The involvement of impaired alpha (α) cell function has been recognized as playing an essential role in several diseases, since hyperglucagonemia has been evidenced in both Type 1 and T2DM. This phenomenon has been attributed to intra-islet defects, like modifications in pancreatic α cell mass or dysfunction in glucagon’s secretion. Emerging evidence has shown that chronic hyperglycaemia provokes changes in the Langerhans’ islets cytoarchitecture, including α cell hyperplasia, pancreatic beta (β) cell dedifferentiation into glucagon-positive producing cells, and loss of paracrine and endocrine regulation due to β cell mass loss. Other abnormalities like α cell insulin resistance, sensor machinery dysfunction, or paradoxical ATP-sensitive potassium channels (KATP) opening have also been linked to glucagon hypersecretion.
- glucagon
- Langerhans’ islets
- type 2 diabetes
- hyperglycaemia
- hypoglycaemia
- α Cell
1. Introduction
2. Alpha Cell Physiology: From Secretion to Regulation
The human pancreas contains 1–2 million islets, each measuring 50–100 μm in diameter and containing ∼2000 cells on average. However, islet cells are only 2% of the overall pancreatic mass. It is notable that up to 65% of the human islet cells are α cells [10][8]. All islet cells originate from the endoderm. Its differentiation into each islet linage is mediated by the pancreatic and duodenal homeobox 1 (Pdx1) and neurogenin-3 (Ngn3) genes. The further evolution of α cells requires both aristaless-related homeobox (Arx) and forkhead box protein A2 (Fox-A2) action in addition to low expression levels of paired box 4 (Pax4). Other factors important for α cell differentiation include MAF BZIP Transcription Factor B (MafB), NK6 Homeobox (Nkx6.1; Nkx6.2), Pax6 [11][9], and RNA Paupar (PAX6 Upstream Antisense RNA). The latter is a novel long noncoding that has been shown to regulate α cell development through alternative splicing of Pax6 [12][10].
Glucagon is the primary α cell hormone product. It is derived from the proteolysis of the 160-aminoacid pre-pro-glucagon peptide coded by its gene located in 2q24.216. This gene is strongly expressed in α cells, the brain, and L-cells of the gut. Under normal conditions, α cells synthetize glucagon via proconvertase 2 post translational proteolysis, while L-cells produce glucagon-like peptide-1 (GLP-1) via proconvertase 1/3 pathway.
The classic model of glucagon secretion regulation is explained by glucose-medicated glucagon exocytosis. Anatomically, pancreatic islets are highly vascularized to ensure a rapid glucose and aminoacidic sensing. A glycaemic drop near to threshold stimulates glucagon release [17][11]. The cellular mechanism behind this glucose-dependent regulation of glucagon secretion is described in Figure 1.
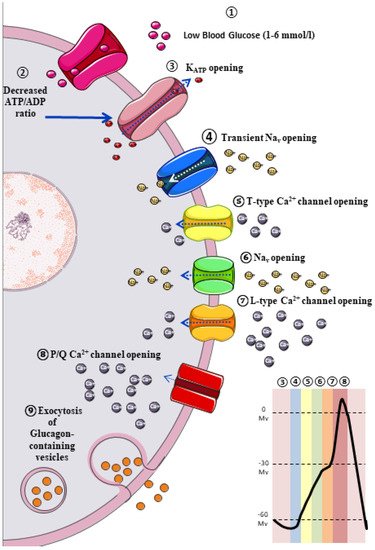
3. Extrinsic Model for the Regulation of Glucagon Secretion: Neurohormonal Mechanism
3.1. Paracrine Regulation
Omar-Hmeadi et al. observed a lack of inhibition in glucagon exocytosis by hyperglycaemia, somatostatin, or insulin in intact islets in α cells from T2DM cadaveric. Instead, hyperglycaemia inhibits α cell exocytosis, but not in the T2DM donor’s α cell or when paracrine inhibition by insulin or somatostatin is blocked. A reduced Surface expression of Somatostatin-receptor-2 in islet from T2DM donors suggests somatostatin resistance, and consequently, elevated glucagon in T2DM may reflect α cell insensitivity to paracrine inhibition during hyperglycaemia [16][12].
3.2. Autocrine Regulation
It is widely known that α cell membranes contain a large number of glucagon receptors (GR) [32][13]. Furthermore, a recent study reported that GR activation increases glucagon gene transcription via cAMP response element-binding (CREB) activation by the PKA-dependent pathway [40][14]. Moreover, several studies have reported that blocking GRs has been reported to improve glucose homeostasis [41,42[15][16][17],43], due to glucagon stimulates its own release acting on GR on α cells [44][18]. In fact, in a study conducted by Liu et al. on αTC1 cells (a pancreatic alpha cell line derived from an adenoma created in transgenic mice), immunofluorescent staining confirmed the presence of GRs on αTC1 cells. After 72 h of treatment with GRs antagonist (to block the effects of the endogenous glucagon), a 44% decrease in αTC1 cell proliferation was observed compared with the control group by counting the cells in the S phase. These results show that glucagon has direct trophic effects on α cells by an autocrine mechanism, and when the pancreatic α-cell number was decreased in db/db mice by a glucagon receptor antagonist, plasma glucagon levels were significantly decreased too [45][19].3.3. Juxtacrine Regulation
The juxtacrine mechanism is a common type of signalling between adjacent cells requiring direct contact between cells. Recently, a juxtacrine connection between α- and β-cells has been documented as another exciting way in glucagon secretion control since the confirmation of Eph/ephrin system between α- and β cells [51,52,53][20][21][22]. The Eph signalling system belongs to the superfamily of transmembrane Tyr kinase receptors. Today, it is well established that Eph allows short-distance cell-cell interaction by binding with their specific ligands (ephrin), primarily affecting cytoskeleton and leading to cell repulsion or adhesion in some circumstances. In this regard, the first studies in this field showed that many processes involving fast changes in cellular morphology were ephrin–Eph dependent [54][23]. More recently, other critical ephrin–Eph signalling-mediated processes have been identified and characterised, e.g., axon guidance, synaptic plasticity, cancer, and processes like juxtacrine hormones release control involving short-distance cell-cell communication. In addition, there is increasing evidence about its influence on cell differentiation, proliferation, and apoptosis regulation [55][24].3.4. Endocrine Regulation
Non-pancreatic hormones also have an active role in α cell regulation. For example, Bagger et al. [66][25] demonstrated that intravenous (IV) glucose administration did not reduce glucagon levels in subjects with T2DM; however, it was achieved with oral glucose administration, suggesting a potential role of intestine lining and incretins on α cell. In addition, GLP-1 and gastric inhibitory polypeptide (GIP) are hormones encoded by the GCG gene (the same encodes glucagon), secreted by enterocytes, with well-studied insulinotropic effects [67][26]. However, considering the difficulty of these experimental studies, its activity on α cells remains disputed [67,68][26][27]. The regulation of glucagon secretion relies on both intrinsic mechanisms in the α cell and extrinsic mechanisms in neighbouring and nearby cells since even autacoids like serotonin exhibit receptors in the α cell membrane inhibiting glucagon release. When α cells have a weak serotonergic tone, they lose their glucose-dependent inhibition of the glucagon-release, generating hyperglucagonemia under hyperglycaemic conditions.4. Hyperglucagonaemia and α Cell Dysfunction in Diabetes
Extensive emerging evidence has been published among the “bi-hormonal theory” in T2DM pathogenesis in which the coexistence of hyperglucagonemia and relative insulin deficiency increase gluconeogenesis and exacerbates peripheral IR [74][28], leading to overt T2DM development. Currently, there is some consensus regarding hyperglucagonemia origin. This is centred on two possible mechanisms: (1) a progressive loss in the regulatory mechanisms in the secretion patterns due to α cells functional alterations [75[29][30],76], or (2) modification in both the islet microarchitecture and cellularity [77,78,79,80][31][32][33][34].
4.1. Structural Alterations in Pancreatic Islet
Conclusive evidence has demonstrated a β cell mass reduction and a concomitant decrease in insulin secretion in subjects with long-standing T2DM [62,63][35][36] and a sensible fall in GABA and serotonin release, which are crucial paracrine regulators of glucagon secretion, as explained previously. However, post-mortem studies have reported an increased α cell mass in subjects with diabetes. Nonetheless, the evidence is not conclusive [63][36]. This finding can be related to a loss of α cell regulating factors or a compensatory mechanism secondary to β cells mass loss [63][36]. Nevertheless, multiple studies have reported that an elevation of Interleukin 6 (IL-6) circulating levels in T2DM adult mice is possibly linked with an expansion of α cell mass and hyperglucagonemia [64][37], suggesting that α cell proliferation in Type 1 Diabetes Mellitus (T1DM) is probably IL-6-dependent [81][38].
4.2. Alpha Pancreatic Cell Dysfunction, Energetic Sensors, and Ionic Channels
Dysfunction in glucose-mediated glucagon releasing mechanisms in α cells is extensively assessed in T2DM [106][39]. In this regard, Zhang et al. [107][40], using islets from T2DM organ donors, reported that glucose did not inhibit glucagon secretion. Furthermore, they replicated this methodology in non-diabetic samples by administering diazoxide, a well-known KATP opener, and oligomycin as an inhibitor of ATP production. Interestingly, in pancreatic islets treated with oligomycin, tolbutamide administration restored glucagon secretion, suggesting that DM hyperglucagonemia is linked to an impaired α cell KATP inhibition [107][40]. A recent in vivo study by Knudsen et al. [108][41] performed on mice lacking fumarase (Fh1) in their β and α cells developed spontaneous T2DM over 10 weeks of age secondary to an impairment of insulin secretion and found a lack of glucagon inhibition on hyper- glycaemic mice. They proposed that hyperglycaemia causes an increment of intracellular sodium in α-cell, secondary to the overactivity of SGLT in these cells. To overcome this, sodium antiporters, as Sodium-H+ transporters, are activated to reduce cellular sodium, conducing to significant cytoplasmic acidification. A subsequent reduction of mitochondrial matrix Ph occurred due to changes in cell electrochemical gradient, carrying an inhibition of oxidative phosphorylation, a decrease of ATP production, a decrease in ATP/ADP ratio, and a consequent opening of KATP, which culminate with an inappropriate glucagon secretion during a hyperglycaemic state. The researchers also hypothesised that mitochondrial and cytoplasmic acidification would lead to a reduction in fumarase activity [108,109][41][42], leading to an accumulation of fumarate in the cytoplasm. Thus, glycolysis may contribute to the worsening of the chaotic state of acidification and KATP activation in α cell during hyperglycaemia. Knudsen et al. [108][41] also set the inhibition of fumarase and disruption of ATP production may be found in other SGLT-expressing cells. It might be related, in part, to the kidney and cardiac failure observed in diabetes, which is also one of the plausible explanations for the cardiovascular risk and renal benefits of SLGT-1 inhibitors in T2DM patients [110,111][43][44]. Impaired Glucokinase (GCK) activity is another metabolic alteration linked to hyperglucagonemia in diabetes. Basco et al. [112][45] developed an in vivo study on GCK-deficient mice reporting an increase in post-prandial glucagon levels with hyper-glucagonemia, an increment in the liver gluconeogenic gene expression, and therefore, a hepatic glucose output increment. This fact may point to evidence of an underexpression/dysfunction of α-cell-GCK in T2DM, suggesting that GCK enhancers are a promissory therapy, not merely for their effects on insulin but also for their inhibitory effects on glucagon secretion. However, a recent trial with a novel GCK activator, HMS5552, did not report any significant improvement in glucagon levels [113][46]. Therefore, islet alterations (such as reduced β-cell mass) and α-cell alterations are typical phenomena of long-standing T2DM and possible transformation into undifferentiated cell forms or other islet cells through glucotoxicity. Otherwise, α cell KATP impairment is hypothesised as a hyperglucagonemia helper and lacks fumarase activity in DM.References
- Brown, A.E.; Walker, M. Genetics of Insulin Resistance and the Metabolic Syndrome. Curr. Cardiol. Rep. 2016, 18, 75.
- Petersen, M.C.; Shulman, G.I. Mechanisms of Insulin Action and Insulin Resistance. Physiol. Rev. 2018, 98, 2133–2223.
- Cantley, J.; Ashcroft, F.M. Q&A: Insulin secretion and type 2 diabetes: Why do β-cells fail? BMC Biol. 2015, 13, 33.
- Unger, R.H.; Orci, L. The essential role of glucagon in the pathogenesis of diabetes mellitus. Lancet 1975, 305, 14–16.
- Reaven, G.M.; Chen, Y.D.; Golay, A.; Swislocki, A.L.; Jaspan, J.B. Documentation of hyperglucagonemia throughout the day in nonobese and obese patients with noninsulin-dependent diabetes mellitus. J. Clin. Endocrinol. Metab. 1987, 64, 106–110.
- Lotfy, M.; Kalasz, H.; Szalai, G.; Singh, J.; Adeghate, E. Recent Progress in the Use of Glucagon and Glucagon Receptor Antagonists in the Treatment of Diabetes Mellitus. Open Med. Chem. J. 2014, 8, 28–35.
- Sandoval, D.A.; D’Alessio, D.A. Physiology of proglucagon peptides: Role of glucagon and GLP-1 in health and disease. Physiol. Rev. 2015, 95, 513–548.
- Wendt, A.; Eliasson, L. Pancreatic α-cells—The unsung heroes in islet function. Semin. Cell Dev. Biol. 2020, 103, 41–50.
- Bramswig, N.C.; Kaestner, K.H. Transcriptional regulation of α-cell differentiation. Diabetes Obes. Metab. 2011, 13 (Suppl. S1), 13–20.
- Singer, R.A.; Arnes, L.; Cui, Y.; Wang, J.; Gao, Y.; Guney, M.A.; Burnum-Johnson, K.E.; Rabadan, R.; Ansong, C.; Orr, G.; et al. The Long Noncoding RNA Paupar Modulates PAX6 Regulatory Activities to Promote Alpha Cell Development and Function. Cell Metab. 2019, 30, 1091–1106.e8.
- Gromada, J.; Franklin, I.; Wollheim, C.B. Alpha-cells of the endocrine pancreas: 35 years of research but the enigma remains. Endocr. Rev. 2007, 28, 84–116.
- Omar-Hmeadi, M.; Lund, P.-E.; Gandasi, N.R.; Tengholm, A.; Barg, S. Paracrine control of α-cell glucagon exocytosis is compromised in human type-2 diabetes. Nat. Commun. 2020, 11, 1896.
- Tian, J.; Dang, H.; Chen, Z.; Guan, A.; Jin, Y.; Atkinson, M.A.; Kaufman, D.L. γ-Aminobutyric acid regulates both the survival and replication of human β-cells. Diabetes 2013, 62, 3760–3765.
- Leibiger, B.; Moede, T.; Muhandiramlage, T.P.; Kaiser, D.; Vaca Sanchez, P.; Leibiger, I.B.; Berggren, P.-O. Glucagon regulates its own synthesis by autocrine signaling. Proc. Natl. Acad. Sci. USA 2012, 109, 20925–20930.
- Yan, H.; Gu, W.; Yang, J.; Bi, V.; Shen, Y.; Lee, E.; Winters, K.A.; Komorowski, R.; Zhang, C.; Patel, J.J.; et al. Fully human monoclonal antibodies antagonizing the glucagon receptor improve glucose homeostasis in mice and monkeys. J. Pharmacol. Exp. Ther. 2009, 329, 102–111.
- Li, X.C.; Zhuo, J.L. Targeting glucagon receptor signalling in treating metabolic syndrome and renal injury in Type 2 diabetes: Theory versus promise. Clin. Sci. 2007, 113, 183–193.
- Petersen, K.F.; Sullivan, J.T. Effects of a novel glucagon receptor antagonist (Bay 27-9955) on glucagon-stimulated glucose production in humans. Diabetologia 2001, 44, 2018–2024.
- Ma, X.; Zhang, Y.; Gromada, J.; Sewing, S.; Berggren, P.-O.; Buschard, K.; Salehi, A.; Vikman, J.; Rorsman, P.; Eliasson, L. Glucagon stimulates exocytosis in mouse and rat pancreatic alpha-cells by binding to glucagon receptors. Mol. Endocrinol. 2005, 19, 198–212.
- Liu, Z.; Kim, W.; Chen, Z.; Shin, Y.-K.; Carlson, O.D.; Fiori, J.L.; Xin, L.; Napora, J.K.; Short, R.; Odetunde, J.O.; et al. Insulin and glucagon regulate pancreatic α-cell proliferation. PLoS ONE 2011, 6, e16096.
- Hutchens, T.; Piston, D.W. EphA4 Receptor Forward Signaling Inhibits Glucagon Secretion From α-Cells. Diabetes 2015, 64, 3839–3851.
- Liu, W.; Kin, T.; Ho, S.; Dorrell, C.; Campbell, S.R.; Luo, P.; Chen, X. Abnormal regulation of glucagon secretion by human islet alpha cells in the absence of beta cells. EBioMedicine 2019, 50, 306–316.
- Reissaus, C.A.; Piston, D.W. Reestablishment of Glucose Inhibition of Glucagon Secretion in Small Pseudoislets. Diabetes 2017, 66, 960–969.
- Kania, A.; Klein, R. Mechanisms of ephrin–Eph signalling in development, physiology and disease. Nat. Rev. Mol. Cell Biol. 2016, 17, 240–256.
- Darling, T.K.; Lamb, T.J. Emerging Roles for Eph Receptors and Ephrin Ligands in Immunity. Front. Immunol. 2019, 10, 1473.
- Bagger, J.I.; Knop, F.K.; Lund, A.; Vestergaard, H.; Holst, J.J.; Vilsbøll, T. Impaired regulation of the incretin effect in patients with type 2 diabetes. J. Clin. Endocrinol. Metab. 2011, 96, 737–745.
- Piro, S.; Mascali, L.G.; Urbano, F.; Filippello, A.; Malaguarnera, R.; Calanna, S.; Rabuazzo, A.M.; Purrello, F. Chronic exposure to GLP-1 increases GLP-1 synthesis and release in a pancreatic alpha cell line (α-TC1): Evidence of a direct effect of GLP-1 on pancreatic alpha cells. PLoS ONE 2014, 9, e90093.
- Holst, J.J.; Christensen, M.; Lund, A.; de Heer, J.; Svendsen, B.; Kielgast, U.; Knop, F.K. Regulation of glucagon secretion by incretins. Diabetes Obes. Metab. 2011, 13 (Suppl. S1), 89–94.
- Patarrão, R.S.; Lautt, W.W.; Macedo, M.P. Acute glucagon induces postprandial peripheral insulin resistance. PLoS ONE 2015, 10, e0127221.
- Moon, J.S.; Won, K.C. Pancreatic α-Cell Dysfunction in Type 2 Diabetes: Old Kids on the Block. Diabetes Metab. J. 2015, 39, 1–9.
- D’Alessio, D. The role of dysregulated glucagon secretion in type 2 diabetes. Diabetes Obes. Metab. 2011, 13, 126–132.
- Campbell-Thompson, M.; Tang, S.-C. Pancreas Optical Clearing and 3-D Microscopy in Health and Diabetes. Front. Endocrinol. 2021, 12, 644826.
- Masini, M.; Martino, L.; Marselli, L.; Bugliani, M.; Boggi, U.; Filipponi, F.; Marchetti, P.; De Tata, V. Ultrastructural alterations of pancreatic beta cells in human diabetes mellitus. Diabetes Metab. Res. Rev. 2017, 33, e2894.
- Willcox, A.; Gillespie, K.M. Histology of Type 1 Diabetes Pancreas. In Type-1 Diabetes; Gillespie, K.M., Ed.; Methods in Molecular Biology; Springer: New York, NY, USA, 2015; Volume 1433, pp. 105–117. ISBN 978-1-4939-3641-0.
- Mateus Gonçalves, L.; Almaça, J. Functional Characterization of the Human Islet Microvasculature Using Living Pancreas Slices. Front. Endocrinol. 2021, 11, 602519.
- Rojas, J.; Bermudez, V.; Palmar, J.; Martínez, M.S.; Olivar, L.C.; Nava, M.; Tomey, D.; Rojas, M.; Salazar, J.; Garicano, C.; et al. Pancreatic Beta Cell Death: Novel Potential Mechanisms in Diabetes Therapy. J. Diabetes Res. 2018, 2018, 9601801.
- Mizukami, H.; Takahashi, K.; Inaba, W.; Tsuboi, K.; Osonoi, S.; Yoshida, T.; Yagihashi, S. Involvement of oxidative stress-induced DNA damage, endoplasmic reticulum stress, and autophagy deficits in the decline of β-cell mass in Japanese type 2 diabetic patients. Diabetes Care 2014, 37, 1966–1974.
- Ellingsgaard, H.; Ehses, J.A.; Hammar, E.B.; Van Lommel, L.; Quintens, R.; Martens, G.; Kerr-Conte, J.; Pattou, F.; Berney, T.; Pipeleers, D.; et al. Interleukin-6 regulates pancreatic alpha-cell mass expansion. Proc. Natl. Acad. Sci. USA 2008, 105, 13163–13168.
- Cai, Y.; Yuchi, Y.; De Groef, S.; Coppens, V.; Leuckx, G.; Baeyens, L.; Van de Casteele, M.; Heimberg, H. IL-6-dependent proliferation of alpha cells in mice with partial pancreatic-duct ligation. Diabetologia 2014, 57, 1420–1427.
- Hamilton, A.; Zhang, Q.; Salehi, A.; Willems, M.; Knudsen, J.G.; Ringgaard, A.K.; Chapman, C.E.; Gonzalez-Alvarez, A.; Surdo, N.C.; Zaccolo, M.; et al. Adrenaline Stimulates Glucagon Secretion by Tpc2-Dependent Ca2+ Mobilization from Acidic Stores in Pancreatic α-Cells. Diabetes 2018, 67, 1128–1139.
- Zhang, Q.; Ramracheya, R.; Lahmann, C.; Tarasov, A.; Bengtsson, M.; Braha, O.; Braun, M.; Brereton, M.; Collins, S.; Galvanovskis, J.; et al. Role of KATP channels in glucose-regulated glucagon secretion and impaired counterregulation in type 2 diabetes. Cell Metab. 2013, 18, 871–882.
- Knudsen, J.G.; Hamilton, A.; Ramracheya, R.; Tarasov, A.I.; Brereton, M.; Haythorne, E.; Chibalina, M.V.; Spégel, P.; Mulder, H.; Zhang, Q.; et al. Dysregulation of Glucagon Secretion by Hyperglycemia-Induced Sodium-Dependent Reduction of ATP Production. Cell Metab. 2019, 29, 430–442.e4.
- Adam, J.; Ramracheya, R.; Chibalina, M.V.; Ternette, N.; Hamilton, A.; Tarasov, A.I.; Zhang, Q.; Rebelato, E.; Rorsman, N.J.G.; Martín-Del-Río, R.; et al. Fumarate Hydratase Deletion in Pancreatic β Cells Leads to Progressive Diabetes. Cell Rep. 2017, 20, 3135–3148.
- Pareek, A.; Chandurkar, N.; Naidu, K. Empagliflozin and Progression of Kidney Disease in Type 2 Diabetes. N. Engl. J. Med. 2016, 375, 1800.
- Zinman, B.; Wanner, C.; Lachin, J.M.; Fitchett, D.; Bluhmki, E.; Hantel, S.; Mattheus, M.; Devins, T.; Johansen, O.E.; Woerle, H.J.; et al. Empagliflozin, Cardiovascular Outcomes, and Mortality in Type 2 Diabetes. N. Engl. J. Med. 2015, 373, 2117–2128.
- Basco, D.; Zhang, Q.; Salehi, A.; Tarasov, A.; Dolci, W.; Herrera, P.; Spiliotis, I.; Berney, X.; Tarussio, D.; Rorsman, P.; et al. α-cell glucokinase suppresses glucose-regulated glucagon secretion. Nat. Commun. 2018, 9, 546.
- Wang, P.; Liu, H.; Chen, L.; Duan, Y.; Chen, Q.; Xi, S. Effects of a Novel Glucokinase Activator, HMS5552, on Glucose Metabolism in a Rat Model of Type 2 Diabetes Mellitus. J. Diabetes Res. 2017, 2017, 1–9.
