Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 1 by Sergey Sedykh and Version 2 by Yvaine Wei.
The structure of HIV-envelope changes as the infection progresses and is one of the biggest obstacles in developing of HIV-vaccine. HIV-infection can cause the production of various natural autoantibodies, including catalytic antibodies hydrolyzing DNA, myelin basic protein, histones, HIV-integrase, HIV-reverse transcriptase, β-casein, serum albumin, and some other natural substrates. Currently, there are various directions for the development of HIV vaccines: stimulation of the immune response on the mucous membranes; induction of cytotoxic T cells, which lyse infected cells and hold back HIV-infection; immunization with recombinant Env proteins or vectors encoding Env; mRNA-based vaccines and some others.
- HIV
- antibodies
- IgG
- neutralizing antibodies
- vaccine
- HIV-vaccine
- catalytic antibodies
- COVID-19
1. Introduction
Since the first clinical detection of AIDS (acquired immunodeficiency syndrome) and the subsequent isolation of HIV (human immunodeficiency virus) in the early 1980s, the HIV epidemic continues to be one of the major health threats in the world, despite four decades of intensive research [1].
A high level of genetic variability in HIV is one of the biggest obstacles in developing a safe and effective vaccine. Unlike many other viruses, HIV is highly variable, with many subtypes and recombinant forms [2][3][2,3]. Low fidelity of HIV reverse transcriptase leads to the rapid generation of mutants carrying base substitutions, insertions, and deletions. Combined with the addition and loss of glycosylation sites, this results in tremendous viral diversity [4][5][6][4,5,6]. A key source of genetic diversity is the viral gene env, which encodes envelope glycoprotein (Env), gp120, and gp41. As is known, gp120 and gp41 subunits on the surface of the virion are trimerized, form spikes, and together are involved in penetration into the target cell. Env has a complex conformation and undergoes significant rearrangements when binding to CD4 and coreceptors [7][8][9][7,8,9]. Glycans make up half the mass of the HIV envelope glycoprotein, forming a topographic landscape that alters the availability of antibody (Ab) binding to the immunogen [10].
Env is also a major target for neutralizing antibodies (nAbs) [11][12][11,12]. The inability of the adaptive immune system to prevent and control infection is explained by the structural variability of the HIV envelope. Protein gp120 consists of five relatively constant (C) regions and five highly variable (V) regions. Most adaptive responses target V-domain epitopes that mutate rapidly [13]. Cytotoxic T-lymphocytes and nAbs provide only temporary protection [14][15][14,15].
Progression of HIV-infection to AIDS occurs over 1 year to more than 2 decades [16]. Some HIV-infected patients develop Abs that can neutralize a wide range of HIV strains [17][18][19][17,18,19]. HIV has mechanisms preventing the production of broadly neutralizing antibodies (bNAbs) (Figure 1), including intense glycosylation of Env glycoproteins, instability of these glycoproteins, and conformational masking of receptor binding sites [20][21][22][20,21,22].
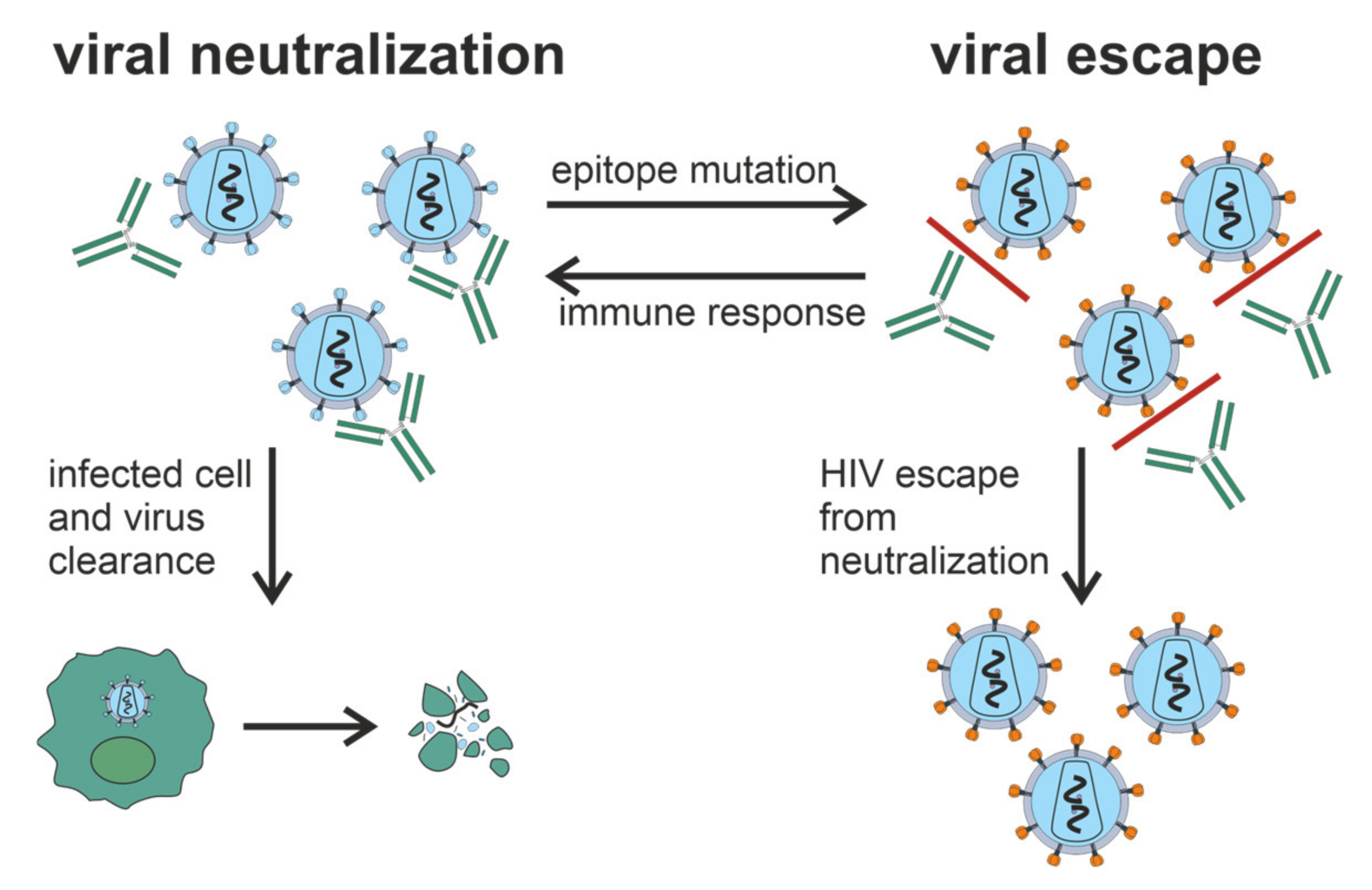

Figure 1. Generation of bNAbs, which neutralize the virus through binding to viral spikes and blocking the entry of the virus into sensitive cells such as CD4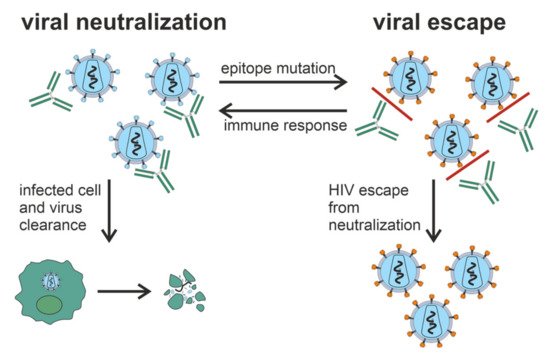

Figure 1. Generation of bNAbs, which neutralize the virus through binding to viral spikes and blocking the entry of the virus into sensitive cells such as CD4
+ T cells, is related to the evasion of virus from the immune response due to the low precision of HIV reverse transcriptase and rapid generation of viral mutants. Glycan shield, immunodominant variable loops, and conformational masking of key viral epitopes protect Env from Ab-response.
T cells, is related to the evasion of virus from the immune response due to the low precision of HIV reverse transcriptase and rapid generation of viral mutants. Glycan shield, immunodominant variable loops, and conformational masking of key viral epitopes protect Env from Ab-response.
Despite the enormous diversity of HIV, there are described a few bNAbs such as b12 [23][24][23,24], 2G12 [24][25][24,25], 4E10 [26], 2F5 [24][27][24,27], VRC01 [28], PG9/PG16, 447-52D [29]. As shown in [30][31][32][30,31,32], b12 Ab isolated from the phage display library can neutralize about 40% of known HIV strains. Another monoclonal antibody (mAb) HJ16, also neutralizes about 40% of viral isolates [33]. Most of the structures of such monoclonal bNAbs are resolved in complexes with Env [8][34][8,34]. These structures can be used to develop immunogens capable of eliciting bNAbs.
Although the detection of circulating autoantibodies (autoAbs) to self-antigens in patients infected by HIV or other viruses does not necessarily reflect the presence of an autoimmune disease, such autoAbs can certainly complicate viral infections. These responses may vary due to the infection of autologous cells by the virus and their consequent targeting by pre-existing and/or induced autoAbs against the virus. Several natural autoAbs were described in HIV-infection [35]: Abs against small nuclear ribonucleoproteins (snRNP) [36], Abs to anticardiolipin (aCL) and antiglycoprotein 1 (aβ2GP1), anti-DNA, and antinuclear Abs [37][38][37,38]. Common to some viral diseases and autoimmune pathologies, catalytic Abs (Abzymes) hydrolyzing DNA [39] and a variety of proteins were described in HIV/AIDS patients: histones [40], myelin basic protein (MBP) [41], HIV integrase [42][43][42,43] and HIV reverse transcriptase [39], β-casein and serum albumin [39]. Such autoAbs may be beneficial for patients, as they expel autologous cells expressing viral antigens from the body. Moreover, one cannot exclude that HIV-infected patients produce natural Abs that recognize both microbial and autologous antigens.
Several vaccines in preclinical tests consisted of single coat proteins or in combination with other HIV proteins and multiepitope synthetic peptides and polypeptides expressed by noninfectious vectors. Numerous HIV vaccine candidates are directed to the induction of nAbs and/or cytotoxic T cells [44]. Despite the induction of robust immune responses, the recombinant glycoprotein gp120 VaxGen [45] and Merck gag/pol/nef (STEP) adenoviral vaccine [46] did not reduce the risk of infection.
2. Neutralizing Antibodies in HIV-Infection
Neutralizing Abs protect cells from a viral particle by binding it. These Abs surround the virus, after which the entire complex containing the virus is removed with the immune system. The induction of nAbs is a key goal of vaccination strategies [47][48][54,55]. During HIV-infection, almost all patients produce Abs to Env, but only a small fraction of these Abs can neutralize the virus [49][50][56,57]. Interestingly, nAbs against viral Env are produced within the first weeks of infection. Still, this early Ab response targets an autologous virus that circulates within each person and is ineffective against heterologous (unrelated) viruses [5]. Among the nAbs, a specific group of Abs neutralizing a wide spectrum of HIV strains are distinguished. These broadly neutralizing antibodies (bNAbs) arise after several years of virus–antibody coevolution in infected patients [51][52][53][58,59,60]; bNAbs usually recognize conserved epitopes on the highly glycosylated envelope glycoprotein (Env) [54][55][61,62].2.1. General Characteristics of nAbs
The first B cell response to HIV-infection appears within 8 days after detectable viremia [56][71]. After the next 5 days, circulating Abs against gp41 are detected, and after another 2 weeks, Abs against gp120 are detected, which primarily target the V3 loop. Autologous nAbs are developed over several months [57][70] and target variable HIV regions by potent but particular molecules [5][58][59][5,72,73]. Over the years, Abs with neutralizing cross-specific potential developed in one third of chronically infected patients and targeted the more conserved regions of Env [60][63]. Heterologous nAbs appear in some patients 1 year after infection reach their peak after 4 years, with no increase after that [19]. High viral load is not a general predictor of Ab neutralization capacity since some patients with high viremia do not develop cross-neutralizing Abs [19][61][19,74]. The breadth of Ab neutralization is usually increased to the end of 2.5 years after infection [62][75]. In the long-term nonprogressor patients, all neutralizing Abs against gp120 were clonally associated with the antibody-diversifying process of somatic hypermutation [63][76]. B cells producing bNAbs have been shown to undergo several rounds of affinity maturation in germinal centers to achieve cross-neutralizing activity [64][65][77,78]. BNAbs develop over time and are maintained by chronic antigen exposure.2.2. Antibodies against Env Epitopes
Several papers are devoted to mapping the Ab-specificity responsible for cross-neutralizing activity [66][67][68][84,85,86]. These nAbs recognize epitopes of monomeric gp120; in some cases, cross-neutralizing activity can be attributed to Abs recognizing linear epitopes in the membrane-proximal outer region (MPER) of gp41 [69][70][87,88]. It has been shown that the quaternary epitope at the end of the trimeric envelope structure, including loops V2 and V3, is often the target of cross-neutralizing Abs [17][32][71][17,32,89]. Figure 2 shows vulnerable targets of HIV: nAbs react with the HIV Env spike, which is composed of three highly glycosylated gp120 molecules, each noncovalently linked to a transmembrane gp41 molecule. To initiate entry of the virus into cells, gp120 binds to the CD4 receptor on the cell surface [72][90]. One of the structurally conserved regions of Env among the various isolates is the CD4 binding site on gp120, which is required for binding with the CD4 receptor on the surface of immune cells, such as T helper cells and monocytes. The second structurally conserved region of Env is the coreceptor binding site of gp120. Both the CD4 binding site and the coreceptor binding site are immunogenic, and numerous mAbs against these Env regions have been isolated from HIV-infected patients. However, most human mAbs against these regions cannot bind the native viral Env trimer and, therefore, cannot neutralize HIV [60][63].
Figure 2. Vulnerable targets on trimeric glycoproteins of HIV spike (gp120 and gp41). Widely neutralizing Abs target the CD4 binding site on gp120, the proximal outer membrane region of gp41, the glycan shield, and epitopes in variable loops 1, 2, and 3 on gp120.
Figure 2. Vulnerable targets on trimeric glycoproteins of HIV spike (gp120 and gp41). Widely neutralizing Abs target the CD4 binding site on gp120, the proximal outer membrane region of gp41, the glycan shield, and epitopes in variable loops 1, 2, and 3 on gp120.
2.3. Hypervariable Domains gp120
HIV gp120 contains five “hypervariable” domains. The first two domains (V1V2) include dramatic insertions and deletions and different glycosylation patterns [73][95]. The V1V2 region regulates the neutralization sensitivity of conserved epitopes such as the coreceptor binding site [8][74][75][8,96,97]. Among viruses of the B subtype, a high ratio of nonsynonymous and synonymous substitutions is characteristic of the V3 region, while in the C subtype, this region remains relatively conserved [76][98]. Abs against V3 play a minimal role in neutralizing primary viruses [31][77][31,99] because the V3 loop is clogged with trimeric Env [75][78][79][97,100,101]. In contrast to V1V2, the role of V4 and V5 in neutralization resistance is unclear, although these regions affect Env conformation and glycan packaging, thereby sterically limiting the availability of neutralization determinants [5][80][81][5,102,103].
3. Autoantibodies in HIV-infection
Autoantibodies can interact with antigens specific to their organism. Pathogenesis of HIV/AIDS involves several factors, not a single virus-controlled destruction of CD4+ T cells [82][104]; among various pathogenic mechanisms there is autoimmunity [35] directed against lymphocytes [83][84][85][105,106,107], platelets [86][87][108,109] and peripheral nerves [88][89][110,111]. Up to 40% of HIV-infected patients are positive for autoAbs against red blood cells [90][91][112,113], which are involved in HIV-associated autoimmune hemolytic anemia [92][93][114,115]. Circulating Abs to small nuclear ribonucleoproteins are presented in the serum of HIV-infected patients [36]. Blood serum of HIV-infected patients contains Abs against anticardiolipin (aCL) and antiglycoprotein 1 (aβ2GP1), anti-DNA, and antinuclear Abs [37][38][37,38]. Exact frequency of autoimmune manifestations and their pathogenesis during HIV-infection remain unknown. Still, it was shown that anti-aCL Abs among HIV-infected patients is much higher than among patients with autoimmune diseases [94][116]. Content of these and similar Abs among HIV-infected patients before antiretroviral therapy ranges from 36% to 67% [37][95][37,117]. The presence of anti-aCL Abs in blood serum is not associated with any clinical manifestations of antiphospholipid syndrome, and it is not easy to explain why [96][118]. Moreover, spontaneous control of HIV replication or subsequent after antiretroviral therapy is associated with lower production of anti-aCL Abs [97][98][119,120].4. Catalytic Antibodies in HIV/AIDS
Autoimmune reactions and the appearance of autoAbs in HIV/AIDS and some other viral infections may be associated with the activation of polyclonal B cells, molecular mimicry between viral and/or microbial antigens and host antigens [99][100][126,127], abnormal expression of immunoregulatory molecules, and anti-idiotypic network [38][101][38,128]. One can propose several physiological functions of the hydrolysis of DNA, histones, and MBP by Abs. ItWe is ssuggested that the most relevant function of such catalytic Abs is the elimination of autoantigens formed due to the destruction of infected cells from the blood. However, itwe cannot be eexcluded the possibility that such autoAbs may also explain some of the observed autoimmune pathologies found in patients with HIV/AIDS. Activation of B-lymphocytes in HIV-infected patients leads to the production of Abs to viral components and autoAbs to various components of human cells [38]. It has been shown that IgG and/or IgM of patients with AIDS hydrolyze not only autoantigens as DNA [102][129], MBP [41], histones [40][103][104][105][40,130,131,132], but also viral enzymes HIV integrase [42][43][106][107][42,43,133,134] and HIV reverse transcriptase [39] and corresponding peptides. Interestingly, IgG of HIV-infected patients efficiently cleaves five human histones (H1 [41][103][41,130], H2a, H2b [104][131], H3, and H4 [105][132]).4.1. Catalytic Antibodies Cleave gp120
Hydrolysis of gp120 is possessed by V domains of the heavy (VH) and light (VL) chains of natural IgG, IgA, and IgM [108][109][135,136]. A rare light chain of Abs, isolated by phage display, can bind and hydrolyze gp120 independently of the heavy chain [110][137]. The most described Abs catalytic sites are located in the VL domains [111][138]. Long-term HIV-infection for 5 years is associated with a modest increase in catalytic IgA against gp120 [109][136]. No accumulation of such Abs was observed at the earlier stages of disease (6 months) [112][139].4.2. Antibodies Hydrolyze HIV Integrase, Reverse Transcriptase, and Corresponding Oligopeptides
IgG and IgM were isolated from the blood serum of HIV-infected patients and subsequently separated on columns with immobilized reverse transcriptase or integrase, respectively, specifically hydrolyzed only recombinant viral reverse transcriptase [39] or integrase [42][43][42,43]. Abs that specifically hydrolyze these HIV enzymes were of potential interest in developing new anti-HIV-drugs for many years. Nearly 40 sites of HIV integrase cleavage by Abs isolated from HIV-infected patients were found. Most sites are located in seven known immunodominant integrase sequences [106][133]. Two 20 mer oligopeptides corresponding to immunogenic integrase sequences contained 9 to 10 clusters of major, moderate, and minor cleavage sites [106][113][133,143]. Each individual Ab preparation from HIV-infected patients had a distinct ratio of hydrolysis sites [106][133].4.3. Antibodies Hydrolyze DNA, Histones, and Myelin Basic Protein
Relative DNase activity of Abs isolated from the blood serum of HIV-infected donors varies significantly from patient to patient, but 96% of the preparations show a detectable level of DNase activity [102][129], which is an intrinsic feature of Abs [39]. Abs against H1, H3, H4, and MBP possess enzymatic cross-reactivity [114][151]: anti-histone H1 IgG hydrolyze H1 as well as MBP, and vice versa, anti-MBP Abs hydrolyze this H1 histone [41]. Similarly, IgG against H2a and H2b histones efficiently hydrolyze these histones and MBP, and anti-MBP Abs cleave MBP, H2a, and H2b histones, but not other control proteins [114][151]. Near 100% of IgG of HIV-infected patients effectively hydrolyze between one and five human histones [40].5. HIV-vaccines: Production of Neutralizing Antibodies
Current HIV prevention and treatment strategies include using antiretroviral drugs for pre-exposure prophylaxis and antiretroviral therapy. This made it possible to transform HIV from a life-threatening disease to a manageable chronic disease [115][156]. However, drugs are expensive, require strict dosing to be effective, and cause side effects. Some HIV-infected patients develop drug resistance; furthermore, drug access remains a significant barrier, especially in low- and middle-income countries. HIV replicates chronically in the host and evades an Ab response, unlike other viral pathogens. Immunity evasion and large genetic variation among HIV strains are significant obstacles to vaccine development. The use of vaccines should provide generation of Abs that can adapt to the Env glycan screen and bind to various viral strains to offer complete protection [116][117][158,159]. An ideal HIV-vaccine would elicit serum Abs that are highly effective and can prevent viral infection at low concentrations and have broad action—making a high percentage of hard-to-neutralize viruses inactive [118][160].5.1. HIV-vaccine Development Strategies
The induction of a protective Ab response remains the main priority in developing HIV-vaccines [119][162]. Unfortunately, most current HIV-vaccine candidates do not elicit nAbs against most circulating viral strains. To replicate chronically in the host, the HIV uses several mechanisms to defend itself against Ab recognition: Env glycoprotein is protected by the shield of glycans. Different variations occur in immunodominant loops, and key viral epitopes are due to the conformational lability [5][21][49][5,21,56] (Figure 3). These defense mechanisms, although very effective, have vulnerabilities; information on the precise location and molecular structure of these vulnerable regions can be useful for the rational design of improved vaccine immunogens.
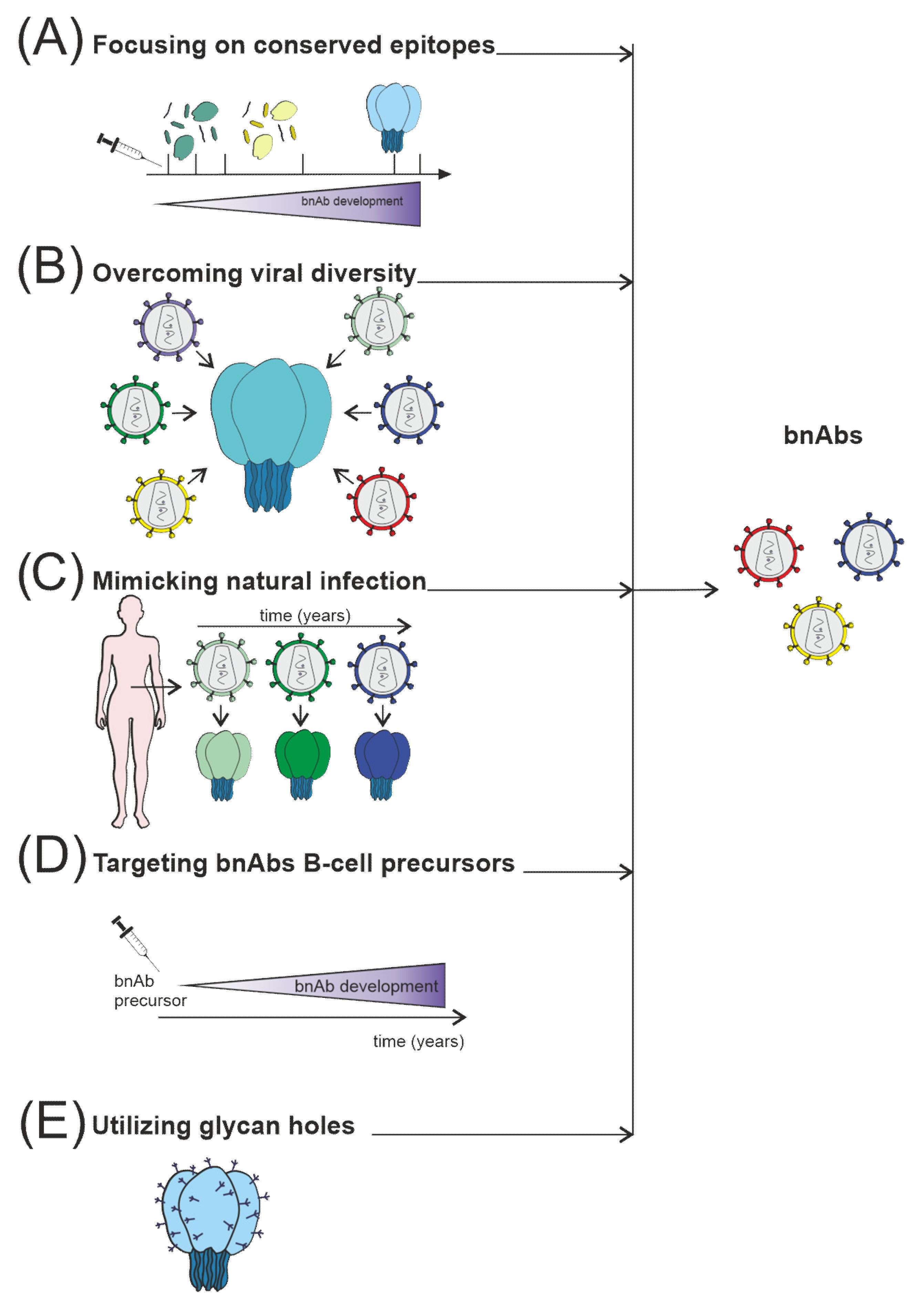
Figure 3. Several strategies are currently used to stimulate nAb responses towards bnAb generation, which consist of (A) directing of Ab response to conserved sites, for example, using fusion peptides; (B) use of mosaic or consensus antigens to overcome the viral diversity of circulating HIV worldwide; (C) clonal immunogens resembling the evolution of the virus in HIV-infected individuals modeling natural infection; (D) targeting immunogens to putative bnAb germline progenitors, followed by immunization leading to the affinity maturation pathway to bnAb developm€; (E) selective removing of glycans to focus Ab responses to a specific site of interest, such as CD4 binding site immunogens.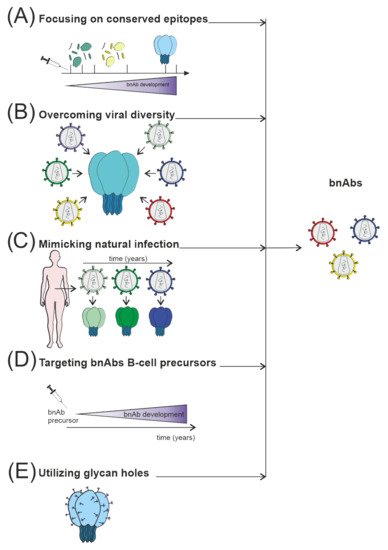

Figure 3. Several strategies are currently used to stimulate nAb responses towards bnAb generation, which consist of (A) directing of Ab response to conserved sites, for example, using fusion peptides; (B) use of mosaic or consensus antigens to overcome the viral diversity of circulating HIV worldwide; (C) clonal immunogens resembling the evolution of the virus in HIV-infected individuals modeling natural infection; (D) targeting immunogens to putative bnAb germline progenitors, followed by immunization leading to the affinity maturation pathway to bnAb developm€; (E) selective removing of glycans to focus Ab responses to a specific site of interest, such as CD4 binding site immunogens.
5.2. Immunogen Structure Design
HIV predominantly induces non-neutralizing or strain-specific Abs during the first months after infection [5][57][5,70]. It was found that approximately 10–20% of HIV-infected patients had bNAbs after a few years of disease [17][60][120][17,63,170], which is the humoral immune response that a vaccine should elicit.
Viral epitopes conserved among most viral strains are more likely to generate cross-reactive Abs. In this regard, research is focused on small numbers of human mAbs isolated from HIV-infected individuals that possess cross-reactive neutralizing activity [49][121][56,81]. However, these conserved viral epitopes are weakly immunogenic or produce Abs with limited reactivity. Minor structural changes may improve specific binding [20][122][20,67]. The crystal structure of b12 mAb bound to the CD4 receptor binding site of the gp120 molecule provides insight into how nAbs gain access to the functionally conserved regions of the Env glycoprotein [34].
