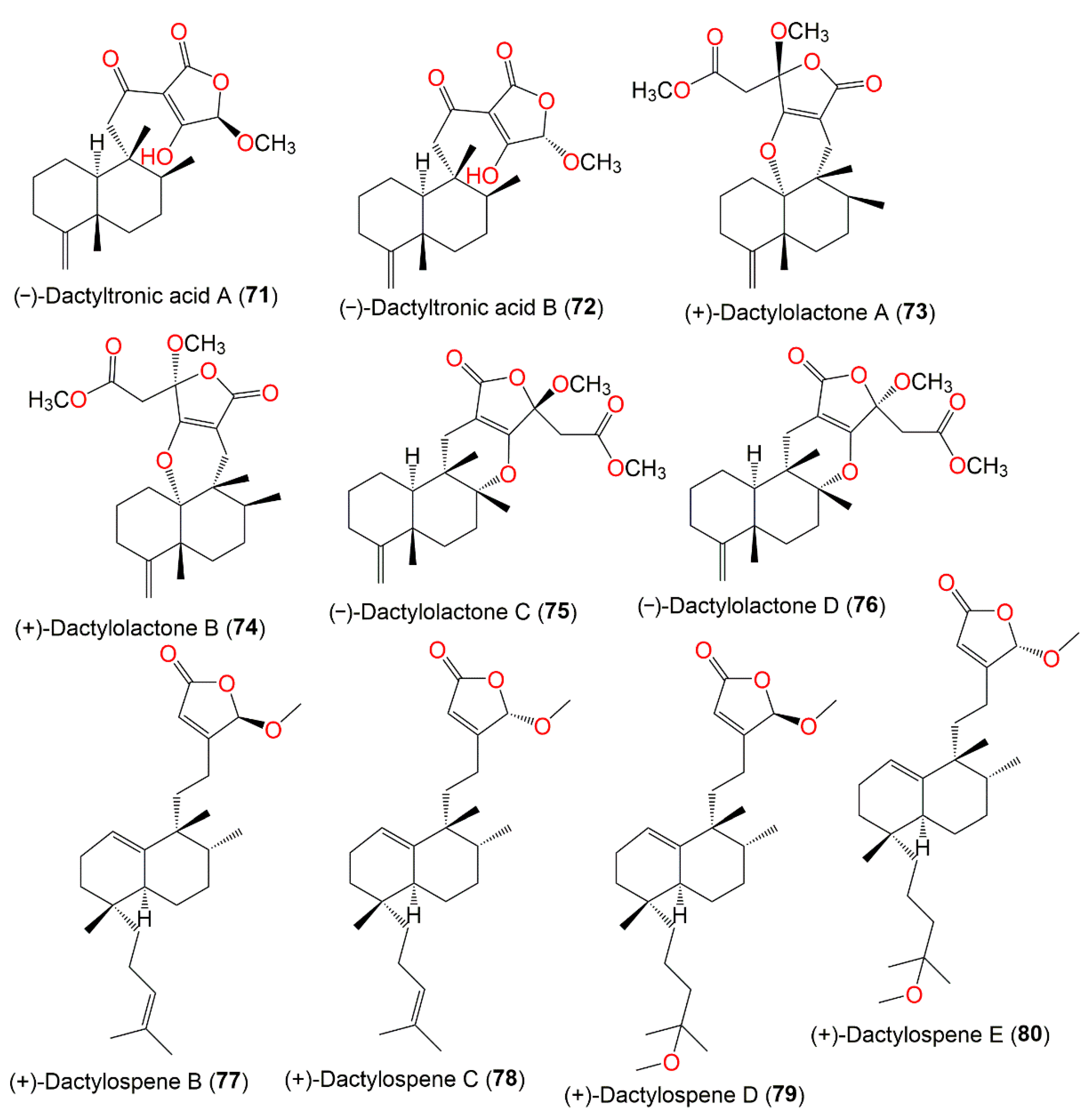
Figure 7.
Structures of compounds
71
–
80
.
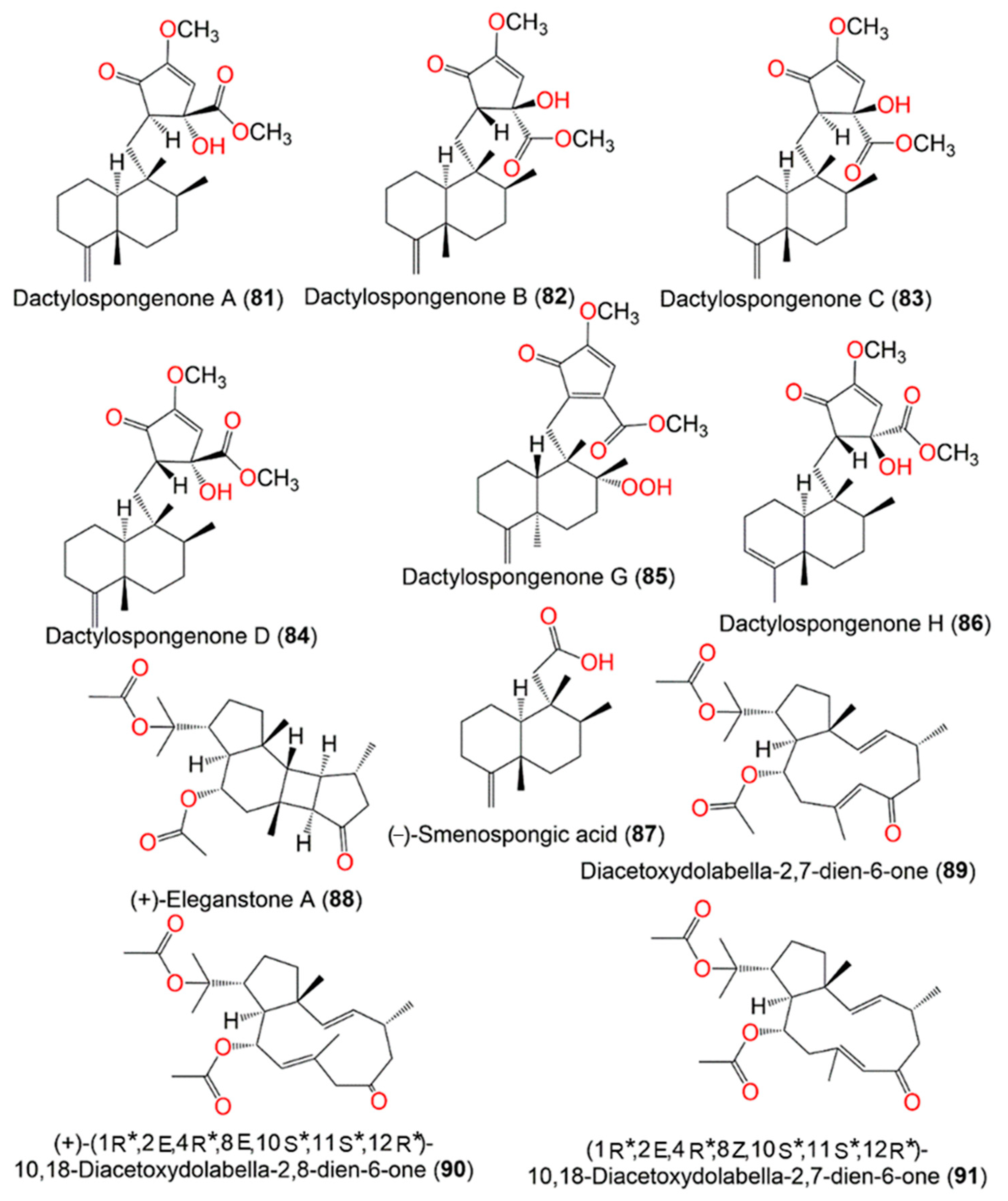
Figure 8.
Structures of compounds
81
–
91
.
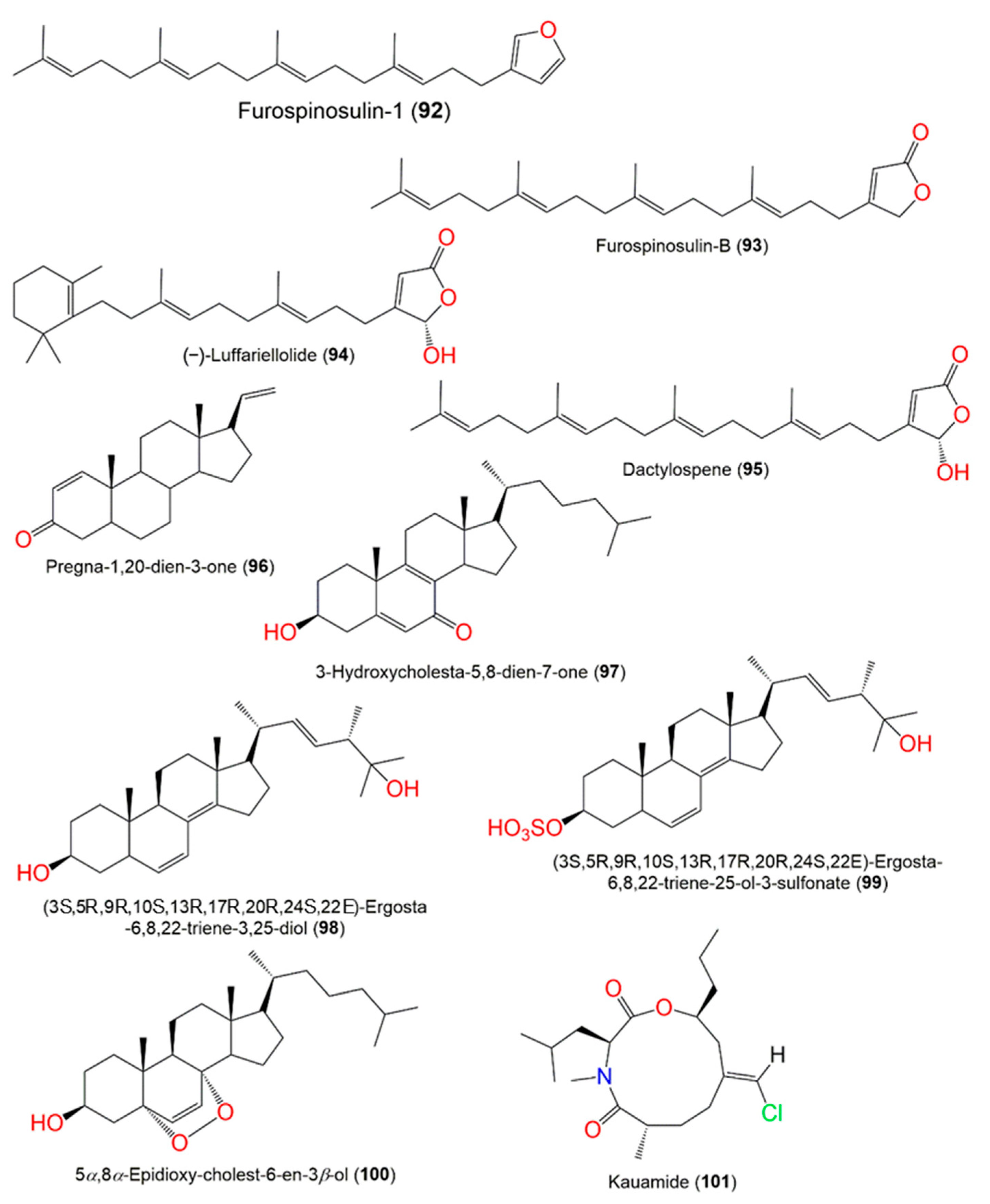
Figure 9.
Structures of compounds
92–101
.
3. Biosynthetic Pathways of D. elegans Metabolites
Several studies reported the biosynthetic pathways of the reported sesquiterpenes from this sponge. In this work, the reported postulated pathways were summarized. It was reported that the observed differences in stereochemistry among the marine sesquiterpene metabolites could be inferred from the precursor binding preferences within a single cyclase enzyme active site
[54][16]. Additionally, this may be due to the existence of various synthase enzymes, whereas each individual enzyme can create a range of diverse metabolites, as well as presumably the potential to change the stereochemical outcomes, relying on the provided substrate nature. Thus, the possibility of enantiomeric metabolites should be considered that emphasize the significance of reporting [α]
D values for these terpenes in cases where they are utilized as a reference in the stereochemical determination
[21,55][17][18].
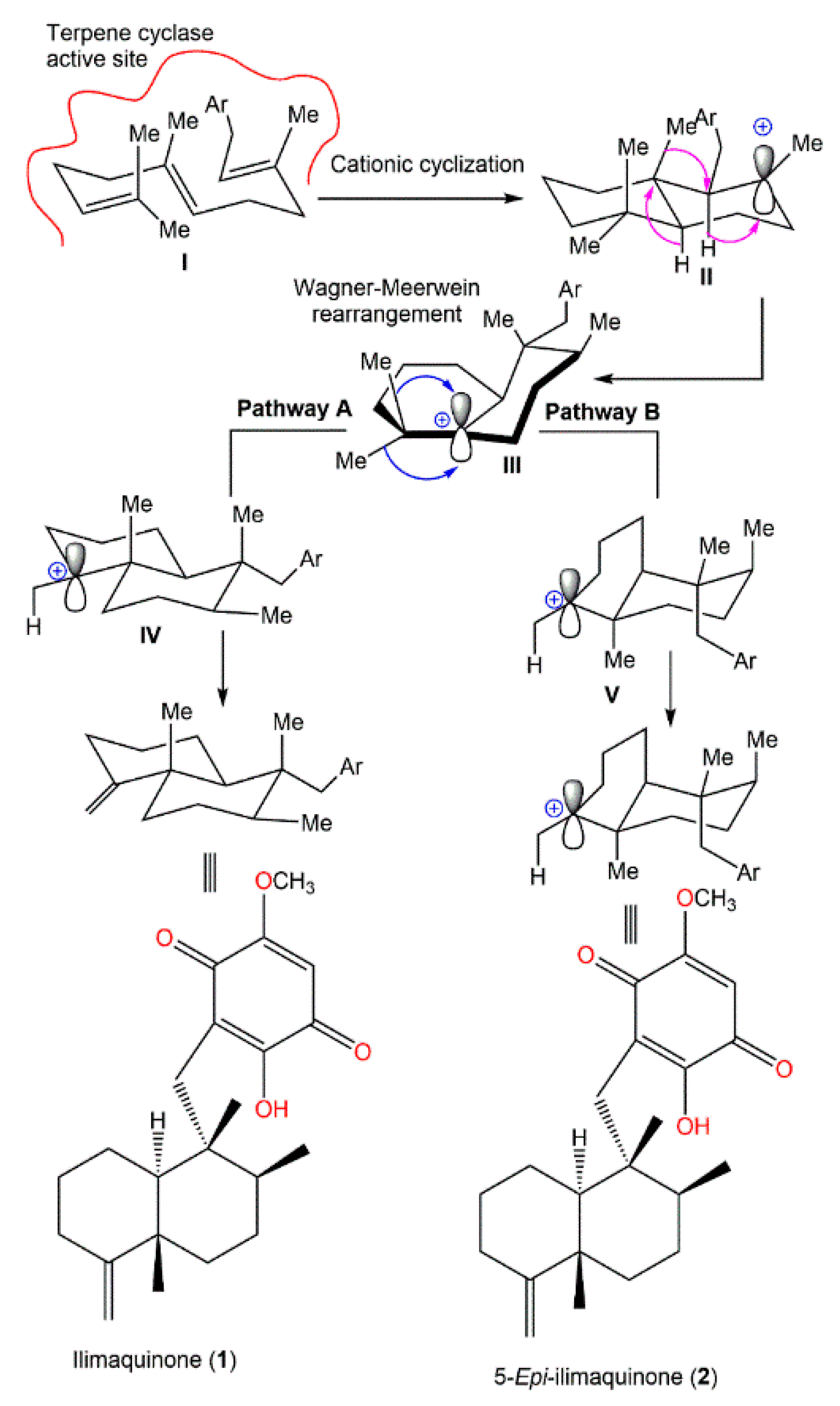
Boufridi et al. hypothesized that the biosynthetic process of
1 and
2 started with the farnesylation of the aromatic ring, which is the quinone moiety’s precursor to obtain
I. This involves the initial folding of
I within the active site of a specific terpene cyclase (
Scheme 1). Successively, two carbocationic intermediates
II and
III are resulted from peri-planar Wagner–Meerwein hydrogen and methyl shifts. Finally,
III may undergo two pathways (A or B) for the formation of
1 and
2 through the loss of a proton from carbocations
IV and
V, respectively
[40][19].
Scheme 1. Postulated biosynthetic pathway for
1 and
2 [40].
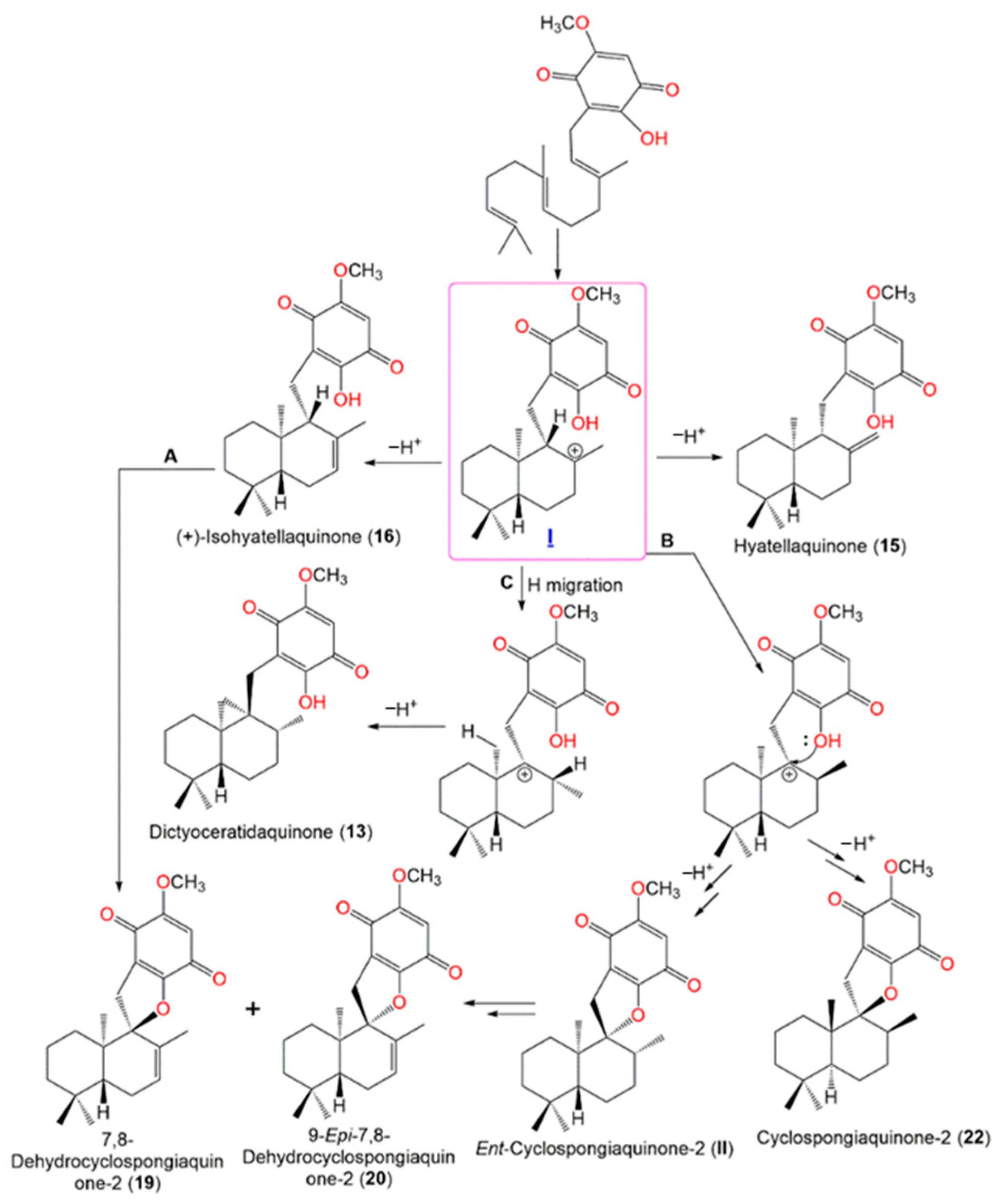
Yong et al. postulated that
13,
15,
16,
19,
20, and
22 may be originated from the cationic intermediate (
I) (
Scheme 2). The introduction of double bond yields
16. The cyclization of
16 (pathway A), or hydride migration with subsequent cyclization forming
22 and its related cyclic intermediate
ent-cyclospongiaquinone-2 (
II), then the formation of double bond yields
19 and
20 (pathway B). Finally, the hydride migration and loss of a proton from the cationic site adjacent methyl form
13 with a cyclopropyl ring (pathway C)
[21][17].
Scheme 2. Postulated biosynthetic pathways for
13,
15,
16,
19,
20, and
22 [21].
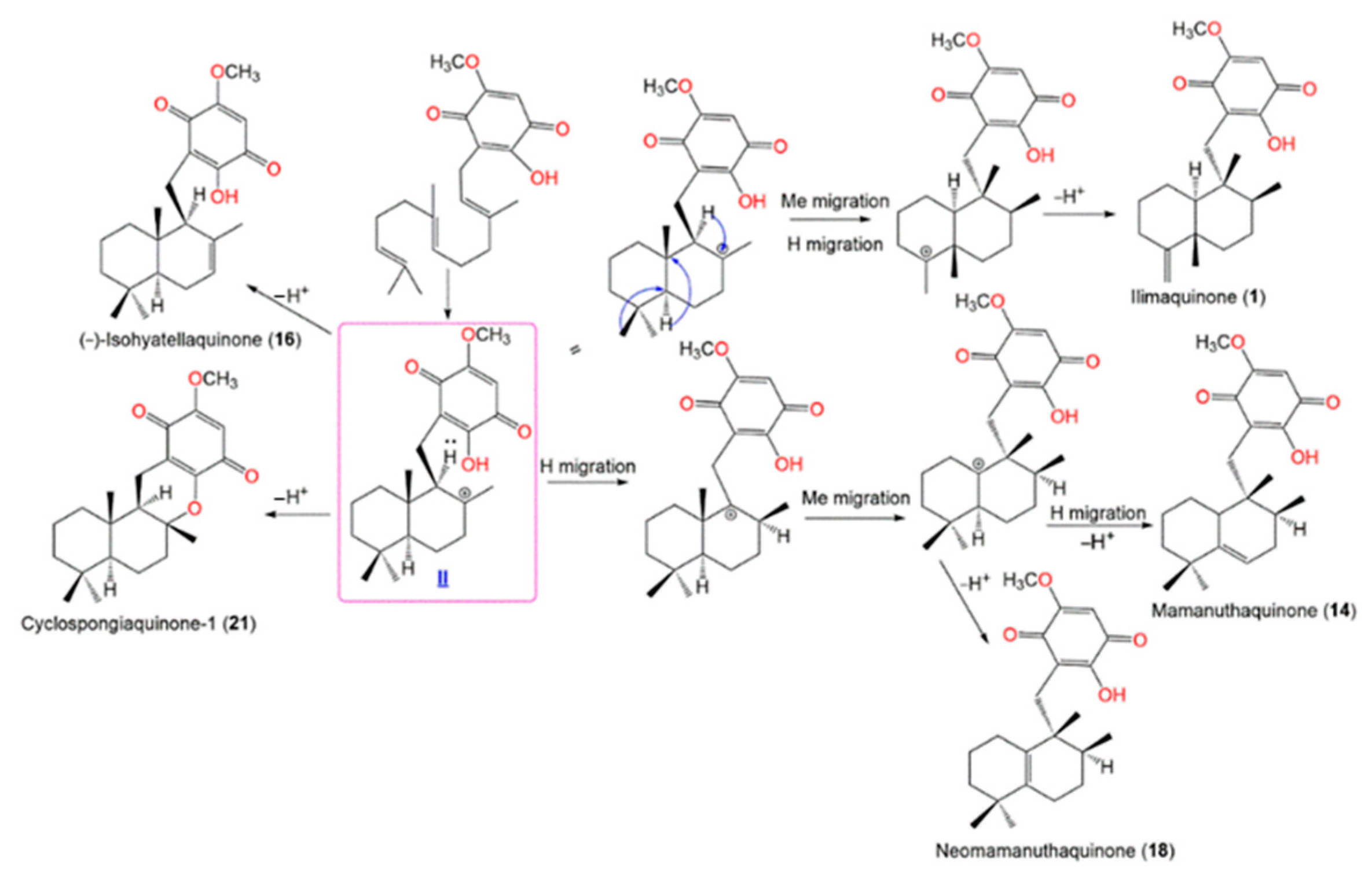
Scheme 3 shows that the farnesyl precursor (
I) initial cyclization gives
II (enantiomeric cation) that undergoes a loss of a proton to give
16 and
21. On the other side, cation
II’s hydride and methyl migrations yield compounds
1,
14, and
18 [21][17].
Scheme 3. Postulated biosynthetic pathways for
1,
14,
16,
18, and
21 [21].
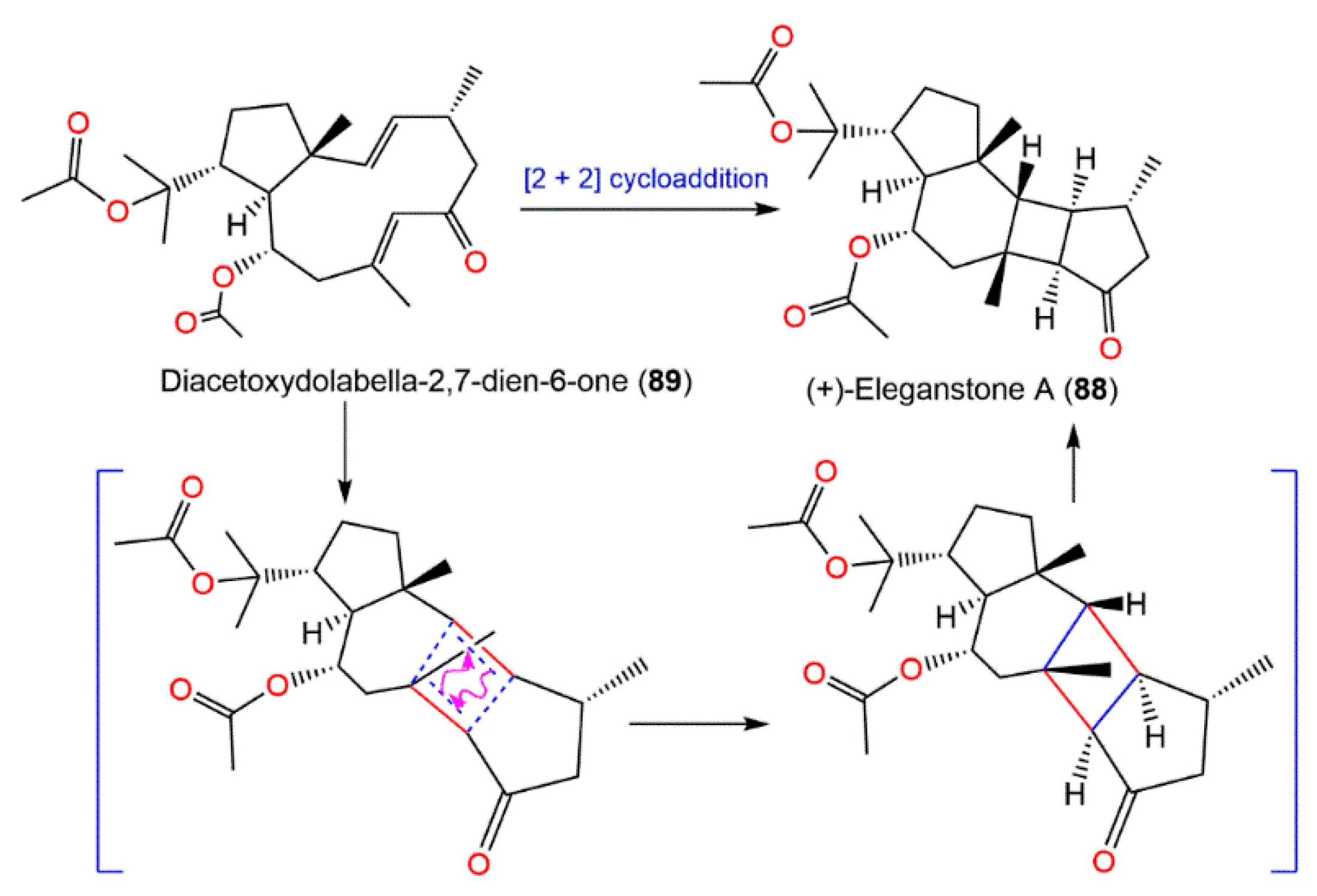
Diacetoxydolabella-2,7-dien-6-one (
89) was proposed to be the biogenetic precursor of
88. (+)-Eleganstone A (
88) may be originated from diacetoxydolabella-2,7-dien-6-one (
89) through intramolecular [2 + 2] cycloaddition. The coupling of Δ
7,8 and Δ
2,3 in
89 via the endo-cycloaddition generates
88 (
Scheme 4)
[36,56][20][21].
Scheme 4. Proposed biosynthetic pathway for
88 [36].
4. Synthesis of D. elegans Metabolites
Some of the reported metabolites from this sponge possessed fascinating bioactivities, such as anticancer. Nevertheless, further biological investigation is limited due to the not enough isolated metabolites. Therefore, research interests have been directed towards synthesis and structural modification of these metabolites to improve the bioactivities and study structural/activity relations, which could help in drug development and discovery. Some of these studies have been highlighted here.
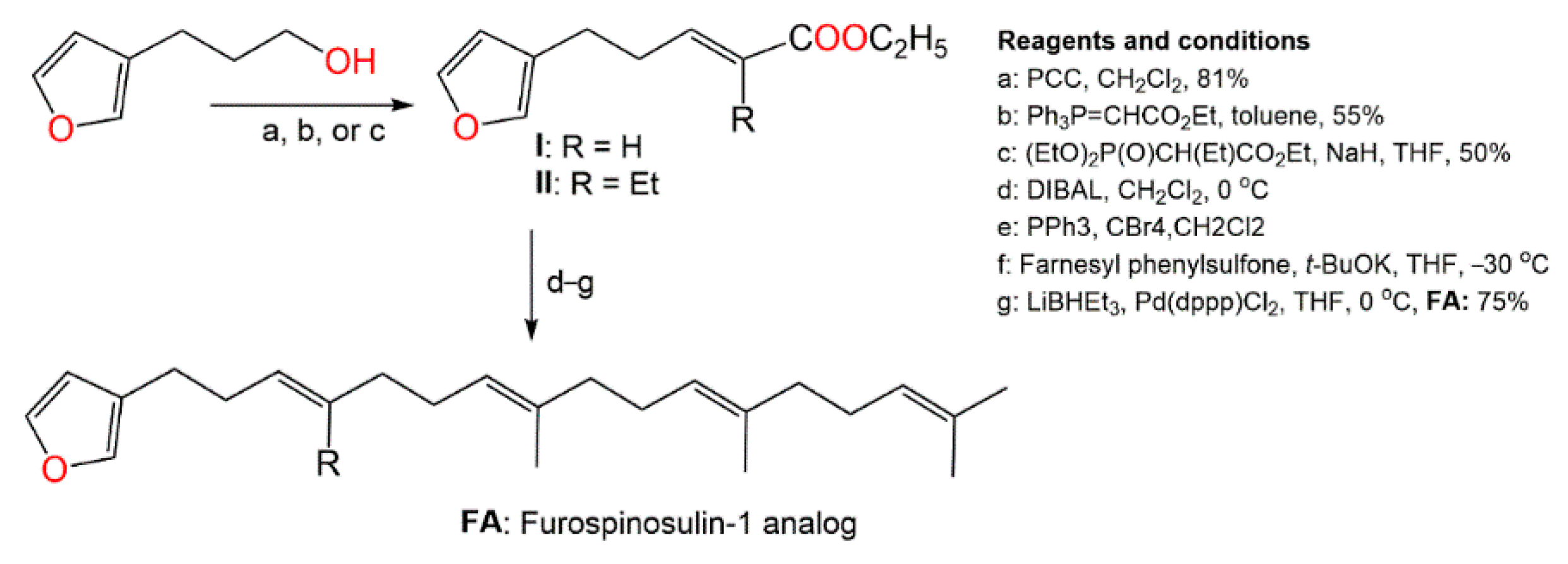
Kotoku et al. synthesized several analogs of furospinosulin-1 (
92) and assessed their selective hypoxia inhibitory potential (
Scheme 5)
[57][22].
Scheme 5. Synthesis of furospinosulin-1 (
92) analogues [57].
It was found that only the analog (
FA) with desmethyl near to the furan ring had an excellent hypoxia-specific growth inhibitory potential such as furospinosulin-1 (
92) and displayed greater in vivo antitumor potential in oral administration (doses 1–10 µM) versus DU145 cells, as well as lower toxicity in normal conditions (dose 300 µM)
[57][22]. Therefore, this analog might be better than furospinosulin-1 for drug candidates.
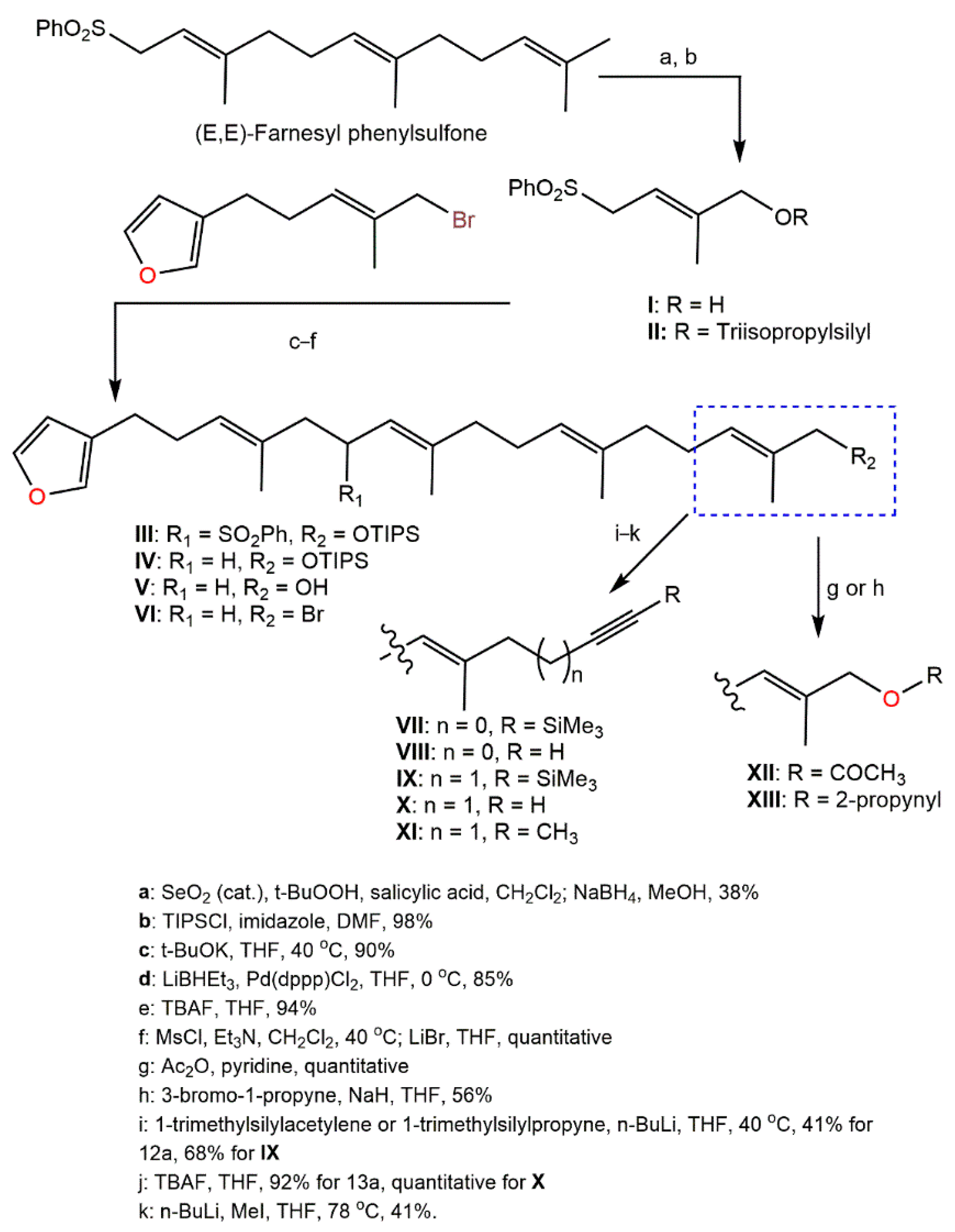
Kotoku et al. reported that the modifications of
92 structure such as elongating the methyl group close to the furan ring, changing the aromatic ring, and side-chain truncation led to a remarkable loss of selective hypoxia growth inhibition capacity, obviously revealing that the entire structure was substantial for the binding with the target molecule
[58][23]; whilst the analog had a longer side chain partially retained hypoxia-selective inhibitory potential. Therefore, Kotoku et al. synthesized and assessed various furospinosulin-1 (
92) tail-modified analogs (
Scheme 6)
Scheme 6. Synthesis of tail-modified analogues of furospinosulin-1 (
92) [58].
The analog
X was found to be much more potent than furospinosulin-1 as it possessed hypoxia-selective growth-inhibitory potential (Conc. 1–300 µM) and had powerful in vivo antitumor effectiveness after oral administration (doses 5–25 mg/kg) without side effects
[58][23].
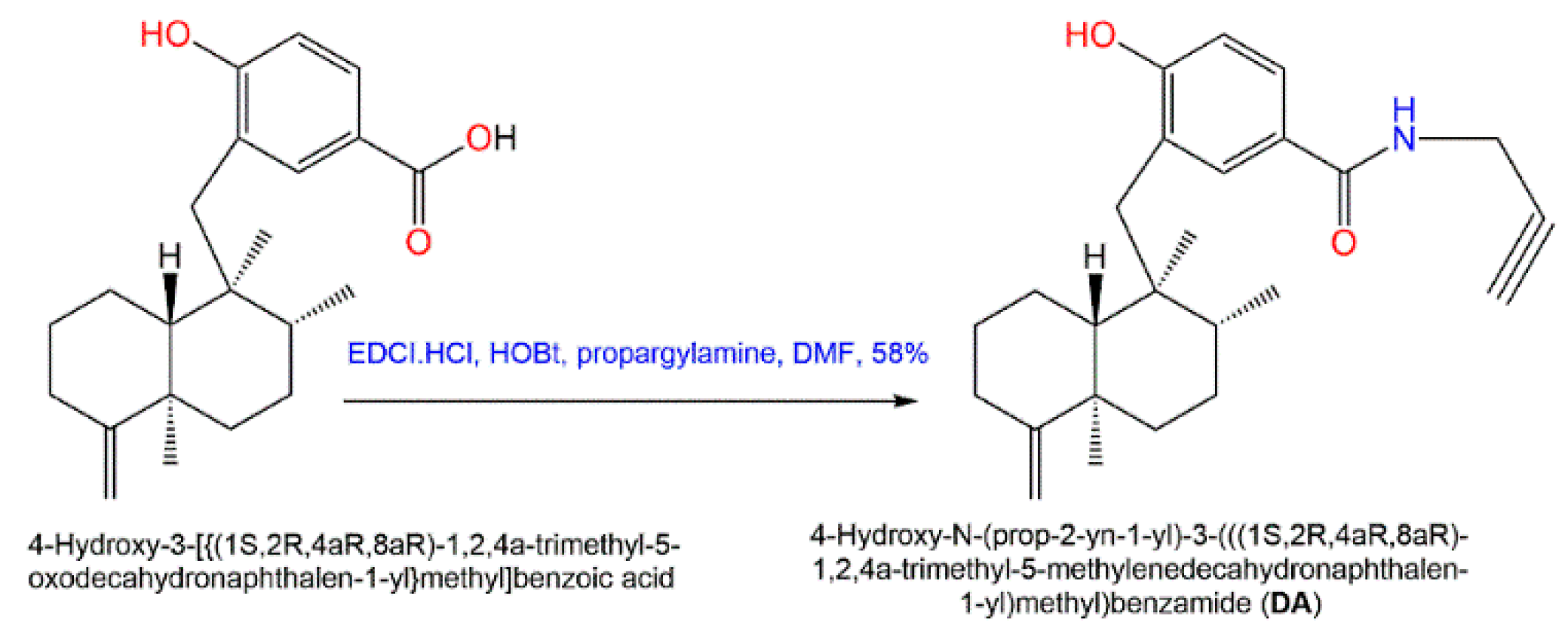
Additionally, in 2015, Sumii et al. assessed the structure–activity relationship of (+)-dictyoceratin A (
49) and (+)-dictyoceratin C (
52) through the synthesis of structural-modified derivatives of (+)-dictyoceratin C (
52) and testing their in-vivo antitumor potential
[47][24]. Thus far, the only methyl ester substitution in
52 with propargyl amide (
DA) had an excellent hypoxia-selective growth prohibition potential at 30 µM relative to
52 (
Scheme 7). Thus, it could be efficient for probe molecules synthesis for target identification of
49 and
52 [47][24].
Scheme 7. Synthesis of potent structurally modified analog of
52 [47].
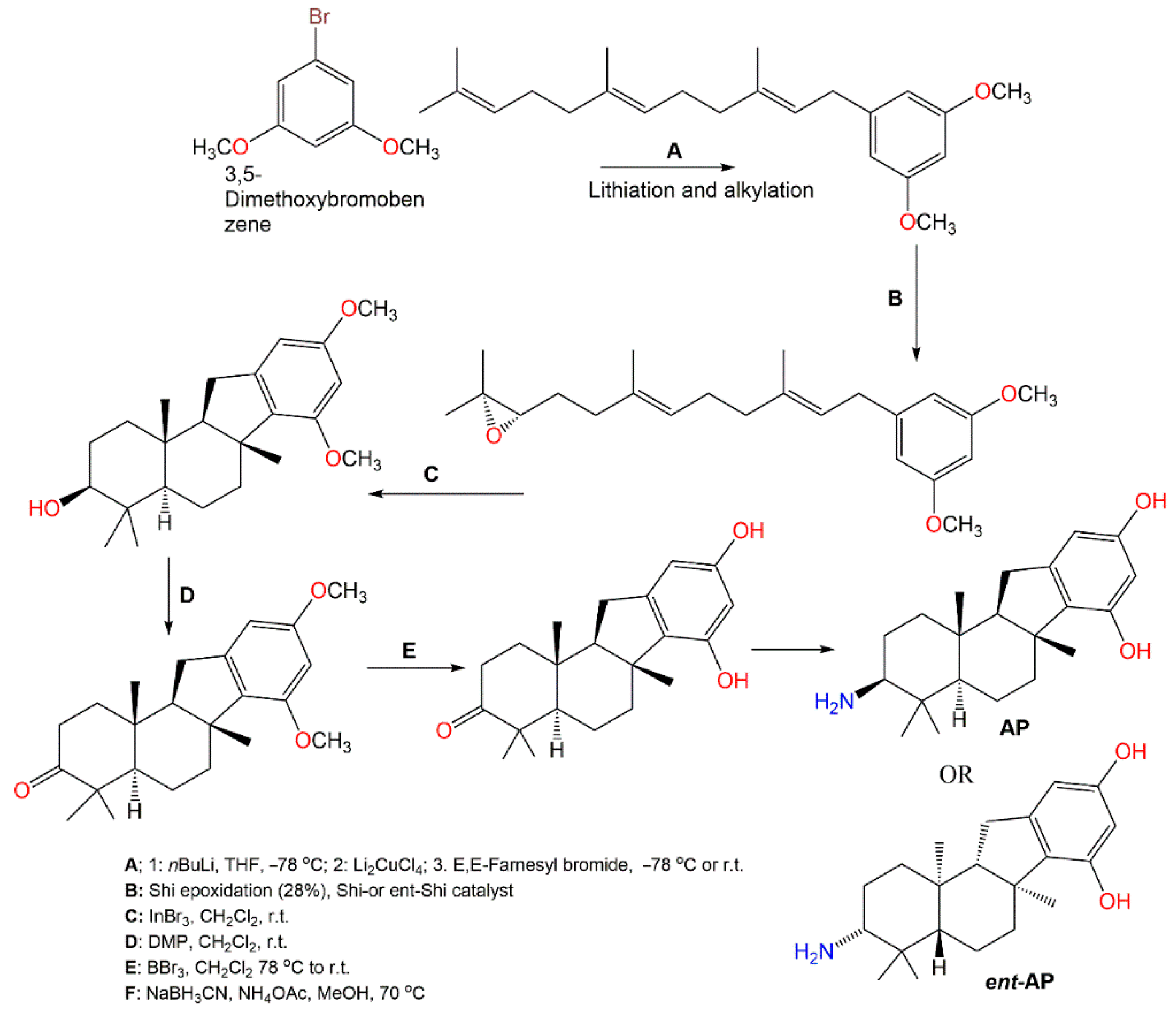
Pelorol (
64), sesquiterpene hydroquinone having C8–C21 cyclization had a potent and selective SHIP1 (Src homology 2-containing inositol 5-phosphatase 1) activating potential
[59][25]. However, this compound had catechol moiety that can be either enzymatic or chemically oxidized to orthoquinones, forming covalent linkages with proteins resulting in losing the activity. Additionally, it is highly water-insoluble, which limits its in vivo bioactivity as a SHIP1 activator. Meimetis et al. synthesized more water-soluble amino analogs
ent-
PA or (±)-PA (
Scheme 8). Synthesis of (±)-PA characterized by generating a tetracyclic ring system through a cation-initiated polyene cyclization
[60][26]. Then, 3,5-Dimethoxybromobenzene lithiation subsequent alkylation with farnesyl bromide produced the 3,5-dimethoxybenzene prenylated intermediate. A racemic epoxide was produced utilizing
m-chloroperbenzoic acid with subsequent steps giving (±)-PA. Repeating the same procedure using
ent-Shi catalyst produced
ent-
PA analog. These analogs in vitro activated SHIP1, prohibited Akt phosphorylation and had potent in vivo anti-inflammation potential (ED50 0.1 mg/kg, oral gavage) using passive cutaneous anaphylaxis mouse model
[60][26]. The results suggested that
ent-PA or (±)-
PA pelorol analogs are promising candidates for further in vivo preclinical investigation as SHIP1-activating therapeutics for treating hematopoietic illnesses, involving aberrant PI3K cell signaling activation.
Scheme 8. Synthesis of SHIP1-activating analogs of pelorol (
64) [60].
5. Activities of D. elegans Extracts and Fractions
BACE1 (β-site of amyloid precursor protein cleaving enzyme) is an enzyme involved in Alzheimer’s disease pathogenesis. The 75% and 100% MeOH C8 fractions of
D. elegans obtained from the coast of Kauai had significant in vitro BACE1 inhibition (%inhibition 66% and 73%, respectively, at Conc. 30 μg/mL)
[61][27]. Li et al. revealed that the CH
2Cl
2/MeOH extract possessed marked IL-6 and TNF-α inhibitory potential
[34][28]. Additionally, the antioxidant potential testing revealed that
D. elegans hexane extract exhibited significant antioxidant potential in comparison to the ascorbic acid
[62][29].