FGF23 is a bone-derived hormone that is essential for regulating vitamin D and phosphate homeostasis.
- fibroblast growth factor 23 (FGF23)
- fibroblast growth factor receptor (FGFR)
- chronic kidney disease
1. The Evolution and Biology of FGF23
- The Evolution and Biology of FGF23
Nearly 50 million years ago, life on earth was flourishing due to the re-population from terrestrial creatures, which progressed life beyond their aquatic habitats [1][2][3][4][5][5,6,7,8,9]. Throughout this period, an abundant amount of gene families, including the fibroblast growth factor (FGF) family, expanded in two phases which managed these new demands on all terrestrial systems, in comparison to their aquatic ancestors [6][7][8][10,11,12]. FGFs have been identified in both invertebrates and vertebrates. Prior to chordate evolution, the FGF gene family expanded from three to six genes via gene duplication, during their first phase of the metazoan linage [9][13]. During the early emergence and evolution of vertebrates, the FGF gene family underwent two large-scale gene duplications during their second phase [9][13]. This resulted in FGFs acquiring more significant roles in biological processes, such as in embryonic development, organogenesis and metabolic homeostasis [9][10][11][13,14,15].
The highly conserved FGF family consists of 22 members in mammals and is divided into three classifications: intracellular, paracrine and endocrine FGFs, based on their mechanisms of action [9][12][13,16]. Their conserved intrinsic core domain, which spans ~120 amino acids in length (~30–60% amino acid identity among all FGF members) promotes ligand binding to a distinct superfamily of receptor tyrosine kinases, known as fibroblast growth factor receptors (FGFRs) [6][9][13][14][15][16][10,13,17,18,19,20]. Intracellular FGFs are not secreted by their producing cells and act in an FGFR-independent manner, which enables them to mediate intracellular signaling events [9][17][18][13,21,22]. To date, their notable functions are regulating voltage-gated Na+ channels and the electrical excitability of neuronal cells [17][19][20][21][21,23,24,25]. Paracrine FGFs and endocrine FGFs (also called FGF19 subfamily) mediate their biological responses in an FGFR-dependent manner [6][9][10,13]. Unlike paracrine FGFs, which bind heparin or heparin sulfate proteoglycans (HPG) as a co-factor to facilitate FGFR activation and function as differentiation factors in development, endocrine FGFs have reduced affinity for HPG due to topological disparities in their heparin-binding region [9][22][23][24][13,26,27,28]. This permits the FGF19 subfamily to escape extracellular matrices and operate over long distances, functioning as circulating hormones [25][26][27][29,30,31]. As an alternative, endocrine FGFs employ αKlotho or βKlotho as their co-receptor to promote efficient FGF:FGFR binding [27][31].
FGF23 is a member of the FGF19 subfamily, which utilizes αKlotho to carry out its physiological functions, targeting the kidney to promote phosphate excretion and enabling the suppression of 1-25-dihydroxyvitamin D synthesis [9][27][28][29][30][31][13,31,32,33,34,35]. The development of this FGF23:αKlotho network is a ramification of vertebrate evolution, where primitive piscine ancestors such as ostracoderms acquired a boney endoskeleton [32][36]. In 2000, FGF23 was ultimately identified in the ventrolateral thalamic nucleus of mice, and its biological significance rapidly followed when a missense mutation of the Fgf23 gene in patients with autosomal dominant hypophosphatemic rickets (ADHR) was later identified [33][34][37,38]. Under physiologic conditions, FGF23 is predominantly produced by osteocytes as a 32 kDa glycoprotein in response to elevations in serum phosphate levels due to dietary loading or serum 1-25-dihydroxyvitamin D, where its N-terminal region shares homologies with other FGF family members and interacts with FGFRs, whereas its C-terminal portion binds αKlotho [30][35][36][37][38][39][34,39,40,41,42,43].
Prior to FGF23 secretion from bone, post-translational modifications allows this protein to circulate in the bloodstream as a full-length mature form (also called intact FGF23), which possess biological activity, or circulate as proteolytic cleaved fragments (Figure 1) [34][36][40][41][38,40,44,45]. This proteolytic cleavage event is executed by subtilisin-like pro-protein convertases, such as furin, which occurs at the consensus sequence Arg176-X-X-Arg179 and is not present in other FGF family members [34][40][42][38,44,46].
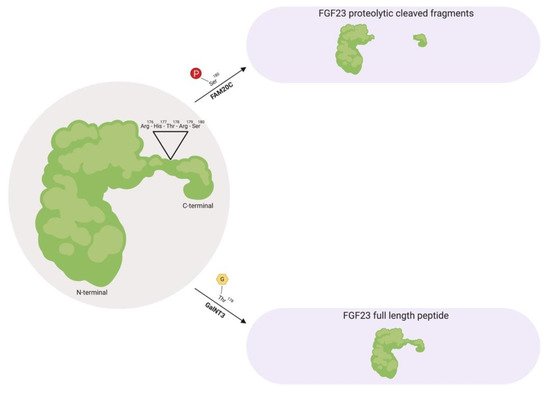
Figure 1. Fibroblast growth factor 23 (FGF23) regulation by post-translational modification. As a full-length biologically active protein in circulation, FGF23 is O-glycosylated by GalNT3 at several residues such as Thr178, which protects FGF23 from proteolytic cleavage by pro-protein convertases such as furin. Vice versa, as proteolytic cleaved fragments in circulation, FAM20C phosphorylates FGF23 at multiple amino acids, such as Ser180. This phosphorylation event impedes O-glycosylation by GalNT3 and allows furin to recognize its Arg176-His177-Thr178-Arg179-Ser180 consensus sequence in FGF23, thus leading to FGF23 cleavage and separation of the N-terminal and C-terminal fragments. GalNT3, polypeptide N-acetylgalactosaminyltransferase 3; FAM20C, secretory protein kinase family with sequence similarity-20 member C; Arg, arginine; His, histidine; Thr, threonine; Ser, serine.
As a circulating biologically active protein, FGF23 is O-glycosylated at several residues, such as Thr178 via polypeptide N-acetylgalactosaminyltransferase 3 (GalNT3), which protects FGF23 from proteolytic cleavage and helps support its intact structure [40][43][44][45][44,47,48,49]. The half-life of this biologically active form of FGF23 is ~45–60 minutes in humans, whereas in rodents, appears to be shorter with ~20–30 minutes in mice and ~5 minutes in rats [46][47][48][50,51,52].
Furthermore, as proteolytic cleaved fragments in circulation, FGF23 is phosphorylated at multiple amino acid residues, such as Ser180 via secretory protein kinase family with sequence similarity-20 member C (FAM20C), which impedes O-glycosylation at Thr178 by GalNT3 and results in its biologically inactive form by separating the respective FGFR and αKlotho binding regions [49][50][53,54]. A firm regulation of these post-translational modification and processing events are essential for maintaining proper homeostatic balances. Missense mutations in the FGF23 consensus sequence or in GalNT3 and/or FAM20C have been shown to be detrimental to physiologic processes, where interfering with FGF23 cleavage results in either elevated serum intact FGF23 levels that results in hypophosphatemia, or diminished serum intact FGF23 levels which results in hyperphosphatemia due to excessive proteolytic cleavage [34][43][51][52][53][54][55][38,47,55,56,57,58,59].
Regarding the metabolic rate of FGF23, renal extraction appears to play a critical role in its metabolism, yet only a minor contribution of FGF23 excretion via the kidney may contribute since FGF23 is not detectable in urine under physiological conditions [48][52]. However, in patients with acute kidney injury (AKI), FGF23 can be measured following urine analysis, where these elevations correspond with all-cause mortality [56][60]. Future investigations are needed in this context to help distinguish the origin of urinary FGF23.
2. FGF23 in Chronic Kidney Disease
- FGF23 in Chronic Kidney Disease
The kidneys are part of the bone-kidney-intestinal axis [32][36]. The crucial role of this axis is to regulate mineral metabolism by altering tubular resorption of serum calcium and phosphate, with the assistance of key modulators FGF23, PTH and 1-25-dihydroxyvitamin D [57][58][85,114]. In an average Western diet, individuals ingest ~1200 mg/day of phosphate, yet a net weight of ~900 mg/day is absorbed into circulation [59][60][61][115,116,117]. The amount of phosphate absorption depends on its exogenous source and bioavailability [62][63][118,119]. Phosphate exists in two forms, organic and inorganic phosphate [57][63][85,119].
Organic phosphate is predominantly found in foods that are rich in protein (non-phytates) such as meat, fish and dairy products, which have a bioavailability of 40–80% [57][63][64][65][66][85,119,120,121,122]. By contrast, organic phosphate can also be obtained from plant-derived foods (phytates) such as cereal and nuts, but they have a bioavailability less than 40% [57][67][68][69][85,123,124,125]. This dissimilarity in phosphate absorption is attributed to monogastric mammals lacking the enzyme phytase, which is acquired to liberate phosphate [57][62][63][70][85,118,119,126]. To increase dietary absorption, phosphate additives are utilized to increase shelf life, alter texture and increase the flavor of food products [71][72][73][74][127,128,129,130]. These inorganic phosphate sources are passively absorbed in the intestine and have a bioavailability of nearly 100% [32][57][75][76][36,85,131,132]. With a wide range of foods containing additives such as polyphosphates and pyrophosphates, the relation of nephrotoxicity to phosphate toxicity (phosphotoxicity) has been the focus of many studies [32][63][77][78][79][80][81][36,119,133,134,135,136,137,138]. Clinical studies have shown that a single dose of 11.5 g of phosphate can accelerate de novo CKD over the course of several months [32][36].
To prevent phosphotoxicity, FGF23 levels rise and fall in correlation with the amount of dietary phosphate absorbed [82][83][139,140]. When an individual absorbs phosphate from foods that are high in its bioavailability, the bone upregulates FGF23 production to induce greater urinary excretion of phosphate and minimizing the efficiency of intestinal phosphate absorption by reducing serum 1-25-dihydroxyvitamin D levels [82][139]. In contrast, when an individual absorbs phosphate from foods that are low in its bioavailability, renal phosphate reabsorption is enhanced, as well as its absorption efficiency from the intestine by the actions of 1-25-dihydroxyvitamin D [82][139]. When renal damage occurs and kidney function declines, the alterations in bone and mineral homeostasis is inescapable. This event is termed chronic kidney disease-mineral bone disorder (CKD-MDB), which refers to renal dysfunction and altered levels of calcium, phosphate, PTH, 1-25-dihydroxyvitamin D and FGF23 [83][84][140,141].
During the early stages of CKD, the expression of the renal FGF23 co-receptor, αKlotho, decreases in response to kidney damage and progressively declines along with the loss of functional nephrons, promoting partial resistance to FGF23′s physiologic actions [82][85][86][87][139,142,143,144]. As a compensatory mechanism, FGF23 levels rise 1000-fold above normal values in an attempt to maintain a neutral phosphate balance [82][41][88][139,145,146]. This compensatory increase in FGF23 promotes the suppression of 1-25-dihydroxyvitamin D production, which in turn, promotes the elevation of PTH causing secondary hyperparathyroidism [89][147]. Throughout these hormonal alterations, serum phosphate levels gradually increase and by end-stage renal disease (ESRD), ultimately result in overt hyperphosphatemia due to renal resistance to FGF23′s actions on impaired kidneys [59][82][89][115,139,147].
These metabolic alterations primarily contribute to associated pathologies that are observed in CKD, such as immune dysfunction, systemic inflammation, anemia, vascular calcification, skeletal muscle dysfunction and cardiac hypertrophy, resulting in premature death [90][91][92][93][94][95][96][148,149,150,151,152,153,154]. Although FGF23 acts in a compensatory manner towards these elevating phosphate levels, clinical CKD studies have demonstrated powerful and dose-dependent associations between elevations in serum levels of FGF23 and CKD-associated pathologies, such as systemic inflammation, anemia and cardiovascular mortality, which is the leading cause of death across all stages of CKD [97][98][99][100][101][2,155,156,157,158]. FGF23 has also been shown to directly promote immune dysfunction, systemic inflammation and cardiac hypertrophy [102][103][104][105].[90,92,95,98]
