Non-alcoholic fatty liver disease (NAFLD) is a challenging disease caused by multiple factors, which may partly explain why it still remains an orphan of adequate therapies. It highlights the interaction between oxidative stress (OS) and disturbed lipid metabolism. Several reactive oxygen species generators, including those produced in the gastrointestinal tract, contribute to the lipotoxic hepatic (and extrahepatic) damage by fatty acids and a great variety of their biologically active metabolites in a “multiple parallel-hit model”. This leads to inflammation and fibrogenesis and contributes to NAFLD progression. The alterations of the oxidant/antioxidant balance affect also metabolism-related organelles, leading to lipid peroxidation, mitochondrial dysfunction, and endoplasmic reticulum stress. This OS-induced damage is at least partially counteracted by the physiological antioxidant response. Therefore, modulation of this defense system emerges as an interesting target to prevent NAFLD development and progression. For instance, probiotics, prebiotics, diet, and fecal microbiota transplantation represent new therapeutic approaches targeting the gut microbiota dysbiosis. The OS and its counter-regulation are under the influence of individual genetic and epigenetic factors as well.
1. Introduction
Non-alcoholic fatty liver disease (NAFLD) is the most widespread and emerging chronic liver disease both in Western and developing countries. It has an estimated prevalence of about 23–25% in the general adult population. The burden varies between and within regions, with the highest prevalence in the Middle East (32%) and South America (30%), and the lowest in Africa (13%)
[1]. Additionally, the prevalence rates in North America, Europe, and Asia were reported to be 24.1%, 23.7%, and 27.4%, respectively
[2], and increased up to 55.5% in type 2 diabetes subjects
[3]. NAFLD is a metabolic disorder with no history of excessive drinking (i.e., less than 14 and 21 standard drinks per week on average for women and men, respectively)
[4] and is characterized by hepatocyte steatosis and fat accumulation. The disease presents with a broad spectrum of histological alterations, ranging from simple steatosis (SS) to the appearance of steatohepatitis (NASH), which is characterized by the presence of lobular inflammation, hepatocyte damage, and ballooning and that can ultimately lead, in a minority of subjects, to hepatic fibrosis
[5]. SS is considered one benign condition with no or little active inflammation and without fibrosis that does not lead to aggravation and progression of the disease. On the contrary, in a small percentage of patients, namely, those with NASH, the disease can be progressive and ultimately evolve towards cirrhosis with its increase in liver-related mortality and/or hepatocellular carcinoma (HCC). Nowadays, NASH represents the second most common indication for liver transplantation in Europe and the US and it is expected within a short time period to overcome hepatitis C
[6]. In addition, it is a matter of concern the growing number of non-cirrhotic subjects in which NASH can progress directly towards HCC
[7].
The diagnosis of NAFLD is usually made through abdominal ultrasound, which, in the studies that have compared it with the gold standard exam (i.e., liver biopsy), has been shown to have good sensitivity and high specificity for the diagnosis of steatosis
[8]. More , especially in the research field, the assessment of steatosis is performed by means of magnetic resonance imaging
[9]. Otherwise, the diagnosis of NASH is made through liver biopsy, using standardized criteria based on the evaluation of the NAS (NAFLD activity score)—which takes into account separate scores for steatosis, balloon-shaped degeneration and inflammation—and fibrosis
[10]. However, most patients with NAFLD do not require a liver biopsy, mainly for ethical reasons. As a matter of fact, biopsy is not indicated especially in those who do not present significant liver disease.
Both SS and NASH are often associated with the presence of common metabolic alterations such as abdominal obesity, atherogenic dyslipidemia, type 2 diabetes, and metabolic syndrome: Indeed, it has been suggested that they represent the hepatic component of all these processes
[3].
The pathogenesis of NAFLD and NASH is multifactorial, and numerous pathophysiological mechanisms have been proposed to explain the process of excessive fat accumulation in the liver and the subsequent possible induction of inflammation and fibrosis
[11].
Among the main biological players and pathways of NAFLD, hepatic steatosis (the first “hit”) has an obvious central role. This is the consequence of an excessive lipid accumulation within the liver, largely resulting from the excessive importation, diminished exportation, and/or reduced breakdown of free fatty acids (FFA) in the liver itself. During periods of excess macronutrient intake, the liver will convert unused carbohydrates and proteins to triglycerides and then store them. During later periods of diminished macronutrient intake or heightened caloric use, these lipids may be broken down and released. Complications of obesity may include hyperlipidemia leading to increased levels of circulating triglycerides and increased delivery and storage of triglycerides in the liver
[12].
On the basis of the traditional “two hits” theory
[13], it is believed that increased insulin resistance is the basis of the first stages of fatty liver, mainly through profound changes to normal systemic lipid metabolism such as increased mobilization of the aforementioned fatty acids (FA) from visceral adipose tissue to the liver. Insulin affects the intestinal-liver-fat axis by regulating blood glucose levels and guiding nutrients from the bloodstream to cells after meals through a complex pathway in which nuclear receptors such as liver X receptor (LXR), farnesoid X receptor (FXR), and nuclear factor erythroid 2-related factor 2 (Nrf2) play an important role
[14]. The development of insulin resistance occurs primarily in muscle and adipose tissue, while liver tissue remains largely spared. When muscle and adipose tissue develop decreased sensitivity to the effects of insulin, less glucose is delivered to these tissues and a catabolic state occurs in which peripheral adipose tissue is broken down and free FA are released into the systemic circulation. To compensate for the elevated blood glucose levels, pancreatic beta cells secrete increasing levels of insulin, resulting in hyperinsulinemia. The liver, having remained largely insulin sensitive and being exposed to increasing levels of blood glucose, serum triglycerides, and insulin, enters a hyper-anabolic state, continuing to synthesize and store lipids
[15]. However, there is currently much debate as to whether insulin resistance and the consequent hyperinsulinemia are only possible causes of steatosis or rather that NAFLD itself promotes a condition of hyperinsulinemia as a result of its degradation slowing down
[16][17][18][16,17,18]. Moreover, some believe that the effects of insulin resistance alone are insufficient to explain the development of true NASH. This is because it is still a matter of debate how lipid accumulation in the liver leads to the hepatocellular damage seen in NASH (which occurs in one out of 10 patients who develop NAFLD)
[19]. Instead, increased oxidative stress (OS) seems to be one of the additional main drivers in the promotion of NAFLD liver damage, playing a fundamental role in the progression from SS to NASH
[20][21][20,21]. This would occur, in particular, as a consequence of the induction of oxygen radical forming cytochrome p-450 microsomal lipoxygenases
[22] and consequent increased lipid peroxidation in which chain reactions between oxygen free radicals and lipids can damage mitochondrial DNA and membrane structures required for beta-oxidation
[23][24][23,24]. As a matter of fact, patients with NASH have been shown to have significant mitochondrial structural abnormalities within their hepatocytes. Whether lipid peroxidation alone is sufficient to produce these abnormalities is not known. In the setting of mitochondrial abnormalities, it has been theorized that the transition to beta-oxidation of FFA seen in NASH produces a further increase in free radical production with consequent hepatocellular damage, inflammation, and eventual fibrosis due to the activation of hepatic stellate cells (HSC)
[25]. In addition, reactive oxygen species (ROS) are also able to inhibit the hepatic secretion of very-low-density lipoproteins (VLDL), thus promoting a further intrahepatic accumulation of lipids
[26]. Antioxidants protect liver tissue from the damaging effects of oxygen radicals and lipid peroxidation by donating electrons and neutralizing these molecules before they react with DNA or proteins. However, they can be depleted in cases of continued OS or insufficient dietary intake. Glutathione (GSH), vitamin E (VitE), vitamin C (VitC), and beta-carotene may all be reduced in the setting of NASH
[27][28][27,28]. Hepatic iron content may also contribute to the oxygen free radical damage seen in NASH. Moreover, hepatic iron levels correlate with insulin resistance and the severity of fibrosis
[29][30][29,30]. However, homozygosity for the homeostatic iron regulator (HFE) gene, which leads to the development of hereditary hemochromatosis, has not been shown to increase the risk of NAFLD
[31]. In addition, no improvement in NAFLD was observed in a study, where participants underwent 6 months of phlebotomy to lower serum ferritin levels. Even those patients with hyperferritinemia at baseline showed no significant difference in the endpoints of hepatic steatosis, alanine aminotransferase (ALT) levels, or insulin sensitivity
[32].
More , alongside this traditional theory, a “multiple hits” hypothesis has been proposed, which, in addition to insulin resistance and OS, identifies as possible pathogenic factors in NAFLD development several other conditions including low-grade chronic inflammation, some genetic and epigenetic mutations, and factors derived from the intestinal microbiota. These elements may be variously associated with NAFLD in the presence or absence of the metabolic syndrome
[11].
In low-grade chronic inflammation—strictly linked to overweight/obesity—lipids and their derivatives contribute to inflammatory liver by the production of abnormal cytokines and adipokines such as interleukin (IL)-6, tumor necrosis factor (TNF)-α, IL-1, leptin, and resistin. These factors inhibit insulin signaling in hepatocytes by activating a suppressor of cytokine signaling (SOCS) proteins, several kinases [such as c-Jun N-terminal kinase (JNK), inhibitor of nuclear factor kappa-B kinase subunit beta (IKK-β), and protein kinase C (PKC)] and protein tyrosine phosphatases [such as protein tyrosine phosphatase 1B (PTP1B) and phosphatase and tensin homolog (PTEN)] that, in turn, impair insulin signaling at the insulin receptor and insulin receptor substrate (IRS) level. Hepatic insulin resistance, in turn, causes impaired suppression of glucose production by insulin in hepatocytes, leading to hyperglycemia. An important and early complication is the induction of hepatic VLDL production, via changes in the rate of apoB synthesis and degradation, de novo lipogenesis, or increased FFA flux from adipose tissue into the liver. Insulin resistance also stimulates the production of C-reactive protein and plasminogen activator inhibitor-1 (PAI-1), both markers of an inflammatory state, and interacts with other complex mechanisms such as hypercholesterolemia, hypertension, hemostatic factors, lipoproteins and their associated receptors structure, homocysteine processing/metabolism, and lipoprotein(a) levels
[33][34][33,34].
Additionally, a role for genetics has been demonstrated, for instance, in twin studies where it contributed roughly to 50% of hepatic fat content and fibrosis
[35]. Genetic variations in hepatic lipid metabolism and insulin signaling have also been demonstrated to contribute to development and progression of NAFLD
[36][37][38][36,37,38]. Obviously, a variety of factors then affects individuals with a genetic predisposition to NAFLD.
The gut microbiome may also contribute to the pathogenesis of NAFLD. One study showed significantly increased intestinal permeability and small intestinal bacterial overgrowth in patients with NAFLD. These patients were shown to have compromised intestinal tight junction integrity
[39][40][39,40]. Colon bacteria can damage intestinal tight junctions through endogenous production of alcohol and acetaldehyde
[41]. Increased intestinal permeability allows endotoxins produced by intestinal bacteria to enter the portal circulation and activate toll-like receptor (TLR)-4 signaling in Kupffer cells, leading to downstream increases in pro-inflammatory cytokines
[42][43][42,43]. TLR-4 can trigger myeloid differentiation primary response 88 (MyD88)-dependent or MyD88-independent pathways, with MyD88-dependent signaling playing a more critical role in the pathogenesis of NASH than in that of alcoholic liver disease
[44][45][44,45]. Both pathways lead to nuclear factor kappa B (NF-κB) activation, with further activation of pro-inflammatory cytokines (being characteristic of MyD88-dependent signaling) and type I interferon (IFN) induction (being characteristic of MyD88-independent signaling)
[46].
All this complexity may partly explain why the development of potential new drugs for NASH has been carried out across multiple channels, such as lipid metabolism regulation, anti-inflammation, and anti-fibrosis, even though this disease basically still remains an orphan of an adequate therapeutic strategy
[47].
2. Evidences on the Role of Oxidative Stress on NAFLD Progression
the researchers already reported that SS may progress to NASH with apparent inflammation, advanced fibrosis, and cirrhosis. In 1965, increased lipid peroxidation levels were reported in carbon tetrachloride- (CCl4-) treated rats, with increased production of ROS. However, it was only in 1972 that ROS started to be hypothesized with a causative role in the progression of liver damage
[48][113]. There is now a large body of evidence that, in the context of NAFLD, impaired redox status and consequent ROS accumulation are indeed the origins of hepatic maladaptive responses to fat accumulation, thereby leading to hepatic metabolic impairment and NASH progression
[49][61]. Moreover, OS-related oxidized phospholipids accumulate and induce mitochondrial dysfunction in hepatocytes
[50][114]. The mitochondrial GSH depletion is also induced by cholesterol accumulation in the progression of SS to NASH
[51][115]. Mitochondrial DNA (mtDNA) released from fatty liver-damaged hepatocytes then causes liver inflammation by TRL-9 activation
[52][116] and its levels tend to decrease in patients with more advanced forms of NAFLD
[53][117]. Thus, increased OS triggers hepatic stress pathways; therefore, maintaining cellular redox homeostasis is a promising strategy for NASH therapy
[54][118].
The hepatocytes are the primary cells affected by lipotoxicity-induced oxidative stress in the liver. However, also non-parenchymal cells (NPC), including HSC, liver sinusoidal endothelial cells (LSEC), and Kupffer cells, are involved in OS-induced liver damage
[55][119]. HSCs are notoriously responsible for extracellular matrix deposition in the development of liver fibrosis. CYP2E1-induced free radicals can activate the transdifferentiating of HSC. On the contrary, antioxidants could prevent the effect of ROS on increasing collagen production
[56][120]. The
NOX1- and
NOX2-deficient mice exhibited improved ROS production and hepatic fibrosis in CCl4 or bile duct ligation-treated models
[57][121]. In addition, mice deficient in antioxidant cytoglobin (
Cygb) are susceptible to OS, inflammation, and fibrosis under diethylnitrosamine (DEN) or a choline-deficient diet
[58][122]. Specifically, LSEC govern the regenerative process initiation, but OS damages their typical phenotype. Aberrant LSEC activation in chronic liver injury can ultimately induce fibrosis
[59][123]. Moreover, OS increases macrophage M1 (i.e., classically activated) polarization and promotes pro-inflammatory cytokines in Kupffer cells
[60][124]. Therefore, a promising line of research for treating NASH progression is to investigate OS targeting, possibly even with cell type-directed strategies.
During liver injury, oxidative stress induces the activation of redox-sensitive transcription factors, such as nuclear factor kappa-light-chain-enhancer of activated B cells (NF-κB) and activator protein-1 (AP-1), leading to an inflammatory response and the activation of cell death pathways in hepatocytes. In NAFLD, ROS regulates NF-kB activation by increasing the expression of pro-inflammatory cytokine TNF-α
[61][125]. NF-κB, a significant regulator of the inflammatory response, plays a vital role in regulating the transcription of genes involved in the establishment of the immune and inflammatory responses
[62][126]. Reduced NF-κB activity by antioxidants has been proposed as a therapeutic target in NASH due to its anti-inflammatory properties
[63][127]. Moreover, in the development of steatohepatitis, Nrf2 acts as a significant regulator of the redox balance and mediates anti-inflammatory and antiapoptotic effects of antioxidants
[64][128]. Upon oxidative stress, the cytoplasmatic transcription factor Nrf2, which regulates the expression of genes encoding antioxidant enzymes and regulators of the GSH (such as glutamate-cysteine ligase catalytic subunit, glutamate-cysteine ligase modifier subunit, GPX7), translocates into the nucleus, where it interacts with specific DNA sequences called antioxidant response elements (ARE) in the promoter of its target antioxidant enzyme genes
[65][129]. Nrf2 expression is increased in the first stage of NAFLD in preclinical models, and pharmacological activation of Nrf2 in mice fed a high-fat and high-fructose diet decreases NASH parameters (insulin resistance, weight, triglycerides, ALT levels) via the transcriptional regulation of genes involved in inflammation, apoptosis, fibrosis, ER stress, and OS
[66][130]. Consistently,
Nrf2-knockout mice treated with MCD show exacerbation of liver inflammation and steatosis, compared to control mice
[67][84]. In addition,
Nrf2-deficient mice fed a HFD develop a more severe NASH phenotype than their wild-type counterparts
[68][131]. Other evidence has shown that the dysfunctional Nrf2 in patients with NASH is tightly involved in the grade of inflammation but not of steatosis
[69][132]. In addition, upregulated Nrf2 in senescent hepatocytes is related to the activation of cocultured HSC. The Nrf2 agonist sulforaphane remarkably inhibits the effect of lipid accumulation-induced hepatocyte senescence on activation of HSC by the Nrf2-ARE pathway
[70][133]. Nrf2 also directly affects lipid metabolism by the activation of genes involved in FA oxidation (acyl-CoA oxidase 2, carnitine palmitoyltransferase 1), triglyceride export (apolipoprotein B), and the lipogenic transcription factor sterol regulatory element binding transcription factor 1 (SREBP-1)
[66][130].
Nfr2 deficiency in HFD-fed mice diminishes phosphorylation of acetyl-CoA carboxylate (ACC), a rate-limiting enzyme of hepatic FA synthesis, and, thus, increases its activity
[68][131]. A further study reported that the dysfunction of redox homeostasis induces hepatocytes to be highly susceptible to proteasome-associated metabolic stress. In comparison, insufficient peroxisome proliferator activating ligand receptors (PPAR) γ/Nrf2-driven antioxidative response is the main factor
[71][134]. Moreover, the interaction between NF-κB and Nrf2 is also a noticeable target for NAFLD progression. Evidence showed that NF-κB p65 subunit represses the Nrf2/ARE system at the transcriptional level by competitive interaction with the binding domain of the CREB-binding protein (CBP)
[72][135]. NF-κB dissociates from inhibitor kappa B (IκB) and then translocates to the nucleus. Nrf2 negatively controls the NF-κB signaling pathway by multiple mechanisms, including inhibiting nuclear translocation of NF-κB and blocking the degradation of IκB-α
[73][136].
Collectively, these data indicate that alterations in antioxidant pathways are associated with NAFLD, suggesting a role of oxidative stress in disease progression.
2.1. Markers of Immunological Responses to Inflammation and Oxidative Stress during NAFLD Evolution
OS in NASH results not only in an inappropriate activation of monocytes/macrophages at the endothelial and atherosclerotic plaque level, as previously mentioned, but also directly in the liver itself. As a matter of fact, a large body of evidence indicates that hepatic macrophages play a key role in directing the evolution of the disease
[74][137]. During chronic liver diseases, liver macrophages consist of ontogenically distinct populations that include liver resident Kupffer cells and monocyte-derived macrophages (MoMFs)
[74][137]. These latter are the main elements responsible for producing pro-inflammatory mediators responsible for the perpetuation for hepatocyte injury and liver inflammation and also for supporting the activation of HSC and extracellular matrix deposition during OS-induced NASH progression to fibrosis and cirrhosis
[74][137]. Consistently, interfering with liver MoMF recruitment ameliorated experimental NASH and reduced fibrosis evolution in a phase 2 clinical trial
[75][138]. What is less clear is how MoMFs interact with other inflammatory cells within the liver and particularly with lymphocytes. In this respect, several studies have demonstrated that lymphocyte-derived cytokines such as IFN-γ, IL-17, and tumor necrosis factor superfamily member 14 (TNFSF14 or LIGHT) can stimulate pro-inflammatory responses of MoMFs
[76][77][78][139,140,141]. However, the presence of more specific interactions between liver lymphocytes, particularly T-cells and MoMFs, has so far received little attention.
Among the signals involved in the OS-elicited interaction of T-cells with other myeloid cells, a new promising field of research is represented by the co-stimulatory molecules’ inducible T-cell co-stimulator (ICOS; CD278) and its ligand ICOSL (CD275, also named B7h, B7-H2, B7RP-1, and LICOS)
[79][142]. ICOS belongs to the CD28 family of co-stimulatory molecules and is selectively expressed by activated T cells, while its ligand ICOSL/ICOSL is constitutively present on the surface of a variety of myeloid cells including dendritic cells, macrophages, and B-cells, but also on endothelial cells, lung epithelium cells, and fibroblasts. The triggering of ICOS on T-cells by ICOSL/ICOSL has been shown to modulate their cytokine secretion pattern
[79][142] and favor regulatory T cell (Treg) differentiation
[80][143]. In addition, ICOS/ICOSL interaction plays an important role in the development and differentiation of follicular T-helper cells (Tfh) in the germinal centers of lymphatic nodes
[79][142]. This action is critical in the selection and survival of B cells expressing high-affinity B cell receptors, as well as in facilitating their differentiation into memory B cell and plasma cells
[79][142]. However, recent reports have shown that ICOS/ICOSL interaction can also trigger reverse signals able to modulate the functions of ICOSL-expressing cells. For instance, in dendritic cells, ICOSL-mediated signals favor maturation and stimulate cytokine secretion and antigen presentation
[81][82][144,145], while in monocytes they prevent the differentiation to osteoclasts stimulated by receptor activator of nuclear factor κ B (RANK) ligand
[83][146]. A recent study has also shown that by binding to αvβ3 integrin on podocytes, ICOSL ameliorates kidney injury and the development of proteinuria
[84][147]. From these observations, some preliminary data have emerged on the possible involvement of ICOS/ICOSL dyad in the evolution of liver inflammation, OS, and fibrosis in NASH
[85][148].
2.2. Mitochondrial Oxidative Injury as a Key Pathway That Links Saturated Fat Intake to the Development and Progression of NAFLD
2.2.1. Mitochondrial Dysfunction and Oxidative Stress in NAFLD/NASH
Liver is an organ that consumes as much as 15% of the body’s oxygen, since it has a huge amount of mitochondria (from 500 to 4000 per hepatocyte), which have a high turnover and are responsible for ATP production
[79][142]. One of the key mechanisms for the genesis and progression of NAFLD is, indeed, mitochondrial dysfunction
[86][87][88][89][149,150,151,152].
High dietary fat consumption and genetic obesity lead to the accumulation of triglycerides and FA in hepatocytes, inducing a compensatory increase in oxidative phosphorylation, tricarboxylic acid cycling, and mitochondrial respiratory chain function. In particular, increased mitochondrial carnitine palmitoyltransferase (CPT)-1 and mitochondrial uncoupling protein (UCP)-2 activity resulting from a HFD reduces the levels of serum markers of liver damage and ROS, playing a protective role against the onset of NAFLD
[90][153]. Nevertheless, in the long term, the liver is no longer able to adaptively compensate for the hepatocyte accumulation of FA, as the increase in mitochondrial respiration becomes bioenergetically inefficient. This leads to structural and functional mitochondrial alterations that determine the onset of NAFLD and, with the perpetuation of oxidative damage, the progression to NASH. As a matter of fact, literature evidence indicates that ROS production, lipid peroxidation, alterations in mitochondrial respiratory chain activity, and changes in mitochondrial membrane composition are all present in individuals with hepatic steatosis and drive NAFLD progression
[91][154]. The progression to advanced stages of NAFLD, including NASH and cirrhosis, is complex, and the role of SFAs in mitochondrial dysfunction is undoubtedly less studied than that of unsaturated FA. However, there is an increasing evidence that also SFAs play a key role in the development of hepatic steatosis and NAFLD progression through changes in mitochondrial function and structure
[91][154].
One of the mechanisms most involved in mitochondrial dysfunction associated with hepatic steatosis is the production of ROS, precisely at the mitochondrial level. ROS are formed when electrons escape from the electron transport chain complexes, interacting with oxygen and forming superoxides. The latter ones damage mitochondria by peroxidizing mitochondrial DNA, phospholipid acyl chains, and respiratory transport chain enzymes. Under normal conditions, redox molecules within the mitochondria matrix neutralize most ROS by converting them to water. However, chronic activation of mitochondrial function in lipid overload leads to excessive electron leakage that exposes the liver to high amounts of OS
[91][92][93][94][154,155,156,157]. Additionally, the results obtained on Huh7.5 cells treated with plasma of NAFLD patients would confirm those issues. Hence, in those cells, an increased mitochondrial ROS release was observed in association with changes of mitochondrial membrane potential
[95][158]. Furthermore, along with the increased mitochondrial ROS production, decreased expression and activity of ROS detoxification mechanisms (e.g., SOD2, catalase, or GSH) have also been reported in in vitro and in vivo experiments. Thus, levels of ROS, lipid peroxidation, and apoptosis are increased in fatty liver disease and are even more so in NASH, suggesting that these processes drive the progression of NAFLD to NASH
[96][90][97][72,153,159].
In addition to inducing ROS production, SFAs can also alter the composition of mitochondrial membranes. In this regard, one of the most studied target molecules is cardiolipin, which constitutes approximately 20% of the total phospholipid content in the inner mitochondrial membrane (IMM). Cardiolipin is critical in many mitochondrial functions. It maintains the fluidity and osmotic stability of the IMM and is crucial in the assembly and stabilization of the enzymatic supercomplexes of the electron transport chain involved in ATP production
[91][98][99][154,160,161]. The most widely represented cardiolipin species in hepatic mitochondria—constituting approximately 55% of hepatic cardiolipin species—is tetralinoleoyl-cardiolipin, in which all four acyl groups are derived from linoleate (linoleic acid methyl ester, C18:2). Linoleate-rich cardiolipin is able to bind with high affinity to a multitude of proteins and enzymes within the IMM, including key enzymes in the mitochondrial respiratory chain, such as cytochrome c
[91][98][100][154,160,162]. Evidence in the literature shows that the linoleate percentage in the IMM cardiolipin pool is lower in subjects with NASH. The precise mechanisms by which changes in cardiolipin content and structure occur, affecting mitochondrial function, are complex and still under investigation. However, it is known that when the acyl chain composition of cardiolipin changes, cytochrome c detaches from cardiolipin. The latter, in turn, translocates to the outer mitochondrial membrane (OMM)
[91][154], destabilizing its lipid composition and promoting pore formation
[101][163]. The final result is an altered mitochondrial function.
A further important characteristic of cardiolipin molecules is that they are very susceptible also to direct ROS attacks
[102][103][104][164,165,166]. Because a HFD increases the levels of ROS and of modified hepatic cardiolipin
[105][106][107][167,168,169], it is reasonable to hypothesize that this is an additional mechanism of mitochondrial damage with regard to NAFLD.
Structural changes in the IMM indirectly reflect on the ER. This is because approximately 80% of mitochondria are in contact with the ER via mitochondria-associated membranes (MAMs) in order to allow exchanges in metabolites and calcium (the latter one critical for cellular homeostasis and signaling). Structural damage of MAM induced by a HFD has been shown to lead to increased calcium influx into mitochondria. This promotes NADH production by tricarboxylic acid cycle enzymes and increases ATP synthesis and ROS production. In addition, increased levels of calcium in the mitochondrial matrix induce further mitochondrial permeability transition pore (mPTP) opening, promoting apoptotic processes in the long term
[91][108][154,170].
Ceramides are also another key element linking SFAs to the progression of NAFLD through mitochondrial dysfunction. These are sphingolipids synthesized from SFAs. Under conditions of hepatic steatosis (and even more so in NASH) when SFA levels are elevated, the production of ceramide substrates is increased. According to several clinical studies, the resulting accumulation of ceramides then induces hepatocyte apoptosis through increased ER stress and formation of mPTPs. In addition, ceramides inhibit mitochondrial function by interacting with the mitochondrial fragmentation factor (MFF), resulting in mitochondrial (and peroxisomal) fragmentation
[79][142].
Finally, another actor involved in the progression of NAFLD to NASH through mitochondrial dysfunction is cholesterol, which determines mitochondrial GSH depletion and sensitizes hepatocytes (in mouse models) to TNF and Fas cell surface death receptor (FAS)-induced apoptosis
[109][171].
Thus, the mechanisms responsible for mitochondrial dysfunction in hepatic steatosis and NAFLD progression are diverse and still not perfectly understood. It is likely that the functional and structural mitochondrial alterations induced by the aforementioned mechanisms mutually interact and reinforce, in a vicious circle leading to a constant evolution of NAFLD
[91][154].
2.2.2. Diagnostic Tools for Mitochondrial Dysfunction in NAFLD
The importance of assessing mitochondrial function in chronic liver diseases, such as NAFLD and its evolved forms, is, therefore, evident, thanks also to new diagnostic tools. One test introduced, although still in the process of implementation, is the 13C breath test (BT)
[108][170]. This is a “dynamic” liver function test based on the use of substrates labeled with the sTable
13C isotope, which is a natural, non-radioactive element. The substrate, inhaled and metabolized at the hepatocellular level, is excreted in breath as
13CO
2, which is measured by mass spectrometry or infrared spectroscopy. Thus, respiratory levels of
13CO
2 can be used as biomarkers of specific metabolic processes (occurring in the cytosol, microsomes, and mitochondria of hepatocytes) and consequently of several chronic liver diseases, including simple hepatic steatosis, NASH, liver fibrosis, cirrhosis, and HCC, and also drug- or alcohol-related hepatitis
[110][172]. In particular, the substrates used in 13C-BT for NAFLD are alpha-ketoisocaproic acid, methionine, and octanoic acid, i.e., substrates capable of measuring mitochondrial oxidation capacity, which is impaired already at an early stage of the disease
[111][173]. Therefore, 13C-BT represents an indirect, convenient, and easy method to evaluate the dynamic liver function in NAFLD with particular concern for the altered mitochondrial function. This may be particularly useful to guide early treatments, with the potential to limit disease progression, but could also be applied during secondary prevention. On the other hand, experimental studies indicate that mitochondrial dysfunction may precede the onset and progression of NAFLD, suggesting a potential use of 13C-BTs also in primary disease prevention
[112][174].
2.3. Gut Microbiota and Oxidative Stress in NAFLD, with Possible Therapeutic Implications
2.3.1. Role of Microbiota in Hepatic Steatosis and Oxidative Stress
Great interest has been addressed by the scientific community to the characterization of the intestinal microbial flora (microbiota) by means of sequencing its genome (microbiome), as its composition has often been found to have important implications in numerous pathological states, including NAFLD. Some observations demonstrated, in fact, quantitative and qualitative differences in the gut microbiota between subjects with or without NAFLD, even more so if the latter is associated with obesity and its related complications such as insulin resistance and diabetes
[113][114][175,176]. More in detail, increased Proteobacteria at the phylum level was seen in NAFLD patients compared to control subjects. Increased Enterobacteriaceae and decreased Rikenellaceae and Ruminococcaceae at the family level were found in NAFLD patients. Increased Escherichia and Dorea and decreased Anaerosporobacter, Coprococcus, Eubacterium, Faecalibacterium, and Prevotella were observed at the genera level in NAFLD patients
[115][177]. Studies in animal models demonstrated a direct role for the intestinal microbiota in the development of NAFLD. Inoculation of B. thetaiotaomicron and M. smithii strains into adult germ-free (GF) C57BL/6 mice increased host adiposity regardless of the amount of dietary intake. These microbial strains promoted monosaccharide uptake from the intestinal lumen, which resulted in the induction of lipogenesis
[116][178]. In a similar study, most wild-type mice fed a HFD developed insulin resistance, OS, and systemic inflammation; however, some mice remained sensitive to insulin and developed lower levels of systemic inflammation and liver FA oxidation (non-responders). GF mice were then inoculated with responder and non-responder gut microbiota and then fed with HFD. Interestingly, mice that received microbiota from a non-responder developed less steatosis and insulin resistance than mice that received the responder microbiota
[117][179]. Following these premises, translational studies were conducted, with particular regard to NAFLD disease. In one study the intestinal microbiota collected from obese donors before and after weight loss was transplanted into GF mice: Mice that received intestinal microbiota before weight loss had higher triglycerides and cholesterol liver levels than mice that received intestinal microbiota after weight loss
[118][180]. Moreover, donor fecal microbiota transplantation studies performed in human subjects confirmed that the gut dysbiosis can per se cause hepatic steatosis mainly by means of weight gain, insulin resistance, and increased OS
[119][120][121][67,181,182]. The hypothesized mechanisms are greater efficiency in energy absorption from food, modification of the intestinal permeability, release of intestinal hormones, and inflammation, with a relevant role for OS induction
[122][123][124][125][126][127][128][183,184,185,186,187,188,189].
2.3.2. Modulation of Gut-Liver Axis: Effects on Oxidative Stress
A very promising line of research is that the microbiota is a dynamic entity that can change over time as a result of the modulation from various factors. In this respect, it can be considered not only as a causative agent of NAFLD, but also as a protective one, leading to possible future therapeutic implications (as regrading expected benefits on the evolution of NAFLD itself). For instance, the microbial composition of the intestinal flora is, first of all, influenced by body weight and some components of the diet, such as fiber, polyphenols, and lipids. The most represented phyla in the gut are Bacteroidetes (50–60%) and Firmicutes (25–30%). While bacteria of the first group feed mainly on vegetable fibers and are able to assimilate few fats from the diet, those of the second group are characterized by their ability to obtain nourishment mainly from dietary fats and sugars while promoting their absorption by the host organism. In obese subjects, weight loss reduces the Firmicutes/Bacteroidetes ratio
[129][190], whereas a diet rich in fat and caloric load increases it
[130][191]. This is true also for NAFLD patients, where the Firmicutes/Bacteroidetes ratio is positively correlated with liver steatosis, too, at least in obese individuals
[131][132][133][192,193,194]. A relatively high abundance of Firmicutes is associated with metabolic endotoxemia due to increased uptake of lipopolysaccharide (LPS), which reaches the circulation and induces OS, inflammation, and impaired insulin signaling
[115][134][135][136][137][177,195,196,197,198]. As a counterevidence, some preliminary studies reported that probiotics (i.e., living microorganisms including Bifidobacterium spp. and Lactobacillus spp.) and prebiotics (i.e., nonviable food components including “insulin-like” fructans) may confer a health benefit that is directly associated with the modulation of the microbiota
[138][139][199,200]. As a matter of fact, in healthy subjects, the daily consumption for 3 weeks of 150 g of goat milk fermented with Lactobacillus fermentum increased the total antioxidant capacity (TAC) and decreased the oxidative markers in human blood and urine (oxidized LDL, F2-isoprostanes, and oxidized/reduced GSH ratio)
[140][201]. Similarly, results were reproduced in individuals with obesity and obesity-related disorders
[137][138][141][142][198,199,202,203]. Translating the evidence from these researches, it can now be assumed that prebiotics and probiotics are capable of regulating the metabolism also of NAFLD subjects
[143][144][145][146][147][148][204,205,206,207,208,209]. For example, studies conducted to identify the effect of novel probiotics on NAFLD induced by a fructose-rich diet underlined the ability of intestinal flora to improve OS. Human subjects fed with such a diet developed hepatic steatosis, and a relevant proportion acquired the clinical features of the true metabolic syndrome
[149][150][151][152][210,211,212,213]. Compared to controls, many individuals indeed presented an increase in plasma concentrations of glucose, insulin, triglycerides, cholesterol, and total levels of markers of OS, as well as in liver mass and lipids. Probiotic treatment lowered almost all of these parameters and reduced lipogenesis
[153][154][214,215].
Other studies emphasized the positive role of probiotics also in modulating the redox state. In NAFLD patients, the consumption of 300 g/day of probiotic yogurt containing Lactobacillus acidophilus and Bifidobacterium lactis significantly improved AST and ALT when compared with the placebo group, generally in parallel with a lowering of cholesterol, triglycerides, and LDL-cholesterol levels; in some cases, there was also an improvement in the intrahepatic fat content and/or waist circumference
[155][156][157][158][216,217,218,219]. The pathophysiological rationale probably resides in an increase in TAC
[159][220], SOD levels
[160][221], and GPx activity
[161][222], which are accompanied by a reduction in lipid oxidation (detected through MDA serum concentrations)
[162][223].
Gut microbiota has a demonstrated beneficial effect on NAFLD/NASH-related OS not only by a direct effect, as previously described, but also through its modulation of other dietary compounds with antioxidant activity. For instance, interesting observations have been made concerning the relationship between microbiota and polyphenols. The composition of the intestinal microbial flora, in fact, seems to be able to act directly on the bioactivity of such substances, which are normal components of the diet, known for their antioxidant effect in vitro, but also for their low bioavailability in vivo
[163][224]. The presence or absence of intestinal germs capable of metabolizing polyphenols could, at least in part, explain the inter-individual variability of the beneficial effects on NAFLD observed as a result of polyphenol dietary supplementations. Because these agents, by changing the intestinal redox state, are, in turn, able to control the different subpopulations of the microbiota, it may well be believed that the microbiota-polyphenol interconnection may represent a new target for studies centered on diseases mediated by OS such as NAFLD
[164][165][225,226].
In NAFLD patients, intestinal microbiota may be modulated also by physical exercise, and this could elicit its antioxidant activities. Lifestyle modifications are indeed one of the traditional treatments in obesity-related diseases and have a proven beneficial role also in the NAFLD context, all the more in patients with excess weight. Apart from dietary interventions, physical exercise interventions are, in fact, a valid way to reach weight loss. With regard to liver disease, physical exercise is capable of modulating hepatic steatosis, improving insulin sensitivity, or affecting body composition independently of weight loss
[166][227]. Only a few studies have considered that the effect of exercise on the metabolic state may be at least partially driven by intestinal microbiota modulation and no clear relation to NAFLD has been yet established. To the best of the researchers knowledge, the impact of exercise specifically on the gut microbiome and its subsequent effects on NAFLD has not been studied in humans. Thus, when researchers suggest that exercise might affect NAFLD (in part) due to modulation of the intestinal microbiome, this is attributed to effects seen mainly on obesity, an important contributor to NAFLD
[167][168][169][170][228,229,230,231]. Interestingly, there is only one rodent study indirectly addressing the modulatory effect of exercise on NAFLD through gut microbiome
[171][232]. In this research, the effect of a controlled exercise program was thoroughly investigated on the gut–liver axis in juvenile rats with promising results, when the mice were introduced to an exercise program after the development of features for obesity and NAFLD induced by a HFD. Exercise effectively counteracted HFD-induced microbial imbalance, leading to intestinal barrier preservation. This, in turn, prevented deregulation of the gut–liver axis and improved bile acid homeostasis, determining the clinical outcomes of NAFLD. It is noteworthy that the exercise protocol also positively increased the citrate synthase enzymatic activity, suggesting an improvement in the oxidative metabolism capacity. In parallel, exercise performance caused a distinct reduction in the HFD-induced hepatic Cyp2e1 expression involved in OS development and derived lipotoxicity. These reports are confirmed also by others: A high-intensity exercise training can reduce postprandial circulating lipid peroxidation levels
[172][233], suggesting that hepatic OS is reduced by aerobic exercise
[173][234]. Thus, with the aforementioned metabolic antioxidant activities (i.e., modulatory capacity over dysbiosis and gut–liver axis activation) at least in part attributable to the action of the microbiota itself, physical exercise may have the potential to be considered as an alternative, indirect microbiome-based therapy for NAFLD, at least for what concerns its obesity-associated variant
[148][174][209,235].
2.4. Relationship of Oxidative Stress and the Development and/or Progression of NAFLD-HCC
Taking into account the aforementioned epidemiological data that clearly indicate that NAFLD/NASH is causing a dramatic increase in the prevalence of HCC development, with NASH being a recognized etiological cause of HCC, it is quite understandable why many studies have attempted to unravel the possible role of OS also in the development of HCC, similarly to what happened for NAFLD and NASH. While increased OS in liver parenchymal cells has been observed and linked to HCC, both the detailed mechanisms and the overall impact of this specific problem have yet to be fully clarified
[175][236]. It is well known that ROS, such as H202, can cause point mutations or larger lesions in the genome
[176][237]. A recent study investigated in detail the role of OS-related enzymes and receptors in liver carcinogenesis, identifying three factors (thioredoxin reductase-1, glutathione reductase, and the transcription factor Nrf2) as major players in the development of HCC
[177][238]. The authors concluded that this process is the result of a complex interaction between several factors. Indeed, although it is well known that anti-oxidant systems constitute an integrated and finely tuned network capable of effectively preventing carcinogenesis by protecting healthy cells, the role of OS is controversial in existing cancers, where ROS are definitely part of the tumor microenvironment (TME)
[178][239] and the antioxidant network probably plays both anti- and pro-cancer roles. In this context, ROS are active players in cancer development, exerting apparently contradictory effects, i.e., stimulation of tumorigenesis and cancer cell proliferation or induction of cell death. Focusing on NAFLD/NASH, an indirect confirmation of the tumorigenic role of OS is the adaptation of cancer cells to antioxidant insults, e.g., by increasing NADPH through the pentose phosphate pathway (PPP), which is also an emerging mechanism of drug resistance exploited by cancer cells
[179][240]. In this context, dissecting the role of NOX (such as NOX1, NOX2, and NOX4), which are both NADPH consumers and ROS generators, would be of great interest.
In any case, an exhaustive analysis of all ROS involved in the transition from NASH to HCC far exceeds the aims of this review. Briefly, the most well-established signaling cascade regulated by ROS is Nrf2. When active in neoplastic cells, Nrf2 signaling restricts ROS damage and favors cancer cell survival under chronic OS. It is believed that some cancer cells actually use Nrf2 signaling as an adaptive mechanism to promote tumor growth
[180][241]. , it was proposed that Nrf2 activation due to the microenvironment could be one of two hits required for HCC, while a mutation in the gene encoding β-catenin, a major genetic aberration observed in a significant subset of NAFLD-associated HCC, provides the second hit
[181][242]. NF-κB is another important pathway in this context. It can have both pro- and antioxidant roles by affecting the expression of target enzymes involved in ROS scavenging and generation
[182][243]. Despite this role of NF-κB in maintaining antioxidant defenses and reducing liver damage, obese and NAFLD patients display increased pro-inflammatory cytokine levels, hepatic NF-κB activation, and risk for HCC development
[183][244]. ROS can also indirectly and directly affect the tumor suppressor p53 activity (with higher expressions causing liver inflammation and NAFLD progression)
[184][245] and hypoxia-inducible factors (HIF) (with a positive correlation with HCC development and resistance to chemotherapeutic agents)
[185][246]. In addition to classical ROS pathways, B-cell lymphoma 2 (BCL-2) proteins are emerging as physiological- and pathophysiological-associated redox molecules in cell survival and death, with a possible role in NAFLD/NASH tumorigenesis. As a matter of fact, BCL-2 proteins can contribute to NAFLD hepatic ROS formation; in addition, ROS levels can trigger cell death through BCL-2 protein modulation
[186][247]. Protein tyrosine phosphatase (PTP) oxidation is another key mechanism of downstream ROS production with a proposed role in the transition from SS to NASH and HCC
[187][248]. Briefly, OS stress inhibition of PTPs serves as a mechanism by which optimal tyrosine phosphorylation is maintained under physiological conditions but, if dysregulated, can contribute to liver dysfunction and HCC development, especially in obese subjects
[188][249].
Among other possible, interesting new molecular mechanisms linking OS and NAFLD tumorigenesis there is the Notch signaling pathway. Selective blocking of Notch homolog 1, translocation-associated (Notch1), inhibits cancer cell growth and deregulates angiogenesis
[189][250]. By performing RNA sequencing of HFD-fed mice, Zhu et al. demonstrated that Notch-active hepatocytes show transcriptional enrichment of ECM-related genes, which may represent a mechanism that persists in the tumorigenic process. Furthermore, the same animal model with Notch active mutation spontaneously forms fully developed liver tumors
[190][251]. Therefore, it can be inferred that the continuous activation of the Notch signaling pathway promotes the occurrence of NAFLD-related HCC. A further indirect evidence comes from the potential inhibition of HCC invasion and metastasis by δ-tocotrienol, an isomer of VitE, via downregulating the Notch1 signaling pathway with reduced biochemical markers of hepatocellular injury and steatosis
[191][252], and by silymarin via inhibiting factors such as Notch intra-cellular domain (NICD), recombining binding protein suppressor of hairless (RBP-Jκ) (i.e., the central player in the transcriptional regulation of Notch target genes and functions) and hairy and enhancer of split-1 (Hes1)
[192][253].
2.5. Biomarkers of Oxidative Stress in NAFLD/NASH
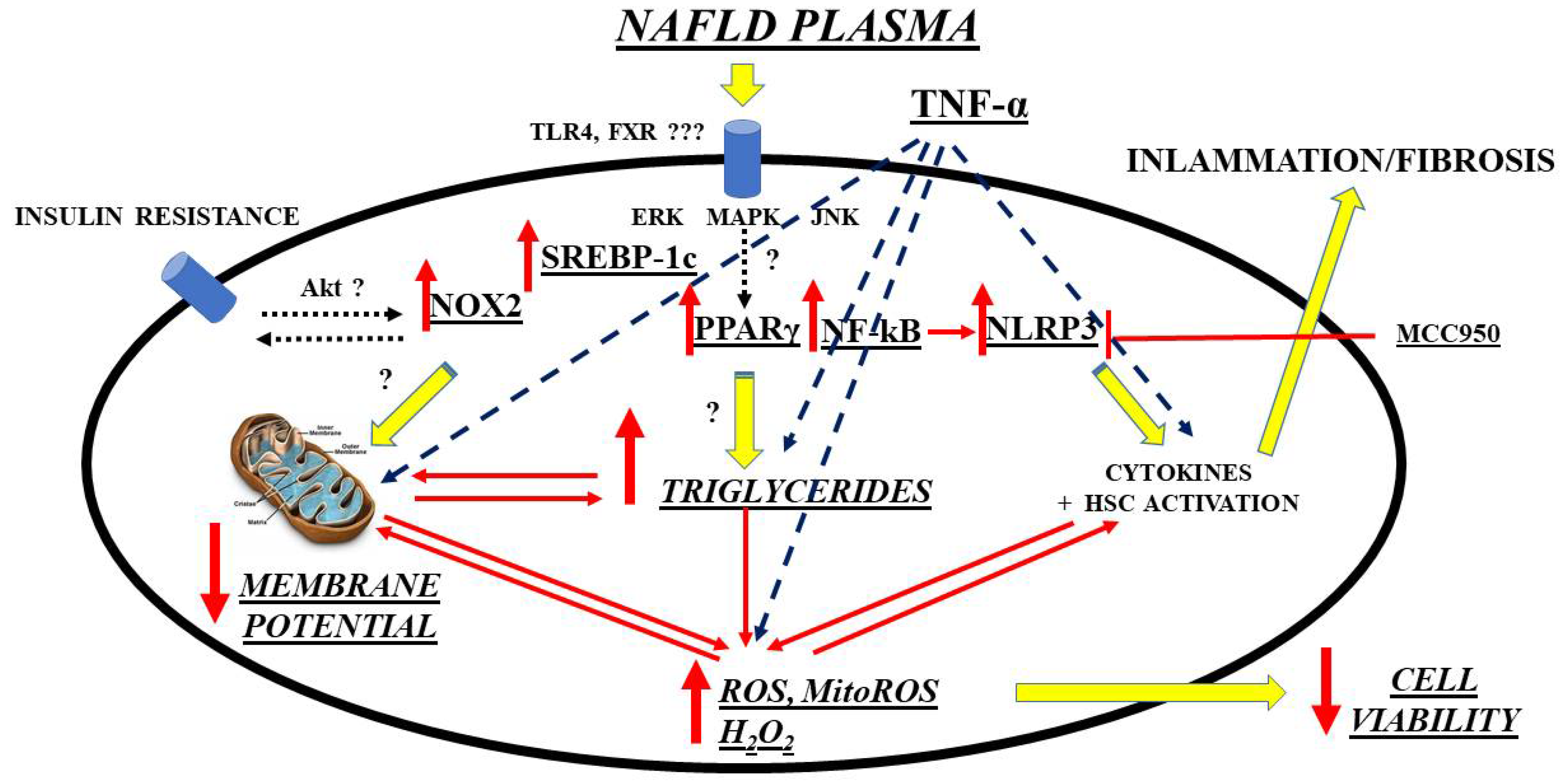
A further interesting and innovative line of research is the investigation of possible new biomarkers that have a diagnostic/prognostic role in relation to OS in NAFLD/NASH and that go alongside those already more validated regarding liver fibrosis. Though not well defined, circulating factors in NAFLD patients could modulate intracellular pathways in the direction predicted by current hypotheses on NAFLD pathogenesis, which has been evidenced by an in vitro study. Hence, exposure of primary human hepatocytes and Huh7.5 cells to plasma of NAFLD patients was able to cause triglycerides’ accumulation and OS through mechanisms involving SREBP-1c, inflammasome nucleotide-binding oligomerization domain-containing protein (NOD), leucine-rich repeats (LRR), pyrin domain-containing protein 3 (NLRP3), and PPAR γ, as a starting point
[95][158]. (
Figure 1).
Figure 1. Mechanisms of damage elicited by NAFLD plasma on hepatocytes. ERK: extracellular signal-regulated kinases; FXR: farnesoid X receptor; HSC: hepatic stellate cells; MAPK: mitogen-activated protein kinase; ROS: reactive oxygen species; MitoROS: mitochondrial ROS; H2O2: hydrogen peroxide; NF-kB: nuclear factor kappa-light-chain-enhancer of activated B cells; NLRP3: NOD-LRR-and pyrin domain-containing protein 3; MCC950: a specific, small-molecule inhibitor of NLRP3 inflammasome; NOX: nicotinamide adenine dinucleotide phosphate (NADPH) oxidases; Akt: Ak strain transforming factor; PPAR: peroxisome proliferator activating ligand receptors; SREBP: sterol regulatory element binding protein; TLR: toll-like receptor; TNF: tumor necrosis factor; JNK: c-Jun N-terminal kinase.
Following these premises, a promising, novel, systemic marker of OS, but also of inflammation and cellular aging, is represented by growth differentiation factor-15 (GDF-15)
[193][194][195][254,255,256]. This is a cytokine upregulated in multiple pathological conditions where OS, endothelial dysfunction, tissue aging, and chronic inflammation are the hallmarks. As a matter of fact, GDF-15 is rapidly produced by various cell types, including hepatocytes, cardiomyocytes, macrophages, adipocytes, and endothelial and smooth cells, among others, in response to pro-inflammatory cytokines or ROS, cellular stress, tissue injury, hypoxia, and/or oncogene activation
[196][257]. More in detail, this factor has been identified as a mitochondrial unfolded protein response (UPR
mt)-associated cell non-autonomous mitokine that regulates systemic energy homeostasis and feeding behavior
[197][258]. Due to the link between mitochondrial dysfunction and liver injury, GDF15 is currently considered a biomarker of diverse chronic liver diseases
[198][259]. First of all, this factor has been demonstrated, as expected, to be a possible prognostic HCC marker because its upregulation as a consequence of mitochondrial oxidative phosphorylation defects and ROS production is associated with liver cancer development, progression, and metastasis
[199][260]. Taking into consideration the same physiopathological considerations, it is not surprising that the factor has also a potential prognostic role in hepatitis B and C chronic viral hepatitis, with an increasing degree of diagnostic accuracy in compensated and decompensated liver cirrhosis
[200][201][202][261,262,263]. Finally, its pathogenic role was demonstrated also in the development and progression of NAFLD. With concern to the latter condition, GDF15 increased the risk of NASH and advanced fibrosis among biopsy-proven NAFLD patients, independently of known metabolic risk factors [(such as age, gender, body mass index (BMI), insulin resistance, and low skeletal muscle mass)]; moreover, the factor was associated with the severity of lobular inflammation and ballooning
[203][204][264,265].
Among other promising biomarkers of OS in NAFLD, the growth arrest-specific gene 6 (Gas6) serum protein and its family of tyrosine kinase receptors, namely, Tyro3, Axl, and MERTK (TAM), can be cited. The Gas6/TAM system (mainly, Axl and MERTK) is already a well-known important player in the progression of liver fibrosis in various hepatic diseases, including NAFLD/NASH
[205][266]. Focusing on OS, it is necessary to go back for a moment to the aforementioned “two hits” theory of NASH. Briefly, steatosis is the first hit that generally is recognized to increase hepatocyte vulnerability to any secondary insult, eliciting an inflammatory response, characterized by lobular inflammation, elevated local and systemic cytokines, activation of HSC, and expansion of liver progenitor cells (LPC). Most probably, both events are strictly interconnected since fat accumulation per se induces OS, inflammatory cytokine synthesis, and LPC expansion
[206][267]. Well, the Gas6/TAM system could be a key element in this interplay
[207][208][268,269]. What is already known is that in the liver, Gas6 and at least its high-affinity receptor Axl, are expressed by many of the previously cited inflammatory elements, such as macrophages, HSC in their myofibroblastic phenotype
[209][270], and LPC
[210][271]. Moreover, in murine models, Gas6 deficiency reduced inflammation and activation of Kupffer cells and myofibroblasts, causing, among other things, delayed liver repair in response to acute injury
[211][272]. Based on these preliminary studies, which suggested a prominent role of Gas6 in the pathogenesis of chronic liver diseases including NAFLD/NASH, new studies followed, still analyzing the possible roles of Gas6 deficiency but this time focusing more on the possible changes in the oxidative state at the hepatic level. For instance, Gas6-deficient mice fed a choline deficient, ethionine-supplemented diet (CDE) showed not only, as expected, reduced liver inflammation and fibrosis, but also attenuated hepatic steatosis. More in detail, Gas6
−/− mice fed with the CDE diet showed a reduced downregulation of some crucial rate-limiting enzymes of mitochondrial FA β-oxidation, such as PPARα and its target genes acyl-CoA oxidase-1 (
ACOX1) and carnitine palmitoyltransferase-1 (
CPT1). Moreover, Gas6 deficiency reversed the CDE-induced repression of stearoyl-CoA desaturase-1 (
SCD1), the rate-limiting step in the biosynthesis of monounsaturated FA. Moreover, Gas6 deficiency at least partially mitigated the increased expression of CD36 hepatic fatty acid translocase (
CD36), an enzyme that mediates uptake and intracellular transport of long-chain FA in hepatocytes and that is known to be abnormally increased in NAFLD. All these mechanisms ultimately accounted for the more efficient FA catabolism and delayed steatosis found in Gas6 knockout mice
[212][273]. On the other hand, CDE diet is long known to promote intrahepatic lipid accumulation because of increased FA uptake and decreased triglycerides synthesis, in addition to the already mentioned inhibition of FA oxidation, and so can be a useful model if NAFLD-associated processes are the study focus
[213][214][215][274,275,276]. This happens probably because the CDE diet has been demonstrated to induce CD36 expression, favoring FA uptake, and to suppress the SCD1 mRNA, preventing synthesis of unsaturated FA
[216][217][218][277,278,279]. Concerning the latter point, a possible explanation for the higher SCD1 expression observed in Gas6
−/− mice is that they have a decreased expression of Kupffer cells-derived TNF-α, which, in turn, has been demonstrated to downregulate SCD1
[216][277]. Moreover, Gas6-deficient mice showed also reduced IL-1β expression, again produced from Kupffer cells. This a cytokine that promotes triglycerides’ storage in hepatocytes by reducing the expression of PPARα and its target genes involved in hepatocyte FA oxidation
[219][280]. Therefore, it can be assumed that reduced IL-1β expression revealed in Gas
−/− mice is an additional mechanism that prevents early CDE-induced downregulation of genes involved in β-oxidation in hepatocytes and steatosis. Of interest, a role of Gas6 in fat accumulation has been reported also in adipogenesis. After exposure to a HFD, fat accumulation in adipose tissues was reduced in Gas6 knockout mice, with no effect on hepatic steatosis. In this study, Gas6 directly promoted the proliferation of Axl-positive preadipocytes and their differentiation into mature adipocytes
[220][281]. For what concerns the liver, it was previously demonstrated that Gas6 and its receptor Axl are not expressed in hepatocytes. Therefore, the contribution of Gas6 to CDE-induced FA accumulation in these cells may not be direct but mediated by Axl-positive cells, such as macrophages, LPC, and HSC in their myofibroblastic phenotype
[209][210][270,271]. By the way, the total content of Axl was higher than in wild-type mice
[212][273]. New insights to the importance of the Gas6/TAM system pathway in the progression of NASH came also from studies involving the Gas6 receptor MERTK. In the metabolic syndrome, and more specifically in NAFLD, accumulation of excess lipoprotein-derived cholesterol in macrophages was shown to activate nuclear liver X receptors (LXR) that, in turn, triggered the induction of the ATP-binding cassette (ABC) transporter, mediating cholesterol efflux
[221][282] and the upregulation of MerTK both in mice
[222][283] and in humans
[223][284]. Both LXR and the aforementioned PPAR influence, among other things, the transcription of genes regulating lipid homeostasis and inflammation. More in detail, PPARγ can be activated by metabolic signals (i.e., polyunsaturated FA and lipoproteins)
[224][285], resulting in lipid uptake through the scavenger receptor CD36 and β-oxidation
[225][286]. LXRs are, in turn, oxysterol-activated transcription factors that sense elevated cellular cholesterol
[226][287]. PPARγ and LXR activities are generally coordinated and control the transcription of many receptors, including MerTK. In MERTK and LXRs, double knockout mice amplified pro-inflammatory responses, and increased susceptibility to OS and atherosclerosis were observed, suggesting that the LXR-dependent regulation of MerTK is important for normal homeostasis including FA metabolism
[227][228][229][288,289,290]. However, further studies are needed to confirm these preliminary data.
Mac-2 binding protein (M2BP), a known important player in cell adhesion, is another NAFLD biomarker candidate
[230][291]. For what concerns liver, a source of M2BP is HSC; since Kupffer cells have an increased M2BP production after autocrine or paracrine stimulation, it was suggested that M2BP secreted from HSC may be taken up by Kupffer cells
[231][292]. Based on these premises, the factor is mainly known as a liver fibrosis marker through inflammation in the extracellular matrix
[230][232][233][291,293,294]. However, there is now increasing evidence for a promising role also, as in the metabolic syndrome context. For instance, serum M2BP levels were higher in subjects with hypertension, dyslipidemia, or abnormal glucose metabolism compared to subjects without such risk factors. Moreover, the protein levels were associated with severity of cardiovascular risk. Subdivision of M2BP levels into quartiles revealed that M2BP was significantly associated with OS and, in particular, with reactive oxygen metabolites
[234][295]. Other studies confirmed that, in normal subjects, M2BP concentrations were not only associated with OS marker derivatives of reactive oxygen metabolites, but also with LDL-cholesterol and triglyceride levels. Furthermore, increased LDL-cholesterol was an independent determinant of M2BP high concentrations and, vice versa, increase in LDL-cholesterol was significantly greater in subjects in whom M2BP concentrations increased during the follow-up period
[235][296]. When focusing on NAFLD patients, there was an indirect association between M2BP levels and subclinical atherosclerosis as determined by brachial-ankle pulse wave velocity
[236][297]. A direct link between OS and M2BP was instead provided in a cohort of Japanese obese men with NAFLD. Independent of the effect of weight loss, a 3-month exercise regimen significantly reduced liver steatosis (by 9.5%) as determined by transient elastography. Moreover, an important decrease in OS parameters during the intervention was observed: Among other parameters, there was also serum M2BP (until −62.4% in the exercise group and −37.7% in the weight-loss group). These reductions paralleled with less significant decreases in insulin resistance (HOMA-IR) and lipid profile (triglycerides and non-esterified FA). Taken together, these data suggest that, in NAFLD, M2BP may be more a marker of organokine balance and systemic inflammation and OS than a surrogate liver steatosis parameter
[237][298] and could find a clinical application primarily in NAFLD patients with obesity and/or metabolic syndrome.
A new line of research in NAFLD biomarkers concerns extracellular vesicles (EVs). Several mechanisms implicated in NAFLD progression, such as inflammation, fibrosis, and angiogenesis, but also OS, all related to metabolic syndrome-associated lipotoxicity, trigger EV production and release by the liver
[238][299]. On the one hand, EVs mediate local intercellular communications between the liver cells, thereby driving disease pathogenesis and, on the other, liver-derived EVs could affect distant tissues and organs upon their release to the bloodstream. Thus, liver-derived EVs have been suggested as biomarkers both for diagnostic (potentially including early stages) and prognostic purposes in NAFLD patients (the so-called liquid biopsy)
[239][240][300,301]. However, the identification of liver-derived EVs in circulation as indicative of metabolic alterations in this organ is still a challenge for basic and clinical researchers. As previously cited, NAFLD is not an isolated condition and generally occurs as a complication of other metabolic disorders. Therefore, multiple tissues may be affected and, consequently, the contribution of extrahepatic EVs during NAFLD cannot be excluded (i.e., adipocyte- or immune cell-derived EVs). Furthermore, most liver cell types produce EVs including hepatocytes, cholangiocytes, HSC, and liver sinusoidal endothelial cells
[241][302]. Nonetheless, as 80% of the liver volume is composed by hepatocytes, their participation to the total pool of liver-derived EVs is likely the most relevant. Based on these premises and focusing on EVs of hepatocyte production, several protein-based EV biomarkers have been introduced for NAFLD liver damage
[242][303] or NASH
[243][244][304,305] with particular regard to the lipotoxicity link with inflammation, although, to date, most studies have focused on characterizing EVs-associated nucleic acids, especially microRNAs (miRNAs). For what concerns OS, some preliminary data exist precisely for EVs-derived miRNAs. It was demonstrated that liver firstly responds to lipid overload and thereafter sends hepatocyte-derived EVs (in particular, let-7e-5p), targeting adipocytes to regulate adipogenesis and lipogenesis. More in detail, these EVs positively correlated with BMI and enhanced adipocyte lipid deposition by increasing lipogenesis and inhibiting lipid oxidation through an axis involving also the peroxisome proliferator-activated receptor gamma coactivator 1-alpha (
Pgc1α). Moreover, taking into account that lipid overload enhances liver geranylgeranyl diphosphate synthase (
Ggpps) expression, which, in turn, regulates EVs’ secretion through Rab27A geranylgeranylation, liver-specific
Ggpps-deficient mice had reduced fat adipose deposition because of reduced EVs’ secretion
[245][306]. Thus, this pilot study highlighted an inter-organ mechanism whereby the liver during NAFLD senses different metabolic states and sends corresponding signals to remodel adipose tissue to adapt to metabolic changes in response to lipid overload. In any case, future studies aiming to examine additional molecular mechanisms possibly involved in EVs’ biogenesis, release, and dysregulation of target cells as well as to identify cargos with potential value as biomarkers for noninvasive diagnosis and monitoring of NAFLD progression are much desirable.
There are many other biomarkers of OS that have been evaluated, or that are under evaluation, in animal and clinical models of NAFLD/NASH
[49][246][247][248][61,68,75,307]. These factors were detected primarily in the liver, plasma, and serum, although a few studies analyzed whole blood samples. Major approaches to measure these biomarkers included ELISA, colorimetry, and immunohistochemistry. Biomarkers of OS measured in experimental models of NAFLD/NASH included lipid oxidation products [thiobarbituric acid reactive substances (TBARS), which were used as MDA, 4-HNE, and 8-isoprostanes], DNA damage products [8-hydroxy-2′-deoxyguanosine (8-OH-dG)], and protein damage products (protein carbonyl, dityrosine, hydroxyproline, and nitrotyrosine) (
Table 1). Among these biomarkers, MDA was the most measured marker of OS. The level or activity of these factors was generally increased in most animal models, with a few exceptions
[249][250][308,309].
Table 1. Oxidative stress markers in animal models of NAFLD/NASH.
| Oxidative Stress Markers |
Disease Status |
Experimental
Model and Species |
Sample |
Changes in
Concentration/
Activity/Expression |
Reference(s) |
[266][267][268][117,325,326,327].
Table 2. Oxidative stress markers in NAFLD/NASH patients.
| Oxidative Stress Markers |
Disease Status |
Sample |
Changes in Concentration/ |
345] (
Table 3 and
Table 4).
Table 3. Antioxidant markers in animal models of NAFLD/NASH.
| Antioxidant Marker |
Disease Status |
Experimental
|
Activity/Expression |
Model and Species |
SampleReference(s) |
| Changes in |
| Concentration/ |
Activity/Expression | Changes in
Concentration/
Activity/Expression |
| TBARS/MDA |
Steatosis, NASH |
| Reference(s) | Reference(s) |
|---|
| TBARS/MDA |
Steatosis, NASH | HFD, HF-HSD, MCD, ob/ob mice, CDHF diet |
Liver |
↑ |
Serum, liver, blood |
↑ |
[269] |
| SOD | [ | 53 | ] |
Steatosis, NASH | [ |
HF, HFD, OLETF rats, MCD | 270] |
| SOD |
Steatosis, NASH | Liver | [ | 87,117, |
↓ |
Serum, plasma, liver |
↓ | 328] |
[286][288] | [344,346] | [251][252] | [310,311] |
| [ | 286 | ] | [ | 288 | ] | [344,346] |
HCD |
↓ |
| Serum |
NS |
| HFD, MCD |
↑ |
HFD, MCD |
NS |
| 4-HNE |
| Blood, serum |
↑ |
NASH |
Liver |
↑ |
[271 |
HFD | ] | [329] |
NS |
| Serum |
NS |
4-HNE |
Steatosis, NASH |
HFD, MCD, HF-HSD, CDHF diet |
Liver |
↑ |
[253][254] | [312,313] |
| Hydroperoxides |
NASH |
Liver |
↑ |
[272] | [330] |
Catalase |
Steatosis, NASH |
HFD, MCD, HCD |
Liver |
↓ |
[53][ |
| Catalase |
Steatosis, NASH | 285 |
Plasma, blood, liver |
↓ |
[53][285] | [117, | ] | [117,343] |
| 343 | ] |
8-OH-dG |
NASH |
MCD, ob/ob mice HFMCD |
Liver |
↑
↓ |
| 8-OH-dG |
Steatosis, NASH |
Liver, plasma
Liver | [ |
↑
NS |
[273] | [331] | 255] | [314] |
| MCD |
↑ |
8-isoprostane |
| Serum |
NS |
NASH |
HFMCD |
Liver |
↑ |
| 8-isoprostane |
NASH |
Plasma | [ |
GPx | NS | 256] |
[267 | [315] |
| ] | [ | 326 |
Steatosis, NASH |
HFD, MCD, HF |
Liver | ] |
↓ |
[267] | [326 | [ |
Protein carbonyl |
Steatosis, NASH |
HFD, MCD
HFD |
Liver |
↑
NS |
[257] | [316] |
| Nitrotyrosine |
| Protein carbonyl |
Steatosis, NASH |
Liver |
↑ |
[268] | [327] |
NASH |
HFD, ob/ob mice |
Liver |
↑ |
[ |
| Nitrotyrosine |
Steatosis, NASH |
Liver |
↑ |
[25] | 258] | [317] |
| Periostin |
NASH |
HFD, ob/ob mice |
| Blood |
| GPx |
Steatosis, NASH | 268 |
Liver, serum |
↓ |
[267][268] | [326,327] | ] | ,327] |
| MCD |
↑ |
| Blood |
↑ |
HFD |
NS |
| Serum |
NS |
NS | Serum, liver |
↑ |
GSH | [259] | [318] |
| Steatosis, NASH |
HFD, HCD, MCD, ob/ob mice |
Liver |
↓ |
[ | 288] | [346] |
| GSH |
Steatosis, NASH |
Liver, blood |
↓ |
[346] |
CYP2E1 |
Steatosis, NASH |
HFD, HF-HSD, CDHF |
Liver |
| Periostin |
Steatosis, NASH |
Serum, plasma, liver
| ↑ |
Serum, plasma |
↑ |
[274 |
OLETF rats, HF MCD | ][275][276][277] |
↑ | [332,333,334,335] | [252][260] | [311,319] |
| Dityrosine |
Steatosis |
HFD |
Liver |
↑ |
[261] | [320] |
| NS |
Hydroxyproline |
| GR |
NASH |
MCD |
Liver |
↓ |
[282] | [340] |
Table 4. Antioxidant markers in NAFLD/NASH patients.
| Antioxidant Marker |
Disease Status |
Sample |
| [ |
| 288 |
| ] |
| Serum |
| ↑ |
| GR |
| Steatosis, NASH |
Serum, blood |
↑ |
[ | 286] | [344] |
| NASH |
MCD/WD |
Liver |
↑ |
| Nitric oxide |
Steatosis, NASH |
Serum, blood |
↑ |
[278][279] | [ | [262] | [321] |
| TRX |
Steatosis |
Serum |
↑ | 336, |
[ | 337] |
287] | [345] |
H | 2 | O | 2 |
NASH |
MCD |
Liver |
| CYP2E1 | ↑ |
Steatosis, NASH |
Liver |
↑ |
| α-Tocopherol | [ | 280 | ] |
Steatosis, NASH | [ |
Serum |
↓ |
[275] | [333] | 250] | [309] |
| [ | 338 | ] |
Lipid peroxide |
NASH |
MCD |
Liver |
↑ |
[263] | [322] |
| NS |
NADPH oxidase |
NASH |
ob/ob mice |
Liver |
↑ |
[264] | [323] |
| Xanthine oxidase |
Steatosis |
HFD |
Liver |
↑ |
[265] | [324] |
For what concerns biomarkers of OS frequently determined in NAFLD/NASH patients, the most studied ones were quite similar to those previously mentioned for experimental models (
Table 2). Overall, the concentrations or activities of these biomarkers were increased in all data examined, although the rise was not significant in some cases
[53]
In addition, many studies, both in human and experimental models of NAFLD/NASH, also measured enzymatic and nonenzymatic liver antioxidant biomarkers [e.g., catalase, SOD, GSH peroxidase (GPx), GSH, thioredoxin reductase (TrxR), α-tocopherol, and ubiquinone], and their activities were decreased in most liver samples, consistently with a loss of their protective abilities due to high OS, while blood, serum, and plasma had more conflicting results
[257][265][267][281][282][283][284][285][286][287][316,324,326,339,340,341,342,343,344,
| NS |
| Ubiquinone |
| Steatosis |
| Serum |
| ↓ |
| [ |
| 289 |
| ] | [ | 347 | ] |
| Bilirubin |
Steatosis, NASH |
Serum |
↓ |
[279] | [337] |
| Ascorbic acid |
Steatosis, NASH |
Serum |
↓ |
[288] | [346] |
| NS |
Data derived from the measurement of all these markers clearly indicate that the presence of OS is strongly associated with NAFLD/NASH. However, these biomarkers provide very limited information on the type, amount, and localization of ROS as well as their targets and involvement in specific pathophysiological processes. Indeed, the cause-and-effect relationship between OS and pathogenesis has not yet been established with certainty, although many studies have provided possible mechanisms supporting the crucial role of OS in the pathogenesis of NAFLD.
3. Conclusions
Starting from the assumption that OS would represent a trigger factor for NAFLD onset and evolution towards NASH and HCC, it is quite evident that the cornerstone of NAFLD treatment can be represented by the modulation of the balance between the antioxidants/oxidants, which is under the influence of individual genetic and epigenetic factors, as well. Various attempts to translate ROS scavenging by antioxidants into experimental and clinical studies have yielded mostly encouraging results. Among several antioxidants, the use of VitE has been particularly examined in Literature, although results obtained are somehow contradictory. Additionally, studies performed with natural polyphenols, which could be useful for NAFLD prevention and treatment thanks to their antioxidant activities, are quite satisfactory. In addition, probiotics/prebiotics, healthy diet, exercise, or fecal microbiota transplantation would represent new therapeutic approaches to modulate microbiota quality and diversity in order to prevent and/or avoid gut damage and consequent liver OS. Nevertheless, it must be said that most of the molecular targets and the exact cellular mechanisms of these compounds remain to be fully elucidated, and additional studies on their therapeutic potential are needed. Finally, given the importance of genetics in the NAFLD onset, precision medicine taking into consideration genetic/epigenetic factors will likely assist in targeting individualized, appropriate treatments.
As a final remark, it should be also highlighted that the finding of the role of any circulating factors in the NAFLD pathogenesis and evolution could open new frontiers for the early diagnosis of the disease and for the prognosis of patients. In this context, circulating EVs could represent the ideal candidate and would deserve further study.