The current understanding of miRNA biology is greatly derived from studies on the guide strands and the passenger strands, also called miRNAs*, which are considered as carriers with no sense for long periods. As such, various studies alter the expression of guide strands by manipulating the expression of their primary transcripts or precursors, both of which are premature miRNAs. In this situation, the regulatory miRNA* species may interfere with the phenotypic interpretation against the target miRNA. However, such methods could manipulate the expression of two functionally synergistic miRNAs of the same precursor, leading to therapeutic potential against various diseases, including cancers. Premature miRNAs represent an underappreciated target reservoir and provide molecular targets for “one-two punch” against cancers. Examples of targetable miRNA precursors and available targeting strategies are provided in this review.
- miRNA*
- post-transcriptional regulation
- premature miRNA intervention
- pre-miRNA annotation
- therapeutic potential
1. Introduction
MicroRNA (miRNA) is a class of small nonprotein-coding RNAs (ncRNAs) that are 15–27-nucleotides (nt) in length, are ubiquitously expressed, and interact with most of the mammalian messenger RNAs (mRNAs) [1]. The interplay between miRNAs and mRNAs leads to decreased translational efficiency and/or mRNA levels, which is the predominant reason behind decreased protein production [2,3,4][2][3][4]. It is estimated that over 60% of mRNAs are conserved targets for endogenous miRNAs, and more mRNAs will be added to this conservative estimation when less conserved sites will be taken into account, indicating the existence of a previously unexpected post-transcriptional RNA signaling network [1]. Extensive screening for functional miRNAs has become easier due to the refinement of the principles that allow the manipulation of miRNA expression; functional miRNAs will gradually overturn the central dogma of molecular biology [5]—evidence regarding involvement of miRNAs in post-transcriptional regulation is mounting, and they are considered as the key components in the maintenance of intracellular homeostasis of many physiological processes [6,7][6][7]. Apart from their intracellular functions, miRNAs are also highlighted as unpredictable, important intercellular communicators, which help in the maintenance of a homeostatic extracellular microenvironment [8]. Dysregulated miRNAs in cell type-specific or cell-state-specific gene expression patterns cause pathophysiological transitions toward neurological [9], cardiovascular [10], metabolic [11], autoimmune [12], and carcinogenic disorders [13], and in turn, provide novel diagnostic, therapeutic, and prognostic opportunities in deciphering diseases [14].
2. Functional Annotation of Premature miRNAs in Cancers
The functional importance of miRNAs is being extensively studied. The functional role of miRNA* species in various diseases is highly valuable in annotating the functionality of miRNA precursors [61][15]. Almost all models of functional miRNAs, from diverse origins and biosynthesis pathways, converge at the step where a hairpin miRNA precursor is produced, and thus, functional annotation of premature miRNA is focused on the last premature form of miRNA, the pre-miRNA. Given the inability to establish direct contact with regulatory protein complexes, the functions of pre-miRNAs are determined by the joint activity of their encompassing regulatory miRNAs. In this entreview, wey, researchers have mainly focused on the pre-miRNAs that produce two mature strands and account for the majority of miRNA precursors. Mining functional information related to miRNA in combination with newly discovered biological implication of its passenger strand is essential for drafting the biological propensity of their parental precursor transcripts. In cancers, pre-miRNAs can be divided into three different subgroups, i.e., oncogenic, tumor suppressive, and ambivalent pre-miRNAs, based on the function of the dual sister strands (Figure 2Figure 1a–c.).
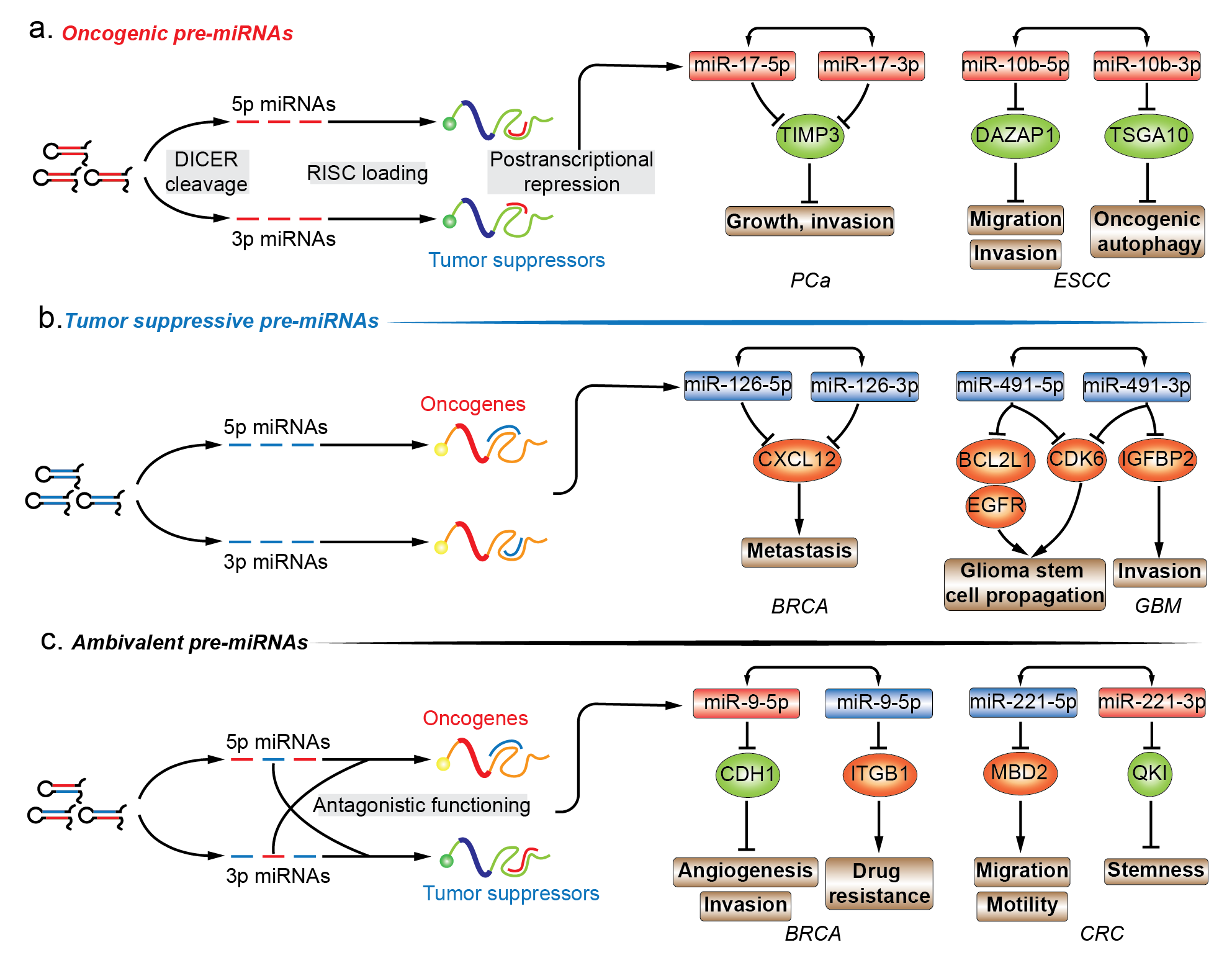
2.1. Oncogenic Pre-miRNAs
Similar to the definition of oncogenic miRNAs (oncomiRs), an oncogenic pre-miRNA produces two oncomiRs, which synergistically contribute to the oncogenic process under a certain cellular context (Figure 21a.; Table 2). For instance, Yang and his colleagues have shown that pre-miR-17 induces more malignant phenotypes in prostate cancer (PCa) and hepatocellular carcinoma (HCC) [72,73][16][17]. In PCa, luciferase reporter assay demonstrated that both miR-17-5p/3p targeted the 3’-UTR of TIMP metallopeptidase inhibitor 3 (TIMP3), and ectopic expression of pre-miR-17 promoted tumor growth and invasion, which was mediated by TIMP3 repression [72][16]. Additionally, pre-miR-17 transgenic mice were prone to develop HCCs. Unlike targeting a common tumor suppressor to augment the regulatory activity in PCa, pre-miR-17 displayed a different mode of action by targeting multiple tumor suppressive pathways in HCC. MiR-17-5p was bound to two putative binding sites of PTEN, while GalNAc transferase 7 (GalNT7) and VIM (namely vimentin) were regulated by the passenger strand miR-17-3p; all these regulatory activities are ceRNA-independent. The combined effect of miR-17-5p/3p led to a proliferative and migratory phenotype [73][17]. Additional examples include oncogenic pre-miR-10b in esophageal squamous cell carcinoma (ESCC) [74[18][19],75], pre-miR-21 in ovarian cancer [98[20][21][22],99,100], and pre-miR-221 in PCa [76[23][24],77], the dual tumor-promoting strands in all these cases have been identified by individual studies. First, starvation-induced miR-10b-5p expression increased the autophagic flux in ESCC during nutrient deprivation, and DAZ associated protein 1 (DAZAP1) was the direct target and mediator of this phenomenon. Decreased DAZAP1, an oncogenic signature, was observed in ESCC tumor samples and was correlated with poor survival, and this enhanced the migratory and invasive capacities in ESCC cell lines [74][18]. MiR-10b-3p expression increased under hypoxic conditions in ESCC cells, and its downregulation, mediated by hypoxia, impaired its migratory and invasive capabilities. This could be because the hypoxia-inducing miR-10b-3p suppressed the expression of targeting testis specific 10 (TSGA10), which contains a putative binding site at the 3’-UTR [75][19]. Second, miR-21-5p is located within chromosomal region of 17q23-25 and its amplification is correlated with shorter progression-free and overall survival in ovarian clear cell carcinoma (OCCC), one of the most lethal types of gynecological malignancies. miR-21-5p amplification demonstrated that tumor suppressor PTEN was a direct target of miR-21-5p, but miR-21-5p antagonization was not able to compromise cell proliferation or invasion of RMG-II cells, which is a newly established OCCC cell line [98][20]. However, in OVCAR-3 cells (low grade serous ovarian papillary adenocarcinoma), miR-21-5p knockdown increased in PTEN upregulation and decreased cell proliferation, migration, and invasion [99][21]. MiR-21-3p was identified as a potential miRNA mediator for the development of cisplatin resistance through microarray analysis performed between A2780 cell line and its cisplatin-resistant derivative, CP70. The acquired cisplatin resistance of A2780 could be induced by miR-21-3p overexpression, which can be witnessed in multiple ovarian cancer cell lines. In response to miR-21-3p upregulation, the decrease of its direct target, the neuron navigator 3 (NAV3), desensitized ovarian cancer cells to cisplatin [100][22]. Unexpectedly in the same paper, the introduction of miR-21-5p mimics sensitized A2780 cells (human ovarian cancer cell line) to cisplatin, and the underlying mechanism was not further investigated in the study [100][22]. The unusual anticancer effect of miR-21-5p contradicts the general notion of its canonical oncogenic role in various cancers, including ovarian cancers. By searching related terms in PubMed, wresearchers found several studies reporting the relationship between miR-21-5p and cisplatin resistance in ovarian cancer, and all the studies demonstrate that miR-21 contributes to the development of cisplatin resistant phenotype [101,102,103,104][25][26][27][28]. It is of particular interest to uresearchers as three out of four studies are performed in A2780 cell line or its variants as well, which indicates a similar cellular background. However, even the same experimental cell line in a different lab can introduce bias in phenotypic performance. Therefore, it is controversial in the function role of miR-21-5p in cisplatin resistance of ovarian cancer, and requires further validation in the same experimental model and at the same time. Lastly, a study showed that pre-miR-221 mediates the malignant transition in PCa. MiR-221-5p expression was elevated in tumor and adjacent tissues of clinical samples, indicating its oncogenic role in prostate carcinogenesis. Ectopic miR-221-5p manipulation proved that miR-221-5p enhanced the migratory and proliferative capabilities of PCa cells in vitro, and suppressers of cytokine signaling 1 (SOCS1), an established tumor suppressor, was validated to be the direct target of 3’-UTR luciferase reporter assay [76][23]. MiR-221-3p is reported to participate in the development or maintenance of castration resistant prostate cancer (CRPC) phenotype, but the underlying mechanism remains unknown [105][29]. Another study has demonstrated that ectopic upregulation of miR-221-3p promoted androgen independent cell growth in LNCaP cell lines. Two potential tumor suppressive targets, HECT domain E3 ubiquitin protein ligase 2 (HECTD2) and RAB1A, were identified by the combination of RNA immunoprecipitation and miR-221-3p target prediction programs. Reduction of HECTD2 and RAB1A or increased miR-221-3p contributed to the development of CRPC phenotype [77][24]. Examples of oncogenic pre-miRNAs elucidate the preliminary therapeutic potential of dual oncomiRs ablation using only one pre-miRNA intervention.2.2. Tumor-Suppressive Pre-miRNAs
In contrast, a tumor suppressive pre-miRNA can be processed into dual tumor suppressive mature miRNAs, which simultaneously target two sets of oncogenes (Figure 21b.; Table 2). Tumor suppressive pre-miR-126 in breast cancer (BRCA) [78][30], pre-miR-144 in lung squamous cell carcinoma (LUSC) [79][31], pre-miR-524 and pre-miR-491 in glioblastoma (GBM) [80,81][32][33] are discussed in this section. To start with, miR-126-5p/3p were downregulated in metastatic BRCA cell lines and clinical samples, and ectopic pri-miR126 expression significantly decreased lung metastasis, implying the negative regulatory potential in cancer metastasis. With focus on cytokines/chemokines, CXCL12 (also known as Sdf-1α) was downregulated in response to forced pri-miR-126 expression. The luciferase reporter assay using CXCL12 3′-UTR construct confirmed that either pri-miR-126 or its mature products, miR-126-5p/3p, suppressed the luciferase activity in BRCA cells. The suppression of the migratory capabilities of mesenchymal stem cells were mediated by downregulation of CXCL12 through miR-126-5p/3p, thereby preventing the development of the metastatic phenotype [78][30]. Secondly, miR-144-5p/3p were downregulated in LUSQ tissues and cell lines, and high miR-144-5p/3p expression was correlated with a favorable outcome for LUSQ patient data in The Cancer Genome Atlas (TCGA) database. Transient miR-144-5p/3p transfection impaired the proliferative, invasive, and migratory abilities of LUSQ cells. Both miR-144-5p/3p were functional substrates of AGO-RISC, and neuronal calcium sensor 1 (NCS1) was a shared target for both strands. The phenotypic influence of neuronal calcium sensor 1 knockdown mimicked the counterpart of miR-144-5p/3p introduction [79][31]. Thirdly, miR-524-5p/3p were exclusively downregulated in the classical subtype of GBM and negatively associated with epidermal growth factor receptor (EGFR) expression, while EGFR amplification was observed in 97% of the classical subtypes and was infrequent in other subtypes [80,106][32][34]. EGFR could recruit the repressive histone modifier to the promoter region of MIR524 and reduced pri-miR-524 production. TEA domain transcription factor 1 (TEAD1) and SMAD2 were validated to be the target of miR-524-5p and -3p, respectively, and HES1 was the target of both the strands. All three targets of pre-miR-524 converge on the activation of the downstream oncogene, MYC, resulting in retention of the migratory, invasive, and proliferative phenotypes [80][32]. Lastly, MIR491 was found to be frequently codeleted with cyclin dependent kinase inhibitor 2A (CDKN2A) located on chromosome 9p21.3 in GBMs, and accordingly, the corresponding miR-491-5p and -3p expression levels were significantly lowered in GBM compared to in the normal brain. Reporter assays confirmed the binding sites within EGFR and BCL2 like 1 (BCL2L1) for miR-491-5p, insulin like growth factor binding protein 2 (IGFBP2) for miR-491-3p, and cyclin dependent kinase 6 (CDK6) for both. The concurrent loss of tumor suppressive miR-491-5p and -3p derepressed multiple oncogenes, consequently exacerbating malignancy in GBM in vitro and in vivo [81][33]. Based on the coordinated effect of the two mature products of pre-miRNA on cancer suppression, supplementation with two tumor suppressors through introduction of a single miRNA precursor might promisingly outweigh a single miRNA amendment for future therapeutic purposes.2.3. Ambivalent Pre-miRNAs
Unlike the aforementioned oncogenic or tumor suppressive pre-miRNA, ambivalent pre-miRNAs can be processed into two mature miRNAs which tend to function in two opposing directions (Figure 21c; Table 2). For example, pre-miR-31 in oral squamous cell carcinoma (OSCC), pre-miR-9 and pre-miR-10b in BRCA consist of a single oncomiRs at the 5′ arm and tumor suppressive miRNA at the 3′ arm, while pre-miR-221 in colorectal cancer (CRC) are constituted in a reverse manner [82,83,84,85,86,87,88,89][35][36][37][38][39][40][41][42]. With respect to pre-miR-31, the oncogenic miR-31-5p in head and neck squamous cell carcinoma (HNSCC) were found to be one of the most highly expressed miRNAs when compared to normal tissues. The elevated miR-31-5p expression was linked to increased oncogenic potential of HNSCC cells by promoting cell growth and migration. The phenotypic effect of miR-31-5p was mediated by the suppression of factor-inhibiting hypoxia-inducible factor (FIH), which is the direct target of miR-31-5p. However, under normoxic conditions, FIH acts as a tumor suppressor by inhibiting oncogenicity [82][35]. The tumor suppressive complementary strand, miR-31-3p, was associated with decreased capability of growth and migration in SAS and Fadu OSCC cell lines. Ras homolog family member A (RHOA) was chosen as a candidate for observing the phenotypic changes in response to miR-31-3p manipulation and the miRNA-target relationship was verified by 3′-UTR luciferase reporter assays. Interestingly, the authors examined the phenotypic influence of forced pre-miR-31 expression, which supplemented two mature products in the form of oncogenic 5′ arm and tumor suppressive 3′ arm, and showed that the phenotypic profiling of ambivalent pre-miR-31 introduction was oncogenic [83][36]. Regarding pre-miR-9, oncogenic miR-9-5p was found to be significantly upregulated in human BRCA compared to normal breast tissues. Besides, c-myc induced an increase of more than 500-fold in the expression of miR-9-5p/3p in mice transgenic models [107,108][43][44]. In human cancer, miR-9-5p displayed a positive correlation with MYCN amplification and tumor grade neuroblastoma, and was elevated in patients with metastatic BRCA. In BRCA cells, 3′-UTR of cadherin 1 (CDH1) was directly targeted by miR-9-5p. CDH1 suppression derepressed β-catenin pathways and induced vascular endothelial growth factor A (VEGFA) expression levels, angiogenesis, mesenchymal transitions, and metastatic phenotypes in BRCA [84][37]. The pairing strand, viz., tumor suppressive miR-9-3p, was screened out from a miRNA mimics library for a possible synthetic sensitizer of MEK inhibitor, viz., AZD6244 in BRCA cell lines. In the presence of miR-9-3p, AZD6244-treated BRCA cells were subjected to vulnerable growth arrest, migration, and inhibition of invasion, which was mediated by miR-9-3p repression of integrin subunit beta 1 (ITGB1) [85][38]. With regards to pre-miR-10b, oncogenic miR-10b-5p was highly expressed in metastatic BRCA, and its upregulation in BRCA cell lines was competent enough to increase the cell motility and invasiveness in vitro and elicit tumor invasion and distant metastasis in vivo. The increase of miR-10b-5p was induced by TWIST1-binding to E-box located at the upstream of MIR10b stem-loop sequences, and the function was executed by direct targeting of 3′-UTR of homeoboxD10 (HOXD10). This is an established tumor suppressor system which impairs the migrating and invading abilities of BRCA cells in vivo [86,109][39][45]. The decreased miR-10b-3p expression was identified in BRCA tumor samples compared with matched peritumor tissue samples, indicating that the loss-of-function may contribute to tumorigenesis. In BRCA cell lines, ectopic introduction of miR-10b-3p inhibited cell viability in vitro and tumor growth in xenograft models. The possible molecular mechanism is the direct repression of the regulatory genes of cell cycle, viz., cyclin A2 (CCNA2), polo like kinase 1 (PLK1) and BUB1, whose expression levels were the most negatively correlated with miR-10b-3p in BRCA samples. The restoration of miR-10b-3p in BRCA cell lines increased subG1- and decreased S-phase cell populations by inhibiting dysregulated cell cycle-associated pathways, and all the three validated targets were associated with poor outcome in BRCA patients [87][40]. In contrast to the above-mentioned oncogenic 5′ arm-derived miRNA, tumor suppressive miR-221-5p were significantly downregulated in metastatic CRC cells, and its downregulation was associated with poor survival of CRC patients belonging to all the stages. Upregulation of miR-221-5p reduced the subcutaneous and orthotopic CRC tumor volumes in mice, and impaired migratory and invasive capability of CRC cells in vitro. Methyl-CpG binding domain protein 2 (MBD2) contains a validated binding site for miR-221-5p, and its knockdown recaptured the phenotypic change of miR-221-5p introduction in CRC cells [88][41]. The oncogenic strand of pre-miR-221 in human CRC, miR-221-3p, was preferentially overexpressed in CRC stem cell-like cells, and its overexpression correlated with the reduced survivability in CRC patients. Overexpressed miR-221-3p promoted the three-dimensional (3D) organoid-forming capacity of human CRC cells in vitro, and miR-221-3p antagonization repressed in vivo engraftment and growth of human CRC patient-derived xenografts (PDX) in mice models. QKI-5 was experimentally validated to be the functional target of miR-221-3p, which impeded the 3D-organoid formation and tumorigenic capability of human CRC PDX cells [89][42]. In certain cellular backgrounds, therapeutic manipulation of ambivalent pre-miRNAs may be infeasible as the expression of the dual sister strands are inevitably interfered simultaneously and unidirectionally, which leads to transcriptional repression of oncogenes as well as tumor suppressors.
3. Conclusion and Future Perspective
Without any doubt, miRNA species have grown to be one of the most important regulatory molecules in maintaining intracellular hemostasis. The evolving knowledge about the biogenesis and function of miRNAs keeps illuminating miRNA biology through unprecedented depth and breadth. The early stage of functional evaluation of miRNA species is focused mostly on the guide strands of miRNA duplex, and the passenger strand is considered to be nonsense and degraded after the guide strand has been loaded into the RISC. With the increase in evidence concerning substantial regulatory activities of miRNA*, the nonsense tag of passenger strand has been gradually torn off. As a consequence, some of the currently available mature miRNA-targeting strategies have been deemed less rigorous in cancer research due to the influence of nonvoluntary negligence in miRNA* expressions. Reviewing the various biogenesis pathways leading to functional miRNAs, impertinence with respect to these inappropriately applied strategies converge on the attempt to manipulate a single miRNA at premature stages. This should be avoided such that simultaneously altered dual strands could either mitigate or exaggerate the performance of the target miRNA-mediated phenotype. One of the major advantages of premature miRNA manipulation rises from the lesson of utilizing such methodology in single miRNA manipulation. If the dual sister strands function synergistically, it would not be more appropriate to use the same strategies for exaggerating the therapeutic performance. The theory of “target-two-sets-of-genes-with-one-pre-miRNA” has been proposed. Currently, only a very small portion of studies are dedicated to establishing the functional relevance of dual sister strands in one study. Functional annotation of a pre-miRNA based on multiple separated studies might face a conflicted situation that ambivalent functions of one of the mature strands are reported. A problem is that the investigations of dual sister strands of a miRNA precursor are mostly not launched in the same lab at the same period of time. It is inevitable that a contradicted phenotype occurs due to the different experimental conditions. However, it can be anticipated that in company with the developing understanding of miRNA* species, functional annotation of pre-miRNA will be taken into account, and annotated pre-miRNA-based therapeutic will sprout with less controversy. Theoretically, a pre-miRNA should be functionally annotated in a single study to rule out the experimental bias, and ideally, phenotypical profiling should be performed before therapeutic interventions. The phenotypic profiling of dual regulatory strands may grant superior performance to the therapeutic interventions on certain premature miRNA, which acts as a “one-two punch” strategy against malignancies. The functional annotation of a miRNA precursor will be generously aided by the availability of annotated miRNA species. WResearchers envision that in the upcoming future, there will be a significant decrease in the inappropriate utilization of premature miRNA inventions on single miRNA manipulations and generous increase in the number of functional miRNA precursor annotations. Being an almost indispensable step of miRNA maturation, the miRNA precursor may deserve a functional tag in the future, acting as a molecular target instead of a mere carrier of regulatory miRNA/miRNA*.
References
- Friedman, R.C.; Farh, K.K.H.; Burge, C.B.; Bartel, D.P. Most mammalian mRNAs are conserved targets of microRNAs. Genome Res. 2009, 19, 92–105.
- Huntzinger, E.; Izaurralde, E. Gene silencing by microRNAs: Contributions of translational repression and mRNA decay. Nat. Rev. Genet. 2011, 12, 99–110.
- Iwakawa, H.; Tomari, Y. The functions of MicroRNAs: mRNA decay and translational repression. Trends Cell Biol. 2015, 25, 651–665.
- Guo, H.; Ingolia, N.T.; Weissman, J.S.; Bartel, D.P. Mammalian microRNAs predominantly act to decrease target mRNA levels. Nature 2010, 466, 835–840.
- Jonas, S.; Izaurralde, E. Towards a molecular understanding of microRNA-mediated gene silencing. Nat. Rev. Genet. 2015, 16, 421–433.
- He, L.; Hannon, G.J. MicroRNAs: Small RNAs with a big role in gene regulation. Nat. Rev. Genet. 2004, 5, 522–531.
- Ebert, M.S.; Sharp, P.A. Roles for MicroRNAs in conferring robustness to biological processes. Cell 2012, 149, 515–524.
- Chen, X.; Liang, H.; Zhang, J.; Zen, K.; Zhang, C.Y. Secreted microRNAs: A new form of intercellular communication. Trends Cell Biol. 2012, 22, 125–132.
- Kosik, K.S. The neuronal microRNA system. Nat. Rev. Neurosci. 2006, 7, 911–920.
- Small, E.M.; Olson, E.N. Pervasive roles of microRNAs in cardiovascular biology. Nature 2011, 469, 336–342.
- Rottiers, V.; Näär, A.M. MicroRNAs in metabolism and metabolic disorders. Nat. Rev. Mol. Cell Biol. 2012, 13, 239–251.
- O’Connell, R.M.; Rao, D.S.; Chaudhuri, A.A.; Baltimore, D. Physiological and pathological roles for microRNAs in the immune system. Nat. Rev. Immunol. 2010, 10, 111–122.
- Berindan-Neagoe, I.; Monroig, P.D.C.; Pasculli, B.; Calin, G.A. MicroRNAome genome: A treasure for cancer diagnosis and therapy. CA Cancer J. Clin. 2014, 64, 311–336.
- Rupaimoole, R.; Slack, F.J. MicroRNA therapeutics: Towards a new era for the management of cancer and other diseases. Nat. Rev. Drug Discov. 2017, 16, 203–221.
- Kozomara, A.; Birgaoanu, M.; Griffiths-Jones, S. miRBase: From microRNA sequences to function. Nucleic Acids Res. 2019, 47, D155–D162.
- Yang, X.; Du, W.W.; Li, H.; Liu, F.; Khorshidi, A.; Rutnam, Z.J.; Yang, B.B. Both mature miR-17-5p and passenger strand miR-17-3p target TIMP3 and induce prostate tumor growth and invasion. Nucleic Acids Res. 2013, 41, 9688–9704.
- Shan, S.W.; Fang, L.; Shatseva, T.; Rutnam, Z.J.; Yang, X.; Du, W.; Lu, W.-Y.; Xuan, J.W.; Deng, Z.; Yang, B.B. Mature miR-17-5p and passenger miR-17-3p induce hepatocellular carcinoma by targeting PTEN, GalNT7 and vimentin in different signal pathways. J. Cell Sci. 2013, 126, 1517–1530.
- Chen, Y.; Lu, Y.; Ren, Y.; Yuan, J.; Zhang, N.; Kimball, H.; Zhou, L.; Yang, M. Starvation-induced suppression of DAZAP1 by miR-10b integrates splicing control into TSC2-regulated oncogenic autophagy in esophageal squamous cell carcinoma. Theranostics 2020, 10, 4983–4996.
- Zhang, Q.; Zhang, J.; Fu, Z.; Dong, L.; Tang, Y.; Xu, C.; Wang, H.; Zhang, T.; Wu, Y.; Dong, C.; et al. Hypoxia-induced microRNA-10b-3p promotes esophageal squamous cell carcinoma growth and metastasis by targeting TSGA10. Aging 2019, 11, 10374–10384.
- Hirata, Y.; Murai, N.; Yanaihara, N.; Saito, M.; Saito, M.; Urashima, M.; Murakami, Y.; Matsufuji, S.; Okamoto, A. MicroRNA-21 is a candidate driver gene for 17q23-25 amplification in ovarian clear cell carcinoma. BMC Cancer 2014, 14, 799.
- Lou, Y.; Yang, X.; Wang, F.; Cui, Z.; Huang, Y. MicroRNA-21 promotes the cell proliferation, invasion and migration abilities in ovarian epithelial carcinomas through inhibiting the expression of PTEN protein. Int. J. Mol. Med. 2010, 26, 521–527.
- Pink, R.C.; Samuel, P.; Massa, D.; Caley, D.P.; Brooks, S.A.; Carter, D.R.F. The passenger strand, miR-21-3p, plays a role in mediating cisplatin resistance in ovarian cancer cells. Gynecol. Oncol. 2015, 137, 143–151.
- Shao, N.; Ma, G.; Zhang, J.; Zhu, W. MIR-221-5p enhances cell proliferation and metastasis through post-transcriptional regulation of SOCS1 in human prostate cancer. BMC Urol. 2018, 18, 1–9.
- Sun, T.; Wang, X.; He, H.H.; Sweeney, C.J.; Liu, S.X.; Brown, M.; Balk, S.; Lee, G.S.; Kantoff, P.W. MiR-221 promotes the development of androgen independence in prostate cancer cells via downregulation of HECTD2 and RAB1A. Oncogene 2014, 33, 2790–2800.
- Chan, J.K.; Blansit, K.; Kiet, T.; Sherman, A.; Wong, G.; Earle, C.; Bourguignon, L.Y.W. The inhibition of miR-21 promotes apoptosis and chemosensitivity in ovarian cancer. Gynecol. Oncol. 2014, 132, 739–744.
- Javanmardi, S.; Tamaddon, A.M.; Aghamaali, M.R.; Ghahramani, L.; Abolmaali, S.S. Redox-sensitive, PEG-shielded carboxymethyl PEI nanogels silencing MicroRNA-21, sensitizes resistant ovarian cancer cells to cisplatin. Asian J. Pharm. Sci. 2020, 15, 69–82.
- Echevarría-Vargas, I.M.; Valiyeva, F.; Vivas-Mejía, P.E. Upregulation of miR-21 in cisplatin resistant ovarian cancer via JNK-1/c-Jun pathway. PLoS ONE 2014, 9.
- Zhang, X.Y.; Li, Y.F.; Ma, H.; Gao, Y.H. Regulation of MYB mediated cisplatin resistance of ovarian cancer cells involves miR-21-wnt signaling axis. Sci. Rep. 2020, 10, 1–8.
- Sun, T.; Wang, Q.; Balk, S.; Brown, M.; Lee, G.S.M.; Kantoff, P. The role of microrna-221 and microrna-222 in Androgen- independent prostate cancer cell lines. Cancer Res. 2009, 69, 3356–3363.
- Zhang, Y.; Yang, P.; Sun, T.; Li, D.; Xu, X.; Rui, Y.; Li, C.; Chong, M.; Ibrahim, T.; Mercatali, L.; et al. MiR-126 and miR-126 * repress recruitment of mesenchymal stem cells and inflammatory monocytes to inhibit breast cancer metastasis. Nat. Cell Biol. 2013, 15, 284–294.
- Uchida, A.; Seki, N.; Mizuno, K.; Misono, S.; Yamada, Y.; Kikkawa, N.; Sanada, H.; Kumamoto, T.; Suetsugu, T.; Inoue, H. Involvement of dual-strand of the miR-144 duplex and their targets in the pathogenesis of lung squamous cell carcinoma. Cancer Sci. 2019, 110, 420–432.
- Zhao, K.; Wang, Q.; Wang, Y.; Huang, K.; Yang, C.; Li, Y.; Yi, K.; Kang, C. EGFR/c-myc axis regulates TGFβ/Hippo/Notch pathway via epigenetic silencing miR-524 in gliomas. Cancer Lett. 2017, 406, 12–21.
- Li, X.; Liu, Y.; Granberg, K.J.; Wang, Q.; Moore, L.M.; Ji, P.; Gumin, J.; Sulman, E.P.; Calin, G.A.; Haapasalo, H.; et al. Two mature products of MIR-491 coordinate to suppress key cancer hallmarks in glioblastoma. Oncogene 2015, 34, 1619–1628.
- Verhaak, R.G.W.; Hoadley, K.A.; Purdom, E.; Wang, V.; Qi, Y.; Wilkerson, M.D.; Miller, C.R.; Ding, L.; Golub, T.; Mesirov, J.P.; et al. Integrated genomic analysis identifies clinically relevant subtypes of glioblastoma characterized by abnormalities in PDGFRA, IDH1, EGFR, and NF1. Cancer Cell 2010, 17, 98–110.
- Liu, C.J.; Tsai, M.M.; Hung, P.S.; Kao, S.Y.; Liu, T.Y.; Wu, K.J.; Chiou, S.H.; Lin, S.C.; Chang, K.W. miR-31 ablates expression of the HIF regulatory factor FIH to activate the HIF pathway in head and neck carcinoma. Cancer Res. 2010, 70, 1635–1644.
- Chang, K.W.; Kao, S.Y.; Wu, Y.H.; Tsai, M.M.; Tu, H.F.; Liu, C.J.; Lui, M.T.; Lin, S.C. Passenger strand miRNA miR-31 regulates the phenotypes of oral cancer cells by targeting RhoA. Oral Oncol. 2013, 49, 27–33.
- Ma, L.; Young, J.; Prabhala, H.; Pan, E.; Mestdagh, P.; Muth, D.; Teruya-Feldstein, J.; Reinhardt, F.; Onder, T.T.; Valastyan, S.; et al. MiR-9, a MYC/MYCN-activated microRNA, regulates E-cadherin and cancer metastasis. Nat. Cell Biol. 2010, 12, 247–256.
- Zawistowski, J.S.; Nakamura, K.; Parker, J.S.; Granger, D.A.; Golitz, B.T.; Johnson, G.L. MicroRNA 9-3p targets 1 integrin to sensitize claudin-low breast cancer cells to MEK inhibition. Mol. Cell. Biol. 2013, 33, 2260–2274.
- Ma, L.; Teruya-Feldstein, J.; Weinberg, R.A. Tumour invasion and metastasis initiated by microRNA-10b in breast cancer. Nature 2007, 449, 682–688.
- Biagioni, F.; Bossel Ben-Moshe, N.; Fontemaggi, G.; Canu, V.; Mori, F.; Antoniani, B.; Di Benedetto, A.; Santoro, R.; Germoni, S.; De Angelis, F.; et al. miR-10b *, a master inhibitor of the cell cycle, is down-regulated in human breast tumours. EMBO Mol. Med. 2012, 4, 1214–1229.
- Yuan, K.; Xie, K.; Fox, J.; Zeng, H.; Gao, H.; Huang, C.; Wu, M. Decreased levels of mir-224 and the passenger strand of miR-221 increase MBD2, suppressing maspin and promoting colorectal tumor growth and metastasis in mice. Gastroenterology 2013, 145, 853–864.e9.
- Mukohyama, J.; Isobe, T.; Hu, Q.; Hayashi, T.; Watanabe, T.; Maeda, M.; Yanagi, H.; Qian, X.; Yamashita, K.; Minami, H.; et al. MiR-221 targets QKI to enhance the tumorigenic capacity of human colorectal cancer stem cells. Cancer Res. 2019, 79, 5151–5158.
- Iorio, M.V.; Ferracin, M.; Liu, C.G.; Veronese, A.; Spizzo, R.; Sabbioni, S.; Magri, E.; Pedriali, M.; Fabbri, M.; Campiglio, M.; et al. MicroRNA gene expression deregulation in human breast cancer. Cancer Res. 2005, 65, 7065–7070.
- Sun, Y.; Wu, J.; Wu, S.H.; Thakur, A.; Bollig, A.; Huang, Y.; Joshua Liao, D. Expression profile of microRNAs in c-Myc induced mouse mammary tumors. Breast Cancer Res. Treat. 2009, 118, 185–196.
- Carrio, M.; Arderiu, G.; Myers, C.; Boudreau, N.J. Homeobox D10 induces phenotypic reversion of breast tumor cells in a three-dimensional culture model. Cancer Res. 2005, 65, 7177–7185.
