Rhus trilobata (RHTR) is a medicinal plant with cytotoxic activity in different cancer cell lines. However, the active compounds in this plant against ovarian cancer are unknown. In this study, we aimed to evaluate the antineoplastic activity of RHTR and identify its active metabolites against ovarian cancer. The aqueous extract (AE) and an active fraction (AF02) purified on C18-cartridges/ethyl acetate decreased the viability of SKOV-3 cells at 50 and 38 μg/mL, respectively, compared with CHO-K1 (> 50 μg/mL) in MTT assays and generated changes in the cell morphology with apoptosis induction in Hemacolor® and TUNEL assays (p ≤ 0.05, ANOVA). The metabolite profile of AF02 showed a higher abundance of flavonoid and lipid compounds compared with AE by UPLC-MS. Gallic acid and myricetin were the most active compounds in RHTR against SKOV-3 cells at 50 and 166 μg/mL, respectively (p ≤ 0.05, ANOVA). Antineoplastic studies in Nu/Nu female mice with subcutaneous SKOV-3 cells xenotransplant revealed that 200 mg/kg/i.p. of AE and AF02 inhibited ovarian tumor lesions from 37.6% to 49% after 28 days (p ≤ 0.05, ANOVA). In conclusion, RHTR has antineoplastic activity against ovarian cancer through a cytostatic effect related to gallic acid and myricetin. Therefore, RHTR could be a complementary treatment for this pathology.
1. Introduction
Ovarian cancer is the sixth most frequent tumor in women and the fourth cause of death in Mexico due to gynecological tumors
[1]. Ovarian tumors can be primary or metastatic and are classified based on their origin as epithelial, germinal, or stromal of the sexual cord tumors
[2]. Epithelial tumors are the most common type of ovarian neoplasm; among them, the serous subtype usually appears more frequently
[2]. Surgical resection is the principal treatment for this disease, followed by antitumoral chemotherapy with cytotoxic or cytostatic drugs
[1][3]. However, the surgent of resistance in neoplastic tissue limits ovarian cancer chemotherapy’s success
[3], making it necessary to search for alternative treatments or new therapeutic agents for this disease. Plants used for alternative medicine represent an option in the search for active compounds for cancer treatment. Recent work with
Rhus trilobata Nutt. (RHTR) demonstrated the presence of antineoplastic agents that could be new therapeutic candidates for the treatment of ovarian cancer that afflicts the female population in Mexico and around the world
[4].
In Mexico, RHTR (
Anacardiaceae family) is used for the treatment of gastrointestinal diseases and cancer. RHTR is known by the common name of
aciditos or
agritos due to the characteristic flavor of its fruit
[4]. Previous studies by Abbott et al. (1966) demonstrated the antineoplastic activity of RHTR in Syrian hamsters xenotransplanted with duodenum adenocarcinoma. Animals were treated with extracts of leaves (100 mg/kg/i.p.) or fruits (400 mg/kg/i.p.) for seven days; tumoral lesions decreased 33% with both extracts in comparison with control conditions
[5]. Subsequently, Pettit et al. (1974) isolated gallic acid (Ga) from RHTR leaves with column chromatography (Sephadex LH-20) and determined its biological activity; Ga decreased KB cell viability (IC
50: 3.1 μg/mL). The authors concluded that the medicinal properties of RHTR correspond to this compound
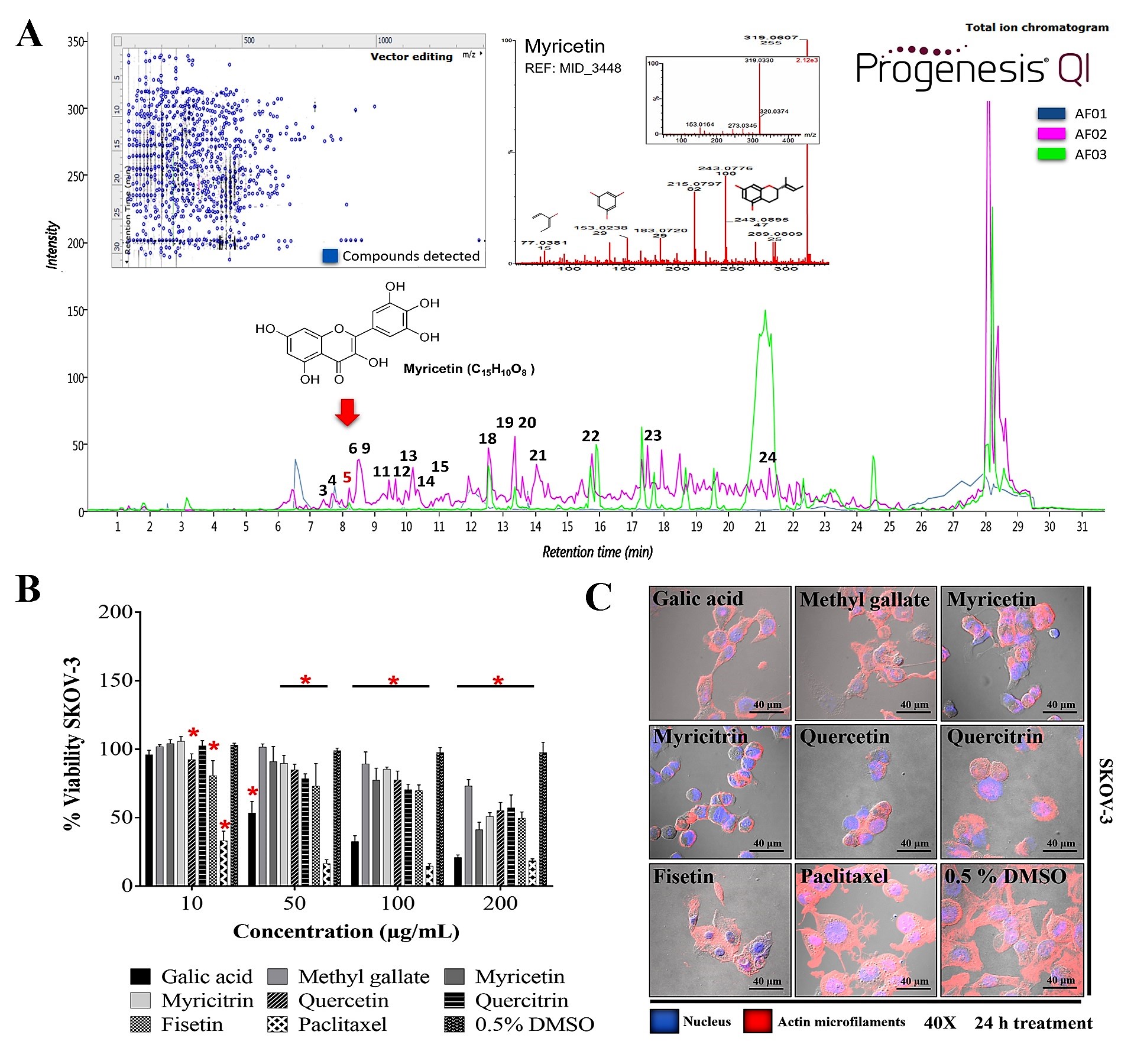
[6]. Studies conducted by our research group revealed that the aqueous extract (AE) of RHTR stems contains quinic acid, myricetin (Myr), Ga, 1,2,3,4,6-pentakis-
O-gallioyl-
β-D-glucose (
β-PGG), quercetin, obtusaquinol, fisetin, margaric acid, and amentoflavone by UPLC-MS
E, which are compounds with biological activity already demonstrated by other studies
[4]. Additionally, AE-RHTR presented a selective activity against CACO-2 cells (IC
50: 5 μg/mL), and low toxicity (LD
50: 1141.5 mg/kg)
[4]. Thus, the biological activity observed in RHTR may be related to compounds such as phenolic acids or flavonoids and the synergic effects between both molecule types.
2. Results and Discussion
2. Biological Activity of AE-RHTR and Fractions in Cell Lines
2.1. Biological Activity of AE-RHTR and Fractions in Cell Lines
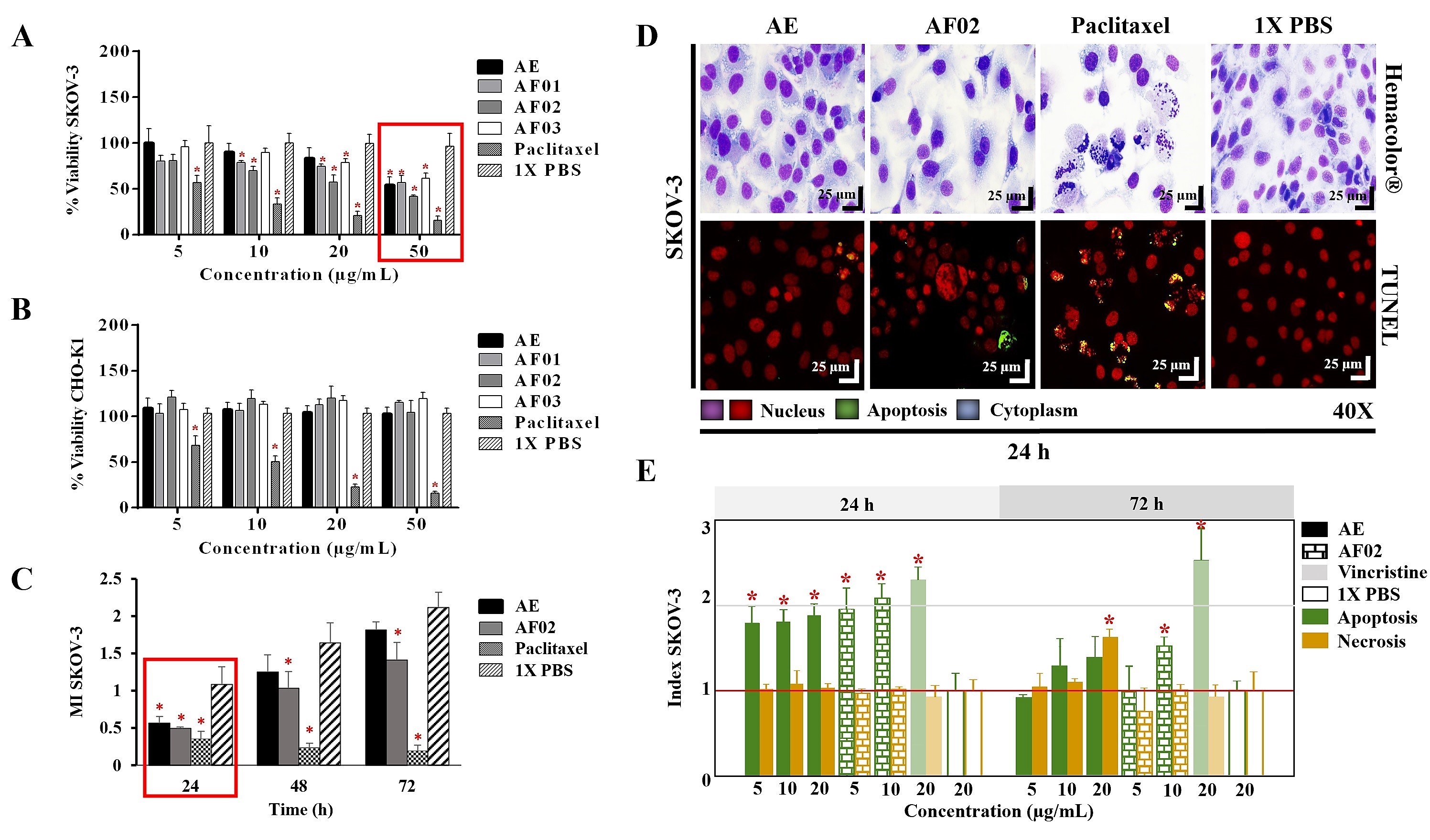
The AE and fractions from RHTR presented biological activity in SKOV-3 cells at 50 μg/mL (
Figure 1A). In comparison, the concentration required to affect the viability of CHO-K1 cells was greater than 50 μg/mL (
Figure 1B), demonstrating a selective effect. AE and AF02 were most active against SKOV-3 cells at 50 and 38 μg/mL, respectively, compared with 1× PBS (vehicle group) (
p < 0.05, Dunnett). However, AE and AF02 were found to only have a 24 h limited inhibitory effect on SKOV-3 cell proliferation (
Figure 1C). Additionally, SKOV-3 cells treated with AE and AF02 had a similar morphology to the vehicle group but with a considerable increase in cytoplasmic vesicles and an absence of cellular mitosis, which suggest a quiescent effect in both treatments (
Figure 1D). The apoptosis assays mainly demonstrated increased caspase-3/7 activity and nuclear DNA-fragmentation in cells treated with AE (8.47% ± 0.9%), AF02 (14.01% ± 3.7%), and paclitaxel (46.42% ± 5.0%) at 24 h in comparison with the vehicle group (2.47% ± 0.4%) (
p < 0.05, ANOVA;
Figure 1D,E). Necrotic events were present during treatments with both samples at 72 h (
Figure 1E), possibly related to stages of late apoptosis and not with true necrosis, which can promote inflammatory processes; however, additional studies are necessary to corroborate this finding. These results resemble those obtained with CACO-2 cells, where cell cycle arrest in G
1 and the appearance of a G
1 subpopulation related to apoptosis were observed. In contrast, in BEAS-2B cells, increasing the concentration up to 800 μg/mL was necessary to observe a similar effect
[4]. Therefore, AE and AF02 were selected to evaluate their antineoplastic activity.
Figure 1. Biological activity of RHTR in ovarian cancer. The IC50 of AE and fractions in SKOV-3 (A) and CHO-K1 (B) cell lines were obtained with dose–response viability curves at 24 h by MTT assay. The antiproliferative (C), cytotoxic, and apoptotic (E) activities of AE and AF02 were determined in SKOV-3 cells by an ApoTox-Glo™ Triplex Assay. The morphological changes with Hemacolor® rapid staining and TUNEL were observed at 24 h of treatment with AE (50 µg/mL) and AF02 (38 µg/mL), respectively (D). The results show the mean ± SD of three biological replicates (n = 3, in triplicates). * p ≤ 0.05 vs. the control group without treatment (1× PBS) (ANOVA). Paclitaxel or vincristine was used as the positive control. MI, mitotic index (MI = Abs sample/Abs control); RHTR, Rhus trilobata; AE, aqueous extract; AF, aqueous fraction (numbers indicate the fraction obtained after fractionation on the solid phase with solvents, as described in the Methodology section).
3. Antineoplastic Activity of RHTR in Mice Xenotransplanted with SKOV-3 Cells
2.2. Antineoplastic Activity of RHTR in Mice Xenotransplanted with SKOV-3 Cells
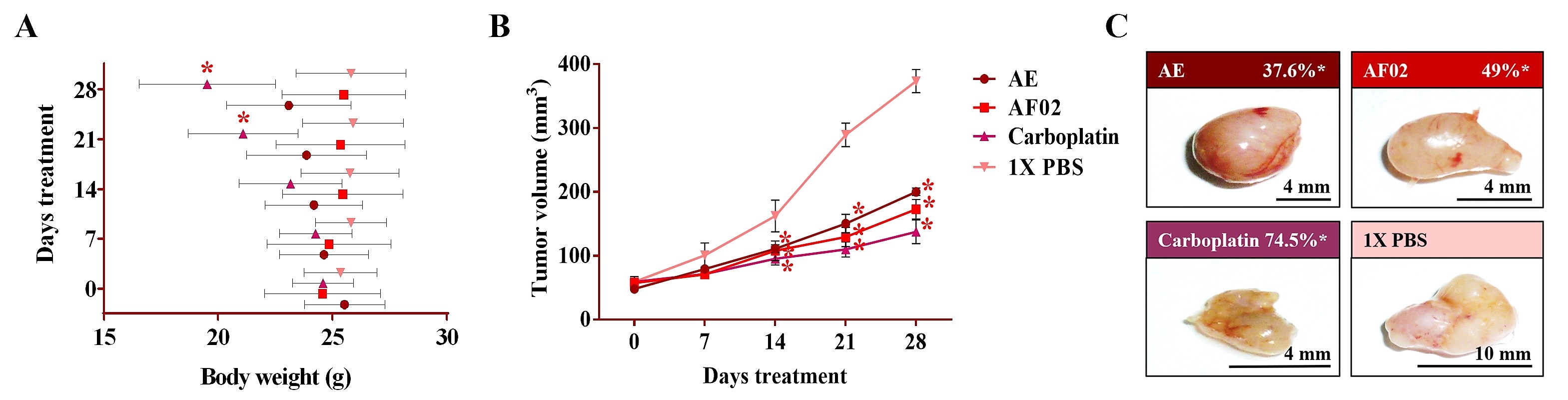
After 24 h of treatment, the rodents showed no behavioral changes. Additionally, the analyses of their bodyweight showed that the group treated with AE experienced a 9.5% decrease, but a 3.7% increase in the group with AF02. However, these changes were not significant compared to the control group treated with 1× PBS vehicle (
p > 0.05, Dunnett;
Figure 2A). At the end of treatments, mice were euthanized for tumor lesions recovery and to perform a macroscopic analysis of the developed lesions (
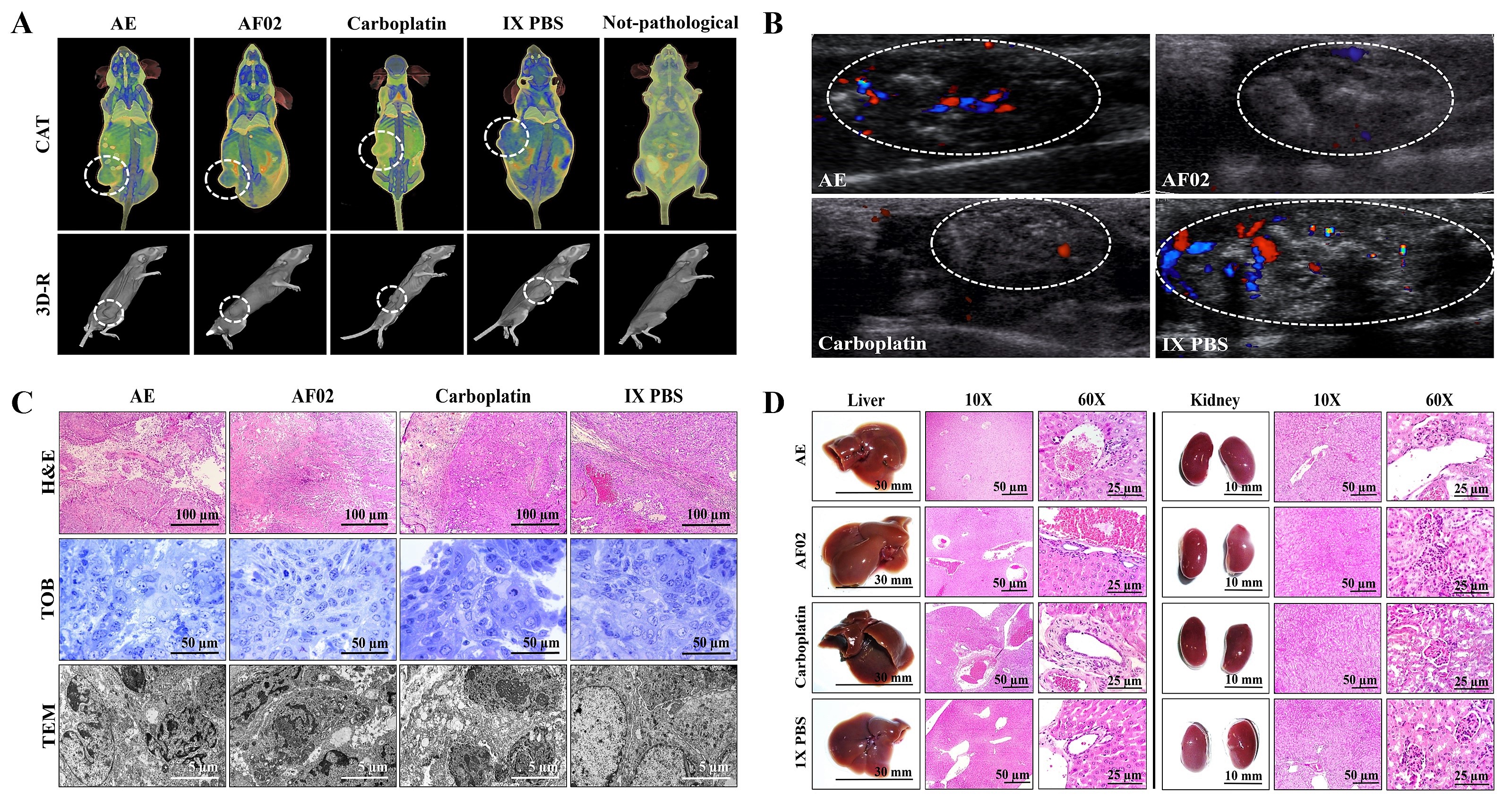
Figure 2C). Similar characteristics in all tumors were present in the treated groups with an ovoid or smooth-surfaced morphology and the presence of vascularity (
Figure 2C). However, minor differences in the color of tumors were found: pink for AE and whitish for AF02; groups treated with carboplatin and vehicle presented a yellow tumor with a nodular surface (
Figure 2C). Analysis of tumoral weight showed that greater masses were found in the vehicle group, followed by AE, AF02, and carboplatin (
p > 0.05, Tukey;
Table 1). These results correlate with the tumor volume of lesions present in rodents along with the treatment time (
Figure 2B). The inhibition percentage in tumoral growth with AE, AF02, and carboplatin was 37.6%, 49%, and 74.5%, respectively, compared with the control group for 14 and 28 days of treatment (
p ≤ 0.05, Dunnett;
Figure 2B,C). These findings demonstrate changes in the disease evolution directly related to the treatments used in the study. Similarly, these results agree with those obtained in tumoral length (
Table 1), which demonstrated a cytostatic effect of the treatments.
Figure 2. Antineoplastic activity of AE and AF02 from RHTR in ovarian cancer. The bodyweight of rodents was monitored with an electronic bascule for 28 days (A). The tumor volume was determined with a Vernier caliper in mice treated with AE and AF02 at 200 mg/kg/i.p./day (Tumoral volume = [Larger diameter × (Shorter diameter)2]/2) (B). Morphological changes in tumor lesions were analyzed at the end of treatments, and the percent inhibition was calculated (C). Results show the mean ± SD of two biological replicates (n = 5). * p ≤ 0.05 vs. the control group without treatment (1× PBS) (ANOVA). The positive control was carboplatin (50 mg/kg/i.p./3 alternating days per week in mice). RHTR, Rhus trilobata; AE, aqueous extract; AF02, aqueous fraction-02 (active fraction obtained with ethyl acetate).
Table 1. Morphological characteristics of ovarian tumor lesions treated with RHTR.
| Treatments |
AE |
AF02 |
Carboplatin |
1× PBS |