Catalytic oxidation is a key technology for the conversion of petroleum-based feedstocks into useful chemicals (e.g., adipic acid, caprolactam, glycols, acrylates, and vinyl acetate) since this chemical transformation is always involved in synthesis processes. Zeolites are microporous, crystalline aluminosilicate materials known since 1756 when the stilbite structure was identified by the Swedish mineralogist Crönstedt. Zeolites and other related porous materials can be supports for organometallic or metallic active species. These materials are the most studied supports due to their combined properties of mechanical and thermal stability that allows it an easy regeneration and recycling.
- hydrocarbon oxidation reactions
- zeolites
- hierarchical zeolites
- immobilized catalyst
1. Industrial Hydrocarbon Oxidation Reactions
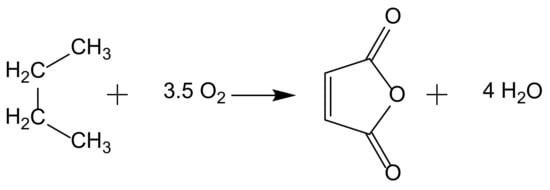
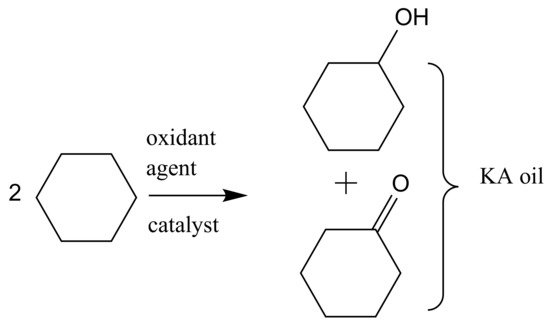
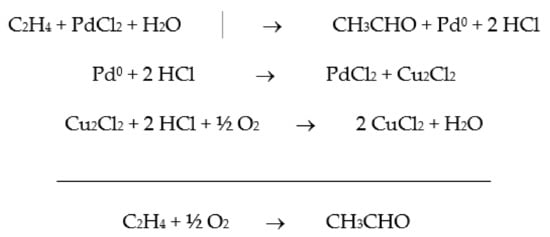
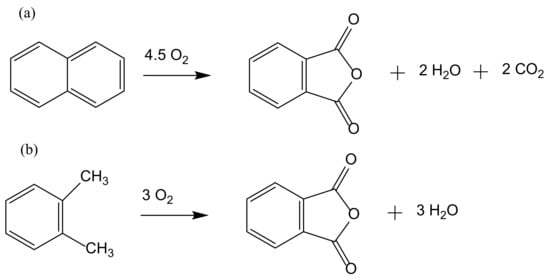
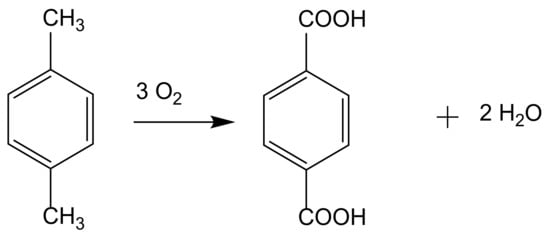
2. From Homogeneous to Heterogenized Catalysts
The development of sustainable methods for the catalytic oxidation reactions of hydrocarbons-alkanes, alkenes, and aromatics is an important scientific challenge with significant technological potential. As mentioned previously, these reactions usually occur in the presence of traditional homogeneous catalysts, such as transition and neat metals or their salts, as well as mineral acids and complexes, due to their high activity and selectivity to the desired products. However, the intensive use of these catalysts is rather controversial due to the difficult separation and recovery of the catalyst from the reaction media. The immobilization of catalytic active species in solid supports is a possible strategy to overcome some of the disadvantages of homogeneous processes. Heterogenized catalysts are easily recovered from the reaction media, without expensive separation processes and large amounts of solvents involved, with the additional advantage of allowing the reuse of the catalyst in several cycles. These are, in fact, the main objectives that one expects to achieve through the immobilization of homogeneous catalysts, but some additional benefits may also be obtained, namely when porous supports are considered. In this case, the confinement effects may enhance the interaction of the substrate with the catalyst. However, the porosity of the support may also impose some diffusional constrains that, especially when voluminous subtracts are considered, can result in an extensive loss of activity. In the case of complexes, the immobilization on solid supports has another additional benefit since it prevents dimerization phenomena that are some of the most common causes of homogeneous catalysts deactivation.
3. Zeolites and Related Materials as Support for the Heterogenization of the Catalysts
3.1. Hierarchical Zeolites
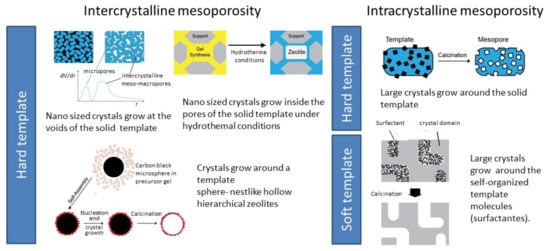
The strictly microporous nature of zeolite structures is responsible for the various types of shape selectivity that are fundamental to increasing the yield of a desired product. A classic example of shape selectivity is an important petrochemical reaction catalyzed by ZSM-5 zeolite (MFI structure): the transformation of m-xylene into o-xylene and, especially, p-xylene, which is the building block to produce polyethylene (PET)-based products. In this case, opposing the thermodynamic equilibrium where the more stable m-xylene is favored, the diffusional limitations for the molecular transport of m-xylene and o-xylene lead to the conversion of these two more voluminous isomers into the most valuable p-xylene [6][7][8,24]. Despite the importance of shape selectivity in several reactions catalyzed by zeolites, their native microporosity can also impose diffusion constraints that will limit the catalytic performance, especially in the presence of bulky molecules. Although a large number of strategies have been proposed and demonstrated, the production of these hierarchical materials can be classified into two major categories: synthesis procedures, also called “bottom-up”, or postsynthesis procedures, also called “top-down”[8][25].3.1.1. Bottom-Up Strategies
To introduce a supplementary pore system, usually mesopores, several strategies involve the addition of hard or soft templates to the synthesis gel, allowing the crystallization of the zeolites around those templates, giving intracrystalline mesoporosity. Alternatively, the crystal can grow at the confined space between particles, originating small crystals where the mesopores appear as the consequence of the particle stacking (intracrystalline mesoporosity), as schematized in Figure 57. In both cases, after synthesis, the templates are removed by combustion, exposing the mesopores.