Selecting the most appropriate inhalation device from the wide range available is essential for the successful management of patients with chronic obstructive pulmonary disease. Although choice is good for healthcare professionals, knowing which inhaler to prescribe is a complex consideration. Among the key factors to consider are quality of disease control, inhaler technique, inhaler resistance and inspiratory flow, inhaler design and mechanisms of drug delivery, insurance and reimbursement restrictions, and environmental impact. In this aentrticley, we offer a simple, practical tool that brings together all these factors and includes hyperlinks to other published resources from the United Kingdom, Belgium, and The Netherlands.
- primary care
- respiratory
- inhaler
- device
1. Introduction
2. Overview of Approaches to Match the Inhaler to the Patient
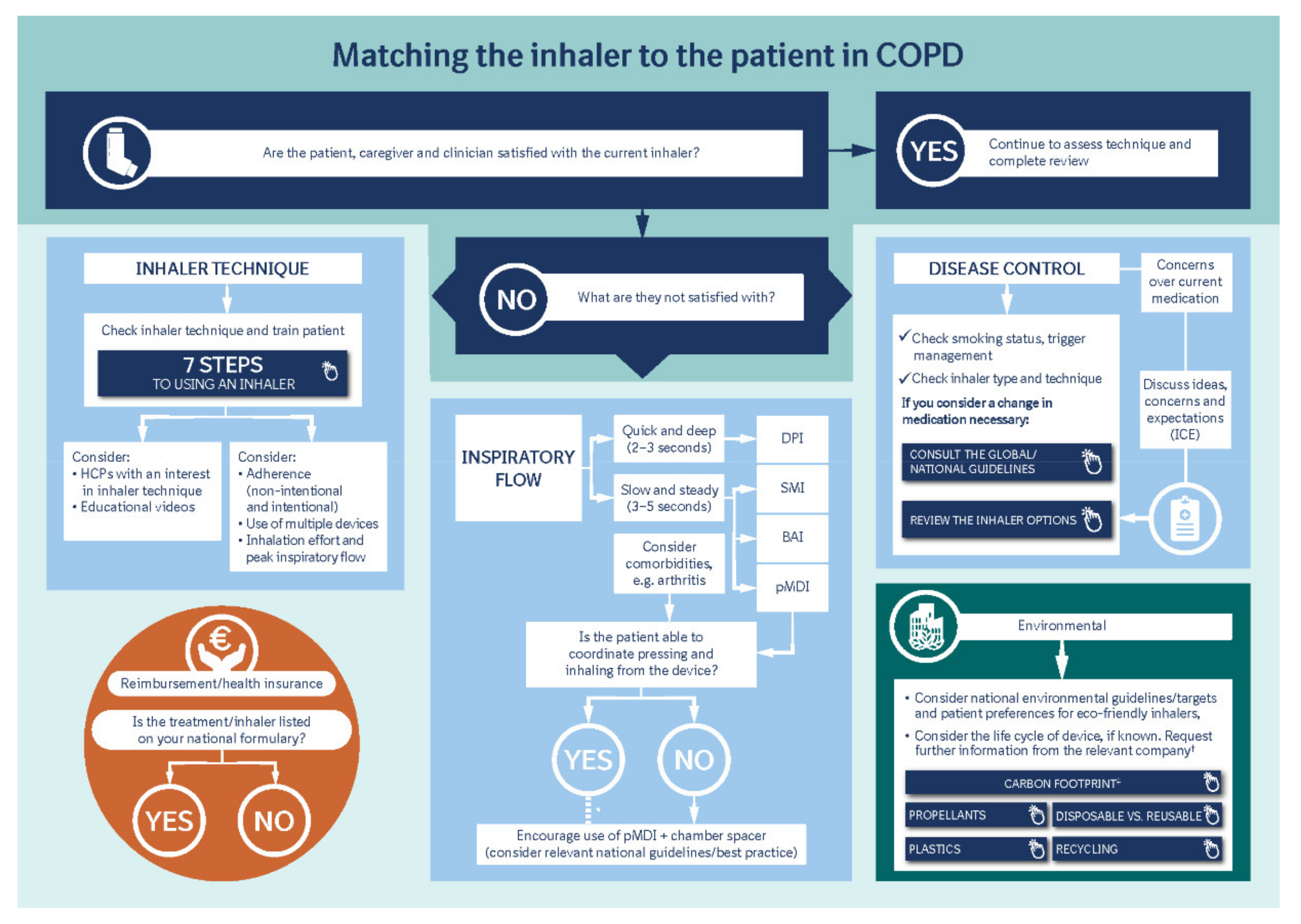
3. Disease Control
4. Inhaler Technique
It is also important to differentiate between intentional and non-intentional non-adherence [12]. Some patients choose not to use their inhaler—due to fear of side effects, for example—which negatively impacts their disease control. Other patients actively engage with disease management but may not receive the full benefits of their medication due to incorrect inhalation technique, or other reasons beyond their control. For example, comorbidities such as arthritis can impact the handling of an inhaler. Ciciliani et al. assessed finger strength across different inhaler types, demonstrating that different finger strengths are required for each device, and therefore concluding that a patient’s manual dexterity and strength need to be taken into consideration when prescribing a device [19].
Another important aspect to consider is device familiarity, as previous studies have demonstrated that patients with COPD who were prescribed inhalation devices requiring a similar technique to their previous devices had better outcomes (such as a lower rate of exacerbations) than patients who were prescribed devices that required a different inhalation technique [20]. In reality, however, some patients may have to overcome the potential confusion associated with learning more than one inhaler technique. Short-acting reliever inhalers, for example, which are typically pressurized metered-dose inhalers (pMDIs), often utilized due to cost or the potential for more effective delivery of bronchodilation in an emergency, may require a different inhalation technique from the inhaler or inhalers used to deliver a patient’s long-acting maintenance therapy.
5. Inspiratory Flow and Aerosol Deposition
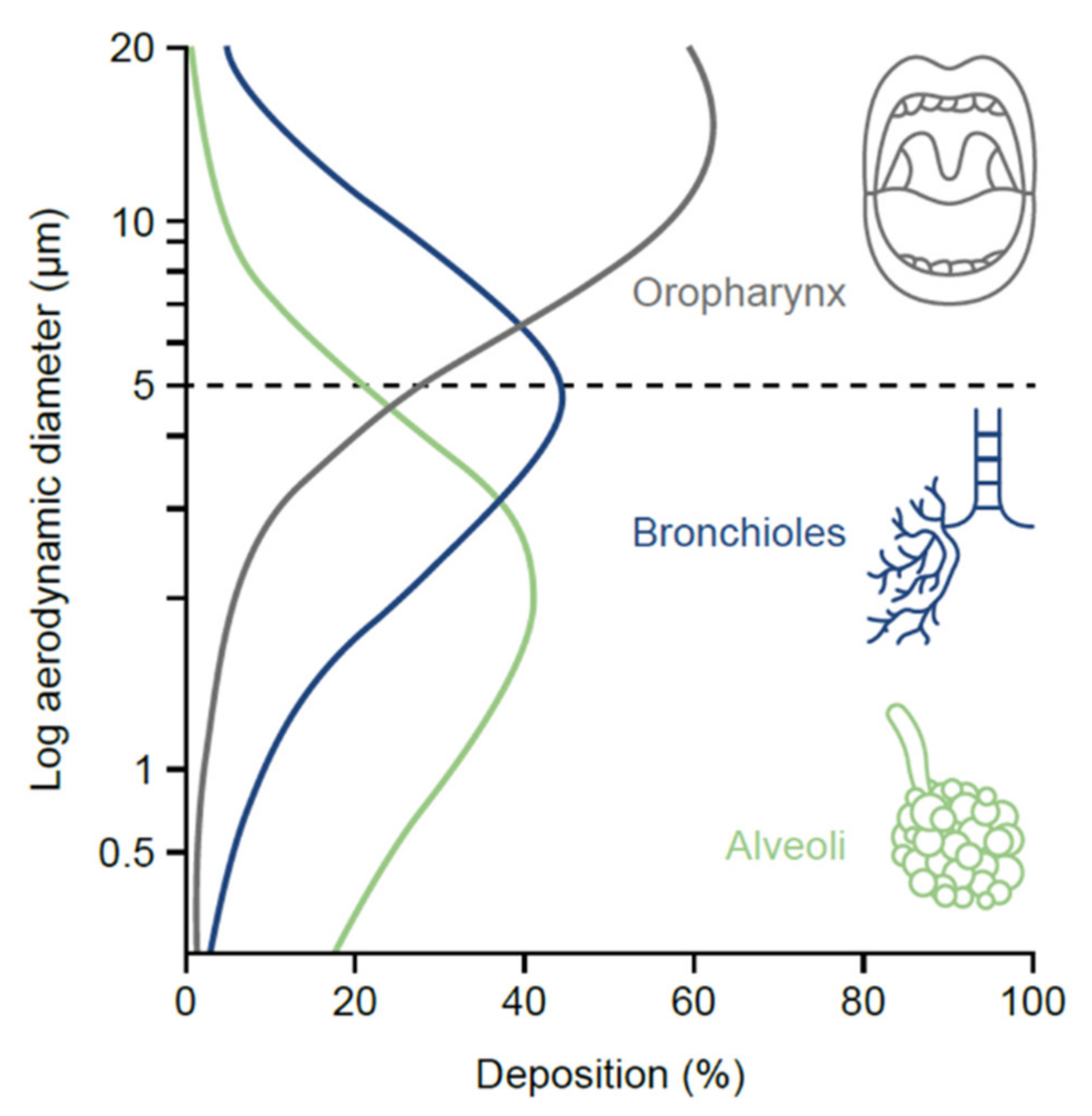
One of the most important particle-related factors that affects aerosol deposition is aerodynamic diameter. This is often referred to in terms of mass median aerodynamic diameter, i.e., the diameter at which 50% of the particles of an aerosol are larger by mass and 50% are smaller [17]. Particles < 5 µm in diameter (fine-particle fraction) have the greatest potential to deposit in the lungs, penetrating beyond the upper airways and traveling to the peripheral airways and alveoli (the majority of devices now generate a significant proportion of particles ≤ 5 µm to ensure optimal drug delivery to the lungs) [17]. As the particles reach the lung periphery, the rate of airflow decreases, increasing drug deposition. If a patient is able to hold their breath for longer than 5 s after inhalation, the drug particles will spend a longer time in the peripheral airways undisturbed by inspiratory or expiratory flow, resulting in higher deposition [25].
6. Inhaler Design and Mechanisms of Drug Delivery
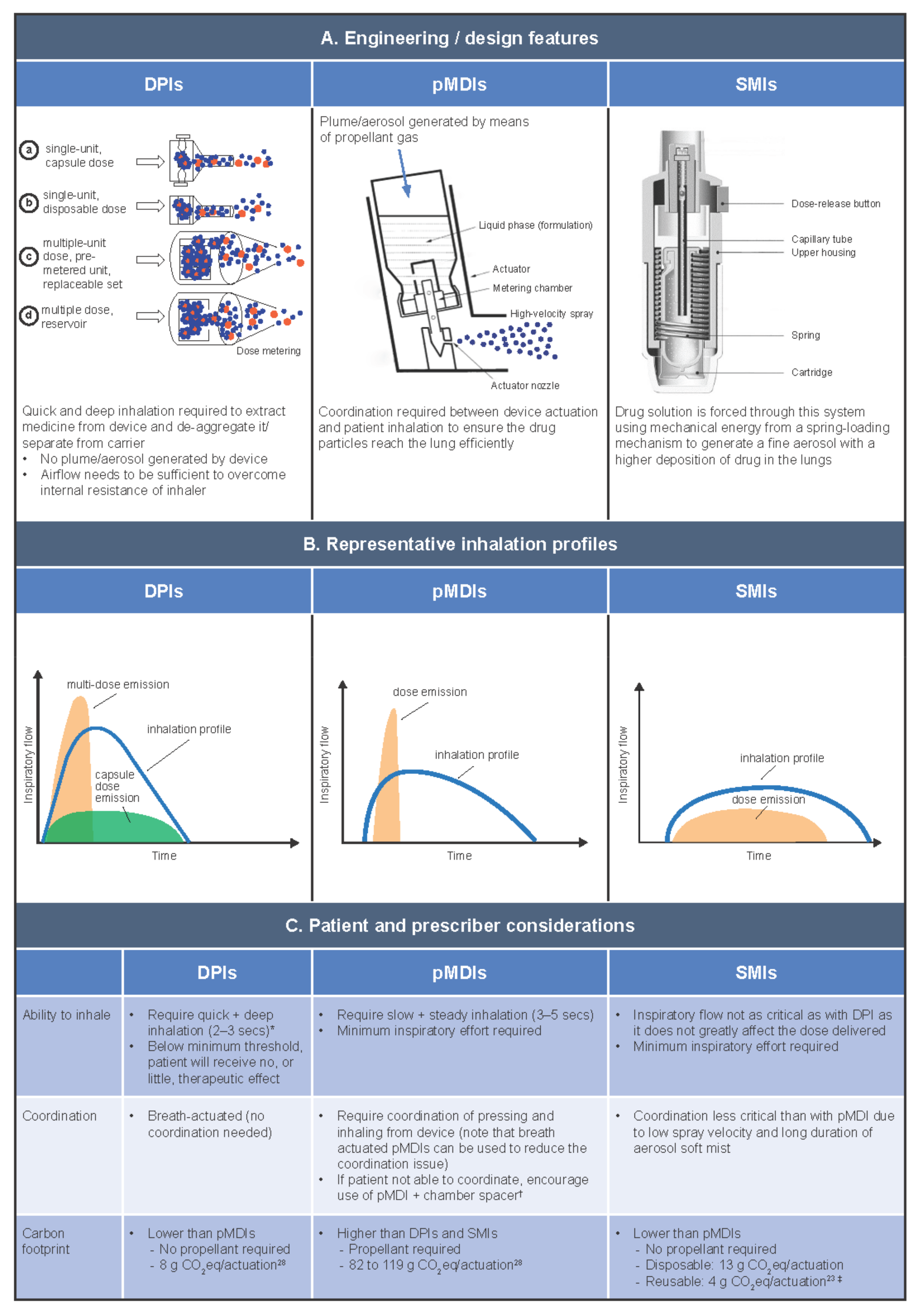
6.1. DPIs
DPIs are portable and compact, and unlike pMDIs, they do not contain propellant gas. There are two basic types of DPI: multidose DPIs and single-dose capsule DPIs. There are also two variations of the multi-dose DPI: those containing a bulk formulation in a reservoir, where the meter is patient controlled; and those containing pre-metered, factory-dispensed doses packaged inside blisters within the device [17], where the meter is not patient controlled. The inspiratory force and airflow generated by the patient when they inhale from a DPI de-aggregates the drug formulation from its carrier molecules, creating the energy to extract the drug from the device and deliver it into the patient. DPIs, therefore, have a minimum threshold of optimum flow, and if the inspiratory flow is too low, a reduced dose is delivered, and the patient may receive sub-optimal clinical benefit [29]. Unlike pMDIs, a patient’s inhalation flow profile when using a DPI is typically quick (> 60 L/min) or, depending on the resistance of the device, slightly slower (e.g., > 30 L/min) [30]. Quicker inhalation is particularly critical for reservoir/blister-type DPIs, which emit the dose earlier compared with capsule DPIs [17]. Regardless of speed, the inhalation from a DPI must be deep to achieve optimal drug delivery [17]. New smaller-particle formulations have been developed for use with DPIs, which may help to optimize drug delivery [31].
As DPIs require a minimum level of inspiratory flow to separate the medication from its carrier molecule, they are less appropriate for use during an exacerbation [26]. Studies suggest that failure to achieve forceful inspiratory flow through the device is a common mishandling error with DPIs, occurring in an estimated 26–38% of cases [7,31,32] and impacting clinical outcomes [7]. In a systematic review of inhaler use errors by Sanchis et al., other common errors associated with DPI use were found to be lack of full expiration before inhalation (42–50%), lack of post-inhalation breath-hold (33–40%), and incorrect device preparation (26–33%) [32].
6.2. pMDIs
pMDIs are compact, contain a metered dose, and can be used even in very breathless patients (for example, during exacerbations); however, they require good hand–lung coordination, which some patients can find challenging. pMDIs have a higher carbon footprint compared with other devices [23,28], due to the global warming potential of the propellant gas they contain (originally chlorofluorocarbon (CFC), later replaced with hydrofluorocarbon), which aerosolizes the drug formulation to produce a high-velocity spray. The use of propellants and the production of the high-velocity spray mean that inhalation from pMDIs must be well timed and well controlled (slow and steady), otherwise the spray may be deposited largely in the mouth or throat. Breath-actuated pMDIs have been developed as an alternative to traditional pMDIs to avoid the problem of poor actuation–inhalation coordination by automatically actuating at low inspiratory flow rates [17]. Training devices that do not contain any medication are also available to improve patients’ pMDI technique alongside verbal inhaler training by HCPs [33]. CFC-containing pMDIs previously caused the freon effect (chilling sensation at the back of the throat after inhalation), which negatively impacted patient inhalation. A small amount of ethanol has since been added to pMDI formulations to alter the taste, increase the temperature, and decrease the velocity at which the medication is released from the device [17].
Even with the right inhalation technique, pMDIs can leave a high deposition of the drug in the patient’s mouth and oropharynx. Spacers can help to alleviate the difficulty of inhaling while pressing for users of pMDIs. As many patients struggle with this technique, HCPs should actively encourage the use of a spacer (in The Netherlands, for example, it is routine practice for all pMDIs to be prescribed with a spacer). The advantages of using a spacer are that the inspiratory effort of the patient is not as critical, and the patient is likely to receive a higher lung deposition than when using a pMDI alone (i.e., less deposition in the mouth and oropharynx). However, spacers are less portable, bring additional cost, and require the correct maintenance [26]. Note that pMDIs that emit smaller particles are associated with improved lung deposition and lower oropharyngeal deposition without the need for a spacer and provide an alternative option compared with traditional pMDIs [34].
6.3. SMIs
SMIs are also portable and compact. In addition, they have a lower dependency on the inspiratory effort of the patient to deliver a high fine-particle fraction and high lung deposition, and do not contain propellants [26]. SMIs atomize the drug solution using mechanical energy imparted by pressure from a spring. When the pressure is released, the solution is forced through a fine nozzle, producing a slow-moving mist of the drug formulation. SMIs deliver the high fine-particle fraction at slow velocity, resulting in good lung deposition even when a patient has a low inhalation flow [35]. As such, it is not necessary for physicians to consider peak inspiratory flow when prescribing an SMI, as the patient’s inspiratory capacity does not affect the dose delivered. There is only one SMI device currently available, which is compatible with a limited range of medications relative to other inhaler types [2,26]. Thus, it is important to consider the medication options and their compatibility with available devices alongside other factors (such as patient skills and cost) when choosing an inhaler [26].
7. Reimbursement
8. Environmental Impact
References
- World Health Organization. Burden of COPD. Available online: https://www.who.int/respiratory/copd/burden/en (accessed on 12 May 2021).
- Global Initiative for Chronic Obstructive Lung Disease. Global Strategy for the Diagnosis, Management, and Prevention of Chronic Obstructive Pulmonary Disease (2021 Report). Available online: https://goldcopd.org/wp-content/uploads/2020/11/GOLD-REPORT-2021-v1.1-25Nov20_WMV.pdf (accessed on 14 April 2021).
- Liew, K.; Wilkinson, A. P280 How do we choose inhalers? patient and physician perspectives on environmental, financial and ease-of-use factors. Thorax 2017, 72, A235.
- Walpole, S.; Fitzpatric, A.; Moffatt, K.; Smith, K.; Potts, A.; Doe, S.; Burns, G.; Tedd, H. P187 Ease of use, effectiveness and environmental impacts: Evaluating inhaler prescriptions, patient preferences and opportunities for improvement. Thorax 2021, 76, A190–A191.
- Matthys, J.; Elwyn, G.; Van Nuland, M.; Van Maele, G.; De Sutter, A.; De Meyere, M.; Deveugele, M. Patients’ ideas, concerns, and expectations (ICE) in general practice: Impact on prescribing. Br. J. Gen. Pract. 2009, 59, 29–36.
- Sriram, K.B.; Percival, M. Suboptimal inhaler medication adherence and incorrect technique are common among chronic obstructive pulmonary disease patients. Chron. Respir. Dis. 2016, 13, 13–22.
- Melani, A.S.; Bonavia, M.; Cilenti, V.; Cinti, C.; Lodi, M.; Martucci, P.; Serra, M.; Scichilone, N.; Sestini, P.; Aliani, M.; et al. Inhaler mishandling remains common in real life and is associated with reduced disease control. Respir. Med. 2011, 105, 930–938.
- Chrystyn, H.; van der Palen, J.; Sharma, R.; Barnes, N.; Delafont, B.; Mahajan, A.; Thomas, M. Device errors in asthma and COPD: Systematic literature review and meta-analysis. NPJ Prim. Care Respir. Med. 2017, 27, 22.
- Bender, B.G. Editorial-How Important Are Inhaler Technique Errors? Chronic Obstr. Pulm. Dis. 2019, 6, 203–205.
- Crompton, G.K.; Barnes, P.J.; Broeders, M.; Corrigan, C.; Corbetta, L.; Dekhuijzen, R.; Dubus, J.C.; Magnan, A.; Massone, F.; Sanchis, J.; et al. The need to improve inhalation technique in Europe: A report from the Aerosol Drug Management Improvement Team. Respir. Med. 2006, 100, 1479–1494.
- Carrion Valero, F.; Maya Martinez, M.; Fontana Sanchis, I.; Diaz Lopez, J.; Marin Pardo, J. Inhalation technique in patients with chronic respiratory diseases. Arch. Bronconeumol. 2000, 36, 236–240.
- Bryant, J.; McDonald, V.M.; Boyes, A.; Sanson-Fisher, R.; Paul, C.; Melville, J. Improving medication adherence in chronic obstructive pulmonary disease: A systematic review. Respir. Res. 2013, 14, 109.
- Takemura, M.; Kobayashi, M.; Kimura, K.; Mitsui, K.; Masui, H.; Koyama, M.; Itotani, R.; Ishitoko, M.; Suzuki, S.; Aihara, K.; et al. Repeated instruction on inhalation technique improves adherence to the therapeutic regimen in asthma. J. Asthma 2010, 47, 202–208.
- Price, D.; Keininger, D.L.; Viswanad, B.; Gasser, M.; Walda, S.; Gutzwiller, F.S. Factors associated with appropriate inhaler use in patients with COPD—Lessons from the REAL survey. Int. J. Chron. Obstruct. Pulmon. Dis. 2018, 13, 695–702.
- Petite, S.E.; Hess, M.W.; Wachtel, H. The role of the pharmacist in inhaler selection and education in chronic obstructive pulmonary disease. J. Pharm. Technol. 2021, 37, 95–106.
- Lareau, S.C.; Hodder, R. Teaching inhaler use in chronic obstructive pulmonary disease patients. J. Am. Acad. Nurse Pract. 2012, 24, 113–120.
- Laube, B.L.; Janssens, H.M.; de Jongh, F.H.; Devadason, S.G.; Dhand, R.; Diot, P.; Everard, M.L.; Horvath, I.; Navalesi, P.; Voshaar, T.; et al. What the pulmonary specialist should know about the new inhalation therapies. Eur. Respir. J. 2011, 37, 1308–1331.
- UK Inhaler Group. Inhaler Standards and Competency Document. Available online: https://www.respiratoryfutures.org.uk/media/69774/ukig-inhaler-standards-january-2017.pdf (accessed on 4 December 2020).
- Usmani, O.S.; Biddiscombe, M.F.; Barnes, P.J. Regional lung deposition and bronchodilator response as a function of beta2-agonist particle size. Am. J. Respir. Crit. Care Med. 2005, 172, 1497–1504.
- Ibrahim, M.; Verma, R.; Garcia-Contreras, L. Inhalation drug delivery devices: Technology update. Med. Devices 2015, 8, 131–139.
- Hänsel, M.; Bambach, T.; Wachtel, H. Reduced environmental impact of the reusable Respimat® Soft Mist™ inhaler compared with pressurised metered-dose inhalers. Adv. Ther. 2019, 36, 2487–2492.
- Labiris, N.R.; Dolovich, M.B. Pulmonary drug delivery. Part I: Physiological factors affecting therapeutic effectiveness of aerosolized medications. Br. J. Clin. Pharmacol. 2003, 56, 588–599.
- Kaplan, A.; Price, D. Matching Inhaler Devices with Patients: The Role of the Primary Care Physician. Can. Respir. J. 2018, 2018, 9473051.
- De Araújo, A.D.; Correia-de-Sousa, J. COPD: Will there be room for nebulisers after the current COVID-19 pandemic? Open Respir. Arch. 2020, 2, 265–266.
- Panigone, S.; Sandri, F.; Ferri, R.; Volpato, A.; Nudo, E.; Nicolini, G. Environmental impact of inhalers for respiratory diseases: Decreasing the carbon footprint while preserving patient-tailored treatment. BMJ Open Respir. Res. 2020, 7, e000571.
- Jeswani, H.K.; Azapagic, A. Life cycle environmental impacts of inhalers. J. Clean. Prod. 2019, 237, 117733.
- Keeley, D.; Scullion, J.E.; Usmani, O.S. Minimising the environmental impact of inhaled therapies: Problems with policy on low carbon inhalers. Eur. Respir. J. 2020, 55, 2001122.
