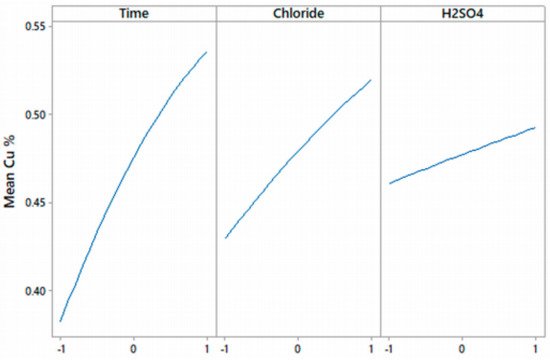
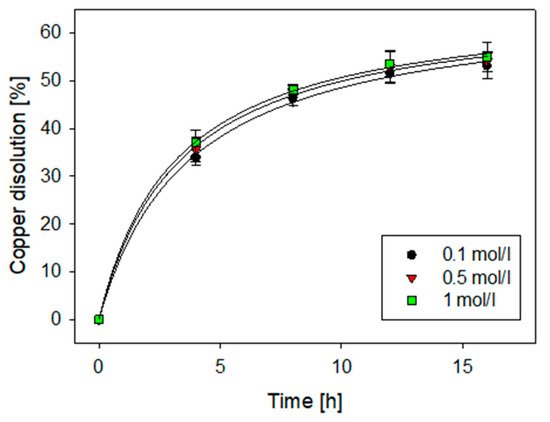
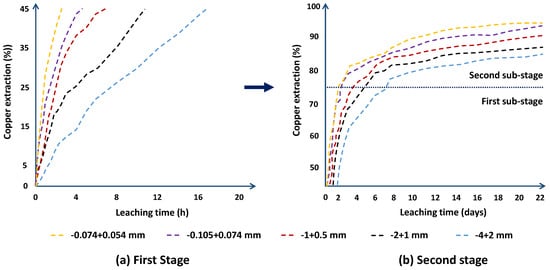
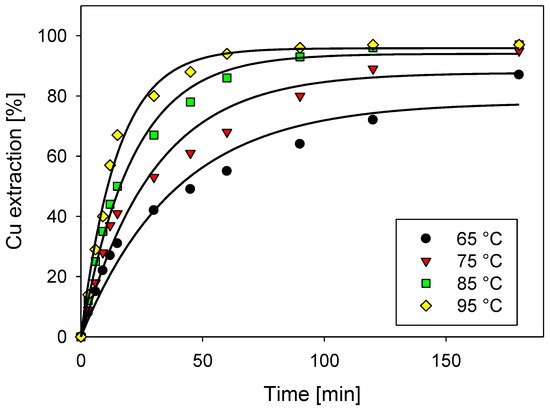
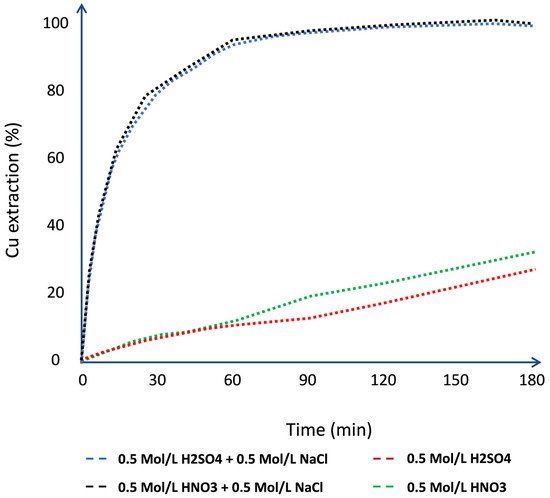
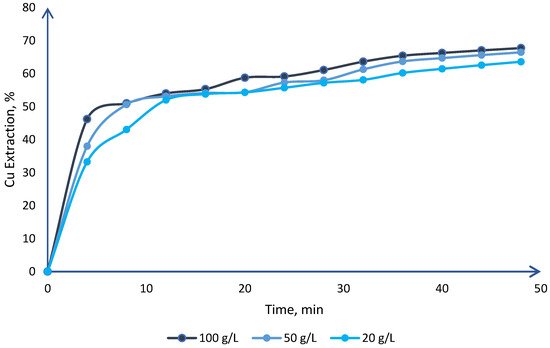
Chalcocite is the most abundant secondary copper sulfide globally, with the highest copper content, and is easily treated by conventional hydrometallurgical processes, making it a very profitable mineral for extraction. Among the various leaching processes to treat chalcocite, chloride media show better results and have a greater industrial boom. Chalcocite dissolution is a two-stage process, the second being much slower than the first. During the second stage, in the first instance, it is possible to oxidize the covellite in a wide range of chloride concentrations or redox potentials (up to 75% extraction of Cu). Subsequently, CuS2 is formed, which is to be oxidized. It is necessary to work at high concentrations of chloride (>2.5 mol/L) and/or increase the temperature to reach a redox potential of over 650 mV, which in turn decreases the thickness of the elemental sulfur layer on the mineral surface, facilitating chloride ions to generate a better porosity of this.
- Cu2S
- CuS
1. Effect on Chloride Concentration
2. Effect on Stirring Speed
3. Effect on Acid Concentration