Malignant pleural mesothelioma (MPM) is an aggressive tumor mainly associated with asbestos exposure and is characterized by a very difficult pharmacological approach. Therefore, it is crucial to better understand the molecular mechanisms involved in MPM development and metastasis, in an attempt to open new scenarios that are useful in the identification of predictive markers and to improve the pharmacological approach against this aggressive cancer.
1. Introduction
Malignant mesothelioma is a rare, aggressive and treatment-resistant tumor that originates from mesothelial cells lining the serous cavity, but the most common type of this tumor is malignant pleural mesothelioma (MPM). MPM development has been closely associated with asbestos exposure
[1]; although asbestos use was banned in most countries, the worldwide incidence of MPM continues to climb, so the understanding of asbestos-induced MPM pathogenesis is crucial in order to combat this aggressive cancer. Asbestos fiber effects are considered to be crucial in driving several pathogenetic mechanisms involved in the MPM onset, such as chronic inflammation, oxidative stress and epithelial-to-mesenchymal transition (EMT)
[2]. Particularly, the critical role of EMT in tumor progression has been well documented in different types of carcinomas (e.g., breast, pancreas and colon)
[3]. Furthermore, EMT and oxidative stress are involved in cancer metabolic changes, and both events are related to the effects mediated by the cytokine transforming growth factor β (TGFβ)
[4]. TGFβ is the main inducer of EMT and can promote reactive oxygen species (ROS) production and downregulate the expression of antioxidant enzymes
[5]. Moreover, many studies showed that miRNAs and other non-coding RNAs are involved in mesothelioma development and the EMT process
[6].
2. Malignant Pleural Mesothelioma
Malignant pleural mesothelioma (MPM) arises from the pleural surface, and it has been associated with previous (usually occupational) asbestos exposure and is more common in males
[7]. The therapeutic approaches against MPM consist of multiple modalities: surgery, chemotherapy and radiotherapy
[8]. Despite the efficacy of chemotherapeutic agents used, such as cisplatinum and pemetrexed, with or without bevacizumab, the frequent chemoresistance of MPM to these treatments causes a median survival from diagnosis of only one to two years
[8]. Recently, immunotherapy was approved for MPM treatment because of the close relationship between the immune system and mesothelial cells and particularly because of the increasing importance of the role of immune TME in asbestos-induced MPM
[9]. Currently, a combination of immune checkpoint inhibitors (ICIs), monoclonal antibodies (mAbs), anti-programmed cell death 1 (PD-1), nivolumab and anti-CTL antigen 4 (CTLA-4) ipilimumab
[10] was approved by the Food and Drug Administration (FDA) for first-line treatment of unresectable MPM based on CheckMate 743, which is the first phase of a randomized trial showing evident improvements in terms of survival with immunotherapy in MPM patients
[10].
Based on the predominant cell type, MPM is classified into three histological sub-types: epithelioid, sarcomatoid and biphasic. The epithelioid form is more common and related to a better prognosis than the non-epithelial one. Phenotypically, MPM cells are a polygonal or cobblestone-like shape in the epithelioid histotype, while a spindle-like form has been associated with the sarcomatoid MPM type
[11]. MPM diagnosis is based on imaging modalities and the evaluation of diagnostic and prognostic biomarkers, such as mesothelin, BRCA-1 associated protein (BAP1), osteo-pontin, fibulin 3 and cytokeratins. Although the role of biomarkers is not well established yet, the latter are used to confirm the MPM histotype through a cytological analysis of pleural effusion and biopsies
[7]. Concerning asbestos-related MPM, Klebe et al. interestingly showed an elevated amosite asbestos fiber content using electron microscopy in sarcomatoid MPM samples and highlighted the usefulness of cytokeratin and calretinin immunohistochemistry in discriminating between different MPM histotypes
[12]. However, the evaluation of asbestos fiber count by microscopy and mesothelioma biomarkers via immunohistochemistry or blood tests is not effective in confirming mesothelioma diagnosis alone.
In various studies, MPM was associated with tumor-suppressing mutations, such as
BAP1, neurofibromatosis type 2 (
NF2),
TP53 and
CDKN2A [1][13][1,13]. Their aberrant expression has been shown to affect MPM cell mobility, proliferation and apoptosis, leading to the onset of the tumor. Finally, MPM has been demonstrated to be triggered by different viral and environmental agents, such as Simian Virus 40 and ionizing irradiation
[14], but the main risk factor is still exposure to asbestos fibers.
3. Asbestos Effects and MPM Development
The term “asbestos” refers to six types of hydrated mineral silicate fibers in nature classified into serpentine (chrysotile) and amphibole (crocidolite, amosite, anthophyllite, actinolite and tremolite) forms. Asbestos toxicity depends on the dose and length/diameter ratio
[15], so amphibole fibers are more dangerous, and their biopersistence is longer than serpentine ones
[1].
Asbestos exposure has been related to different diseases, such as asbestosis (a form of pulmonary fibrosis) and MPM, the latter of which is clinically evident after a long latency period of about 40 years
[16], and different pathogenetic mechanisms promoted by asbestos exposure were demonstrated to be involved in MPM development. After inhalation, asbestos fibers migrate to the pleura by inducing irritation, damage and fibrosis
[1][15][16][17][18][1,15,16,17,18]. Moreover, asbestos fibers can induce chronic inflammation, involving TNF-α and IL-1β release by macrophages, and, consequently, macrophagic frustrated phagocytosis
[18], mitosis aberrations and the generation of free radicals, the latter of which is involved in promoting DNA, lipid and protein damage. Moreover, asbestos exposure induces the release of different molecules, such as growth factors or cytokines (such as, TGFβ, HGF and VEGF), which are widely involved in asbestos-related pathologies onset. These asbestos-induced mechanisms promote cell necrosis and the release of damage-associated molecular patterns (DAMPs), such as high mobility group box 1 protein (HMGB1) involved in survival, proliferation and autophagy, particularly in mesothelial asbestos-exposed cells
[8][19][8,19]. Therefore, asbestos fibers were demonstrated to allow, through these events, the creation of a microenvironment prone to the cancer onset and progression. Thus, asbestos fibers can induce epithelial and mesenchymal plasticity; therefore, asbestos-related diseases have been associated with epithelial-to-mesenchymal transition
[20][21][22][20,21,22].
4. Epithelial-to-Mesenchymal Transition
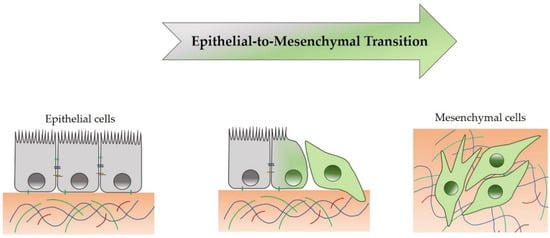
The epithelial-to-mesenchymal transition (EMT) is a morphogenetic reversible process, during which an epithelial cell loses its own features by reducing some epithelial proteins expression (e.g., E-cadherin, β-catenin, cytokeratins-5/6 and ZO-1) and by acquiring a mesenchymal phenotype, thereby increasing mesenchymal proteins expression (e.g., N-cadherin, Fibronectin, Vimentin, α-SMA, Snail, Slug, TWIST, ZEB-1/2 and MMP-2/9)
[3][21][23][3,21,23]. Therefore, epithelial cells lose the apical-basal cell polarity and disassemble intercellular (tight junctions and desmosomes) and cell-ECM junctions (e.g., integrins and fibronectin)
[21]. During EMT, a complete mesenchymal state is rarely reached, but an intermediate state between epithelial and mesenchymal status is reached, as shown in
Figure 1.
Figure 1.
Transition from epithelial cell toward a mesenchymal phenotype.
EMT is crucial in physiological events, such as embryogenesis (type I) and wound healing (type II). However, EMT has also been demonstrated to be involved in cancer (type III) and fibrosis development
[3]. In tumor progression, EMT makes cancer cells proficient in mobility, invasiveness, apoptosis resistance and extracellular matrix (ECM) production
[3][21][23][3,21,23], thus contributing to the tumor onset.
Mesenchymal-to-epithelial transition (MET) is the reverse process of EMT, in which cells with a mesenchymal phenotype acquire an apical-basal cell polarity, reorganize the cytoskeleton and return to an epithelial phenotype
[21]. Both EMT and MET occur in physiological and pathological conditions, and their deregulation has been shown to confer to cancer cell aggressiveness, which is particularly crucial for the establishment of metastasis during tumor progression
[3].
5. EMT and TME Crosstalk in MPM: A Possible Therapeutic Approach
The therapeutic approaches against MPM are surgery, chemotherapy, radiotherapy and recently, immunotherapy
[8][9][8,9]. Unfortunately, conventional strategies are not always successful. For example, chemotherapy itself has been reported to induce irreversible EMT through endoplasmic reticulum stress, which consequently activates the unfolded protein response both in lung adenocarcinoma cell lines A549 and H358 and primary lung adenocarcinoma samples from patients, providing an interesting mechanism that might contribute to tumor chemoresistance
[24][85].
Therefore, the introduction of new possible therapeutic approaches is necessary. In this sense, the FDA authorization of the ICIs for first-line treatment of unresectable MPM
[10] constituted a meaningful improvement in MPM therapy. However, not all MPM patients have a favorable benefit/risk ratio using ICIs, so we need to identify new strategies to treat this type of tumor. Therapeutically, TGFβ-induced signaling pathways could also be potential targets because of their association with induction of the EMT and immunosuppression of TME. In this regard, the inhibition of protein kinase CK-2 by CX-4945 with the use of Ginsenoside 20 (R)-Rg3 in A549 blocked TGFβ-induced EMT
[25][55], along with the use of Galunisertib or Vacosertib (inhibitors of TGFβRI)
[26][86], constituted further potential options in MPM treatment. Moreover, as indicated above, the use of Hsp70 inhibitors or RNA mimics could inhibit EMT and be a potential strategy in MPM treatment
[27][28][29][70,75,77].
Interestingly, considering the role of TME, the hypothetical use of mesothelin (MSLN)-targeted CAR T-cells, in combination with checkpoint blockade, could constitute an attractive approach
[30][87]. Recently, Wirawan et al. found that lysine-specific demethylase 1 (LSD1/KDM1), a histone-modifying enzyme, controls mesenchymal phenotype and apoptosis through SNAIL and FAK-AKT-GSK3β
[31][88]. Consequently, the combinatorial therapy of cisplatin and LSD1 inhibitors could be a solution to cisplatin resistance in MPM
[31][88].
Finally, gene therapy could also represent a future strategy by using vectors with a suicide gene or interferons to support the patient’s immune system against tumor cells
[32][89].
6. Conclusions
Malignant pleural mesothelioma (MPM) is such an aggressive tumor that it is currently very difficult to treat. Conventional therapies and current diagnostic biomarkers are not effective. Thus, the identification of new diagnostic markers to identify the early stages of tumor, and the individuation of novel drug targets, to have other available therapeutic approaches, are crucial goals. To reach this aim, it is necessary to understand MPM pathogenetic mechanisms induced by exposure to the main risk factor, particularly asbestos fibers. This rentryview shows how, among events that have been demonstrated to be involved in the MPM onset and progression, EMT is particularly crucial: this process mediates the effects of asbestos fibers on human mesothelial cells through a mechanism mediated by crosstalk between oxidative stress and TGFβ, which can cooperate in the MPM development and metastasis.
Taken as a whole, these mechanisms provide insights that can help improve the diagnosis and treatment of MPM, with the aim of achieving new ways to counteract this aggressive cancer.