In light of expanding incidences of keratinocyte carcinoma (KC) with many patients developing multiple tumors, the demand for new treatment modalities is high. With the approval of cemplimab for locally advanced and metastasizing basal cell carcinoma and squamous cell carcinoma, KC is now included as an indication for systemic immunotherapy. At present, however, systemic KC therapy remains limited by the severe side effects associated with treatment. Immunotherapy might be more broadly applied if locally administered. Localized to the skin, KCs are easily accessible to topical drugs and physical interventions such as laser. There is an increasing appreciation of lasers’ potential to activate an immune response. Further enhancement of the laser-based immune activation might be obtained by combining laser and immunotherapeutic agents, known as laser immunotherapy.
- keratinocyte carcinoma
- basal cell carcinoma (BCC)
- squamous cell carcinoma (SCC)
- laser immunotherapy
- laser
- immunotherapy
- ablative fractionated laser (AFL)
- immune response
- immunohistochemistry
1. Introduction
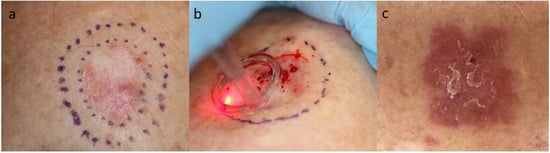
2. Rationales for Immunotherapy in Keratinocyte Carcinoma
3. Keratinocyte Carcinoma Immunotherapy: Current Status
References
- Goldmann, L. Biomedical Aspects of the Laser: The Introduction of Laser Applications into Biology and Medicine; Springer: Berlin/Heidelberg, Germany, 1967.
- Shah, S.M.; Konnikov, N.; Duncan, L.M.; Tannous, Z.S. The effect of 595 nm pulsed dye laser on superficial and nodular basal cell carcinomas. Lasers Surg. Med. 2009, 41, 417–422.
- Ortiz, A.E.; Anderson, R.R.; Avram, M.M. 1064 nm long-pulsed Nd:YAG laser treatment of basal cell carcinoma. Lasers Surg. Med. 2015, 47, 106–110.
- Haedersdal, M.; Erlendsson, A.M.; Paasch, U.; Anderson, R.R. Translational medicine in the field of ablative fractional laser (AFXL)-assisted drug delivery: A critical review from basics to current clinical status. J. Am. Acad. Dermatol. 2016, 74, 981–1004.
- Wenande, E.; Anderson, R.R.; Haedersdal, M. Fundamentals of fractional laser-assisted drug delivery: An in-depth guide to experimental methodology and data interpretation. Adv. Drug Deliv. Rev. 2020, 153, 169–184.
- Cives, M.; Mannavola, F.; Lospalluti, L.; Sergi, M.C.; Cazzato, G.; Filoni, E.; Cavallo, F.; Giudice, G.; Stucci, L.S.; Porta, C.; et al. Non-Melanoma Skin Cancers: Biological and Clinical Features. Int. J. Mol. Sci. 2020, 21, 5394.
- Donia, M.; Ellebaek, E.; Øllegaard, T.H.; Duval, L.; Aaby, J.B.; Hoejberg, L.; Køhler, U.H.; Schmidt, H.; Bastholt, L.; Svane, I.M. The real-world impact of modern treatments on the survival of patients with metastatic melanoma. Eur. J. Cancer 2019, 108, 25–32.
- Chang, J.; Zhu, G.; Cheung, C.; Li, S.; Kim, J.; Chang, A.L.S. Association Between Programmed Death Ligand 1 Expression in Patients With Basal Cell Carcinomas and the Number of Treatment Modalities. JAMA Dermatol. 2017, 153, 285–290.
- Lipson, E.J.; Lilo, M.T.; Ogurtsova, A.; Esandrio, J.; Xu, H.; Brothers, P.; Schollenberger, M.; Sharfman, W.H.; Taube, J.M. Basal cell carcinoma: PD-L1/PD-1 checkpoint expression and tumor regression after PD-1 blockade. J. Immunother. Cancer 2017, 5, 23.
- Migden, M.R.; Khushalani, N.I.; Chang, A.L.S.; Lewis, K.D.; Schmults, C.D.; Hernandez-Aya, L.; Meier, F.; Schadendorf, D.; Guminski, A.; Hauschild, A.; et al. Cemiplimab in locally advanced cutaneous squamous cell carcinoma: Results from an open-label, phase 2, single-arm trial. Lancet Oncol. 2020, 21, 294–305.
- Gambichler, T.; Schröter, U.; Höxtermann, S.; Susok, L.; Stockfleth, E.; Becker, J.C. Decline of programmed death-1-positive circulating T regulatory cells predicts more favourable clinical outcome of patients with melanoma under immune checkpoint blockade. Br. J. Dermatol. 2019, 182, 1214–1220.
- Kaporis, H.G.; Guttman-Yassky, E.; Lowes, M.A.; Haider, A.S.; Fuentes-Duculan, J.; Darabi, K.; Whynot-Ertelt, J.; Khatcherian, A.; Cardinale, I.; Novitskaya, I.; et al. Human Basal Cell Carcinoma Is Associated with Foxp3+ T cells in a Th2 Dominant Microenvironment. J. Investig. Dermatol. 2007, 127, 2391–2398.
- Zhang, B.; Chikuma, S.; Hori, S.; Fagarasan, S.; Honjo, T. Nonoverlapping roles of PD-1 and FoxP3 in maintaining immune tolerance in a novel autoimmune pancreatitis mouse model. Proc. Natl. Acad. Sci. USA 2016, 113, 8490–8495.
- Kamada, T.; Togashi, Y.; Tay, C.; Ha, D.; Sasaki, A.; Nakamura, Y.; Sato, E.; Fukuoka, S.; Tada, Y.; Tanaka, A.; et al. PD-1+ regulatory T cells amplified by PD-1 blockade promote hyperprogression of cancer. Proc. Natl. Acad. Sci. USA 2019, 116, 9999–10008.
- Wojas-Krawczyk, K.; Kalinka, E.; Grenda, A.; Krawczyk, P.; Milanowski, J. Beyond PD-L1 Markers for Lung Cancer Immunotherapy. Int. J. Mol. Sci. 2019, 20, 1915.
- Chalmers, Z.R.; Connelly, C.F.; Fabrizio, D.; Gay, L.; Ali, S.M.; Ennis, R.; Schrock, A.; Campbell, B.; Shlien, A.; Chmielecki, J.; et al. Analysis of 100,000 human cancer genomes reveals the landscape of tumor mutational burden. Genome Med. 2017, 9, 1–14.
- Goodman, A.M.; Kato, S.; Cohen, P.R.; Boichard, A.; Frampton, G.; Miller, V.; Stephens, P.J.; Daniels, G.A.; Kurzrock, R. Genomic landscape of advanced basal cell carcinoma: Implications for precision treatment with targeted and immune therapies. OncoImmunology 2018, 7, e1404217.
- Hartevelt, M.M.; Bavinck, J.N.B.; Kootte, A.M.M.; Vermeer, B.J.; Vandenbroucke, J.P. Incidence of skin cancer after renal transplantation in The Netherlands. Transplantation 1990, 49, 506–509.
- Omland, S.H.; Gniadecki, R.; Hædersdal, M.; Helweg-Larsen, J.; Omland, L.H. Skin cancer risk in hematopoietic stem-cell transplant recipients compared with background population and renal transplant recipients a population-based cohort study. JAMA Dermatol. 2016, 152, 177–183.
- Vaddepally, R.K.; Kharel, P.; Pandey, R.; Garje, R.; Chandra, A.B. Review of Indications of FDA-Approved Immune Checkpoint Inhibitors per NCCN Guidelines with the Level of Evidence. Cancers 2020, 12, 738.
- Migden, M.R.; Rischin, D.; Schmults, C.D.; Guminski, A.; Hauschild, A.; Lewis, K.D.; Chung, C.H.; Hernandez-Aya, L.; Lim, A.M.; Chang, A.L.S.; et al. PD-1 Blockade with Cemiplimab in Advanced Cutaneous Squamous-Cell Carcinoma. N. Engl. J. Med. 2018, 379, 341–351.
- Zhao, C.Y.; Hwang, S.J.E.; Anforth, R.; Carlos, G.; Chou, S.; Carlino, M.; Fernández-Peñas, P. Incidence of Basal Cell Carcinoma and Squamous Cell Carcinoma in Patients on Antiprogrammed Cell Death-1 Therapy for Metastatic Melanoma. J. Immunother. 2018, 41, 343–349.
- Falchook, G.S.; Leidner, R.; Stankevich, E.; Piening, B.; Bifulco, C.; Lowy, I.; Fury, M.G. Responses of metastatic basal cell and cutaneous squamous cell carcinomas to anti-PD1 monoclonal antibody REGNJ. Immunother. Cancer 2016, 4, 70.
- Ikeda, S.; Goodman, A.M.; Cohen, P.R.; Jensen, T.J.; Ellison, C.K.; Frampton, G.; Miller, V.; Patel, S.P.; Kurzrock, R. Metastatic basal cell carcinoma with amplification of PD-L1: Exceptional response to anti-PD1 therapy. NPJ Genom. Med. 2016, 1, 16037.
- Cohen, P.R.; Kato, S.; Goodman, A.M.; Ikeda, S.; Kurzrock, R. Appearance of New Cutaneous Superficial Basal Cell Carcinomas during Successful Nivolumab Treatment of Refractory Metastatic Disease: Implications for Immunotherapy in Early Versus Late Disease. Int. J. Mol. Sci. 2017, 18, 1663.
- Cannon, J.G.; Russell, J.S.; Kim, J.; Chang, A.L.S. A case of metastatic basal cell carcinoma treated with continuous PD-1 inhibitor exposure even after subsequent initiation of radiotherapy and surgery. JAAD Case Rep. 2018, 4, 248–250.
- Winkler, J.; Schneiderbauer, R.; Bender, C.; Sedlaczek, O.; Fröhling, S.; Penzel, R.; Enk, A.; Hassel, J.C. Anti-programmed cell death-1 therapy in nonmelanoma skin. Br. J. Dermatol. 2017, 176, 498–502.
- Jørgensen, N.; Kaae, J.; Grauslund, J.; Met, Ö.; Nielsen, S.; Pedersen, A.; Svane, I.; Ehrnrooth, E.; Andersen, M.; Zachariae, C.; et al. Vaccination against PD-L1 with IO103 a Novel Immune Modulatory Vaccine in Basal Cell Carcinoma: A Phase IIa Study. Cancers 2021, 13, 911.
- Peris, K.; Fargnoli, M.C.; Garbe, C.; Kaufmann, R.; Bastholt, L.; Seguin, N.B.; Bataille, V.; Marmol, V.D.; Dummer, R.; Harwood, C.A.; et al. Diagnosis and treatment of basal cell carcinoma: European consensus–based interdisciplinary guidelines. Eur. J. Cancer 2019, 118, 10–34.
- Dummer, R.; Urosevic, M.; Kempf, W.; Hoek, K.; Hafner, J.; Burg, G. Imiquimod in basal cell carcinoma: How does it work? Br. J. Dermatol. 2003, 149, 57–58.
- Vidal, D.; Matías-Guiu, X.; AlOmar, A. Open study of the efficacy and mechanism of action of topical imiquimod in basal cell carcinoma. Clin. Exp. Dermatol. 2004, 29, 518–525.
- MacFarlane, D.; El Tal, A. Cryoimmunotherapy: Superficial basal cell cancer and squamous cell carcinoma in situ treated with liquid nitrogen followed by imiquimod. Arch Dermatol. 2011, 147, 1326–1327.
- Gaitanis, G.; Nomikos, K.; Vava, E.; Alexopoulos, E.; Bassukas, I.; Alexopoulos, E. Immunocryosurgery for basal cell carcinoma: Results of a pilot, prospective, open-label study of cryosurgery during continued imiquimod application. J. Eur. Acad. Dermatol. Venereol. 2009, 23, 1427–1431.
