There is already a societal awareness of the growing impact of nanoscience and nanotechnology, with nanomaterials (with at least one dimension less than 100 nm) now incorporated in items as diverse as mobile phones, clothes or dentifrices. In the healthcare area, nanoparticles of biocompatible materials have already been used for cancer treatment or bioimaging enhancement. Nanotechnology in dentistry, or nanodentistry, has already found some developments in dental nanomaterials for caries management, restorative dentistry and orthodontic adhesives.
1. Introduction
Nanoscience and nanotechnology, which deals with science and technology at the nanoscale, have developed in such a way that it has already been used in several areas of knowledge, with social applications and economic impact [1]. From the Internet of Nano Things [2] to health benefits [3], exploiting the nanoworld has made a big impact. This is no different in dentistry, although still in its infancy with respect to other areas of healthcare [4]. The authors of ref. [5] provided a good and updated account of nanomaterials in a diversity of dentistry applications. Among those, dental materials have received increasing attention [6], dental nanomaterials for caries management [7], restorative dentistry [8] and orthodontic adhesives [9]. Additionally, another importance to nanodentistry is silver nanoparticles, due to their antibactericidal properties [10].
2. Nanodentistry
Nanodentistry deals mainly with nanobiomaterials applied to dentistry and is a growing field of research with high potential for clinical translation
[4][5][11][12][13][14][15][16][17][4,5,13,14,15,16,17,18,19]. It is a term also employed when characterization techniques in the nanoscale regime are employed for dental materials and tissues. The so-called dental nanobiomaterials are fabricated by well-developed nanofabrication processes (see more details in ref.
[13][15]). Naturally occurring nanostructures, particularly nanobiostructures present in biotissues, are subject to intense attention from researchers worldwide, not only for imaging but also for seeing those nanostructures in situ and working is out of the scope of the research.
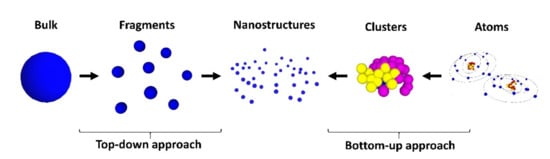
Nanofabrication processes are generally classified by two approaches: the top-down and the bottom-up. As the terms indicate, the former means that, by starting from bulk materials, nanosized structures are formed. Conversely, starting with atoms or molecules, nanostructures can be formed. Figure 1 shows examples of top-down and bottom-up routes, indicating some of the techniques that have been used to achieve nanostructures. From an economic point of view, the bottom-up approach is cheaper than the top-down.
Figure 1. Top-down and bottom-up approaches used in nanofabrication.
As a result of the growth and development of the nanofabrication processes, it is possible nowadays to have access to organic or inorganic nanostructures, which can be dielectric, such as TiO2 or rare-earth-doped materials, semiconducting, as ZnO, or metallic nanoparticles, as Au or Ag, and of different structures and shapes, such as nanoparticles, nanoshells or nanorods, dimensions, and so on. These make the physical properties of these nanomaterials very morphology dependent.
3. From Basics to Applications in Nanodentistry
Optical Coherence Tomography (OCT) exploits the broad spectral width of a low coherence optical source, in combination with a Michelson-type interferometer, to retrieve high resolution (submicrometer) images with low penetration depth (few mm), relying on the interference between the backscattered light from tissue and the reflected beam from the reference arm. The OCT technique was pioneered in 1991 when Huang and co-workers first described a tomographic image of the eye retina
[18][25]. Since then, scientific and technological developments in the OCT technique evolved exponentially, and the reader is referred to the authoritative book by Fujimoto and Drexler
[19][26]. Instead of giving an extensive list of references in OCT and its diversity of applications in medical and non-medical areas, the reader is also referred to the public webpage
www.octnews.org, a trusted and always updated webpage for OCT developments.
Conceptually, OCT is a very simple method: a spectrally broadband optical beam, which can be generated by coherent or incoherent sources and travels through a Michelson-interferometer, with the beam being split to the reference arm and the other arm which has the sample under study. When ultrashort Fourier transformed limited pulses are employed, they are not chirped. Otherwise, the optical sources employed are chirped. The reflected light from the surface and inner parts (including scattered light) of the biological samples thus interferes with the light returned from the reference arm, providing an interference pattern that carries the image information. The spatial resolution is provided in two directions: the axial resolution of the system is determined by the optical source and is inversely proportional to its bandwidth. The lateral resolution depends on the employed focusing optics, either lenses or microscope objectives. A very important point is the role of absorption and scattering. For this reason, an OCT system performs better in the 800 nm–1300 nm spectral window, also known as the NIR-I or NIR-II window
[20][27]. Below 800 nm (towards the visible) absorption may hinder light penetration, and therefore working in the NIR-I and NIR-II allows deeper penetration. Furthermore, tissue scattering is also reduced as the light wavelength increases from the visible to the NIR, which is another advantage. The pioneer OCT systems operated in the so-called temporal or time-domain OCT (TD-OCT), but the technological development led to the exploitation of Fourier-domain methods, which can be spectral-domain OCT (SD-OCT) and swept-source OCT (SS-OCT).
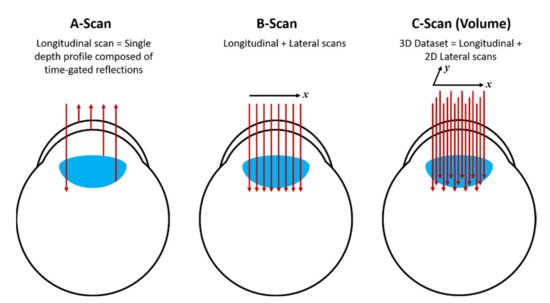
The results obtained by OCT can be displayed by the so-called A-scan, whereby a single depth profile is shown (without lateral displacement of the beam); B-scan, whereby the beam is laterally displaced and the collected data is composed of a set of 2D frames and finally, the C-scan, which provides a 3D image by acquiring a volume dataset of B-scans.
Figure 2 shows diagrammatically each scheme, applied to OCT in the eye as an example. For further details and alternative schemes for OCT, the reader should consult ref.
[19][26].
Figure 2.
Diagram showing the A-scan, B-scan and C-scan obtained from the OCT method.
4. OCT in Nanodentistry: Image Enhancement with Gold Nanoparticles
The applications of OCT in dentistry have been reviewed on a number of occasions
[21][22][23][28,29,30]. Our group in Brazil has employed OCT for dental applications in the areas of cariology, endodontics, periodontology, aesthetics and dental materials, as exemplified by refs.
[24][25][26][27][28][29][30][31][32][33][34][31,32,33,34,35,36,37,38,39,40,41]. OCT in nanodentistry can be seen from two perspectives: (a) the OCT technique operates such as to improve the sensitivity or detect in the nanometer regime or (b) the OCT technique can be employed to study nanostructures in dental materials or dental tissues (hard or soft).
In
[35][36][37][42,43,44], a technique called nanosensitive OCT (nsOCT) was developed and demonstrated, which exploits proper manipulation of the micrometer resolution data from Fourier-domain OCT to resolve the images in the nanoscale (10′s nm) regime and was recently applied to assess wound healing within the cornea
[37][44]. Further, J. Yi and co-workers developed a so-called inverse spectroscopic OCT (ISOCT), which, upon quantification of the wavelength-dependent backscattering coefficient μ
b(λ) and the scattering coefficient μ
s,), quantify the mass-density correlation function to achieve detectable structural changes ranging from ~30 to ~450 nm. The authors validated their findings by numerical simulations, tissue phantom studies, and ex-vivo colon tissue measurements, which were cross-validated by scanning electron microscopy. For further details of both methods, the readers should assess the given references.
From the second perspective, OCT can be employed to assess nanostructures within the sample. A commercially available OCT system was employed. In reference
[38][46], within an international collaborative work, a pioneer application of OCT in nanodentistry was reported, whereby gold nanoparticles (AuNP) were formed in situ and immediately used as a contrast agent for dental OCT. This development was essential to imaging the desired region.
To perform the work, an innovative in situ photothermal reduction procedure was developed and implemented, which allowed the production of spherical AuNP inside dentinal layers and tubules. A three-step procedure was performed: First, gold ions were dispersed in the primer of a commercially available dental bonding system. Secondly, the modified adhesive system was applied to the dentin, and the dental bonding materials were photopolymerized using a commercially available photopolymerizer, simultaneously with the formation of AuNP. The presence of AuNP was confirmed by scanning electron microscopy in the hybrid layer and dentinal tubules. The diameter of the AuNP was determined to be in the range of 40 to 120 nm, while it is known that the dentin tubule’s diameters ranged from 500–4000 nm.