Cancer Survivor (CS), the most widely used definition is: “being a CS, starts on the day of diagnosis and continues until the end of life. Three cancer survivorship phases can be distinguished: “acute survivorship” (i.e., early-stage or time during curative treatment), “permanent survivorship” (i.e., living with cancer or also called the palliative stage), and “extended survivorship” (i.e., cured but not free of suffering). Chronic pain is one of these and occurs in 40% of CSs. Chronic pain is defined by the International Association for the Study of Pain (IASP) as pain that persists or recurs for longer than three month. Unrelieved pain can have considerable adverse consequences on a CSs’ quality of life.
- cancer survivor (CS)
- pain
- stress
- sleep
- diet
- smoking
- alcohol
- physical activity
1. Introduction
2. Pain
3. Lifestyle Behaviour
3.1. Stress
| Lifestyle Factor | First Author, Year Published, Study Type | Included Population | Number of Included Studies (n1) and Participants (n2) | Detail of Lifestyle Factor/Intervention Assessed | Main Results in Context of the Specified State-of-the-Art | Level of Evidence [46] |
|---|---|---|---|---|---|---|
| Alcohol consumption | Leysen et al., 2017, Systematic review with meta-analysis [5] | Breast Cancer Survivors | n1 = 2 (1 CS and 1 C) and n2 = 2519 |
Alcohol use | Alcohol (OR 0.94, 95% CI [0.47, 1.89], p = 0.86, I2 = 67%) was not a predictor for pain, Inconsistent and low evidence |
3b |
| Diet | Kim et al., 2018, Systematic review of systematic reviews [47] | Breast Cancer Survivors with AIA | n1 = 3 (systematic review of RCT), and n2_Omega-3 = 817, and n2_VD = 453 | Omega-3 Fatty Acids, and Vitamin D | Significant effects were found for omega-3 fatty acids (MD −2.10, 95% CI [−3.23, −0.97]), and vitamin D (MD 0.63, 95% CI [0.13, 1.13]) on pain, Low evidence |
1a |
| Yilmaz et al., 2021, Systematic review [48] | Cancer Survivors | n1 = 2 (uncontrolled clinical trial) and n2 = 77 | Nutritional supplements: vitamin C, chondroitin, and glucosamine | Lack of evidence | 2a | |
| Obesity | Leysen et al., 2017, Systematic review with meta-analysis [5] | Breast Cancer Survivors | n1 = 7 (4 CS and 3 C) and n2 = 5573 |
BMI | BMI > 30 (OR 1.34, 95% CI [1.08, 1.67], p = 0.008, I2 = 33%,) was a predictor for pain, Consistent and low evidence |
3b |
| Timmins et al., 2021, Systematic review [49] | Cancer Survivors | n1 = 16 (3 CS, 11 C, and 2 retrospective chart review) and n2 = 14,033 | Obesity | According to the SORT: the association between obesity and CIPN was good-to-moderate patient-centred evidence | 3b | |
| Physical Activity | Boing et al., 2020, Systematic review with meta-analysis [50] | Breast Cancer Survivors with AIA | n1 = 3 (2 RCT, 1 pilot study), and n2 = 118 | Exercise | Significant effect was found on pain (SMD −0.55, 95 % CI [−1.11, −0.00], p = 0.05 I2 = 80%), Low Evidence |
1b |
| Kim et al., 2018, Systematic review of systematic reviews [47] | Breast Cancer Survivors with AIA | n1 = 2 (systematic review of RCT), and n2 = 262 | Aerobic Exercise | No significant effect was found on pain (MD −0.80, 95% CI [−1.33, 0.016]), Low evidence | 1a | |
| Lavín-Pérez et al., 2021, Systematic review with meta-analysis [51] | Cancer Survivors | n1 = 7 (RCT), and n2 = 355 | Exercise (HIT) | Significant effect was found on pain (SMD −0.18, 95% CI [−0.34, −0.02], p = 0.02, I2 = 4%), Moderate evidence | 1a | |
| Lu et al., 2020, Systematic review with meta-analysis [52] | Breast Cancer Survivors with AIA | n1 = 6 (RCT), and n2 = 416 | Exercise | Significant effect was found on pain (SMD −0.46, 95% CI [−0.79, −0.13], p = 0.006, I2 = 63%), Moderate evidence |
1a | |
| Timmins et al., 2021, Systematic review [49] | Cancer Survivors | n1 = 5 (2 C and 3 CS), and n2 = 3950 | Low physical activity | According to the SORT: the association between physical inactivity and CIPN was of moderate evidence | 3b | |
| Sleep | Leysen et al., 2019, Systematic review with meta-analysis [53] | Breast Cancer Survivors | n1 = 4 (2 CS and 2 C) and n2 = 1907 | Sleep Disturbances | Pain was a predictor for sleep disturbances (OR 1.68, 95% CI [1.19, 2.37], p = 0.05, I2 = 55%, after subgroup analysis OR 2.31, 95% CI [1.36, 3.92], p = 0.002, I2 = 27%) |
3b |
| Smoking | Leysen et al., 2017, Systematic review with meta-analysis [5] | Breast Cancer Survivors | n1 = 2 (1 CS and 1 C) and n2 = 2519 |
Smoking status | Smoking (OR 0.75, 95% CI [0.62, 0.92], p = 0.005, I2 = 0%) was not a predictor for pain, Consistent and low evidence | 3b |
| Stress | Syrowatka et al., 2017, Systematic review [37] |
Breast Cancer Survivors | n1 = 12 (6 CS and 6 C) and n2 = 7842 | Distress | Pain was significantly associated with distress: 9/12 studies (75%) | 3b |
| Intervention | Chang et al., 2020, Systematic review with meta-analysis [45] | Breast Cancer Survivors | n1 = 5 (RCT) and n2 = 827 |
Mindfulness-Based interventions | No significant effect was found on pain (SMD −0.39, 95% CI, [−0.81, 0.03], p = 0.07, I2 = 85%), Moderate evidence | 1a |
| Cillessen et al., 2019, Systematic review with meta-analysis [54] | Cancer Patients and Survivors | n1 = 4 (RCT) and n2 = 587 |
Mindfulness-Based interventions | Significant effect was found on pain (ES 0.2, 95% CI [0.04, 0.36], p = 0.16, I2 = 0%), Moderate evidence | 1a | |
| Martinez-Miranda [21] | Breast Cancer Survivors | n1 = 2 (RCT) and n2 = 134 |
Patient Education | No significant effect was found on pain (SMD −0.05, 95% CI [−0.26, 0.17], p = 0.67, I2 = 0%, Low evidence |
1a | |
| Silva et al., 2019, Systematic review [55] | Cancer Survivors | n1 = 4 (4 quasi-experimental studies), and n2 = 522 | Promoting healthy behaviour by mHealth apps | Effect found on pain was inconsistent and of low quality of evidence | 2b |
3.2. Sleep
3.3. Diet
3.3.1 Dietary intake
3.3.2. Obesity
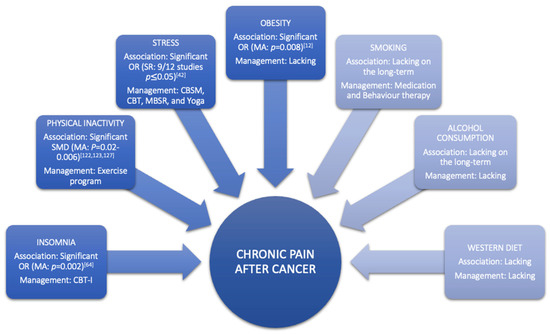
Obesity is a condition characterised by an increase in body fat [83,84][75][76]. At the neurobiological level, obesity is considered to cause pain through various mechanisms, including inflammation and hormone imbalance [85][77]. At the mechanical level, obesity can also cause pain by structural overloading [84[76][78],86], which can lead to altered body posture and joint misuse [87][79]. The latest review in taxane- and platinum-treated CSs demonstrated a good-to-moderate relationship between obesity and higher severity or incidence of chemotherapy-induced peripheral neuropathy (CIPN), with moderate evidence showing diabetes did not increase incidence or severity of CIPN [88][80]. Furthermore, a systematic review with meta-analyses of Leysen et al., (2017) demonstrated that breast CSs with a BMI > 30 have a higher risk (odds ratio = 1.34, 95% CI 1.08–1.67) of developing pain (Table 1, Figure 2) [12]. However, more research is needed to determine the long-term impact of obesity among the expanding population of CSs [89][81]. Studies looking at the link between changes in body mass index, fat mass, inflammatory markers, and chronic pain might help us better comprehend the relationship between these variables in the CS population. Additionally, well-designed, high-quality randomised controlled trials on the effect of combined weight loss/pain therapies are required to inform patients and clinicians on how to personalise the approach to reduce chronic pain prevalence, intensity, or severity in CSs through obesity management (Figure 2).
3.4. Smoking
Pain might be one of the barriers to smoking cessation in CSs [98][82]. An observational study by Aigner et al., (2016) demonstrated that when patients experience higher pain levels, they usually smoke a larger number of cigarettes during these days and initiate fewer attempts to quit smoking [98][82]. This can be explained by the fact that nicotine produces an acute analgesic effect, making it much harder for them to stop due to the rewarding sensation they experience [99][83]. Despite its short-term analgesic effect, tobacco smoking sustains pain in the long-term [93][84]. This underlines the importance of incorporating anti-smoking medications in CSs with pain to avoid relapse during nicotine withdrawal [99][83]. Moreover, pain management should be added to the counselling aspect to enhance the patient’s knowledge, which in turn, might improve their adherence to the whole smoking cessation program [98][82]. Furthermore, the 5As (Ask, Advise, Assess, Assist, Arrange) approach, which assesses the willingness of the patient to quit smoking, is no longer recommended since studies have demonstrated that smokers who did not feel ready to quit smoking at the same rate as those who wanted to [100][85]. The model with the most promising results might be “opt-out”, during which health care providers offer counselling and pharmacotherapy to all smokers, which is more ethical [101][86]. However, research on how to integrate this approach in current cancer care for CSs is needed.
3.5. Alcohol Consumption
The impact of alcohol use on pain is poorly investigated in CSs, but according to one systematic review of two cohort studies, the risk of developing pain can be reduced by alcohol use (Table 1) [12]. This finding might be misleading due to the fact that alcohol has an acute analgesic effect [111][87]. In non-cancer populations, studies demonstrated that this analgesic effect diminishes over time, and there is an association between chronic pain and alcohol consumption [112][88]. This pain might be evoked by developing alcoholic neuropathy, musculoskeletal disorders, or alcohol withdrawal [112][88]. Conversely, chronic pain increases the risk of alcohol abuse [113][89]. Nevertheless, psychosocial factors are also highly present in patients with alcohol abuse and can be attributed to abnormalities in the reward system of the brain [114][90]. Additionally, a recently published study demonstrated that chronic pain patients with high levels of pain catastrophising are more likely to be heavy drinkers [115][91]. General advice on alcohol consumption after cancer is currently not possible due to the high variability of results in different CSs. Therefore, health care providers should tailor their advice according to cancer types and patients [116][92]. Within that view, an overview of recommendations regarding individualised alcohol consumption for each CS type could support clinicians in doing so, yet such evidence-based recommendations are currently lacking (Figure 2).
3.6. Physical Activity
the evidence demonstrated that supervised physical activity reduces cancer-related fatigue, depression, and increases quality of life, cardiovascular and musculoskeletal fitness in CSs [14,15,16]. Additional beneficial effects of physical activity were also seen on musculoskeletal pain and stiffness in breast CSs taking aromatase inhibitors for a long period (4. Future Directions for Scientists
First, it is recommended that researchers make a clear distinction between CSs’ phases when initiating and reporting studies in CSs. Currently, the term CS is too globally used, making it difficult to compare or combine results of studies due to their high heterogeneity. An individual in palliative care has different needs than an individual that is cured of cancer; however, both are CSs according to the most widely used definition [4]. A distinction between the different phases has been described by Mullan et al. in 1985 [138]. Unfortunately, these terms are not frequently used in the literature [138] even though a clear distinction between phases could help clinicians to communicate more easily and to provide the appropriate care to patients’ needs according to their phase in the survival of cancer.
Second, most studies were performed on Caucasian breast CSs with high socio-economic status. This population is more likely to have a higher adherence and willingness to change their lifestyle habits [139]. However, to reach a better understanding of barriers for lifestyle changes, research needs to be performed among CS populations with diverse socio-economic backgrounds. This way, oncological care for CSs can be more tailored to patients of different gender, race, and socio-economic capacities.
Third, future studies regarding lifestyle factors in CSs should more thoroughly account for possible confounders. Indeed, research studying a particular lifestyle factor should not only be adjusted for age, gender, education, and so forth, but also for other established lifestyle factors, which might be a considerable confounder. Furthermore, the effects of lifestyle factors in CSs are most often observed over a short period, preventing to draw conclusions regarding long-term impact of lifestyle factors in CSs. More research is warranted to observe the long-term effects of pain management and healthy lifestyle interventions in CSs.
5. Future Directions for Clinicians
The literature indicates that implementing healthy lifestyle habits in CSs has low compliance rates [140]. A barrier that might cause low adherence to healthy lifestyle behaviours is the burdensome treatment format of most behavioural interventions [73,74]. Therefore, stepped care models might provide clinicians with a possible solution to improve the feasibility and deliver care efficiently [141]. In existing stepped care models, the first step is typically a form of self-management therapy (e.g., recommendations) with the possibility to progress to the highest step of six to eight individual sessions with a specialist, if needed [142,143]. For example, a recent study in CSs demonstrated that more than 50% of CSs with insomnia benefit form a one-hour group-delivered session that empowers CSs by teaching them about sleep health and provides specific information on how to adapt their sleep behaviours [142]. Interestingly, they found that CSs who had experienced sleep problems for a shorter period and perceived less burden from their sleep problems were most likely to benefit from the one-hour program, suggesting that it is crucial to identify CS with sleep problems as soon as possible to enhance the efficacy of low-intensity interventions [142]. However, further research is warranted before implementing stepped care for the other lifestyle factors. In addition, systematic reviews demonstrated promising findings for virtual therapy, suggesting that virtual interventions might be a possible option to enhance access to care, which solves the distance issue [69,137,144].
Furthermore, to reduce the treatment burden, clinicians should perform early screenings and identify negative predictors to improve patients’ self-efficacy to sustain a healthy lifestyle. Developing evidence-based guidelines, including algorithms with practical triage and referral plans to other healthcare professionals, will improve survivorship care. Enhancing the productivity of oncological care by 2025 is of utmost importance because there will be a shortage of oncologists due to the growing cancer population [145]. Besides that, many clinicians have difficulties providing the ideal pain management plan and delivering health promotion guidance due to a lack of knowledge [22]. Supplementary support and educational interventions should be organized for health care providers to enhance their expertise and confidence in this field.
Another recommendation for future clinical practice is considering the use of pain neuroscience education as a way to decrease the threatening nature of pain, catastrophic thinking and fear-avoidance beliefs in CSs [146]. Cancer patients indicate themselves that they have insufficient knowledge regarding pain during or after cancer, what the possibilities of pain relief are and how they can access support when needed [24,25]. When comparing pain knowledge between CSs, healthy controls and caregivers, CSs had the lowest pain knowledge of the three groups [147]. Education about pain is underused in the field of oncology and non-existent in the survivorship phase [148]. Pain neuroscience education can clear the path for more active approaches to pain management, including providing lifestyle interventions. Manuals with guidelines for clinicians on how to explain pain following cancer [146], including accounting for perceived injustice during pain neuroscience education [149], are available to support clinicians in doing so.
Lastly, this state-of-the-art paper underlines once more the complexity of managing chronic pain in CSs. As discussed previously, adopting a healthy lifestyle might have a beneficial influence on the chronic pain of CSs. Unfortunately, there is currently a lack of research about the effectiveness of modifiable lifestyle factors on pain. Moreover, pain in CSs should be targeted on cognitive, behavioural, sensory and emotional levels due to its complexity [18]. Therefore, all pain interventions should be multidisciplinary and personalized for each CS [19].
6. Conclusions
Emerging evidence shows that CSs find it challenging to receive optimal treatment plans for their burdens, and support or reinforcement to maintain a healthy lifestyle. Therefore, it is crucially important to prepare clinicians well, so they can provide guidance along and after primary treatment. For chronic pain in CSs, it is primordial to identify factors that contribute to the transition of acute to chronic pain in CSs because chronic pain remains underrecognized and mistreated in this population. Furthermore, a proper definition between CSs’ phases should be developed for optimal research and treatment. In the clinical field, new psychosocial factors and modifiable lifestyle factors should be targeted to improve pain relief in CSs.
Modifiable lifestyle factors and their impact on pain have been discussed in depth in this paper and are, for instance, stress, insomnia, diet, obesity, smoking, alcohol consumption and physical activity. First, an inappropriate stress response promotes pain by dysregulating the autonomic, endocrine, and immune response creating a problematic back loop because pain is a manageable risk factor for distress. The stress response can be managed by CBSM, CBT, MBSR and yoga. Second, sleep and pain also form a vicious cycle (sleep problems exacerbate response to nociceptive stimuli and pain can disturb sleep quality) that CBT-I can break. Third, guidelines recommend prudent diets in CSs. However, more research is needed to unravel the role of nutrition and obesity in CSs. Fourth, alcohol consumption and smoking are both negative lifestyle behaviours that impact patients’ general health. Smoking cessation should consist of behaviour therapy and medication. Last, physical activity demonstrates its beneficial impact in several systematic reviews. However, the adherence is low and new treatment strategies such as motivational interviewing or BGA should be investigated in CSs to increase treatment outcomes in the long-term.
In the future, there will be an insufficient number of professionals (oncologists) due to the growing cancer population [150,151]. Therefore, it is a priority that researchers refine current treatment plans and define the benefits of modifiable lifestyle factors and their impact on chronic pain in CSs.
References
- Tevaarwerk, A.; Denlinger, C.S.; Sanft, T.; Ansbaugh, S.M.; Armenian, S.; Baker, K.S.; Broderick, G.; Day, A.; Demark-Wahnefried, W.; Dickinson, K.; et al. Survivorship, Version 1.2021: Featured Updates to the NCCN Guidelines. J. Natl. Compr. Cancer Netw. 2021, 19, 676–685. Levit, L.A.; Balogh, E.; Nass, S.J.; Ganz, P. Delivering High-Quality Cancer Care: Charting a New Course for a System in Crisis; National Academies Press: Washington, DC, USA, 2013.
- Paice, J.A.; Portenoy, R.; Lacchetti, C.; Campbell, T.; Cheville, A.; Citron, M.; Constine, L.S.; Cooper, A.; Glare, P.; Keefe, F. Management of Chronic Pain in Survivors of Adult Cancers: American Society of Clinical Oncology Clinical Practice Guideline. J. Clin. Oncol. 2016, 34, 3325–3345. Bluethmann, S.M.; Mariotto, A.B.; Rowland, J.H. Anticipating the “Silver Tsunami”: Prevalence Trajectories and Comorbidity Burden among Older Cancer Survivors in the United States. Cancer Epidemiol. Biomark. Prev. 2016, 25, 1029–1036.
- Moloney, N.A.; Pocovi, N.C.; Dylke, E.S.; Graham, P.L.; De Groef, A. Psychological Factors Are Associated with Pain at All Time Frames After Breast Cancer Surgery: A Systematic Review with Meta-Analyses. Pain Med. 2021, 22, 915–947. Zale, E.L.; Maisto, S.A.; Ditre, J.W. Interrelations between pain and alcohol: An integrative review. Clin. Psychol. Rev. 2015, 37, 57–71.
- Wang, L.; Guyatt, G.H.; Kennedy, S.A.; Romerosa, B.; Kwon, H.Y.; Kaushal, A.; Chang, Y.; Craigie, S.; de Almeida, C.P.B.; Courban, R.J.; et al. Predictors of persistent pain after breast cancer surgery: A systematic review and meta-analysis of observational studies. CMAJ 2016, 188, E352–E361. Boissoneault, J.; Lewis, B.; Nixon, S.J. Characterizing chronic pain and alcohol use trajectory among treatment-seeking alcoholics. Alcohol 2019, 75, 47–54.
- Leysen, L.; Beckwée, D.; Nijs, J.; Pas, R.; Bilterys, T.; Vermeir, S.; Adriaenssens, N. Risk factors of pain in breast cancer survivors: A systematic review and meta-analysis. Support. Care Cancer 2017, 25, 3607–3643. Maleki, N.; Oscar-Berman, M. Chronic Pain in Relation to Depressive Disorders and Alcohol Abuse. Brain Sci. 2020, 10, 826.
- Demark-Wahnefried, W.; Rogers, L.Q.; Alfano, C.M.; Thomson, C.A.; Courneya, K.S.; Meyerhardt, J.A.; Stout, N.L.; Kvale, E.; Ganzer, H.; Ligibel, J.A. Practical clinical interventions for diet, physical activity, and weight control in cancer survivors. CA Cancer J. Clin. 2015, 65, 167–189. Nieto, S.J.; Green, R.; Grodin, E.N.; Cahill, C.M.; Ray, L.A. Pain catastrophizing predicts alcohol craving in heavy drinkers independent of pain intensity. Drug Alcohol Depend. 2021, 218, 108368.
- Meneses-Echávez, J.F.; González-Jiménez, E.; Ramírez-Vélez, R. Effects of supervised exercise on cancer-related fatigue in breast cancer survivors: A systematic review and meta-analysis. BMC Cancer 2015, 15, 77. Rock, C.L.; Doyle, C.; Demark-Wahnefried, W.; Meyerhardt, J.; Courneya, K.S.; Schwartz, A.L.; Bandera, E.V.; Hamilton, K.K.; Grant, B.; McCullough, M.; et al. Nutrition and physical activity guidelines for cancer survivors. CA Cancer J. Clin. 2012, 62, 243–274.
- Kessels, E.; Husson, O.; van der Feltz-Cornelis, C.M. The effect of exercise on cancer-related fatigue in cancer survivors: A systematic review and meta-analysis. Neuropsychiatr. Dis. Treat. 2018, 14, 479–494. Meneses-Echávez, J.F.; González-Jiménez, E.; Ramírez-Vélez, R. Effects of supervised exercise on cancer-related fatigue in breast cancer survivors: A systematic review and meta-analysis. BMC Cancer 2015, 15, 77.
- Fuller, J.T.; Hartland, M.C.; Maloney, L.T.; Davison, K. Therapeutic effects of aerobic and resistance exercises for cancer survivors: A systematic review of meta-analyses of clinical trials. Br. J. Sports Med. 2018, 52, 1311. Kessels, E.; Husson, O.; van der Feltz-Cornelis, C.M. The effect of exercise on cancer-related fatigue in cancer survivors: A systematic review and meta-analysis. Neuropsychiatr. Dis. Treat. 2018, 14, 479–494.
- Bobyrov, V. Bases of Bioethics and Biosafety: Study Guide for Stud. of Higher Med. Est; Hoва Kнига: Vinnytsia, Ukraine, 2012. Fuller, J.T.; Hartland, M.C.; Maloney, L.T.; Davison, K. Therapeutic effects of aerobic and resistance exercises for cancer survivors: A systematic review of meta-analyses of clinical trials. Br. J. Sports Med. 2018, 52, 1311.
- Maindet, C.; Burnod, A.; Minello, C.; George, B.; Allano, G.; Lemaire, A. Strategies of complementary and integrative therapies in cancer-related pain-attaining exhaustive cancer pain management. Support. Care Cancer 2019, 27, 3119–3132. Kim, T.H.; Kang, J.W.; Lee, T.H. Therapeutic options for aromatase inhibitor-associated arthralgia in breast cancer survivors: A systematic review of systematic reviews, evidence mapping, and network meta-analysis. Maturitas 2018, 118, 29–37.
- Glare, P.A.; Davies, P.S.; Finlay, E.; Gulati, A.; Lemanne, D.; Moryl, N.; Oeffinger, K.C.; Paice, J.A.; Stubblefield, M.D.; Syrjala, K.L. Pain in cancer survivors. J. Clin. Oncol. 2014, 32, 1739. Lu, G.; Zheng, J.; Zhang, L. The effect of exercise on aromatase inhibitor-induced musculoskeletal symptoms in breast cancer survivors: A systematic review and meta-analysis. Support. Care Cancer 2020, 28, 1587–1596.
- Bennett, M.I.; Kaasa, S.; Barke, A.; Korwisi, B.; Rief, W.; Treede, R.D. The IASP classification of chronic pain for ICD-11: Chronic cancer-related pain. Pain 2019, 160, 38–44. Boing, L.; Vieira, M.C.S.; Moratelli, J.; Bergmann, A.; Guimarães, A.C.A. Effects of exercise on physical outcomes of breast cancer survivors receiving hormone therapy—A systematic review and meta-analysis. Maturitas 2020, 141, 71–81.
- Brown, M.R.; Ramirez, J.D.; Farquhar-Smith, P. Pain in cancer survivors. Br. J. Pain 2014, 8, 139–153. Ballard-Barbash, R.; Friedenreich, C.M.; Courneya, K.S.; Siddiqi, S.M.; McTiernan, A.; Alfano, C.M. Physical activity, biomarkers, and disease outcomes in cancer survivors: A systematic review. J. Natl. Cancer Inst. 2012, 104, 815–840.
- Pachman, D.R.; Barton, D.L.; Swetz, K.M.; Loprinzi, C.L. Troublesome symptoms in cancer survivors: Fatigue, insomnia, neuropathy, and pain. J. Clin. Oncol. 2012, 30, 3687–3696. Hasenoehrl, T.; Palma, S.; Ramazanova, D.; Kölbl, H.; Dorner, T.E.; Keilani, M.; Crevenna, R. Resistance exercise and breast cancer-related lymphedema-a systematic review update and meta-analysis. Support Care Cancer 2020, 28, 3593–3603.
- Sun, V.; <monospace> </monospace>Borneman, T.; Piper, B.; Koczywas, M.; Ferrell, B. Barriers to pain assessment and management in cancer survivorship. J. Cancer Surviv. 2008, 2, 65–71. Geneen, L.J.; Moore, R.A.; Clarke, C.; Martin, D.; Colvin, L.A.; Smith, B.H. Physical activity and exercise for chronic pain in adults: An overview of Cochrane Reviews. Cochrane Database Syst. Rev. 2017, 1, Cd011279.
- Chow, R.; Saunders, K.; Burke, H.; Belanger, A.; Chow, E. Needs assessment of primary care physicians in the management of chronic pain in cancer survivors. Support. Care Cancer 2017, 25, 3505–3514. Lavín-Pérez, A.M.; Collado-Mateo, D.; Mayo, X.; Liguori, G.; Humphreys, L.; Copeland, R.J.; Jiménez, A. Effects of high-intensity training on the quality of life of cancer patients and survivors: A systematic review with meta-analysis. Sci. Rep. 2021, 11, 15089.
- Oldenmenger, W.H.; Geerling, J.I.; Mostovaya, I.; Vissers, K.C.; de Graeff, A.; Reyners, A.K.; van der Linden, Y.M. A systematic review of the effectiveness of patient-based educational interventions to improve cancer-related pain. Cancer Treat. Rev. 2018, 63, 96–103. Ijsbrandy, C.; Ottevanger, P.B.; Gerritsen, W.R.; van Harten, W.H.; Hermens, R. Determinants of adherence to physical cancer rehabilitation guidelines among cancer patients and cancer centers: A cross-sectional observational study. J. Cancer Surviv. 2021, 15, 163–177.
- Binkley, J.M.; Harris, S.R.; Levangie, P.K.; Pearl, M.; Guglielmino, J.; Kraus, V.; Rowden, D. Patient perspectives on breast cancer treatment side effects and the prospective surveillance model for physical rehabilitation for women with breast cancer. Cancer 2012, 118, 2207–2216. Kampshoff, C.S.; Jansen, F.; van Mechelen, W.; May, A.M.; Brug, J.; Chinapaw, M.J.; Buffart, L.M. Determinants of exercise adherence and maintenance among cancer survivors: A systematic review. Int. J. Behav. Nutr. Phys. Act. 2014, 11, 80.
- McGuire, D.B. Occurrence of cancer pain. J. Natl. Cancer Inst. Monogrphs 2004, 2004, 51–56. Ormel, H.L.; van der Schoot, G.G.F.; Sluiter, W.J.; Jalving, M.; Gietema, J.A.; Walenkamp, A.M.E. Predictors of adherence to exercise interventions during and after cancer treatment: A systematic review. Psychooncology 2018, 27, 713–724.
- Martínez-Miranda, P.; Casuso-Holgado, M.J.; Jiménez-Rejano, J.J. Effect of patient education on quality-of-life, pain and fatigue in breast cancer survivors: A systematic review and meta-analysis. Clin. Rehabil. 2021, 35, 1722–1742. Spencer, J.C.; Wheeler, S.B. A systematic review of Motivational Interviewing interventions in cancer patients and survivors. Patient Educ. Couns. 2016, 99, 1099–1105.
- Boland, E.G.; Ahmedzai, S.H. Persistent pain in cancer survivors. Curr. Opin. Support Palliat. Care 2017, 11, 181–190. Turner, R.R.; Steed, L.; Quirk, H.; Greasley, R.U.; Saxton, J.M.; Taylor, S.J.; Rosario, D.J.; Thaha, M.A.; Bourke, L. Interventions for promoting habitual exercise in people living with and beyond cancer. Cochrane Database Syst. Rev. 2018, 9.
- Ong, W.Y.; Stohler, C.S.; Herr, D.R. Role of the Prefrontal Cortex in Pain Processing. Mol. Neurobiol. 2019, 56, 1137–1166. Veenhof, C.; Köke, A.J.; Dekker, J.; Oostendorp, R.A.; Bijlsma, J.W.; van Tulder, M.W.; van den Ende, C.H. Effectiveness of behavioral graded activity in patients with osteoarthritis of the hip and/or knee: A randomized clinical trial. Arthritis Rheum. 2006, 55, 925–934.
- Sullivan, M.J.; Scott, W.; Trost, Z. Perceived injustice: A risk factor for problematic pain outcomes. Clin. J. Pain. 2012, 28, 484–488. Cillessen, L.; Johannsen, M.; Speckens, A.E.M.; Zachariae, R. Mindfulness-based interventions for psychological and physical health outcomes in cancer patients and survivors: A systematic review and meta-analysis of randomized controlled trials. Psychooncology 2019, 28, 2257–2269.
- Sullivan, M.J.; Davidson, N.; Garfinkel, B.; Siriapaipant, N.; Scott, W. Perceived injustice is associated with heightened pain behavior and disability in individuals with whiplash injuries. Psychol. Inj. Law 2009, 2, 238–247. Duan, L.; Xu, Y.; Li, M. Effects of Mind-Body Exercise in Cancer Survivors: A Systematic Review and Meta-Analysis. Evid. Based Complement. Altern. Med. 2020, 2020, 7607161.
- Carriere, J.S.; Donayre Pimentel, S.; Yakobov, E.; Edwards, R.R. A Systematic Review of the Association Between Perceived Injustice and Pain-Related Outcomes in Individuals with Musculoskeletal Pain. Pain Med. 2020, 21, 1449–1463. Mendoza, M.E.; Capafons, A.; Gralow, J.R.; Syrjala, K.L.; Suárez-Rodríguez, J.M.; Fann, J.R.; Jensen, M.P. Randomized controlled trial of the Valencia model of waking hypnosis plus CBT for pain, fatigue, and sleep management in patients with cancer and cancer survivors. Psychooncology 2017, 26, 1832–1838.
- Leysen, L.; Cools, W.; Nijs, J.; Adriaenssens, N.; Pas, R.; van Wilgen, C.P.; Bults, R.; Roose, E.; Lahousse, A.; Beckwée, D. The mediating effect of pain catastrophizing and perceived injustice in the relationship of pain on health-related quality of life in breast cancer survivors. Support. Care Cancer 2021, 29, 5653–5661. Marzorati, C.; Riva, S.; Pravettoni, G. Who Is a Cancer Survivor? A Systematic Review of Published Definitions. J. Cancer Educ. 2017, 32, 228–237.
- Carriere, J.S.; Martel, M.O.; Kao, M.C.; Sullivan, M.J.; Darnall, B.D. Pain behavior mediates the relationship between perceived injustice and opioid prescription for chronic pain: A Collaborative Health Outcomes Information Registry study. J. Pain Res. 2017, 10, 557–566. Mullan, F. Seasons of survival: Reflections of a physician with cancer. N. Engl. J. Med. 1985, 313, 270–273.
- Turk, D.C.; Okifuji, A. What factors affect physicians’ decisions to prescribe opioids for chronic noncancer pain patients? Clin. J. Pain 1997, 13, 330–336. Paxton, R.J.; Jones, L.A.; Chang, S.; Hernandez, M.; Hajek, R.A.; Flatt, S.W.; Natarajan, L.; Pierce, J.P. Was race a factor in the outcomes of the Women’s Health Eating and Living Study? Cancer 2011, 117, 3805–3813.
- Paice, J.A. Chronic treatment-related pain in cancer survivors. Pain 2011, 152, S84–S89. Blanchard, C.M.; Courneya, K.S.; Stein, K. Cancer survivors’ adherence to lifestyle behavior recommendations and associations with health-related quality of life: Results from the American Cancer Society’s SCS-II. J. Clin. Oncol. 2008, 26, 2198–2204.
- Derksen, J.W.G.; Beijer, S.; Koopman, M.; Verkooijen, H.M.; van de Poll-Franse, L.V.; May, A.M. Monitoring potentially modifiable lifestyle factors in cancer survivors: A narrative review on currently available methodologies and innovations for large-scale surveillance. Eur. J. Cancer 2018, 103, 327–340. Stinson, K.; Tang, N.K.; Harvey, A.G. Barriers to treatment seeking in primary insomnia in the United Kingdom: A cross-sectional perspective. Sleep 2006, 29, 1643–1646.
- Gopalakrishna, A.; Longo, T.A.; Fantony, J.J.; Van Noord, M.; Inman, B.A. Lifestyle factors and health-related quality of life in bladder cancer survivors: A systematic review. J Cancer Surviv. 2016, 10, 874–882. Matthews, E.E.; Arnedt, J.T.; McCarthy, M.S.; Cuddihy, L.J.; Aloia, M.S. Adherence to cognitive behavioral therapy for insomnia: A systematic review. Sleep Med. Rev. 2013, 17, 453–464.
- Riba, M.B.; Donovan, K.A.; Andersen, B.; Braun, I.; Breitbart, W.S.; Brewer, B.W.; Buchmann, L.O.; Clark, M.M.; Collins, M.; Corbett, C.; et al. NCCN Clinical Practice Guidelines in Oncology: Distress Management, v3.2019. Natl. Compr. Cancer Network. 2019, 17, 1229–1249. Available online: https://www.nccn.org/professionals/physician_gls/pdf/distress.pdf (accessed on 14 December 2021). Bower, P.; Gilbody, S. Stepped care in psychological therapies: Access, effectiveness and efficiency. Narrative literature review. Br. J. Psychiatry 2005, 186, 11–17.
- Fagundes, C.; LeRoy, A.; Karuga, M. Behavioral Symptoms after Breast Cancer Treatment: A Biobehavioral Approach. J. Pers. Med. 2015, 5, 280–295. Zhou, E.S.; Michaud, A.L.; Recklitis, C.J. Developing efficient and effective behavioral treatment for insomnia in cancer survivors: Results of a stepped care trial. Cancer 2020, 126, 165–173.
- Liu, Y.Z.; Wang, Y.X.; Jiang, C.L. Inflammation: The Common Pathway of Stress-Related Diseases. Front. Hum. Neurosci. 2017, 11, 316. Lynch, F.A.; Katona, L.; Jefford, M.; Smith, A.B.; Shaw, J.; Dhillon, H.M.; Ellen, S.; Phipps-Nelson, J.; Lai-Kwon, J.; Milne, D.; et al. Feasibility and Acceptability of Fear-Less: A Stepped-Care Program to Manage Fear of Cancer Recurrence in People with Metastatic Melanoma. J. Clin. Med. 2020, 9, 2969.
- Reis, J.C.; Antoni, M.H.; Travado, L. Emotional distress, brain functioning, and biobehavioral processes in cancer patients: A neuroimaging review and future directions. CNS Spectr. 2020, 25, 79–100. Ma, Y.; Hall, D.L.; Ngo, L.H.; Liu, Q.; Bain, P.A.; Yeh, G.Y. Efficacy of cognitive behavioral therapy for insomnia in breast cancer: A meta-analysis. Sleep Med. Rev. 2021, 55, 101376.
- Syrowatka, A.; Motulsky, A.; Kurteva, S.; Hanley, J.A.; Dixon, W.G.; Meguerditchian, A.N.; Tamblyn, R. Predictors of distress in female breast cancer survivors: A systematic review. Breast Cancer Res. Treat 2017, 165, 229–245. Hernandez Silva, E.; Lawler, S.; Langbecker, D. The effectiveness of mHealth for self-management in improving pain, psychological distress, fatigue, and sleep in cancer survivors: A systematic review. J. Cancer Surviv. 2019, 13, 97–107.
- National Comprehensive Cancer Netwerk. Survivorship 2021 (Version 3. 2021). 2021. Available online: https://www.nccn.org/login?ReturnURL=https://www.nccn.org/professionals/physician_gls/pdf/survivorship.pdf (accessed on 8 September 2021).Roberts, A.L.; Fisher, A.; Smith, L.; Heinrich, M.; Potts, H.W.W. Digital health behaviour change interventions targeting physical activity and diet in cancer survivors: A systematic review and meta-analysis. J. Cancer Surviv. 2017, 11, 704–719.
- Thornton, L.M.; Andersen, B.L.; Blakely, W.P. The pain, depression, and fatigue symptom cluster in advanced breast cancer: Covariation with the hypothalamic-pituitary-adrenal axis and the sympathetic nervous system. Health Psychol. 2010, 29, 333–337. Yang, W.; Williams, J.H.; Hogan, P.F.; Bruinooge, S.S.; Rodriguez, G.I.; Kosty, M.P.; Bajorin, D.F.; Hanley, A.; Muchow, A.; McMillan, N.; et al. Projected supply of and demand for oncologists and radiation oncologists through 2025: An aging, better-insured population will result in shortage. J. Oncol. Pract. 2014, 10, 39–45.
- Addison, S.; Shirima, D.; Aboagye-Mensah, E.B.; Dunovan, S.G.; Pascal, E.Y.; Lustberg, M.B.; Arthur, E.K.; Nolan, T.S. Effects of tandem cognitive behavioral therapy and healthy lifestyle interventions on health-related outcomes in cancer survivors: A systematic review. J. Cancer Surviv. 2021, 1–24. Chow, R.; Saunders, K.; Burke, H.; Belanger, A.; Chow, E. Needs assessment of primary care physicians in the management of chronic pain in cancer survivors. Support. Care Cancer 2017, 25, 3505–3514.
- Traeger, L.; Penedo, F.J.; Benedict, C.; Dahn, J.R.; Lechner, S.C.; Schneiderman, N.; Antoni, M.H. Identifying how and for whom cognitive-behavioral stress management improves emotional well-being among recent prostate cancer survivors. Psychooncology 2013, 22, 250–259. Nijs, J.; Wijma, A.J.; Leysen, L.; Pas, R.; Willaert, W.; Hoelen, W.; Ickmans, K.; Wilgen, C.P.V. Explaining pain following cancer: A practical guide for clinicians. Braz. J. Phys. Ther. 2019, 23, 367–377.
- Tang, M.; Liu, X.; Wu, Q.; Shi, Y. The Effects of Cognitive-Behavioral Stress Management for Breast Cancer Patients: A Systematic Review and Meta-analysis of Randomized Controlled Trials. Cancer Nurs. 2020, 43, 222–237. Binkley, J.M.; Harris, S.R.; Levangie, P.K.; Pearl, M.; Guglielmino, J.; Kraus, V.; Rowden, D. Patient perspectives on breast cancer treatment side effects and the prospective surveillance model for physical rehabilitation for women with breast cancer. Cancer 2012, 118, 2207–2216.
- Danhauer, S.C.; Addington, E.L.; Cohen, L.; Sohl, S.J.; Van Puymbroeck, M.; Albinati, N.K.; Culos-Reed, S.N. Yoga for symptom management in oncology: A review of the evidence base and future directions for research. Cancer 2019, 125, 1979–1989. McGuire, D.B. Occurrence of cancer pain. J. Natl. Cancer Inst. Monogrphs 2004, 2004, 51–56.
- Matchim, Y.; Armer, J.M.; Stewart, B.R. Mindfulness-based stress reduction among breast cancer survivors: A literature review and discussion. Oncol. Nurs. Forum 2011, 38, E61–E71. Lexmond, W.; Jäger, K. Psychomteric Properties of the Dutch Version of the Revised Neurophysiology of Pain Questionnaire; Vrije Universiteit Brussel: Brussels, Belgium, 2019; p. 36.
- Chang, Y.C.; Yeh, T.L.; Chang, Y.M.; Hu, W.Y. Short-term Effects of Randomized Mindfulness-Based Intervention in Female Breast Cancer Survivors: A Systematic Review and Meta-analysis. Cancer. Nurs. 2021, 44, E703–E714. Bennett, M.I.; Bagnall, A.M.; Closs, S.J. How effective are patient-based educational interventions in the management of cancer pain? Systematic review and meta-analysis. Pain 2009, 143, 192–199.
- The Joanna Briggs Institute Levels of Evidence and Grades of RecommendationWorking Party Joanna Briggs Institute Levels of Evidence and Grades of Recommendation. 2014. Available online: https://jbi.global/sites/default/files/2019-05/JBI-Levels-of-evidence_2014_0.pdf (accessed on 22 November 2021).Nijs, J.; Roose, E.; Lahousse, A.; Mostaqim, K.; Reynebeau, I.; De Couck, M.; Beckwee, D.; Huysmans, E.; Bults, R.; van Wilgen, P.; et al. Pain and Opioid Use in Cancer Survivors: A Practical Guide to Account for Perceived Injustice. Pain Physician 2021, 24, 309–317.
- Kim, T.H.; Kang, J.W.; Lee, T.H. Therapeutic options for aromatase inhibitor-associated arthralgia in breast cancer survivors: A systematic review of systematic reviews, evidence mapping, and network meta-analysis. Maturitas 2018, 118, 29–37. Maindet, C.; Burnod, A.; Minello, C.; George, B.; Allano, G.; Lemaire, A. Strategies of complementary and integrative therapies in cancer-related pain-attaining exhaustive cancer pain management. Support. Care Cancer 2019, 27, 3119–3132.
- Yilmaz, S.T.; Elma, Ö.; Deliens, T.; Coppieters, I.; Clarys, P.; Nijs, J.; Malfliet, A. Nutrition/Dietary Supplements and Chronic Pain in Patients with Cancer and Survivors of Cancer: A Systematic Review and Research Agenda. Pain Physician 2021, 24, 335–344. Glare, P.A.; Davies, P.S.; Finlay, E.; Gulati, A.; Lemanne, D.; Moryl, N.; Oeffinger, K.C.; Paice, J.A.; Stubblefield, M.D.; Syrjala, K.L. Pain in cancer survivors. J. Clin. Oncol. 2014, 32, 1739.
- Timmins, H.C.; Mizrahi, D.; Li, T.; Kiernan, M.C.; Goldstein, D.; Park, S.B. Metabolic and lifestyle risk factors for chemotherapy-induced peripheral neuropathy in taxane and platinum-treated patients: A systematic review. J. Cancer Surviv. 2021, 1–15.
- Boing, L.; Vieira, M.C.S.; Moratelli, J.; Bergmann, A.; Guimarães, A.C.A. Effects of exercise on physical outcomes of breast cancer survivors receiving hormone therapy—A systematic review and meta-analysis. Maturitas 2020, 141, 71–81.
- Lavín-Pérez, A.M.; Collado-Mateo, D.; Mayo, X.; Liguori, G.; Humphreys, L.; Copeland, R.J.; Jiménez, A. Effects of high-intensity training on the quality of life of cancer patients and survivors: A systematic review with meta-analysis. Sci. Rep. 2021, 11, 15089.
- Lu, G.; Zheng, J.; Zhang, L. The effect of exercise on aromatase inhibitor-induced musculoskeletal symptoms in breast cancer survivors: A systematic review and meta-analysis. Support. Care Cancer 2020, 28, 1587–1596.
- Leysen, L.; Lahousse, A.; Nijs, J.; Adriaenssens, N.; Mairesse, O.; Ivakhnov, S.; Bilterys, T.; Van Looveren, E.; Pas, R.; Beckwée, D. Prevalence and risk factors of sleep disturbances in breast cancersurvivors: Systematic review and meta-analyses. Support. Care Cancer 2019, 27, 4401–4433.
- Cillessen, L.; Johannsen, M.; Speckens, A.E.M.; Zachariae, R. Mindfulness-based interventions for psychological and physical health outcomes in cancer patients and survivors: A systematic review and meta-analysis of randomized controlled trials. Psychooncology 2019, 28, 2257–2269.
- Hernandez Silva, E.; Lawler, S.; Langbecker, D. The effectiveness of mHealth for self-management in improving pain, psychological distress, fatigue, and sleep in cancer survivors: A systematic review. J. Cancer Surviv. 2019, 13, 97–107.
- Roth, T. Insomnia: Definition, prevalence, etiology, and consequences. J. Clin. Sleep Med. 2007, 3, S7–S10.
- Savard, J.; Ivers, H.; Villa, J.; Caplette-Gingras, A.; Morin, C.M. Natural course of insomnia comorbid with cancer: An 18-month longitudinal study. J. Clin. Oncol. 2011, 29, 3580–3586.
- Johnson, J.A.; Rash, J.A.; Campbell, T.S.; Savard, J.; Gehrman, P.R.; Perlis, M.; Carlson, L.E.; Garland, S.N. A systematic review and meta-analysis of randomized controlled trials of cognitive behavior therapy for insomnia (CBT-I) in cancer survivors. Sleep Med. Rev. 2016, 27, 20–28.
- Miller, K.D.; Siegel, R.L.; Lin, C.C.; Mariotto, A.B.; Kramer, J.L.; Rowland, J.H.; Stein, K.D.; Alteri, R.; Jemal, A. Cancer treatment and survivorship statistics, 2016. CA Cancer J. Clin. 2016, 66, 271–289.
- Hall, D.L.; Mishel, M.H.; Germino, B.B. Living with cancer-related uncertainty: Associations with fatigue, insomnia, and affect in younger breast cancer survivors. Support. Care Cancer 2014, 22, 2489–2495.
- Carpenter, J.S.; Elam, J.L.; Ridner, S.H.; Carney, P.H.; Cherry, G.J.; Cucullu, H.L. Sleep, fatigue, and depressive symptoms in breast cancer survivors and matched healthy women experiencing hot flashes. Oncol. Nurs. Forum 2004, 31, 591–5598.
- Savard, J.; Davidson, J.R.; Ivers, H.; Quesnel, C.; Rioux, D.; Dupere, V.; Lasnier, M.; Simard, S.; Morin, C.M. The association between nocturnal hot flashes and sleep in breast cancer survivors. J. Pain Symptom Manag. 2004, 27, 513–522.
- Gupta, P.; Sturdee, D.W.; Palin, S.L.; Majumder, K.; Fear, R.; Marshall, T.; Paterson, I. Menopausal symptoms in women treated for breast cancer: The prevalence and severity of symptoms and their perceived effects on quality of life. Climacteric 2006, 9, 49–58.
- Desai, K.; Mao, J.J.; Su, I.; Demichele, A.; Li, Q.; Xie, S.X.; Gehrman, P.R. Prevalence and risk factors for insomnia among breast cancer patients on aromatase inhibitors. Support. Care Cancer 2013, 21, 43–51.
- Finan, P.H.; Goodin, B.R.; Smith, M.T. The association of sleep and pain: An update and a path forward. J. Pain 2013, 14, 1539–1552.
- Haack, M.; Simpson, N.; Sethna, N.; Kaur, S.; Mullington, J. Sleep deficiency and chronic pain: Potential underlying mechanisms and clinical implications. Neuropsychopharmacology 2020, 45, 205–216.
- Qaseem, A.; Kansagara, D.; Forciea, M.A.; Cooke, M.; Denberg, T.D. Management of Chronic Insomnia Disorder in Adults: A Clinical Practice Guideline From the American College of Physicians. Ann. Intern. Med. 2016, 165, 125–133.
- Perlis, M.L.; Jungquist, C.; Smith, M.T.; Posner, D. Cognitive Behavioral Treatment of Insomnia: A Session-by-Session Guide; Springer Science and Business Media: New York, NY, USA, 2008.
- Zhou, E.S.; Partridge, A.H.; Syrjala, K.L.; Michaud, A.L.; Recklitis, C.J. Evaluation and treatment of insomnia in adult cancer survivorship programs. J. Cancer Surviv. 2017, 11, 74–79.
- Mindell, J.A.; Bartle, A.; Wahab, N.A.; Ahn, Y.; Ramamurthy, M.B.; Huong, H.T.; Kohyama, J.; Ruangdaraganon, N.; Sekartini, R.; Teng, A.; et al. Sleep education in medical school curriculum: A glimpse across countries. Sleep Med. 2011, 12, 928–931.
- Thomas, A.; Grandner, M.; Nowakowski, S.; Nesom, G.; Corbitt, C.; Perlis, M.L. Where are the Behavioral Sleep Medicine Providers and Where are They Needed? A Geographic Assessment. Behav. Sleep Med. 2016, 14, 687–698.
- Stinson, K.; Tang, N.K.; Harvey, A.G. Barriers to treatment seeking in primary insomnia in the United Kingdom: A cross-sectional perspective. Sleep 2006, 29, 1643–1646.
- Matthews, E.E.; Arnedt, J.T.; McCarthy, M.S.; Cuddihy, L.J.; Aloia, M.S. Adherence to cognitive behavioral therapy for insomnia: A systematic review. Sleep Med. Rev. 2013, 17, 453–464.
- Mohammadi, S.; Sulaiman, S.; Koon, P.B.; Amani, R.; Hosseini, S.M. Association of nutritional status with quality of life in breast cancer survivors. Asian Pac. J. Cancer Prev. 2013, 14, 7749–7755.
