Yeasts, and particularly
Saccharomyces cerevisiae, used in ethanol production are most efficient in warm (e.g., 20–30 °C) and acidic (pH between 4.5–6.5) environments
[4][5]. During microbial fermentation, yeasts produce glycerol, as both an osmoprotectant
[5][6][7][6,7,8] and for metabolic recovery of NAD+. Yeast also acidifies their environment to promote growth through proton secretion, secretion of organic acids (discussed below), removal of buffering agents, and dissipation of carbon dioxide
[4][5]. The most common yeast used in alcoholic fermentation,
Saccharomyces cerevisiae, is preferred for rapid and efficient conversion of sugar solutions to solutions with correspondingly high ethanol concentrations. The latter solutions are readily distilled and dehydrated to yield products suitable for blending with gasoline. Dehydration processes to produce anhydrous ethanol include heterogeneous azeotropic distillation using solvents (e.g., benzene), extractive distillation with solvents and entrainers (e.g., salts), adsorption using molecular sieves, and pervaporation membranes (e.g., zeolite, silica, etc.)
[8][9]. During fermentation with yeast, value-added co-products (e.g., α-glycerylphosphorylcholine) can accumulate along with ethanol, while fusel alcohols can be recovered during distillation and are often added to ethanol used in fuel applications. Common lower value co-products can also accumulate (e.g., acetic acid, succinic acid, and glycerol) that are more difficult to valorize, and nuisance coproducts can accumulate (methanol, hydrogen sulfide, and methyl mercaptan). A portion of the volatile co-products are volatile, toxic, and/or contribute to odors that can be co-distilled with the ethanol product. Multi-stage distillation and other purification technologies are required to remove these compounds. A group of alcohols and aldehydes (e.g., aldehydes, butanols, propanols, etc.) are naturally synthesized from amino acids through the Ehrlich pathway
[9][10] and simple sugars during fermentation
[10][11][11,12]. In addition to alcohols, organic acids can also accumulate (e.g., acetic acid, succinic acid, and lactic acid)
[2][12][13][14][3,13,14,15] because of yeast metabolic processes and even metabolism by adventitious bacteria present during ethanolic fermentation. Secondary fermentation (two-stage fermentation) of stillage can affect the contents of these compounds.
2. Thin Stillage and Distillers’ Grains
Following ethanolic fermentation and distillation processes, the by-product stillage contains much of the protein, oil, fiber, and non-starch carbohydrate that were not available to the yeast during fermentation. A common process for using these components starts with the separation of whole stillage into a liquid portion with suspended solids (thin stillage) and wet solids (distillers’ wet grains), using centrifugation, vibratory separation, or a press [22][23][23,24]. These by-products can then be further processed (i.e., drying and fractionation) or extracted into specific components.
Without any further processing, all or a portion of thin stillage can be returned or backset to the next fermentation. This practice replaces some of the water required to soak incoming feedstocks intended for fermentation [24][25]. Protein is a major nutrient remaining in the stillage, and many methods have been developed to recover stillage proteins. Grain thin stillage contains approximately 37% protein (w/w dry basis) [25][26], and research has been conducted to develop stillage protein concentrates. Physical clarification techniques using additives [26][27][27,28], gas flotation [28][29][29,30], centrifugation [30][31], and filtration [31][32][32,33] have been tested as approaches to produce protein-enriched solids from thin stillage. Another approach for improving stillage protein quality and concentration is through a two-stage fermentation strategy [14][33][15,34], where it is also possible to upgrade glycerol to higher value compounds. Where thin stillage is not suited for use as a feed, it may be used as a nitrogen-rich fertilizer for crops [34][35]. Another strategy is to pair thin stillage valorization with a protein extraction process. Protein extraction from oilseed meal requires the use of large volumes of solution to dissolve proteins before precipitation. Thin stillage has some dissolved protein, but it can be used as a solution for protein extraction from oilseed meal [35][36]. An economical use of thin stillage that avoids the need for evaporation while providing the benefit of the stillage as a nutrient solution involves simply providing stillage in the water for cattle. In this way, the stillage becomes a nutrient-rich water source [36][37].
Once thin stillage is separated by dewatering distillers’ wet grains [37][38][38,39], the thin stillage can then be dried to a concentrated syrup called distillers’ solubles (DS), which is useful as an animal feed component. Remaining solids or distillers’ wet grains (DWG) can be dried to produce distillers’ dried grains (DDG) for storage and shipping. An alternative practice is to add DS to the grains as they dry to produce dried distillers’ grains with solubles (DDGS), a product that is commonly used with cattle feed [39][40]. Compared to wet feed products, DDG and DDGS have extended shelf-life and are more easily shipped. The sale of fermentation by-products for use as cattle feed generally provides 10–20% of the total revenue of ethanol production facilities [40][41] while avoiding revenue losses that would be incurred if co-product disposal was necessary. Fractionation of the DDGS can concentrate protein and generate fractions with high fiber contents to produce additional protein and fiber products [41][42]. In some rations, the higher fiber content of DDGS is undesirable. Producing a higher protein- and fat-content feed ingredient can improve the value of this product stream. In addition to the use of stillage products in animal feed, a portion DDGS proteins can be more readily solubilized and extracted for a wide variety of industrial uses (e.g., biopolymer production) [42][43].
DDG, produced during first-generation biofuels processes, can be used as a substrate for a second fermentation after pre-treatment that converts unhydrolyzed and unprocessed cellulose into fermentable sugars [43][44].
Finally, oil is another product of bioethanol production that is often poorly utilized. For corn, methods of oil extraction from thin stillage have been patented [44][47], and other oil extraction methods from corn DDGS have been developed [45][48].
3. α-Glycerylphosphorycholine
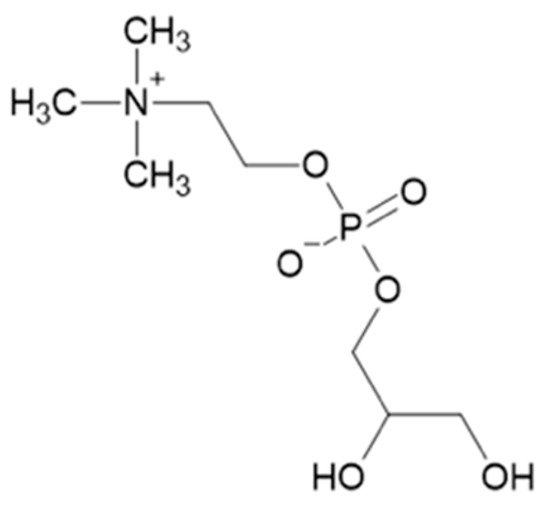
α-Glycerylphosphorylcholine (α-GPC) (
Figure 1) is a yeast metabolite and precursor involved in the synthesis of acetylcholine and membrane phospholipids
[46][49].
Figure 1. Chemical structure of α-glycerylphosphorylcholine.
Fermentation of ginger feedstocks using
Schizosaccharomyces pombe yeast
[47][72] has also resulted in the production of a nootropic compound (e.g., 6-paradol)
[47][72]; however, these fermentations are often slower and produce by-products
[48][49][73,74] that can make purification costly. Nonetheless, the ability to isolate and purify these nootropic coproducts can add significant revenue for bioethanol producers, by providing ethanol producers with new products that meet the needs of other markets. For example, nootropics (e.g., α-GPC) had a value of 7.21 billion USD in 2020, and they are expected to grow with a CAGR of 80% until 2028
[50][75]. Accumulation of α-GPC in the mash has been observed during alcoholic fermentation of cereal crops, such as wheat, barley, and oat, although the concentration produced can differ among cultivars
[2][12][3,13]. For example, fermentation of 28 barley and 12 oat cultivars resulted in the accumulation of between 0.84 g/L to 1.81 g/L for barley and 0.62 g/L to 0.88 g/L for oat, depending on cultivar
[2][3]. Meanwhile, fermentation of wheat resulted in α-GPC accumulation of approximately 1.68 g/L
[12][13]. Oyeneye et al. also found that treatment of the grain with phospholipase A1, an enzyme that readily hydrolyses phosphatidyl choline and lysophosphatidyl choline, produced a beer with higher α-GPC than other treatments. This treatment effect strongly suggests that α-GPC might accumulate as a result of phosphatidyl choline hydrolysis. Pre-treatment of the feedstock (e.g., soaking, germination, incubation temperature, etc.) can also influence α-GPC yield (unpublished data). Therefore, alcoholic fermentation of cereal feedstocks can be highly advantageous due to the inexpensive processes involved and because α-GPC can be concurrently produced with ethanol during fermentation. α-GPC is a naturally produced endogenous choline derivative; however, it is rarely found at high concentrations in nature. Therefore, there is great potential in developing alternative, inexpensive, and sustainable means for commercial production to supply this compound.
4. Fusel Alcohols
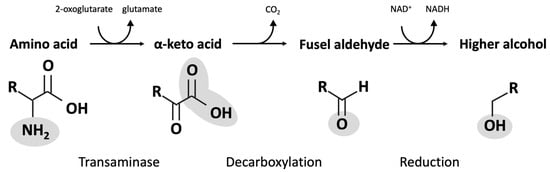
A series of primary alcohols are found as natural co-products, generated during the fermentation process. These compounds are largely produced by metabolic processes, called the Ehrlich pathway in yeast, that recover nitrogen required for growth and metabolism. Enzymes of the pathway catalyze the transfer of amines between amino acids and ketones and the decarboxylation of α-keto acids to produce aldehydes, and they reduce aldehydes to form fusel alcohols (
Figure 23). Products from the Ehrlich pathway can be influenced by the presence of certain amino acid intermediates (
Table 2).
Figure 2. Ehrlich pathway conversion of amino acids into fusel alcohols.
Table 2. Amino acids intermediates and products of the Ehrlich pathway. Reconstructed from Hazelwood et al., (2008) [9].
| Amino Acid |
α-Keto Acid |
Fusel Aldehyde |
Fusel Alcohol |
| Isoleucine |
α-Ketomethylvalerate |
Methylvaleraldehyde |
Active amyl alcohol |
| Leucine |
α-Keotisocaproate |
Isoamylaldehyde |
Isoamyl alcohol |
Methionine
Phenylalanine
Threonine |
α-Keto-γ-(methylthio)butyrate
Phenylpyruvate
2-Ketobutyrate |
Methional
Phenylethanal
Propanal |
Methionol
Phenylethanol
Propanol |
| Tryptophan |
3-Indole pyruvate |
3-Indole acetaldehyde |
Tryptophol |
| Tyrosine |
p-Hydroxyphenylpyruvate |
p-Hydroxyphenylacetaldehyde |
p-Hydroxyphenylethanol or tyrosol |
| Valine |
α-Ketoisovalerate |
Isobutanal or isovaleraldehyde |
Isobutanol |
During ethanol distillation, fusel alcohols concentrate in the distillation column as they are less volatile than the ethanol water azeotrope. The removal of these compounds is not required for fuel production but is essential for ethanol destined for food or pharmaceutical applications. If these higher alcohols are not removed, they can reach their limit of solubility and increase vapour pressures in the column. In turn, the increase in vapour pressure can cause boiling and flooding and thus interfere with distillation. In bioethanol plants with continuous distillation processes, the fusel alcohols are distributed in the distillation or rectifier column, where the product at the top of the column is purified alcohol (~95% by wt.), and at the bottom is mostly water. Fusel alcohols are most volatile at lower ethanol concentrations and tend to collect in the intermediate region where ethanol concentration is approximately 45% (
v/v)
[51][76]. In continuous distillation columns, fusel alcohol removal is accomplished by drawing a solution of water, ethanol, and fusel alcohols from the center of the rectifier (
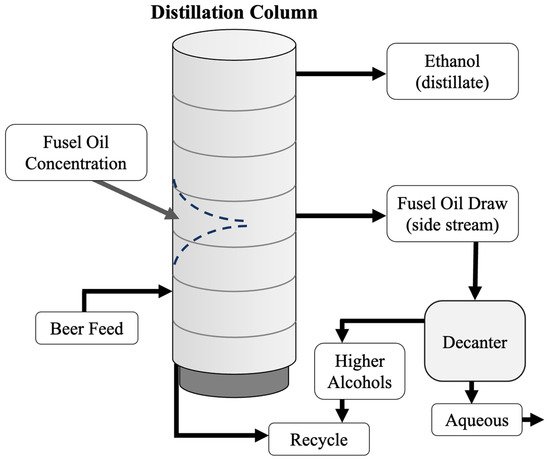
Figure 34).
Figure 3. Distillation column with a fusel alcohol draw near the middle of the column. The fusel alcohols can be removed from the distillation process and decanted, reusing the higher alcohols and removing the aqueous phase. Without a fusel alcohol draw, the fusel alcohols will fall in the column and form a two-phase system. This can interfere with the distillation efficiency.
In batch distillation, fusel alcohols form azeotropes with water that can accumulate at the bottom of the column to become concentrated late distilling fractions called “tails” that evaporate when the column temperature rises at the end of a distillation.
Fusel alcohols can also be purified and esterified to yield a range of valuable esters. Isoamyl alcohol is the primary component in fusel alcohols and can be used in the production of organic esters for industrial purposes. For example, isoamyl acetate is formed by esterification of isoamyl alcohol with acetic acid, and it has applications in commercial products such lacquers and as a food flavouring additive, with a banana-, caramel-, or pear-like flavour profile
[51][52][53][76,77,78]. As such, the recovery of isoamyl alcohol can add significant value to the bioethanol producer, as this compound has three-times the market value of fuel ethanol
[54][81]. Unfortunately, isolating and purifying isoamyl alcohol from fusel alcohols is challenging, due to the formation of heterogenous azeotropes of several alcohols with water during distillation. Multiple decanters have been used to collect fusel alcohol draws at alternating positions in the distillation column, to separate and purify fusel alcohols, although these methods typically do not afford products that are sufficiently pure for commercial application
[51][55][54][76,79,81]. Esterification of fusel alcohols, a process called reactive distillation, has also been proposed and demonstrated successfully at a bench-scale to simultaneously obtain highly enriched alcohol esters
[51][52][76,77]. Another method to aid in fusel alcohol separation is the addition of water to promote phase separation of higher alcohols from the aqueous phase (containing methanol, ethanol, propanol, and isopropanol). The organic phase can then be further distilled to enrich isoamyl alcohol
[51][55][76,79].
Enrichment and purification of individual fusel alcohols and their esters can produce value-added compounds, suitable for inclusion in flavours and fragrances. The accumulation of these alcohols depends greatly on yeast genetics
[51][52][53][76,77,78] and fermentation medium components (e.g., sugar source)
[51][52][76,77]. For example, the production of isopropanol can be manipulated through the addition of exogenous acetone to the mash
[51][52][76,77]. Yeast genetics determines the production of volatile compounds, such as fusel alcohols, that contribute to the product flavours and aromas
[52][56][57][77,83,84]. Identifying yeasts that appropriately influence the flavour profiles of the final product is of great importance for the distillation of materials such as beers, ciders, and spirits. Alteration of fusel compounds is possible, although the complete suppression of fusel alcohol formation by selection of yeast is not, due to the biosynthesis of the α-keto acid present in the Ehrlich pathway (
Figure 23)
[56][58][59][83,85,86]. Overall, modifications can influence the production of alcohol by-products, and they can add considerable value to the bioethanol producer if these compounds can be further isolated and purified.
5. Two-Stage Fermentation of Thin Stillage
In additional to physical processing, stillage can be fermented by a range of organisms. The biorefinery of thin stillage has been accomplished via fermentation with a consortium of LAB (e.g.,
Lactobacilli) selectively recovered from a stillage storage tank (
Figure 45)
[13][14][14,15]. Fermentation was effective in modifying the stillage, allowing the efficient separation of a protein-rich fraction. The consortium organisms belong to species that are classified as Generally Recognized as Safe (GRAS) and are routinely used in the food industry. Clarification of thin stillage via LAB fermentation may result from a combination of gas production that leads to anoxic gas flotation
[60][87], production of exopolysaccharides that cause particle aggregation
[13][14], and/or allowing sufficient time for settling/floatation of aggregated particles or separation of the particles from the solution with decanting and desludging centrifuges. The resulting protein-rich slurry contains much of the original thin stillage protein, as well as proteins produced by the LAB. Drying the slurry creates a concentrate of up to 60% protein
[61][88]. The bacteria-rich protein concentrate can then serve as a probiotic animal feed supplement
[62][89].
Figure 4. Wheat thin stillage before (left) and after (right) 48 h of secondary fermentation by lactic acid bacteria. The initially homogenous solution separates into an upper layer composed of, mostly, clear liquid and a protein rich slurry in the bottom layer.
5.1. Organic Acids
5.1. Organic Acids
Many of the organic acids (e.g., acetic acid, succinic acid, and lactic acid) produced by yeast are acids that typically arise from glycolysis (e.g., acetic acid and lactic acid) and the citric acid cycle (e.g., succinic acid)
[63][90]. These compounds are typically produced commercially via fermentation microbial processes
[63][64][65][90,91,92] for use in the food, beverage, and manufacturing industries. The efficiency of production of these organic acids is dependent on the microorganisms, feedstock, fermenter productivity, and development of efficient recovery processes
[66][67][68][93,94,95].
Due to its pH tolerance and simple nutrient requirements,
Saccharomyces cerevisiae has been investigated and genetically modified for organic acid production
[69][96]. Organic acids are not traditionally produced by
Saccharomyces cerevisiae fermentation. The production of these organic acids could produce considerable additional value (
Table 1), but a portion of the organic acids (e.g., acetic acid and lactic acid) seen in commercial ethanol beers are typically attributable to nuisance organisms present during bioethanol production. At modest concentrations, these organic acids can inhibit fermentation
[4][70][71][5,97,98].
5.2. Conversion of Glycerol to 1,3-Propanediol
In addition to the production of organic acids, during fermentation, glycerol is also produced by yeast to protect cells against lysis and regenerate NAD
+ needed for glycolysis
[5][6][7][6,7,8]. Interestingly, glycerol in the fermentation mash can be further upgraded through its conversion to 1,3-propanediol using Lactobacilli
[13][14][14,15]. This compound is useful for the production of textiles, carpets, adhesives, moldings, etc.
[72][73][100,101].
5.3. Bacteriocins
Bacteriocins are another potential value-added product that could be purified after a second two-stage fermentation of ethanol stillage with
Lactobacillus. Bacteriocins are antimicrobial proteins produced by most bacteria
[74][75][107,108].Some bacteriocins exhibit broad-spectrum antagonistic effects. For example, bacteriocins produced by Gram-negative bacteria typically affect closely related species, whereas Gram-positive bacteria can produce bacteriocins that exhibit a broader spectrum of activity
[76][110]. These antimicrobial compounds might have utility as natural food preservatives or for pharmaceutical applications
[76][77][110,111].
Unlike traditional antibiotics, which typically act as enzyme inhibitors
[78][116], bacteriocins elicit adverse effects by inhibiting bacterial cell growth, by disrupting essential functions (e.g., translation and transcription)
[79][117], and by targeting the cell surface and altering membrane permeability
[76][79][80][81][82][83][110,117,118,119,120,121] (e.g., formation of membrane channels). Bacteriocin-producing organisms simultaneously express bacteriocin-immunity proteins that protect producing organisms from their own toxins
[84][85][122,123]. Bacteria can acquire or lose immunity against specific bacteriocins through horizontal gene transfer
[86][87][88][89][90][91][92][124,125,126,127,128,129,130].
6. Spent Yeast
Spent yeast is another by-product from the brewing and biofuels industry. The spent yeast is typically removed at the end of the fermentation, although a small amount can be retained for subsequent fermentation batches
[93][131]. Due to its protein content, discarded yeast is typically used as inexpensive animal feed materials
[93][131]. However, spent yeast also contains valuable nutrients, such as vitamins (e.g., vitamins B
1, B
2, B
3, B
6, B
9, and B
12)
[94][95][96][132,133,134], minerals (e.g., Na, K, Ca, Mg, Fe, Mn, Zn, Cu, Se, Cr, and Mo)
[96][97][134,135], proteins (e.g., mannoproteins and hydrolysates)
[97][135], carbohydrates (e.g., β-glucans)
[97][135], antioxidants (e.g., glutathione)
[98][136], and phenolic compounds (e.g., gallic acid and (±)-catechin)
[94][132]. Retrieval of these compounds can have added value in animal feeds, nutritional supplements, and functional foods (e.g., flavor enhancers) and non-food additives (e.g., cosmetics)
[93][99][131,137]. Hydrolysis of spent yeast can yield a complex mixture of oligopeptides, peptides, and free amino acids, also known as hydrolysates
[99][137]. Total solids recovery of hydrolysates, proteins, and α-amino nitrogen content in dried spent yeast was reported to be 50%, 55.9%, and 4.8%, respectively
[99][137].