Carbon nanotubes (CNTs) are considered a promising nanomaterial for diverse applications owing to their attractive physicochemical properties such as high surface area, superior mechanical and thermal strength, electrochemical activity, and so on.
- Crabon nanotubes
- Agricultural
- Biomedical
1. Introduction
Table 1. CNTs have been recognized as an attractive material that can be utilized in a variety of fields depending on mechanical [3], electrical [4], and thermal characteristics [5]. CNTs are well suited for biological applications, where a high aspect ratio is required [6]. Initially, CNTs’ works were primarily focused on electronic devices [7], displays [8], transistors [9][10], and so on, using the electrical characteristics of this nanomaterial. However, CNTs are considered a suitable material for several applications, ranging from biomedical [11] to agricultural technology [12][13][14]. The possible applications of CNTs are shown in
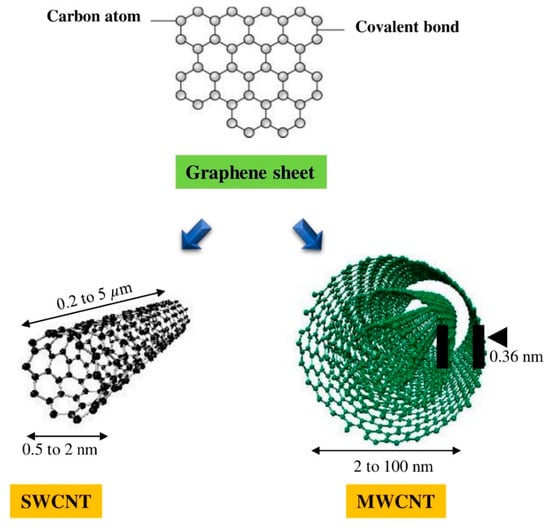
Figure 1.
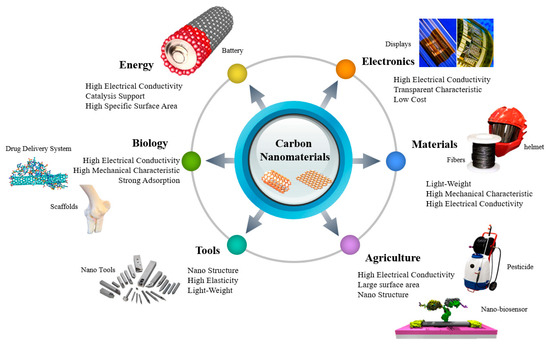
Figure 2.
Table 1. Comparative study between single-walled carbon nanotubes (SWCNTs) and multi-walled carbon nanotubes (MWCNTs) [2].
Figure 3b,c.
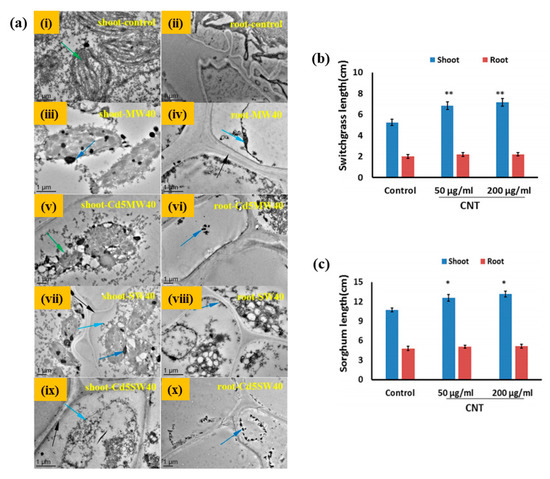
Figure 34.
a
i
v
iv
vii
ix
iii
vi
vii
viii
x
iv
vii
ix) [29]. (
ii
b
c
p
p < 0.01) [31].
Table 13.
|
Type of CNMs |
Plant |
Treatment |
Effect |
Reference |
|||||||||||||||
|
MWCNTs and oxidized MWCNTs (o-MWCNTs) |
Brassica juncea (mustard) seeds |
23 × 10−3 and 46 × 10−3 mg/mL of MWCNTs for 5 and 10 days and 2.3 × 10−3 and 6.9 × 10−3 mg/mL of o-MWCNTs for 5 and 10 days, respectively |
After 10 days, seedlings treated with low concentration of o-MWCNTs developed the highest shoot (4.2 cm) and root (5.8 cm) length. Seeds treated with a low concentration of MWCNTs also showed shoot about 1.5 times and root about two times longer than original seeds |
[20][64] |
|||||||||||||||
[102] |
Fullerol and MWCNTs |
Tomato seeds |
|||||||||||||||||
SWCNTs | Ethoprophos, terbufos, thiometon, tefluthrin, iprobenfos, vinclozolin, octachlorodipropyl ether, isofenphos, phenthoate, chlorfenapyr, propiconazol, Ethyl-p-nitrophenylthionobenzenephosphonate (EPN), and λ-cyhalothrin |
50 mg/L and exposure ranged from 0 to 60 minutes (0, 5, 10, 30, or 60 min) |
Teas | (green tea, oolong tea, white tea, and flower tea) |
When exposed for a short period of 5 min, the germination rate was higher than that of the control group and showed no harm to germination |
- | 75–118 |
[58] |
[103] |
||||||||||
SWCNTs | Hexachlorcyclohexan, dichlorodiphenyldichloroethylene, dichlorodiphenyldichloroethane, and dichlorodiphenyltrichloroethane | Lake water | 2 g | 88–111 |
[59] |
[104] |
|||||||||||||
CNTs–silicon dioxide | Diazinon, fenthion, parathion, and chlorpyrifos | River water and agricultural wastewater, pear, grape, and eggplant | 50 mg | 79–99 |
[60] |
[105] |
2.2. Energy and Environmental Applications
Works on CNTs in the field of bioresources are being studied as a material capable of overcoming the limitations of existing carbon materials or improving performance by using the high electrical conductivity of CNTs. As CNTs showed a high specific surface area, much research has been conducted into CNTs as an adsorbent for the removal of different contaminants such as Zn
2.2. Energy and Environmental Applications
2+
2+ [64]. CNTs nanocomposites have a wide range of applications depending on the type and combination of the target materials. Here, we have briefly described the nanotechnological applications of CNMs-based materials, including the battery, wastewater treatment, fuel cell, and energy storage, by considering some attractive works.
4.2.1. Battery
Despite the rapid development of lithium-ion batteries, which have high power and energy density properties [65], numerous reports have focused on the application of CNTs for the energy sector [66][67][68]. The energy efficiency of CNTs is intensely affected by the synthesis method, shape, and structure. Maurin et al. showed that lithium was intercalated between the graphene layers of the MWCNTs prepared by arc discharge using micro-raman spectroscopy [69]. CNTs produced by the arc discharge method had a reversible capacity of 125 mA·hg
−1 at a low current density [70], which has limited the practical application in lithium-ion batteries to some extent [71]. However, CNTs synthesized by chemical vapor deposition (CVD) showed the high reversible capacity of 340–640 mA·hg
−1 at a low current density [72][73][74]. A comparative study was performed by Yang et al. using short CNTs (S-CNTs) and long CNTs (L-CNTs) synthesized through co-pyrolysis, as well as the CVD method, respectively, to evaluate the reversible capacity of both samples at a low current density. The reversible capacity of S-CNTs anode material was 266 and 170 mA·hg
−1
−2, respectively, which were twice that of L-CNTs anode materials. The surface film and charge-transfer resistant of S-CNTs anode materials were 1.7 Ω and 3–4 Ω, respectively, which is much lower than the L-CNTs (14 Ω, and 31.2–61.2 Ω) anode materials, indicating higher electrochemical activity [75]. The holes in the graphene sheet allow lithium to diffuse better inside the CNTs and increase the capacity. The conductive SWCNTs were able to store about five times more lithium ions than semiconducting SWCNTs [76]. The high conductivity of CNTs also provides enhanced electron transfer with nanostructured anode material [77]. However, long-term stability has remained a challenging task. The electrochemical performance is highly dependent on the nanostructure, shape, and surface properties [78][79][80][81][82]. Lee et al. have developed CNT–Si composite anode with extremely stable long-term cycling and a discharge capacity of 2364 mA·hg
−1
−3
0.85
0.05
0.10
2
−1 with excellent capacity retention for 500 cycles at 1C [83]. The electrochemical performances of CNTs-based Li-ion batteries are given in
Table 46.
|
CNMs |
Method |
Current Density |
Initial Discharge Capacity (mA·h/g) |
Cycles |
Residual Reversible Capacity (mA·h/g) |
Reference |
|
CNTs–SnSb0.5 |
CVD |
50 mA/g |
549 |
30 |
369 |
[67][111] |
|
CNTs–LiCoO2 |
CVD |
0.2 C |
118 |
[21][65] |
||
|
Single-walled carbon nanohorns (SWCNHs) |
Barley, Corn, Rice, Soybean, Switchgrass, Tomato |
25, 50, and 100 μg/mL for 2 and 6 days |
The highest germination rate was recorded for barley, corn, rice, and switchgrass seeds exposed to 100 μg/mL SWCNHs and the highest germination rate was observed at 25 μg/mL SWCNHs in tomato seeds |
[22][66] |
||
|
MWCNTs |
Broccoli |
10 mg/L MWCNTs, 100 mM NaCl, and 100 mM NaCl + 10 mg/L MWCNTs |
| SWCNTs | MWCNTs |
|---|---|
| Single layer of graphene | Multiple layer of graphene |
| Expensive | Cheaper |
| Thermal conductivity in the range of 6000 W/m·K | Thermal conductivity in the range of 3000 W/m·K |
| Semiconducting and metallic properties (excellent field emission capability) | Low physical properties |
| Bulk synthesis is difficult | Easy to synthesis in bulk |
| Easily twisted | Difficult to twist |
| Catalyst needed for synthesis | Manufactured without catalyst |
| Low purity | High purity |
| Less accumulation body | Greater accumulation in body |
| More defection during the functionalization | Less defection, but hard to improve |
2. Application of CNTs
2.1. Agriculture Applications
The unique properties of nanomaterials such as small size, large surface area, and reactivity provide excellent opportunities for its use in the agricultural sector. The foremost applications of CNTs in the agricultural field include seed germination, early plant growth, pesticides, and biosensor diagnostics and analysis. The potential toxicity of nanomaterials has not yet been widely investigated [16][17][18]. Here, we described the potential utilization of CNTs in the agricultural sector by considering some selected, but significant works.
2.1.1. CNTs in Plant Growth
The applications of the nanomaterials as a promoter for plant and crop growth have received a significant amount of interest from the scientific community. It has been noted that CNTs can penetrate the thick seed coat and activate the water uptake process, which might be responsible for rapid seed germination and early growth [19]. Mondal and coworkers measured the seeds germination rate of
Brassica juncea
50 (time for 50% germination), was noted in the presence of a low concentration of oxidized MWCNTs compared with the control. They observed that the moisture content was significantly high in oxidized MWCNTs-treated seeds than in the untreated condition, indicating that oxidized MWCNTs facilitated the water-absorbing potential of the seeds for rapid regeneration. The high water content in oxidized MWCNTs-treated seeds was the result of the easy penetration ability of these functionalized CNTs. However, the exact mechanism for the rapid growth of seeds in oxidized MWCNTs is still unclear. It is well known that aquaporins facilitate the water uptake inside the cells. The efficiency of aquaporin is profoundly affected by several factors like pH; concentrations of the heavy metal ions; osmotic pressure; and water channel expression genes such as plasma membrane intrinsic protein (PIP), small basic intrinsic protein (SIP), and so on. Aquaporin also reduces the flow of different ions through membranes and controls the electrochemical potential of the membrane. This potential of aquaporins is expected to be the key reason for the rapid regeneration of seeds in the presence of oxidized MWCNTs [20]. Several studies have been done to explore the effects of the various carbon nanomaterials (CNMs), including MWCNTs, fullerenes, and carbon nanohorns on different plants such as tomato, rice, cucumber, onion, radish, corn, soybean, switchgrass, and broccoli [21][22][23][24][25][26][27]. It was noted that 50–100 mg/L concentrations of CNMs are sufficient to penetrate the seeds for fast germination and growth rates [21][22]. Various factors such as size, shape, surface structure, solubility, and concentrations, as well as the presence of the functional groups, have significant contributions towards the toxicity and pathology caused by CNTs in the germination of seeds [17][28]. Functionalized carbon nanotubes (F-CNTs) also have an important aspect of being used as a nanomaterial to alter the seed germination and growth rates. Chang and coworkers have evaluated the toxic effects of CNTs (SWCNTs and MWCNTs) combined with cadmium (Cd) on wheat seedling growth. A significant reduction in total root length, root surface area, average root diameter, numbers of root hairs, and the dry weight of shoots and roots was observed in Cd-combined CNTs treatment groups than with Cd, as well as SWCNTs and MWCNTs treatment, indicating that Cd-combined CNTs remarkably inhibited wheat growth and development. Furthermore, a decrease in tubulins in the root was also noted. However, an enhancement in glutathione S-transferase and cytochrome P450 in the shoots and roots was observed in Cd-combined CNTs treatment groups, suggesting the improved defense ability of wheat seedling. It was interesting to see that the accumulation of Cd in shoot and root tissues was profoundly affected by the concentrations of CNTs. These results suggested that CNTs facilitated the toxicity of Cd to the wheat seedling. Therefore, the toxicity of CNTs should be remarkably considered with food security in the future with exposure of crops to Cd [29]. Transmission electron microscopy (TEM) morphologies of wheat plant cells under different conditions are shown in
Figure 3a. The results indicated that CNTs had the potential to destroy the cell structure, and Cd highly influenced this ability. A comparative study has been done by Cano and coworkers to evaluate the effects of CNTs at various concentrations (0, 10, and 100 mg/kg) for the germination and growth of corn seeds. For this, they have taken pure SWCNTs, OH-functionalized, and surfactant stabilized SWCNTs [30]. The microwave-induced heating approach was explored to determine CNTs in different parts of the germinated seeds. They noted that the accumulation of F-CNTs in roots, stems, and leaves was independent of the functional groups present in CNTs, but dependent on the volume and composition of the soil. No significant difference in the plant physiological stress was observed between SWCNTs and the control. The effects of CNMs on plant and crop growth are also summarized in
Table 1. Bioenergy crops are a suitable candidate for use in energy production. For bioenergy applications, plants should produce a high amount of biomass and resist adverse environmental conditions. The effects of CNMs on seed germination, biomass accumulation, and salt stress response of bioenergy crops (sorghum and switchgrass) were studied by Pandey et al. [31]. A significant enhancement in the germination rate was observed in CNTs-treated crops compared with the control, indicating the positive effect of nanomaterial towards crop growth. Approximately 73.68% and 31.57% enhancement in shoot biomass was noted in switchgrass seedlings with the exposure of CNTs for 10 days at concentrations of 50 and 200 μg/mL, respectively. A significant reduction in salt (NaCl)-induced stress symptoms was noted in CNMs-treated media compared with the control, demonstrating that CNMs have the potential to protect the plants against salt-induced stress in the saline growth medium. The effects of CNTs on the growth rate of switchgrass and sorghum seedlings at different concentrations after 10 days of exposure are given in
20 | 118 | [68][112] |
||||
|
CNTs |
arc discharge |
2 C |
300 |
300 |
255 |
[70][114] |
The MWCNTs-treated plants had positive effects on growth compared with the control and NaCl alone application | [ | 23][67] |
||||
|
MWCNTs |
Barley, Soybean, Corn |
25, 50, and 100 μg/mL for 2 and 6 days |
After six days, all seeds treated with MWCNT reached a germination rate of 100% compared with control seeds reaching a germination rate of 63% |
[24][68] |
. Zhang et al. have fabricated Ti/SnO
2-Sb-CNTs electrodes for anodic oxidation of dye-containing wastewater through the pulse electrodeposition method. The CNTs-modified electrode exhibited a larger surface area compared with that without CNTs, which provides a more active area for electrochemical oxidation of organic pollutants. The CNTs-modified electrode was 4.8 times more durable compared with that without CNTs. The modified electrode has a higher kinetic rate constant, chemical oxygen demand (COD), total organic carbon (TOC) removals, and current mineralization efficiency, which are 1.93, 1.27, 1.26, and 1.38 times higher, respectively, than those of the unmodified electrode. The CNTs-based electrode exhibited 1.15 times more permeation flux compared with the electrode without CNTs [92]. The electrochemically activated CNTs filters were developed for wastewater treatment [93]. Thus, the solutions for implementing water reuse, seawater desalination, and water purification more efficiently and cost-effectively are expected to emerge from the use of nanotechnology with CNTs. The applications of CNMs in wastewater treatment are also summarized in
Table 5. It was noted that phenolic compounds are often explored in the commercial manufacturing of several products such as resins, polymeric materials, ion exchange resin, dyes, drugs, and explosives, among others. Owing to the extensive uses of phenolic products, a large amount of phenol is discharged from industries in the water, which causes toxicity and can damage the cellular proteins. Therefore, the removal of phenolic compounds from the contaminated water on a large scale is necessary for a healthy life. For this, CNTs with rich pore structure, analytic abilities, high surface area, and sharp curvatures show great potential for the removal of the phenolic compounds from the contaminated water through π–π, electrostatic, hydrophobic, and hydrogen bonding interactions [94]. Ma et al. have prepared CNTs/Fe@C hybrids material for the removal of the binary dye from the contaminated water through the one-pot method with a high specific surface area (186.3 m
2
Figure 8a. Cooperative adsorption was noted in the MB–MO dye system through the developed hybrids material. An enhancement in the adsorption capacity was observed in the MB–MO dye system by 30% and 35%, with a decrease in the equilibrium time by 25% and 50%. Meanwhile, the MB–NR dye system exhibited a competitive adsorption tendency. The adsorption isotherm of MO and MB from the prepared hybrids material is shown in
Figure 8b. These results suggested that the prepared hybrids had the efficiency to be used as a promising adsorbent for the large-scale applications in binary dye systems, which exhibited a cooperative and competitive adsorption tendency to address the dye pollution effectively [95]. Lee and coworkers fabricated MWCNTs-based polyaniline (PANi)/polyether sulfone (PES) membranes by in situ polymerization of aniline in the presence of MWCNTs for the effective removal of natural organic matter (NOM) in water. The fabricated membranes exhibited 30 times greater efficiency than the PES membrane. This enhancement was attributed to the synergistic effects of the MWCNTs/PANi complex. The electrostatic interactions between the membrane surface and NOM facilitate the adsorption capacity of the developed membrane. The fabricated membrane exhibited 100% water flux recovery and 65% total fouling ratio after treatment with 0.1 M HCl/0.1 M NaOH solution for 1 h [96]. The extending exploration of SWCNTs raises environmental concerns. Qu et al. have evaluated the microbial communities’
(Zoogloea
Rudaea
Mobilicocus
Burkholderia, Singulisphaera
Labrys
Mucilaginibacter) responses of SWCNTs in phenol containing wastewater media. The enhancement in the phenol removal rates was observed in the SWCNTs-treated batch in 20 days initially. However, as the phenol concentrations increased to 1000 mg/L after 60 days, a decrease in the phenol removal rate was noted even at the higher concentration of SWCNTs (3.5 g/L). It was noted that SWCNTs protected the microbes from inactivation by generating more bound extracellular polymeric substances (EPSs), which form a protective layer for the microbes. A significant decrease in the bacterial community structure was observed after the addition of SWCNTs. This phenomenon is associated with the change in sludge settling, aromatic degradation, and EPS generation. These results demonstrated that SWCNTs exhibited the protective response for sludge microbes in phenol containing wastewater media and enabled the important information related to the potential effects of SWCNTs on wastewater treatment processes [97].
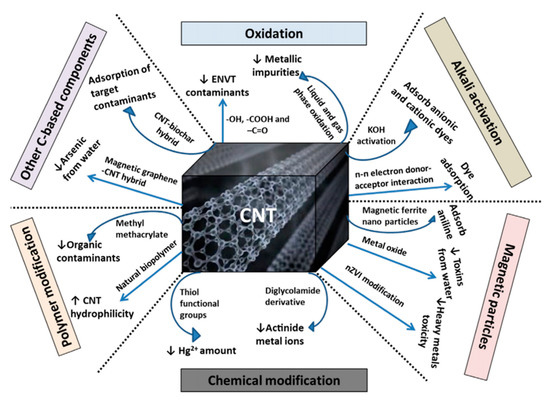
Figure 78. Schematic diagram representing different modification processes of CNTs for contaminant removal from water and wastewater (C: carbon; CNT: carbon nanotube; ENVT: environmental; Hg: mercury; KOH: potassium hydroxide) [85].
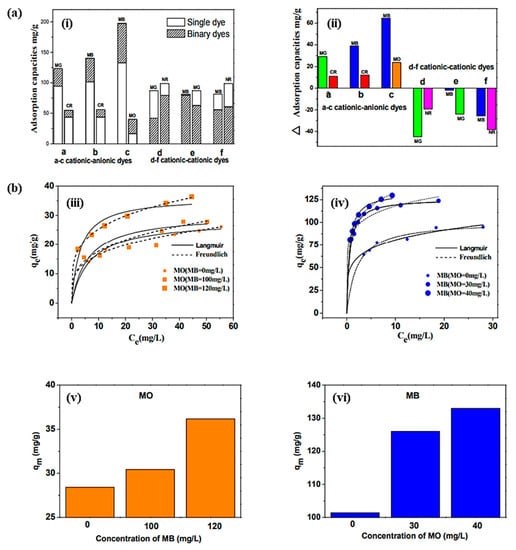
Figure 89.
a
i
ii
b
iii
iv
v
vi) increased with the concentration of the other dye [95].
Table 57.
|
Applications |
Desirable Nanomaterials Properties |
Type of CNMs |
Efficiency of the CNMs |
Reference |
|||||
|
Catalysts |
Higher catalyst loads and stability, stronger metal–support interactions, high dispersion, high stability and activity, low cost |
Ruthenium/MWCNT-COOH-Na2CO3 |
98.3% and 70.3% aniline and total organic carbon (TOC) removals |
[84][128] |
|||||
|
Ruthenium/MWCNT-COOH |
89.9% and 53.7% aniline and TOC removals |
[84][128] |
|||||||
|
Mass Transfer |
Facilitate contaminant mass transfer, large surface areas, high electrochemical efficiency, degrade organics with much higher current Efficiency and lower energy consumption |
CNTs |
The efficiency was 340–519% higher than the conventional reactor, and the energy consumption was only 16.5–22.3% of the conventional reactor |
[87][131] |
|||||
|
MWCNTs |
arc discharge |
0.2 mA cm−2 |
117 |
||||||
|
Adsorption |
Large specific surface areas, high chemical and thermal stabilities, high aspect ratios, exceptional mechanical strength, diverse contaminant–CNT interactions |
SWCNTs, MWCNTs | 30 |
The maximum zinc adsorption capacities of SWCNTs and MWCNTs were 43.66 and 32.68 mg/g, respectively, in the initial zinc ion concentration range (10–80 mg/L) |
[90] |
113 |
[134] |
[71][115] | |
|
MWCNTs |
Tomato plants |
50 and 200 μg/mL |
The CNT-treated tomato plants produced twice as many flowers as the control plants |
[25][69] |
|||||
|
CNTs |
Rice |
50, 100, and 150 μg/mL |
CNTs at appropriate concentrations (~100 μg/mL) promoted rice seed germination and root growth |
[26][70] |
|||||
|
SWCNTs and functionalized SWCNTs |
Cucumber, Onion |
28, 160, 900, and 5000 mg/L for 2 and 3 days |
Non-functionalized CNTs enhanced root elongation in onion and cucumber, the effects were more pronounced at 24 h than at 48 h |
[27][71] |
|||||
|
SWCNTs (non-functionalized, OH-functionalized, or surfactant stabilized) |
Corn |
0, 10, and 100 mg/kg |
Root length was significantly higher in plants exposed to non-functional SWNT 100 mg/kg and plant root uptake also followed the trend of non-functionalized > surfactant stabilized > OH-functionalized |
[30][74] |
The biosensor is a device that quantitatively measures the molecules reacting in a solution having analytes to be measured by utilizing their reacting properties with a specific substance. The excellent physicochemical potentials make CNMs an ideal material for sensing applications to detect the pathogens [32][33]. In comparison with the commercially available sensors such as metal oxides, silicon, and so on, CNTs-based biosensors have significant advantages, such as high sensitivity (large surface area ratio), excellent luminescence properties, fast response time, and high stability [34]. Different types of sensors are explored for monitoring the pollutant/species present in the medium. Biosensors are utilized to detect compounds such as aromatic and organic compounds and halogenated pesticides. Solid-state electrochemical sensors are suitable for the chemical gas sensor from their sensitivity, reproducibility, and power consumption. The basic principle of a biosensor for soil diagnosis is to determine the relative activity of favorable and unfavorable microbe’s presence in the soil based on differential oxygen consumption owing to respiration. Surface plasmon resonance (SPR) phenomenon is also explored for the development of the biosensor from metallic nanoparticles [35]. Nano-biosensors are being rapidly explored in the agricultural sector and food processing. CNTs-based optical sensors were developed to monitor the real-time detection of pathogenic bacteria [36], organophosphate chemical warfare agents and pesticides [[37], toxic materials, and proteins [38]. The one-dimensional (1D) properties of CNTs facilitate the ultrasensitive detection of analyte because all atoms are surface atoms, and minor perturbations in the chemical environment can dramatically change the electrical or optical properties [39]. This property plays a vital role in the monitoring of the optical sensor under various circumstances [40]. Among different biosensors, electrochemical biosensors are the most popular because of their excellent conductivity and electro-catalysis, high surface, and volume ratio [41]. The transfer of the electrons occurred in these biosensors [42][43][44]. CNMs have the potential to improve the response characteristics and can act as the immobilization matrices for the bio-receptors [45]. A significant decrease in the response time was observed in MWCNTs-coated electrodes used as a sensor [46]. An enhancement in the detection limits was noted in Au-MWCNTs nanocomposite, and it can detect concentrations up to 0.1 nM [47]. Enzymes are considered as a suitable substrate for the development of the biosensors. CNTs have been utilized as a support for the immobilization of enzymes in nanostructured devices. Scholl and coworkers have developed the thin film of CNTs for enhancing the enzymatic potential of penicillinase for biosensing applications. The presence of CNTs in the developed film not only altered the catalytic potential of penicillinase, but also facilitated their enzymatic activity. ConCap responses curves for penicillin G detection through the fabricated films are shown in
Figure 4. Recently, Yang et al. [48] have reported a composite skin patch with a high-performance flexible sensor consisting of Ag/CNT/PDMS for monitoring of the heartbeat as well as breath during active labor (
Figure 5). Owing to the presence of CNTs, the wrinkled patch is highly sensitive and conductive. This could potentially be used in prophylactic medicine for monitoring of fever or hyperthermia caused by specific pathogens. The biosensors developed with CNTs indicate regular steps of the distinct output signal for all concentration ranges compared with the control. These changes may directly influence the potential and performance of the developed sensor in terms of their sensitivity and coefficient of determination (R
2) [49].
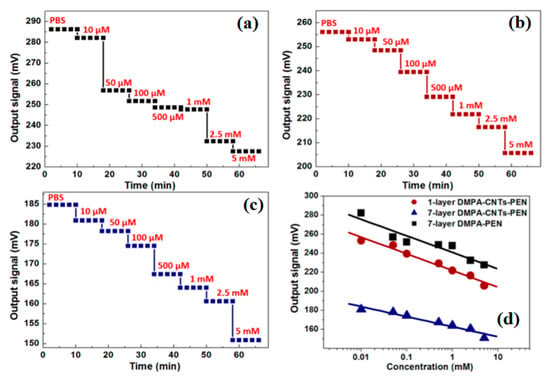
Figure 45.
a
b
c
d) Calibration curve of ConCap responses for the LB films correlated with penicillin concentrations [49].
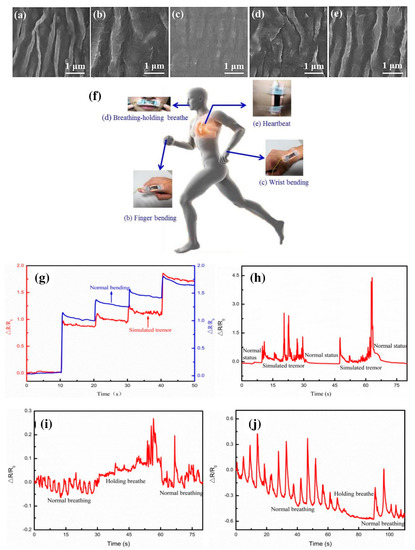
Figure 56.
a
e
f
g
h–j) Signal from wrist, upper lip, and chest showing the significant change in peak [48].
2.1.3. Pesticide Analysis
The high adsorption properties of CNTs are utilized for extraction techniques such as solid-phase extraction (SPE) and solid-phase micro-extraction (SPME) [50]. SPE technology is one of the most widely used extraction methods for environmental, food, and biological sample pretreatment. Several studies have been done showing the potential of MWCNTs as a promising adsorbent for the pre-concentration of cobalt, nickel, and lead ions [51][52]; organophosphate (OP) pesticides [53]; and chloro-phenols [54]. The recoveries of the analyte were also altered by the amount of MWCNTs and the treatment conditions, indicating that, by varying the sample conditions, they could be extended to other analytes and other types of food samples [55]. SPE sorbent, based on nanoparticles, demonstrates the potential for adequate enrichment and sensitive analysis of metal ions in a variety of media [56][57]. The effects of the CNMs in the SPE technique are also given in
Table 2. An enhancement in the extraction efficiency was noted in SWCNTs- or MWCNTs-coated SPME fiber. The development of fiber coating technology for high-efficiency extraction of the analyte is considered an exciting research direction in SPME [58]. Higher extraction efficiency, precision, and accuracy were observed in SWCNTs-coated fiber from the targeted samples [56]. It has been noted that CNTs-coated fibers have more extraction efficiency than the commercially available PDMS [59][60]. Saraji et al. synthesized CNTs/SiO
2 nanohybrids for SPME coating and evaluated their extraction efficiency for some organophosphorus pesticides (OPPs) in vegetables, fruits, and water samples [61]. Gas chromatography-corona discharge ion mobility spectrometry was applied for the detections of the OPPs. Significant enhancement in the adsorption capacity and mass transfer rate was observed in CNTs/SiO
2-coated SPME compared with the commercial SPME fibers (PA, PDMS, and PDMS-DVB), indicating their improved extraction efficiency. For water samples, the detection limits range was 0.005–0.020 μg/L, and the quantification limits were 0.010 and 0.050 μg/L, with excellent linearity in the range of 0.01–3.0 μg/L for the samples. The spiking recoveries range was from 79 (±9) to 99 (±8). Therefore, the developed materials have the potential and can be applied for the analysis of OPPs in real samples [62]. The influence of the CNMs in the SPME technique is also summarized in
Table 3. Feria and colleagues have determined the presence of different types of pesticides in virgin olive oils using MWCNTs and carboxylated c-SWCNTs. It was interesting to note that the c-SWCNTs exhibited better sorbent capabilities than those of MWCNTs owing to the presence of carboxyl functional groups in their structure, which facilitates better interactions between pesticides and CNTs. A comparison of the performance of c-SWCNTs and MWCNTs for the detection of different pesticides from virgin oil samples is shown in
Figure 6a. The bar diagram demonstrates the better sorbent potential of c-SWCNTs than MWCNTs for different kinds of pesticides from the selected samples owing to the presence of the different functional groups. The effect of the number of c-SWCNTs (10 and 50 mg) on the analytical signal for different pesticides is shown in
Figure 6b. An enhancement in the peak area was observed by increasing the number of c-SWCNTs for all analytes up to 30 mg. Furthermore, a decrease in the peak value was noted after a 30 mg dose of c-SWCNTs owing to non-quantitative elution of the retained analytes [63].
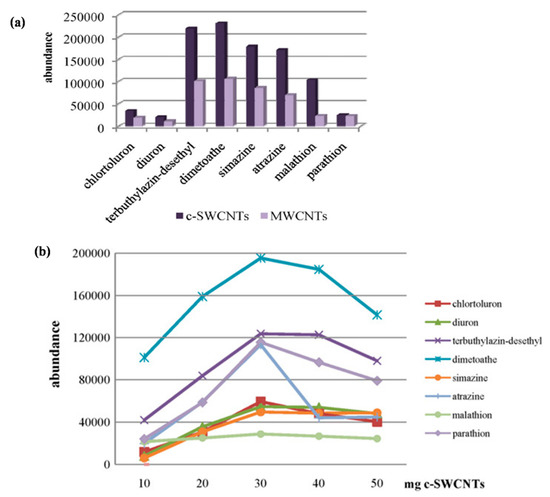
Figure 67.
a
b) Influence of the amount of c-SWCNTs packed in the solid-phase extraction (SPE) cartridge for the preconcentration of the selected pesticides from virgin olive oil samples [63].
Table 24.
|
CNTs |
Analyte |
Sample |
CNTs amount (mg) |
Recovery (%) |
Reference |
|
MWCNTs |
Disulfoton sulfoxide, ethoprophos, disulfoton, terbufos sulfone, cadusafos, dimethoate, terbufos, chlorpyrifos-methyl, fenitrothion, malaoxon, pirimiphosmethyl, malathion, chlorpyrifos, disulfoton sulfone, and fensulfothion |
||||
[ | |||||
] | |||||
[101] | |||||
Table 35.
CNTs | Analyte | Sample | CNTs amount | (mg) | Recovery | (%) | Reference | ||||||||||||||
Water |
(run-off, mineral, and tap water) |
130 |
67–107 |
[50][94] |
|||||||||||||||||
|
GO–MCNTs-diethylenetriamine | |||||||||||||||||||||
MWCNTs | Polybrominated diphenyl ethers (PBDEs) | River water, waste water, milk | 20 mg | 90–119 |
[57] |
Cr(III), Fe(III), Pb(II), and Mn(II) ions |
Wastewater |
30 |
95 |
[51][95] |
|||||||||||
|
MWCNTs |
Organophosphate |
Garlic |
1.2 |
97–104 |
[53][97] |
||||||||||||||||
|
MWCNTs |
4-Chlorophenol, 3-chlorophenol, dichlorophenol, trichlorophenol, and pentachlorophenol |
River water |
300 |
93–117 |
[54][98] |
||||||||||||||||
|
MWCNTs |
Tolclofos-methyl, fenitrothin, malathion, phorate, diazinon, isocarbophos, and quinalphos phenamiphos |
Peanut oil |
100 |
86–115 | |||||||||||||||||
[ | ] | [99] |
|||||||||||||||||||
|
Short CNTs |
|||||||||||||||||||||
|
Flocculation |
CVD |
Exceptional adsorption capabilities and efficiencies, larger surface area, affinity towards target compounds |
0.8 mA cm−2 |
CNTs | 491 |
Demonstrated the ability to successfully coagulate colloidal particles in the brewery wastewater |
30 |
[91][135] |
170 |
[75][119] |
|||||||||||
|
MWCNTs |
Fe |
Ethoprophos, diazinon, fenitrothion, malathion, and phosmet |
2O3/CNT–graphene foam |
Agricultural soil, forestal soil, and ornamental soil |
CVD | 100 |
200 mA/g |
1190 |
|||||||||||||
|
Electrode |
Effective compound adsorption and oxidation, high energy efficiency, fast reaction rate, electrochemical oxidation | 10 |
900 |
[ |
Ti/SnO254–91 | 78][122] |
|||||||||||||||
-Sb-CNT electrode | 80.12% and 46.01% COD and TOC removals |
[92][136] |
CNTs–cobalt oxide |
|
0.1 C |
1250 |
100 |
530 |
[81][125] |
||||||||||||
|
Zn2SnO4/CNT |
|
100 mA/g |
1925.4 |
30 |
703.8 |
[82][126] |
2.2.2. Wastewater Treatment
Nanotechnology plays a vital role in water purification. CNTs can be used for the purification of wastewater [84]. Adsorption and degradation/detoxification is the key strategy for the removal of contaminant from the samples through CNTs. The functionalization of the material can improve the efficiency of CNTs for contaminants. It is possible to target a specific contaminant through the well-modified CNTs. A schematic representation of CNTs’ modifications for the removal of contaminant from water and wastewater is shown in
Figure 7. Design or modification of CNTs’ properties may also assist in the separation of materials following the contaminant treatment process. Nanoparticle separation is facilitated by incorporating a magnetic component into CNTs [85]. It is easy to control the potential and current in the electrochemical technique for wastewater treatment [86][87]. Yang et al. have used a seepage carbon nanotube electrode (SCNE) reactor to improve the electrochemical wastewater treatment efficiency. The innovative concept behind the reactor design was that the overall mass transfer would be significantly improved via contaminant migration through the porous carbon nanotube electrode. The current efficiency of the SCNE reactor was 340–519% higher than those of the conventional reactor, and the energy utilization to mineralize the equal weight of organic content was only 16.5–22.3% of the conventional reactor. The developed reactor has the potential for application in wastewater treatment [88]. The electrocoagulation is also useful for removing effluents from the polluted water [89]. These applications utilize the advantages of CNTs’ properties such as high reactivity, strong adsorption, and high specific surface area [89][90][91]
2.2.3. Microbial Fuel Cells (MFCs)
Microbial fuel cell (MFC) technology produces hydrogen or electrons by a bacterial oxidizing process from substances such as wastewater. This is the basic concept of generating electricity through an anode–cathode system. For this, the cathode should have excellent compatibility with microorganisms and possess a large specific surface area per unit volume, as well as excellent durability as chemically safety materials [98]. CNTs have received much attention for cathodic applications owing to their superior and tunable physiochemical potential. The electronic signal is also affected by the temperature of the medium [99]. The high limiting current density and electrochemical performance were observed in the deformed CNTs owing to the higher specific surface area generated by deformation [100]. The modification is required in CNMs to achieve the proper catalytic surface area for better electrochemical performance [101][102][103]. The change in the aspect ratio and surface area of CNTs was performed using a metal catalyst such as platinum (Pt) [104][105][106]. The CNTs/Pt composites exhibited better powder density (~8.7% higher) than the pure Pt catalyst when the chemical oxygen content of the substrate reached 100 mg/L. The significant enhancement in the electrical properties was observed in nitrogen-doped CNTs [107]. The nitrogen-doped CNTs exhibited a maximum power density of 1600 ± 50 mW·m
2.2.3. Microbial Fuel Cells (MFCs)
−2, which is significantly higher than the commonly used Pt catalyst for cathode application. For a better reaction process, the surface area and durability of the anodic materials should be high [108][109]. The CNTs/polyaniline (PANi) composites showed an enhanced electrochemical activity at a higher content of CNTs in the medium [110]. The CNTs-coated anode demonstrated ~62% higher voltage output than the untreated anode [111]. The performance of the anodic materials can also be improved by using the three-dimensional (3D) structure of graphene oxide (GO)/CNTs and melamine sponge composites [112]. The 3D graphene oxide (GO)/CNTs and melamine sponge display the highest electrochemical performance at a thickness of 1.5 mm. The porous structure facilitates the biocompatibility of the composites. These results provide valuable insights into the active anode–cathode development for MFC applications. The effects of the carbon-based electrode on MFCs are also given in
Table 68.
|
Type of electrode |
MFCs |
Type of MFCs |
Effect |
Power Density (Max.) mW/m |
Reference |
|
|
Anode |
Cathode |
|||||
|
Graphite fiber |
Carbon nanotube/Pt |
Effluent from an air-cathode MFC |
Single chamber MFCs |
|||
Table 7. The hydrogen storage ability of CNTs is shown in
Figure 9. The interaction energy plays a vital role in the storage of hydrogen. The results indicated that CNTs could effectively store hydrogen under cryogenic conditions, which is not suitable for mobile applications. This is because of the reduced interaction energy (1 kcal/mol) between hydrogen and the CNTs. For significant, but reversible storage under ambient conditions, the interaction energies should be around 7 kcal/mol. The interaction energy can be tuned by doping with heteroatoms or by incorporating light metal ions in CNTs [130].

Figure 910.
Table 89.
|
CNMs |
Storage (wt.%) |
Temperature (K) |
Pressure (MPa) |
Reference |
||||||||||
|
CNTs |
9.6 |
77 |
10 |
[115][159] |
||||||||||
The cathode had a maximum power density of about two times higher than that of the carbon cloth cathode | ||||||||||||||
|
CNTs |
1.5 | 329 |
296 |
12.5 |
[116][160] |
[100][144] |
||||||||
|
Carbon paper |
CNTs/ Poly-pyrrole |
Anaerobic digester sludge collected from Indah Water Konsortium treated Palm oil mill effluent (POME) |
||||||||||||
|
SWCNTs | Two cubic shaped chambers |
COD removal of the system using CNT/PPy was 96% |
4.5 |
77 |
6 |
[117][161] |
113.5 |
[101][145] |
||||||
|
Carbon cloth |
N-CNTs on carbon cloth |
Acetate-laden synthetic wastewater |
Air-cathode cylindrical-shaped MFCs, dual chamber |
The maximum power density was about 9% higher than that of Pt-carbon on carbon cloth |
135 |
[103][147] |
||||||||
|
Chemically activated carbon |
5.6 |
77 |
4 |
[118][162] |
Carbon paper |
|||||||||
|
Carbon with boron |
CNTs/Pt |
5.9 | Palm oil mill effluent (POME- Selangor, Malaysia) sludge |
298 |
Two cylindrical H-shaped chambers |
10 |
The composite electrode increased the power output of MFC by 8.7~32% compared with Pt electrode |
169.7 |
[119][163] |
[105][149] |
||||
|
Carbon paper |
Chemically activated carbon nanofibers |
|||||||||||||
|
SWCNTs | Palm oil mill effluent (POME) anaerobic (Selangor, Malaysia) sludge |
1.73 |
Two cylindrical and H-shaped chambers |
COD removal was approximately 82.3% and could generate up to 3.17 times more power than carbon paper |
61.3 |
[106][150] |
||||||||
77 |
10 |
[120][164] |
Carbon fiber |
Nitrogen-doped CNTs (N-CNTs) |
20% domestic wastewater collected from a municipal wastewater treatment plant of Shanghai, China |
Air-cathode single chamber MFCs |
The power density drop rate was low, so electricity can be produced more permanently than the platinum catalyst |
1600 ± 50 |
||||||
|
SWCNTs-SnO2 |
2.4 |
623 | [ |
5 |
[121]][151] | |||||||||
[165] |
MWCNT/ rGO-biofilm |
carbon fiber brush |
||||||||||||
|
Si-doped SWCNTs |
2.5 |
S. putrefaciens CN32 cell suspension was inoculated on bacteria |
298 |
H-type dual-chamber |
Composite electrodes provide higher maximum power density than individual MWCNTs and rGO |
10 |
[122][166] |
789 |
[108][152] |
|||||
|
Vertically Aligned CNTs |
Cr/Au film |
|||||||||||||
|
Un-doped SWCNTs |
1.4 |
Acetate-fed microbial electrolytic cells (MEC) with Geobacter-enriched bacterial community from anaerobic digestion sludge Dual chamber MFCs; anode and cathode chambers |
298 |
61.3% of Coulombic efficiency |
270 |
10 |
[109][153] |
|||||||
|
Randomly Aligned CNTs |
Cr/Au film |
73% of Coulombic efficiency |
540 |
|||||||||||
|
Spin/spray layer-by-layer CNTs | ||||||||||||||
|
Cr/Au film |
73% of Coulombic efficiency |
540 |
||||||||||||
|
CNTs/polyaniline (PANI) |
Pt |
Bacteria |
E. coli-based MFCs |
Composite electrodes containing 20 wt.% CNTs provide high discharge performance and high power output |
42 |
[110][154] |
||||||||
|
CNTs |
CNTs/Pt |
Bacteria |
Air-cathode MFCs |
COD removal was 95% and the maximum coulombic efficiency was 67% |
65 |
[111][155] |
||||||||
|
rGO-CNT sponges |
- |
Anaerobic sludge |
Aerobic chamber and anoxic chamber |
Produced higher durability |
Max. current density of 335 A m−3 |
[112][156] |
||||||||
2.2.4. High-Efficiency Electrical Devices
For energy applications, it is crucial to increase the energy density of the material without compromising other electrochemical properties [113]. CNTs are not only light in weight, but also have a sufficient area for hydrogen storing in their tubular structure, which can increase the charge storage capacity per unit mass [114][115][116][117][118][119][120][121][122]. CNTs can also be utilized in other electrochemical applications [123][124] and supercapacitor preparation [125][126][127][128]. An increased surface area of CNMs is required for energy applications with pore sizes of 0.7 to 0.9 nm, which are suitable for the ions approach. It has been proved that hydrogen is stored in the pores formed in the space between the tubes, and the adsorbed hydrogen molecules are subjected to a stable surface suction force. Approximately 3.3 wt.% and 0.7 wt.% hydrogen adsorption was noted within the tube (10, 10) and interstitial space of CNTs, respectively [116]. A hierarchical structure is required to obtain the high output characteristics, which are connected in a vast pore region for the fast ion diffusion even at a high current density. The maximum power density can also be improved by using the cetrimonium bromide (CTAB) with CNTs [129]. A porous three-dimensional structure was formed by intercalating the CNTs into graphite in a vertical direction to improve the maximum energy density. A significant enhancement in the maximum energy density was observed in this structure, which was 117.2 Wh/L at a maximum power density of 424 kW/L per volume, and a maximum energy density of 110.6 Wh/kg at a maximum power density of 400 kW/kg per weight. This kind of structure is light in weight, which provides additional advantages to make small portable electronic products such as automobile batteries, rechargeable batteries, and notebook computers. The hydrogen storage capacity of different types of CNTs is given in
