Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Catherine Yang and Version 1 by Chao Wu.
ATP-binding cassette (ABC) transporters, a large class of transmembrane proteins, are widely found in organisms and play an important role in the transport of xenobiotics. Insect ABC transporters are involved in insecticide detoxification and Bacillus thuringiensis (Bt) toxin perforation. The complete ABC transporter is composed of two hydrophobic transmembrane domains (TMDs) and two nucleotide binding domains (NBDs).
- insect
- ABC transporters
- structure
- Bt Insecticidal Activity
1. Introduction
ATP-binding cassette (ABC) proteins comprise an extensive and variable transporter superfamily within P-loop motif and are found in all living organisms [1,2,3][1][2][3]. Studies on ABC transporters began in the early 1970s with the biochemical characterization of substrate-binding protein-dependent transport in Escherichia coli that was directly energized by hydrolysis of ATP [4,5][4][5]. In 1982, cytoplasmic membrane-associated transporter genes in the histidine transport system of Salmonella typhimurium (coded by the hisP gene) and maltose-maltodextrin transport system of E. coli (coded by the malK gene) were cloned [6,7][6][7]. Concurrently, in mammalian cells, the gene encoding permeability, glycoprotein (P-gp, a large glycosylated membrane protein related to multi-drug resistance) was identified and cloned in 1985 [8,9][8][9]. Eventually, substrate-binding transport proteins with ATP-binding subunits were found to constitute a large superfamily of transport proteins and termed ABC transporters in 1990 [10]. On the basis of differences in the ATP-binding sites among insect ABC transporters, the superfamily can be divided into eight subfamilies (ABCA to ABCH) [11].
2. Structure and Mechanism of ABC Transporters
Structural models of ABC transporters are based on the crystal structure of different bacterial proteins that act as importers such as vitamin B12 transporter BtuCDF from E. coli and exporters such as the multidrug exporter Sav1866 from Staphylococcus aureus or related flippases such as MsbA lipid flippase from E. coli [29,30,31][12][13][14]. On the basis of their architecture and biochemical activity, the ABC importers have been divided into type I and type II [12,32,33][15][16][17]. The energy coupling factor (ECF) transporters, which differ structurally and functionally from other ABC importers, are sometimes considered as type III ABC importers [34,35,36,37][18][19][20][21]. However, ABC importers have only been confirmed in prokaryotes, not in eukaryotes [11,15][11][22]; therefore, in this review, we focus only on ABC exporters.
The structure of ABC transporters is highly conserved among most eukaryotic organisms, including insects. A functional ABC transporter is characterized by the presence of a P-type traffic ATPase, which comprises two cytosolic nucleotide-binding domains (NBDs) and two transmembrane domains (TMDs) [1,12,38][1][15][23] (Figure 1A). The four domains of a functional transporter (2TMDs-2NBDs) are combined in a single polypeptide, forming a full transporter (FT), whereas a half transporter (HT) contains one TMD and one NBD, which are sometimes encoded as separate polypeptides and then fused into multidomain proteins. For ATP binding and hydrolysis, the HT must become a functional transporter by forming homo- or heterodimeric complexes. The NBD contains several highly conserved nucleotide-binding sequences such as the Walker A and B motifs, common in nucleotide-binding proteins (the Walker B motif also provides the catalytic base); d-loop, which contains an aspartate residue and is responsible for forming a salt bridge; Q-loop, which contains a glutamate residue and acts as the attacking nucleophile in ATP hydrolysis; H motif, which has an invariant histidine active site that may be involved in maintaining the stability of the pre-hydrolytic state; and an α-helical region that has the ABC signature sequence (LSGGQ motif) [12,13,39][15][24][25]. The ABC exporter fold, a prominent quaternary structure in TMDs in all ABC exporters, is characterized by 12 transmembrane helices and acts as a switch between different conformational changes and initiates substrate translocation [12][15].
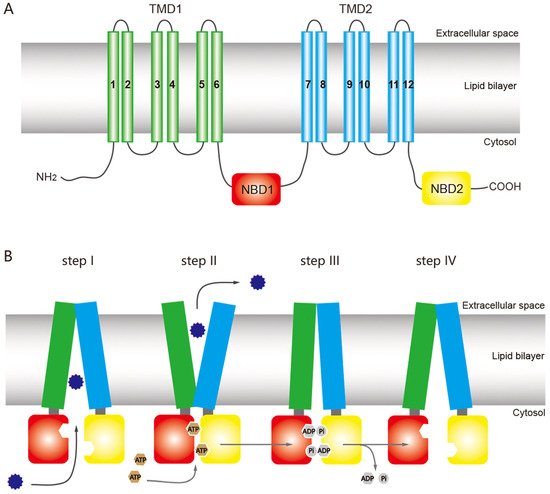

Figure 1. General structure of an ATP-binding cassette (ABC) full transporter (ABC exporter) and the ATP-switch model for the transport mechanism of ABC transporters. (A) Typical ABC full transporter with two transmembrane domains (TMDs), TMD1 (green) and TMD2 (sky blue), and two nucleotide-binding domains (NBDs), NBD1 (red) and NBD2 (yellow). Each transmembrane domain (TMD) contains six transmembrane helices. The “long” multidrug-resistance associated proteins (MRPs) of the ABCC subfamily contains an additional TMD (TMD0) at the N terminus [46][26]. (B) The ATP-switch model [14][27] includes (I) binding of the substrates (12-point blue circle) to the TMDs; (II) subsequent structural changes to the NBDs (red and yellow), hydrolysis of ATP (brown circles), followed by closed dimer formation of the NBDs and major conformational change in the TMDs, which initiates substrate translocation; (III) the ATP is hydrolyzed (gray circles), releasing ADP and Pi, and (IV) finally destabilization of the closed dimer restores its initial open dimer configuration for another new cycle. This figure is drawn by following the previous report of ABC transporter by Dermauw & Van Leeuwen [14][27].
ABC transporters have a common mechanism for exporting substrates across the membrane by hydrolyzing ATP as a pump, but other models have been proposed for the ABC transporter mechanism based on structural and biochemical evidence, including the ATP-switch [39][25], alternating site [40][28], constant contact [41[29][30],42], and thermodynamic models [43][31]. Among these models, the ATP-switch model provides a reasonable framework for the transport mechanism [39,44,45][25][32][33] in which repeated communication between NBDs and TMDs occurs in both directions and involves only non-covalent conformational changes. The transport process is initiated by the binding of the substrate to the TMDs, and subsequent structural changes are transmitted to the NBDs, which include ATP-binding and closed dimer formation of the NBDs. Then the closed NBD dimer induces a substantial conformational change in the TMDs. This conformational change initiates translocation of the substrate through a rotation of the TMDs and opening toward the extracellular milieu. Finally, the ATP is hydrolyzed, releasing ADP and Pi and destabilizing the closed dimer conformation to restore its open dimer configuration for another new cycle [39,44,45][25][32][33] (Figure 1B).
3. ABC Transporter Subfamilies in Insects
In recent years, with the large-scale development of genome sequencing technology, the sequencing results have shown that ABC transporter genes are highly conserved in many insects (Figure 2). Aside from some important discoveries on the function of some ABC transporters in insects, however, knowledge on the role and function of these proteins is still limited. ABC transporters of numerous important agricultural pests and model insects, such as D. melanogaster, B. mori, Helicoverpa armigera and Plutella xylostella, have been reported (Table 1).
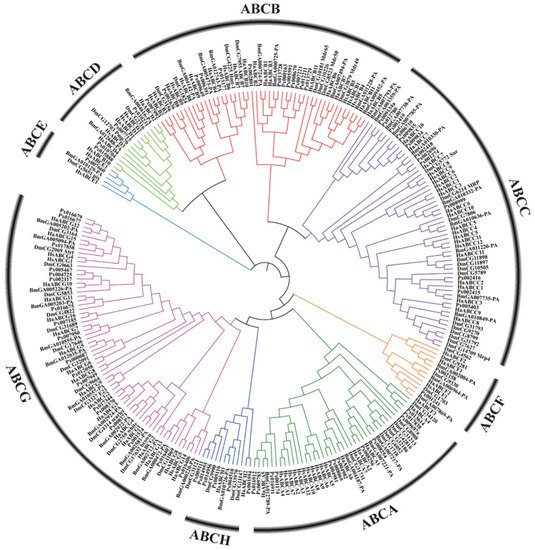

Figure 2. Phylogenetic tree based on amino acid sequences of 262 ABC transporters (Supplementary Material 1) from several insects and humans. The sequences were aligned using MUSCLE. The evolutionary history was inferred using the neighbor-joining method and MEGA-X with 1000 bootstrap replicates. All positions with less than 95% site coverage were eliminated. Species codes: Ha, Helicoverpa armigera; Bm, Bombyx mori; Px, Plutella xylostella; Hs, Homo sapiens; Dm, Drosophila melanogaster.
Table 1.
Distribution of genes among ABC transporter subfamilies for different arthropods and
Homo sapiens
.
| Organisms | A | B | C | D | E | F | G | H | Total | References |
|---|
| Homo sapiens | 12 | 11 | 12 | 4 | 1 | 3 | 5 | 0 | 48 | [1] | ||
| Drosophila melanogaster | 10 | 8 | 14 | 2 | 1 | 3 | 15 | 3 | 56 | [1] | ||
| Anopheles gambiae | 9 | 5 | 13 | 2 | 1 | 3 | 16 | 3 | 52 | [47] | [34] | |
| Daphnia pulex | 4 | 7 | 7 | 3 | 1 | 4 | 24 | 15 | 65 | [48] | [35] | |
| Pediculus humanus humanus | 2 | 6 | 5 | 2 | 1 | 3 | 13 | 6 | 40 | a | [49] | [36] |
| Apis melifera | 3 | 5 | 9 | 2 | 1 | 3 | 15 | 3 | 41 | [50] | [37] | |
| Bombyx mori | 7 | 9 | 11 | 2 | 1 | 3 | 16 | 2 | 51 | b | [50,51,52] | [37][38][39] |
| Tribolium castaneum | 10 | 6 | 35 | 2 | 1 | 3 | 13 | 3 | 73 | [20] | [40] | |
| Tetranychus urticae | 9 | 4 | 39 | 2 | 1 | 3 | 23 | 22 | 103 | [53] | [41] | |
| Chrysomela populi | 5 | |||||||||||
| Helicoverpa zea | ||||||||||||
| 7 | ||||||||||||
| 11 | ||||||||||||
| 11 | ||||||||||||
| 2 | ||||||||||||
| 1 | 3 | 17 | 2 | 54 | [ | 52 | ] | [ | 39 | ] | ||
| Acyrthosiphon pisum | 11 | 9 | 16 | 2 | 1 | 4 | 19 | 9 | 71 | [63,64] | [51][52] |
a Includes two uncharacterized ABC transporters. b The number of B. mori ABC transporter genes in the different subfamilies is mainly based on the latest report [52][39], but were reported previously [50][37].
4. ABC Transporter Roles in Bt Insecticidal Activity
The ABC transporters that act as Bt-toxin receptor belong to subfamilies A, B, C and G (Table 2). With the long-term cultivation of Bt cotton, field resistance to Cry2Ab toxin evolved in H. armigera. The resistance of two H. armigera strains to Cry2Ab was an independent evolutionary event involving different deletion mutations, which were located in different exons of the same ABCA2 gene [68][53]. A homologous deletion in HaABCA2 was also detected in resistant lines of H. punctigera Cry2Ab. A similar situation was also found in resistant lines of Pectinophora gossypiella; a loss of PgABCA2 exon 6 was caused by alternative splicing in resistant larvae of laboratory-selected strains in Arizona and field-selected strains in India [118][54]. In addition, insect ABCA2 was expressed in the midgut where the Cry2Ab toxin binds [68,119][53][55]. A bioassay of two strains of H. armigera with CRISPR/cas9 knockout of HaABCA2 showed that HaABCA2 plays an important role in mediating resistance to Cry2Aa and Cry2Ab [120][56].
Table 2.
Bt toxins and their possible corresponding ABC transporters as receptors in insects.
| Bt toxin | Receptor | Target pest | Reference | ||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
| Cry1Aa | ABCC2 | Bombyx mori | [121] | [57] | |||||||||||||||
| Cry1Ab | ABCC2 | Bombyx mori | [122] | [58] | |||||||||||||||
| ABCG1 | Ostrinia furnacalis | [123] | [59] | ||||||||||||||||
| Cry1Ac | ABCC2 | Helicoverpa armigera | [25] | [60] | |||||||||||||||
| ABCC2 | Heliothis virescens | [124] | [61] | ||||||||||||||||
| ABCG1 | Ostrinia furnacalis | [123] | [59] | ||||||||||||||||
| ABCG1 | Plutella xylostella | [125] | [62] | ||||||||||||||||
| ABCC2 | Plutella xylostella | [126] | [63] | ||||||||||||||||
| ABCC2 | Spodoptera exigua | [127] | [64] | ||||||||||||||||
| 8 | 29 | 2 | 1 | 3 | 14 | 3 | 65 | [ | 54 | ] | [42] | ||||||||
| ABCC3 | Spodoptera frugiperda | [128] | [65] | Lygus hesperus | 11 | 6 | 12 | ABCC22 | 1 | 3 | 19 | 11 | 65 | Spodoptera exigua | [127] | [ | [55] | [43] | |
| Cry1Ca | 64 | ] | Lepeophtheirus salmonis | 3 | 4 | 11 | 3 | 1 | 4 | 2 | 5 | 33 | |||||||
| Cry1Fa | ABCC2 | Ostrinia nubilalis | [129] | [66] | [ | 56 | ] | [ | 44 | ] | |||||||||
| Laodelphax striatellux | 2 | 6 | 5 | 2 | 1 | 2 | 14 | 8 | |||||||||||
| Cry2Aa | ABCA2 | Helicoverpa armigera | 40 | [120] | [56] | [ | 57 | ] | [ | 45] | |||||||||
| Bemisia tabaci | 8 | 3 | 6 | 2 | 1 | 3 | 23 | 9 | 55 | ||||||||||
| Cry2Ab | ABCA2 | Helicoverpa armigera | [68] | [53] | [ | 58 | ] | [ | 46 | ] | |||||||||
| Plutella xylostella | 15 | 14 | 21 | 3 | 1 | 3 | 19 | 6 | 82 | [59, | |||||||||
| ABCA2 | Helicoverpa punctigera | [68] | [53] | 60 | ] | [ | 47 | ] | [48] | ||||||||||
| Manduca sexta | 7 | 9 | 11 | 2 | 1 | 3 | 13 | 3 | 52 | [60] | |||||||||
| ABCA2 | Pectinophora gossypiella | [118] | [54 | [ | 48 | ] | |||||||||||||
| ] | Danaus plexippus | 8 | 16 | 12 | 3 | 1 | 3 | 16 | 3 | 62 | [60] | [48] | |||||||
| Cry3Aa | ABCB1 | Chrysomela tremuela | [130] | [67] | Heliconius melpomene | 10 | 11 | 15 | 2 | 1 | 3 | 17 | 3 | ||||||
| Cry3Bb1 | 62 | [ | 60 | ] | [ | 48 | ] | ||||||||||||
| ABCB1 | Diabrotica virgifera virgifera | [ | 131] | [68] | Helicoverpa armigera | 7 | 11 | 11 | 2 | 1 | 3 | 17 | 2 | 54 | [52,61,62] | [39][49][50] | |||
A Chrysomela tremuela strain survived and reproduced on transgenic poplar trees that expressed a high level of Cry3Aa Bt toxin, and its Cry3Aa resistance was an autosomal recessive trait. Candidate resistance genes were analyzed using midgut transcriptome of larvae, demonstrating that a mutation of the ABCB homolog of P-gp was closely linked to the resistance of Cry3Aa, and named CtABCB1. A 4-bp deletion in CtABCB1 introduced a frame shift with a premature stop codon, resulting in the loss of transporter signature motifs 1 (TpM1) and transmembrane domain 2 (TMD2) [130][67]. A CtABCB1 homologous gene in western corn rootworm (Diabrotica virgifera virgifera) was also shown to be genetically linked to Cry3Bb1 resistance [131][68]. Perhaps the insect ABCB1 transporter is a receptor specific for Cry3 toxin, and structural changes in this transporter may be associated with Cry3 toxin resistance of the insect.
Some ABCC transporters are a functional receptor for more than one Cry1A toxin. The binding of Cry1Ac toxin to ABCC2 on membrane vesicles can lead to membrane perforation. Bretschneider et al. studied the relationship between the ABCC2 of Heliothis virescens and cytotoxicity of three Cry1A toxins (Cry1Aa, Cry1Ab, Cry1Ac) in Spodoptera frugiperda Sf9 cells and found that ABCC2 is the central target of Cry1A toxin action [132][69]. Heterologous expression in D. melanogaster has been used to validate the involvement of ABCC2 in Cry1Ac toxin binding [133][70]. An inactivating mutation, such as mis-splicing of ABCC2 or a mutation causing a single amino acid change, could reduce binding of Cry1Ac toxin, conferring high levels of Bt resistance in the target pest [25,124,134][60][61][71]. In a Cry1Ac-resistant strain of P. xylostella with a 30-bp deletion in exon 20, was expected to cause the removal of the 12th transmembrane (TM) domain and carboxyl terminal of TM12 that is located outside the cell. The mutation may result in the lack of function of a core ATP-binding loop [126][63]. In addition, an ABCC transporter is also the receptor of Cry1Ca and Cry1Fa toxins [127,129][64][66]. A cytotoxicity assay showed that the binding affinity of ABCC transporters to Cry toxin was largely linked to the susceptibility of receptor-expressing cells to Cry toxin and that the extracellular loop (ECL) structures determine the specificity of ABCC to Cry toxins [135][72].
Bt resistance mediated by ABCC transporters involves not only different forms of gene mutations, but also the regulation of ABCC gene expression. The study of Bt resistance in hybrid larvae of B. mori revealed a trans-regulatory mechanism involved in the allele-specific expression of ABCC2 in response to Cry1Ab toxin, which may play an important role in insect Bt resistance [122][58]. The binding of Cry1Ac toxin to ABCC transporters that causes midgut membrane perforation is regulated by a constitutively and transcriptionally activated upstream gene (MAP4K4) in the MAPK signaling pathway [125][62]. In addition, Forkhead box protein A (FOXA) upregulates expression of ABCC2 and ABCC3 genes in Sf9 cells [128][65]. ABCC2 and ABCC3 are important receptors of Cry1Ac toxin, and their expression level has a significant influence on insect Bt resistance; thus, FOXA may be involved in the regulation of insect resistance to Cry1Ac toxin.
ABCG1 (white) gene is one of the most widely studied members of the ABCG protein subfamily. The white protein in P. xylostella is located on the cell membrane, and the expression of Pxwhite gene in the midgut of a Bt-resistant population is significantly lower than in a susceptible population. After RNAi silencing of the midgut Pxwhite gene, the sensitivity of the larvae to Bt-Cry1Ac toxin significantly decreased [136][73]. Genetic linkage analysis confirmed that the decrease was closely linked to the Bt-Cry1Ac resistance of the moth. Downregulated expression of ABCG subfamily genes in Ostrinia furnacalis is also related to its resistance to Cry1Ab and Cry1Ac toxins [123][59].
References
- Dean, M.; Rzhetsky, A.; Allikmets, R. The human ATP-binding cassette (ABC) transporter superfamily. Genome Res. 2001, 11, 1156–1166.
- Dassa, E.; Bouige, P. The ABC of ABCS: A phylogenetic and functional classification of ABC systems in living organisms. Res. Microbiol. 2001, 152, 211–229.
- Holland, I.B.; Cole, S.P.; Kuchler, K.; Higgins, C.F. ABC Proteins—From Bacteria to Man; Academic Press: Cambridge, MA, USA, 2003; p. 647.
- Berger, E.A. Different mechanisms of energy coupling for the active transport of proline and glutamine in Escherichia coli. Proc. Natl. Acad. Sci. USA 1973, 70, 1514–1518.
- Berger, E.A.; Heppel, L.A. Different mechanisms of energy coupling for the shock-sensitive and shock-resistant amino acid permeases of Escherichia coli. J. Biol. Chem. 1974, 249, 7747–7755.
- Higgins, C.F.; Haag, P.D.; Nikaido, K.; Ardeshir, F.; Garcia, G.; Ames, G.F. Complete nucleotide sequence and identification of membrane components of the histidine transport operon of S. typhimurium. Nature 1982, 298, 723–727.
- Gilson, E.; Higgins, C.F.; Hofnung, M.; Ames, F.L.; Nikaido, H. Extensive homology between membrane-associated components of histidine and maltose transport systems of Salmonella typhimurium and Escherichia coli. J. Biol. Chem. 1982, 257, 9915.
- Juliano, R.L.; Ling, V. A surface glycoprotein modulating drug permeability in Chinese hamster ovary cell mutants. Biochim. Biophys. Acta 1976, 455, 152–162.
- Riordan, J.R.; Deuchars, K.; Kartner, N.; Alon, N.; Trent, J.; Ling, V. Amplification of P-glycoprotein genes in multidrug-resistant mammalian cell lines. Nature 1985, 316, 817–819.
- Hyde, S.C.; Emsley, P.; Hartshorn, M.J.; Mimmack, M.M.; Gileadi, U.; Pearce, S.R.; Gallagher, M.P.; Gill, D.R.; Hubbard, R.E.; Higgins, C.F. Structural model of ATP-binding proteing associated with cystic fibrosis, multidrug resistance and bacterial transport. Nature 1990, 346, 362–365.
- Merzendorfer, H. Chapter one–ABC transporters and their role in protecting insects from pesticides and their metabolites. Adv. Insect Physiol. 2014, 46, 1–72.
- Chang, G.; Roth, C.B. Structure of MsbA from E. coli: A homolog of the multidrug resistance ATP binding cassette (ABC) transporters. Science 2001, 293, 1793–1800.
- Dawson, R.J.; Locher, K.P. Structure of a bacterial multidrug ABC transporter. Nature 2006, 443, 180–185.
- Hvorup, R.N.; Goetz, B.A.; Niederer, M.; Hollenstein, K.; Perozo, E.; Locher, K.P. Asymmetry in the structure of the ABC transporter-binding protein complex BtuCD-BtuF. Science 2007, 317, 1387–1390.
- Rees, D.C.; Johnson, E.; Lewinson, O. ABC transporters: The power to change. Nat. Rev. Mol. Cell Biol. 2009, 10, 218–227.
- Ter Beek, J.; Guskov, A.; Slotboom, D.J. Structural diversity of ABC transporters. J. Gen. Physiol. 2014, 143, 419–435.
- Holland, I.B.; Blight, M.A. ABC-ATPases, adaptable energy generators fuelling transmembrane movement of a variety of molecules in organisms from bacteria to humans. J. Mol. Biol. 1999, 293, 381–399.
- Erkens, G.B.; Berntsson, R.P.; Fulyani, F.; Majsnerowska, M.; Vujicic-Zagar, A.; Ter Beek, J.; Poolman, B.; Slotboom, D.J. The structural basis of modularity in ECF-type ABC transporters. Nat. Struct. Mol. Biol. 2011, 18, 755–760.
- Xu, K.; Zhang, M.; Zhao, Q.; Yu, F.; Guo, H.; Wang, C.; He, F.; Ding, J.; Zhang, P. Crystal structure of a folate energy-coupling factor transporter from Lactobacillus brevis. Nature 2013, 497, 268–271.
- Eitinger, T.; Rodionov, D.A.; Grote, M.; Schneider, E. Canonical and ECF-type ATP-binding cassette importers in prokaryotes: Diversity in modular organization and cellular functions. FEMS Microbiol. Rev. 2015, 35, 3–67.
- Wang, T.; Fu, G.; Pan, X.; Wu, J.; Gong, X.; Wang, J.; Shi, Y. Structure of a bacterial energy-coupling factor transporter. Sci. Found. China 2014, 497, 272–276.
- Hopfner, K.P. Invited review: Architectures and mechanisms of ATP binding cassette proteins. Biopolymers 2016, 105, 492–504.
- Gottesman, M.M.; Ambudkar, S.V. Overview: ABC transporters and human disease. J. Bioenerg. Biomembr. 2001, 33, 453–458.
- Higgins, C.F. ABC transporters: From microorganisms to man. Annu. Rev. Cell Biol. 1991, 8, 67–113.
- Higgins, C.F.; Linton, K.J. The ATP switch model for ABC transporters. Nat. Struct Mol. Biol. 2004, 11, 918–926.
- Deeley, R.G.; Westlake, C.; Cole, S.P. Transmembrane transport of endo- and xenobiotics by mammalian ATP-binding cassette multidrug resistance proteins. Physiol. Rev. 2006, 86, 849–899.
- Dermauw, W.; Van Leeuwen, T. The ABC gene family in arthropods: Comparative genomics and role in insecticide transport and resistance. Insect Biochem. Mol. Biol. 2014, 45, 89–110.
- Senior, A.E.; al-Shawi, M.K.; Urbatsch, I.L. The catalytic cycle of P-glycoprotein. FEBS Lett. 1995, 377, 285–289.
- Sauna, Z.E.; Kim, I.W.; Nandigama, K.; Kopp, S.; Chiba, P.; Ambudkar, S.V. Catalytic cycle of ATP hydrolysis by P-glycoprotein: Evidence for formation of the E.S reaction intermediate with ATP-gamma-S, a nonhydrolyzable analogue of ATP. Biochemistry 2007, 46, 13787–13799.
- Siarheyeva, A.; Liu, R.; Sharom, F.J. Characterization of an asymmetric occluded state of P-glycoprotein with two bound nucleotides: Implications for catalysis. J. Biol. Chem. 2010, 285, 7575–7586.
- Zhang, X.C.; Han, L.; Zhao, Y. Thermodynamics of ABC transporters. Protein Cell 2016, 7, 17–27.
- George, A.M.; Jones, P.M. Perspectives on the structure-function of ABC transporters: The switch and constant contact models. Prog. Biophys. Mol. Biol. 2012, 109, 95–107.
- Linton, K.J.; Higgins, C.F. Structure and function of ABC transporters: The ATP switch provides flexible control. Pflugers Arch. 2007, 453, 555–567.
- Roth, C.W.; Holm, I.; Graille, M.; Dehoux, P.; Rzhetsky, A.; Wincker, P.; Weissenbach, J.; Brey, P.T. Identification of the Anopheles gambiae ATP-binding cassette transporter superfamily genes. Mol. Cells 2003, 15, 150–158.
- Sturm, A.; Cunningham, P.; Dean, M. The ABC transporter gene family of Daphnia pulex. BMC Genom. 2009, 10, 170.
- Lee, S.H.; Kang, J.S.; Min, J.S.; Yoon, K.S.; Strycharz, J.P.; Johnson, R.; Mittapalli, O.; Margam, V.M.; Sun, W.; Li, H.M.; et al. Decreased detoxification genes and genome size make the human body louse an efficient model to study xenobiotic metabolism. Insect Mol. Biol. 2010, 19, 599–615.
- Liu, S.; Zhou, S.; Tian, L.; Guo, E.; Luan, Y.; Zhang, J.; Li, S. Genome-wide identification and characterization of ATP-binding cassette transporters in the silkworm, Bombyx mori. BMC Genom. 2011, 12, 491.
- Xie, X.; Cheng, T.; Wang, G.; Duan, J.; Niu, W.; Xia, Q. Genome-wide analysis of the ATP-binding cassette (ABC) transporter gene family in the silkworm, Bombyx mori. Mol. Biol Rep. 2012, 39, 7281–7291.
- Pearce, S.; Clarke, D.; East, P.; Elfekih, S.; Gordon, K.; Jermiin, L.; McGaughran, A.; Oakeshott, J.; Papanikolaou, A.; Perera, O. Genomic innovations, transcriptional plasticity and gene loss underlying the evolution and divergence of two highly polyphagous and invasive Helicoverpa pest species. BMC Biol. 2017, 15, 63.
- Broehan, G.; Kroeger, T.; Lorenzen, M.; Merzendorfer, H. Functional analysis of the ATP-binding cassette (ABC) transporter gene family of Tribolium castaneum. BMC Genom. 2013, 14, 6.
- Dermauw, W.; Osborne, E.J.; Clark, R.M.; Grbic, M.; Tirry, L.; Van Leeuwen, T. A burst of ABC genes in the genome of the polyphagous spider mite Tetranychus urticae. BMC Genom. 2013, 14, 317.
- Strauss, A.S.; Wang, D.; Stock, M.; Gretscher, R.R.; Groth, M.; Boland, W.; Burse, A. Tissue-specific transcript profiling for ABC transporters in the sequestering larvae of the phytophagous leaf beetle Chrysomela populi. PLoS ONE 2014, 9, e98637.
- Hull, J.J.; Chaney, K.; Geib, S.M.; Fabrick, J.A.; Brent, C.S.; Walsh, D.; Lavine, L.C. Transcriptome-based identification of ABC transporters in the western tarnished plant bug Lygus hesperus. PLoS ONE 2014, 9, e113046.
- Carmona-Antonanzas, G.; Carmichael, S.N.; Heumann, J.; Taggart, J.B.; Gharbi, K.; Bron, J.E.; Bekaert, M.; Sturm, A. A Survey of the ATP-binding cassette (ABC) gene superfamily in the salmon louse (Lepeophtheirus salmonis). PLoS ONE 2015, 10, e0137394.
- Sun, H.; Pu, J.; Chen, F.; Wang, J.; Han, Z. Multiple ATP-binding cassette transporters are involved in insecticide resistance in the small brown planthopper, Laodelphax striatellus. Insect Mol. Biol. 2017, 26, 343–355.
- Tian, L.; Song, T.; He, R.; Yang, Z.; Xie, W.; Wu, Q.; Wang, S.; Zhou, X.; Zhang, Y.J.B.G. Genome-wide analysis of ATP-binding cassette (ABC) transporters in the sweetpotato whitefly, Bemisia tabaci. BMC Genom. 2017, 18, 330.
- Guo, Z.J.; Kang, S.; Zhu, X.; Xia, J.X.; Wu, Q.J.; Wang, S.L.; Xie, W.; Zhang, Y.J. The novel ABC transporter ABCH1 is a potential target for RNAi-based insect pest control and resistance management. Sci. Rep. 2015, 5, 13728.
- Qi, W.; Ma, X.; He, W.; Chen, W.; Zou, M.; Gurr, G.M.; Vasseur, L.; You, M. Characterization and expression profiling of ATP-binding cassette transporter genes in the diamondback moth, Plutella xylostella (L.). BMC Genom. 2016, 17, 760.
- Bretschneider, A.; Heckel, D.G.; Vogel, H. Know your ABCs: Characterization and gene expression dynamics of ABC transporters in the polyphagous herbivore Helicoverpa armigera. Insect Biochem. Mol. Biol. 2016, 72, 1–9.
- Jin, M.; Liao, C.; Chakrabarty, S.; Zheng, W.; Wu, K.; Xiao, Y. Transcriptional response of ATP-binding cassette (ABC) transporters to insecticides in the cotton bollworm, Helicoverpa armigera. Pestic. Biochem. Phys. 2019, 154, 46–59.
- Wang, X.H.; Fang, X.D.; Yang, P.C.; Jiang, X.T.; Jiang, F.; Zhao, D.J.; Li, B.L.; Cui, F.; Wei, J.N.; Ma, C.A.; et al. The locust genome provides insight into swarm formation and long-distance flight. Nat. Commun. 2014, 5, 1–9.
- Lu, H.; Xu, Y.; Cui, F. Phylogenetic analysis of the ATP-binding cassette transporter family in three mosquito species. Pestic. Biochem. Physiol. 2016, 132, 118–124.
- Tay, W.T.; Mahon, R.J.; Heckel, D.G.; Walsh, T.K.; Downes, S.; James, W.J.; Lee, S.F.; Reineke, A.; Williams, A.K.; Gordon, K.H.J. Insect resistance to Bacillus thuringiensis toxin Cry2Ab is conferred by mutations in an ABC transporter subfamily a protein. PLoS Genet. 2015, 11, e1005534.
- Mathew, L.G.; Ponnuraj, J.; Mallappa, B.; Chowdary, L.R.; Zhang, J.; Tay, W.T.; Walsh, T.K.; Gordon, K.H.J.; Heckel, D.G.; Downes, S.; et al. ABC transporter mis-splicing associated with resistance to Bt toxin Cry2Ab in laboratory- and field-selected pink bollworm. Sci. Rep. 2018, 8, 13531.
- Tabashnik, B.E. ABCs of insect resistance to Bt. PLoS Genet. 2015, 11, e1005646.
- Wang, J.; Wang, H.; Liu, S.; Liu, L.; Tay, W.T.; Walsh, T.K.; Yang, Y.; Wu, Y. CRISPR/Cas9 mediated genome editing of Helicoverpa armigera with mutations of an ABC transporter gene HaABCA2 confers resistance to Bacillus thuringiensis Cry2A toxins. Insect Biochem. Mol. Biol. 2017, 87, 147–153.
- Tanaka, S.; Endo, H.; Adegawa, S.; Iizuka, A.; Imamura, K.; Kikuta, S.; Sato, R. Bombyx mori ABC transporter C2 structures responsible for the receptor function of Bacillus thuringiensis Cry1Aa toxin. Insect Biochem. Mol. Biol. 2017, 91, 44–54.
- Chen, Y.; Li, M.; Islam, I.; You, L.; Wang, Y.; Li, Z.; Ling, L.; Zeng, B.; Xu, J.; Huang, Y.; et al. Allelic-specific expression in relation to Bombyx mori resistance to Bt toxin. Insect Biochem Mol. Biol. 2014, 54, 53–60.
- Zhang, T.; Coates, B.S.; Wang, Y.; Wang, Y.; Bai, S.; Wang, Z.; He, K. Down-regulation of aminopeptidase N and ABC transporter subfamily G transcripts in Cry1Ab and Cry1Ac resistant Asian corn borer, Ostrinia furnacalis (Lepidoptera: Crambidae). Int. J. Biol. Sci. 2017, 13, 835–851.
- Xiao, Y.; Zhang, T.; Liu, C.; Heckel, D.G.; Li, X.; Tabashnik, B.E.; Wu, K. Mis-splicing of the ABCC2 gene linked with Bt toxin resistance in Helicoverpa armigera. Sci. Rep. 2014, 4, 6184.
- Gahan, L.J.; Pauchet, Y.; Vogel, H.; Heckel, D.G. An ABC Transporter Mutation Is Correlated with Insect Resistance to Bacillus thuringiensis Cry1Ac Toxin. PLoS Genet. 2010, 6, e1001248.
- Guo, Z.; Kang, S.; Chen, D.; Wu, Q.; Wang, S.; Xie, W.; Zhu, X.; Baxter, S.W.; Zhou, X.; Jurat-Fuentes, J.L.; et al. MAPK signaling pathway alters expression of midgut ALP and ABCC genes and causes resistance to Bacillus thuringiensis Cry1Ac toxin in diamondback moth. PLoS Genet. 2015, 11, e1005124.
- Baxter, S.W.; Badenes-Perez, F.R.; Morrison, A.; Vogel, H.; Crickmore, N.; Kain, W.; Wang, P.; Heckel, D.G.; Jiggins, C.D. Parallel evolution of Bacillus thuringiensis toxin resistance in Lepidoptera. Genetics 2011, 189, 675–679.
- Park, Y.; Gonzalez-Martinez, R.M.; Navarro-Cerrillo, G.; Chakroun, M.; Kim, Y.; Ziarsolo, P.; Blanca, J.; Canizares, J.; Ferre, J.; Herrero, S. ABCC transporters mediate insect resistance to multiple Bt toxins revealed by bulk segregant analysis. BMC Biol. 2014, 12, 46.
- Li, J.; Ma, Y.; Yuan, W.; Xiao, Y.; Liu, C.; Wang, J.; Peng, J.; Peng, R.; Soberon, M.; Bravo, A.; et al. FOXA transcriptional factor modulates insect susceptibility to Bacillus thuringiensis Cry1Ac toxin by regulating the expression of toxin-receptor ABCC2 and ABCC3 genes. Insect Biochem. Mol. Biol. 2017, 88, 1–11.
- Coates, B.S.; Siegfried, B.D. Linkage of an ABCC transporter to a single QTL that controls Ostrinia nubilalis larval resistance to the Bacillus thuringiensis Cry1Fa toxin. Insect Biochem. Mol. Biol. 2015, 63, 86–96.
- Pauchet, Y.; Bretschneider, A.; Augustin, S.; Heckel, D.G. A P-glycoprotein is linked to resistance to the Bacillus thuringiensis Cry3Aa toxin in a Leaf BEETLE. Toxins Basel 2016, 8, 362.
- Flagel, L.E.; Swarup, S.; Chen, M.; Bauer, C.; Wanjugi, H.; Carroll, M.; Hill, P.; Tuscan, M.; Bansal, R.; Flannagan, R.; et al. Genetic markers for western corn rootworm resistance to Bt toxin. G3 Bethesda 2015, 5, 399–405.
- Bretschneider, A.; Heckel, D.G.; Pauchet, Y. Three toxins, two receptors, one mechanism: Mode of action of Cry1A toxins from Bacillus thuringiensis in Heliothis virescens. Insect Biochem. Mol. Biol. 2016, 76, 109–117.
- Stevens, T.; Song, S.; Bruning, J.B.; Choo, A.; Baxter, S.W. Expressing a moth abcc2 gene in transgenic Drosophila causes susceptibility to Bt Cry1Ac without requiring a cadherin-like protein receptor. Insect Biochem. Mol. Biol. 2017, 80, 61–70.
- Atsumi, S.; Miyamoto, K.; Yamamoto, K.; Narukawa, J.; Kawai, S.; Sezutsu, H.; Kobayashi, I.; Uchino, K.; Tamura, T.; Mita, K.; et al. Single amino acid mutation in an ATP-binding cassette transporter gene causes resistance to Bt toxin Cry1Ab in the silkworm, Bombyx mori. Proc. Natl. Acad. Sci. USA 2012, 109, E1591–E1598.
- Endo, H.; Tanaka, S.; Adegawa, S.; Ichino, F.; Tabunoki, H.; Kikuta, S.; Sato, R. Extracellular loop structures in silkworm ABCC transporters determine their specificities for Bacillus thuringiensis cry toxins. J. Biol. Chem. 2018, 293, 8569–8577.
- Guo, Z.J.; Kang, S.; Zhu, X.; Xia, J.X.; Wu, Q.J.; Wang, S.L.; Xie, W.; Zhang, Y.J. Down-regulation of a novel ABC transporter gene (Pxwhite) is associated with Cry1Ac resistance in the diamondback moth, Plutella xylostella (L.). Insect Biochem. Mol. Biol. 2015, 59, 30–40.
More
