Inflammatory bowel disease (IBD) is a chronic autoimmune condition affecting the gastrointestinal (GI) tract. IBD includes Crohn’s disease (CD) and ulcerative colitis (UC), with UC characterized by inflammation of colonic mucosa and submucosa starting at the rectum and extending through the colon. The precise etiology of UC is unknown but is thought to involve a combination of environmental and genetic factors. Chief among these is the intestinal microbiome, which has been extensively studied both for its role in disease pathogenesis and possible treatment. In this review, we will discuss the microbial changes that have been described in UC, its interplay with host immune function, and evidence supporting its role as a potential therapeutic. We will also discuss parallels between UC, the microbiome and colitis-associated cancer (CAC).
- ulcerative colitis
- UC
- inflammatory bowel diseases
- IBD
- pediatrics
- paediatrics
- probiotics
- prebiotics
- synbiotics
- antibiotics
- fecal microbiota transplant
- faecal microbiota transplant
- FMT
- colitis-associated cancer
- CAC
- colorectal cancer
- CRC
- dysbiosis
1. Background
2. Microbiome-Immune Interactions in UC
2.1. Immune System Perturbations in UC
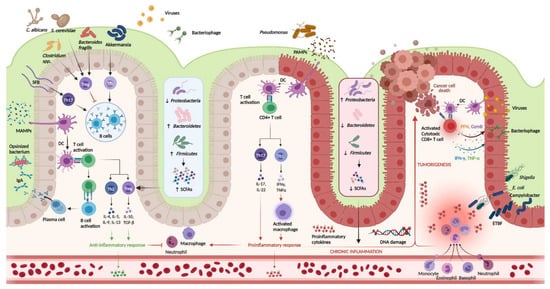
FIGURE 1. Host-igure 1mmune interactions in ulcerative colitis. Host-immune interactions in ulcerative colitis. Intestinal microbiota interact with the immune system through various pathways. In the healthy colon, DCs sample MAMPs and present antigens on major histocompatibility complex class II to naive CD4+ T cells. Naive CD4+ T cells become activated and differentiate into various T cell subtypes depending on the presence of specific cytokines within the local microenvironment. Anti-inflammatory Th subtypes comprise Th2 and Treg cells. CD4+ T cells also activate plasma cells which secrete immunoglobulin A (IgA) which is essential for microbial opsonization. Proinflammatory Th subtypes consist of Th1 cells and Th17 cells, which are upregulated in the diseased colon via interactions between DCs and PAMPs. Chronic inflammation contributes to DNA damage and tumorigenesis. Invading viruses stimulate CD8+ cytotoxic T cell activation via antigen-MHC I interactions. However, CD8+ T cells can also assist in cancer cell death. Disruptions in the mucosal barrier provides avenues for microbial translocation, including ETBF, which has been implicated in colitis-associated cancer. Finally, the production of SCFA is increased in the healthy colon (mediated by increased density of Firmicutes and Bacteroidetes phyla), while increased density of the Proteobacterium phylum is associated with lower concentrations of SCFA and colonic inflammation. DC, dendritic cell; DNA, deoxyribonucleic acid; ETBF, enterotoxigenic
Intestinal microbiota interact with the immune system through various pathways. In the healthy colon, DCs sample MAMPs and present antigens on major histocompatibility complex class II to naive CD4+ T cells. Naive CD4+ T cells become activated and differentiate into various T cell subtypes depending on the presence of specific cytokines within the local microenvironment. Anti-inflammatory Th subtypes comprise Th2 and Treg cells. CD4+ T cells also activate plasma cells which secrete immunoglobulin A (IgA) which is essential for microbial opsonization. Proinflammatory Th subtypes consist of Th1 cells and Th17 cells, which are upregulated in the diseased colon via interactions between DCs and PAMPs. Chronic inflammation contributes to DNA damage and tumorigenesis. Invading viruses stimulate CD8+ cytotoxic T cell activation via antigen-MHC I interactions. However, CD8+ T cells can also assist in cancer cell death. Disruptions in the mucosal barrier provides avenues for microbial translocation, including ETBF, which has been implicated in colitis-associated cancer. Finally, the production of SCFA is increased in the healthy colon (mediated by increased density of Firmicutes and Bacteroidetes phyla), while increased density of the Proteobacterium phylum is associated with lower concentrations of SCFA and colonic inflammation. DC, dendritic cell; DNA, deoxyribonucleic acid; ETBF, enterotoxigenic
Bacteroides fragilis
; IFN-γ, interferon-gamma; IgA, immunoglobulin A; MAMPs, microbe-associated molecular patterns; PAMPs, pathogen-associated molecular patterns; SCFAs, short chain fatty acids; SFB, segmented filamentous bacteria; Th, T helper; Treg, T regulatory; TNF-α, tumor necrosis factor-alpha. Created in
Biorender.com (accessed: 1 August 2021) [9].
(accessed: 1 August 2021) [16].
2.1.1. Physical Barrier
2.1.2. Immunoglobulin A
2.1.3. Innate and Adaptive Immunity
2.2. Intestinal Microbiota Composition in Ulcerative Colitis
TABLE 1. Intestinal microbiota alterations in ulcerative colitis and impacts on host immune, intestinal function.
| Gut Microbiota Alterations in UC | Consequences for Mammalian Host Health | ||||
|---|---|---|---|---|---|
| Life Domain | Taxonomic Classification | Compositional Changes of Gut Microbiota | Functional Changes of Gut Microbiota | Impact on Host Immune Function |
Impact on Host Intestinal Function |
| Bacteria | Phyla | 16S ribosomal RNA gene sequencing ↓α- diversity in UC as compared with HC [63][71][70,78] ↑β-diversity in UC (UC bacteriome clusters differently form HC) [63][71][70,78] ↓relative abundance of Firmicutes and Bacteroidetes [13][63][64][67][74][20,70,71,74,81] |
shotgun metagenomics sequencing ↑l-arginine biosynthesis (I, IV), biotin biosynthesis II, transfer RNA charging [71][78] Super pathway of polyamine biosynthesis in patients with risk factors for developing UC as compared with HC [71][78] ↑amino acid and protein metabolism (in UC as compared with HC): l-lysine fermentation to acetate and butanoate, creatinine degradation II, ketogenesis, protein N-glycosylation [70][77] ↑proteolytic and elastase activity in pre- and post-UC as compared with HC Correlated with the protease-producing bacterial species altered in UC- Proteobacteria and Bacteroides-↑elastase from B. vulgatus) [71][78] ↓glycerol and glycerophospholipids in UC as compared with HC Positive correlation between bacterial species and carbohydrate-degradation pathways [75][82] |
Ruminococcus, Eubacterium, Roseburia, and Akkermansia, Anaerostipes hadrus ↓butyrate production = ↓Treg cells differentiation ↓maturation of Treg cells in the colonic epithelium increased levels of proinflammatory cytokines [64][65][71][72][76][71,72,78,79,83] Enterobacteriaceae ↑colonic epithelial cells invasion ↑levels of proinflammatory cytokine IL-8 and TNF-α [77][84] Fusobacteria ↑tumorigenesis in the colon [65][72] Faecalibacterium prausnitzii ↑production of IL-12, IFNγ and reduction of IL-10 levels in blood cells [78][85] Adlercreutzia ↓synthesis of isoflavones, phenolic compounds with antimicrobial and anti-inflammatory properties [71][78] |
Ruminococcus bromii, Eubacterium rectale, Roseburia, and Akkermansia ↓butyrate production = impaired epithelial barrier function ↑epithelial permeability and commensals translocation [13][64][65][20,71,72] ↑colonic inflammation with crypt abscess [77][84] ↑of deciduous epithelial and/or blood cells in stools of patients with UC or CAC, gut barrier injury, impaired cell cycle [75][82] |
| ↑Proteobacteria [13][63][64][65][68][20,70,71,72,75] | |||||
| Families | ↓Clostridiaceae [64][65][71,72] ↑Enterobacteriaceae [79][86] |
||||
| Genera | ↓Clostridium clusters IV, XIVa [65][72] ↓Ruminococcus, Eubacterium, Roseburia, Akkermansia [64][71][71,78] ↓Adlercreutzia, Bilophila, Bifidobacterium [71][78] ↓Bacteroides, Lachnospira, Phascolarctobacterium, Coprococcus, Odoribacter, Butyricimonas [68][79][75,86] |
||||
| ↑Escherichia-Shigella, Fusobacterium, Campylobacter, Helicobacter [64][68][71][71,75,78] ↑Actinobacillus [71][78] ↑Streptococcus, Anaerostipes Enterococcus, Actyinomyces, Lactobacillus, Acetobacter, Rothia, Pseudomonas, Collinsella [68][75] |
|||||
| Species | ↓Faecalibacterium prausnitzii [65][76][80][72,83,87] ↓Anaerostipes hadrus [72][79] ↑Flavonifractor plautii, Coprococcus catus, Parabacteroides merdae [71][78] |
||||
| Fungi | Phyla | Stool ITS2 gene sequencing ↓α-diversity in UC (not in CD) [63][70] ↑β-diversity between UC in flare as compared with UC in remission and to HC [63][70] ↑ ratio of Basidiomycota/Ascomycota in UC in flare as compared with UC in remission and to HC [63][70] ↑correlation between fungi and bacteria in UC as compared with CD and HC [63][70] Colonic mucosa: ↓fungi load in UC as compared with HC No significant changes in α-diversity UC mycobiota clusters differently from HC No changes in the ratio of Basidiomycota/Ascomycota [81][88] |
N/A | Saccharomyces cerevisiae and Candida Albicans = ↑IL-6 production [63][70] ↓Saccharomyces cerevisiae = ↓IL-10 production (anti-inflammatory cytokine) [63][70] Aspergillus ↑aflatoxin production, a carcinogenic mycotoxin [81][88] Positive correlation between Wickerhamomyces and Penicillium with the expression of TNF-α and IL-17A, respectively (in colonic mucosa) [81][88] Negative correlation between Sporobolomyces and IL-6 and between Trametes and IL-1β (in colonic mucosa) [81][88] |
Aspergillus Potential for aspergillosis, with consequent abdominal pain and GI bleeding [81][88] |
| Genera | ↓Saccharomyces in UC fecal samples [63][70] ↑Aspergillus in UC mucosa specimen [81][88] |
||||
| Species | ↓Saccharomyces cerevisiae in UC fecal samples [63][70] ↑Candida albicans in UC fecal samples [63][70] Trend toward an increase in mucosal specimen [82][89] |
||||
| Virus | Orders | Metagenomics sequencing of viral-like particles ↓α-diversity (virome species richness and evenness) in UC mucosal samples [83][90] ↑abundance Caudovirales bacteriophages in UC mucosal samples [83][90] ↑β-diversity; UC mucosal virome clusters differently from HC [83][90] ↑virome dissimilarity between UC subjects (not observed in HC subjects) [83][90] |
↓integral component of membrane, DNA binding, ATP-binding cassette (ABC) transporter and integrase core domain in UC as compared with HC [83][90] ↑Pathways related to the phage lysis of bacteria: DNA template negative regulation of transcription, beta-lactamase, glutamine amidotransferase, glycosal hydrolases, type II/IV secretion system and multicopper oxidase in UC as compared with HC [83][90] |
↑bacteriophage = ↑bacterial lysis, PAMPs production, TLRs overstimulation, ↑intestinal inflammation [83][90] ↑transfer of bacterial genetic material (i.e., antibiotic resistance genes) [83][90] ↑phages can stimulate IFN-γ via the nucleotide-sensing receptor TLR9 [84][91] |
↑bacteriophages = ↑bacterial lysis, ↑intestinal inflammation, potential implication in abdominal pain, diarrhea [83][84][90,91] |
| Families | ↓Anelloviridae (eukaryotic virus) [83][90] ↑Microviridae (single-stranded DNA phage), Myoviridae, Podoviridae (double-stranded DNA phages) [83][90] Pneumoviridae (eukaryotic virus) [83][90] |
||||
| Genera | ↓Coccolithovirus, Minivirus Orthopoxvirus (vertebrate-infecting virus) (all eukaryotic viruses) [83][90] ↑Phix174microvirus, P1virus, Lambdavirus, T4virus, P22virus (all Caudovirales bacteriophages) Orthopneumovirus [83][90] |
||||
| Species | ↓α-diversity of Caudovirales species in UC mucosal samples [83][90] ↑Escherichia and Enterobacteria bacteriophages [83][90] Lactobacillus, Escherichia, and Bacteroides bacteriophages [84][91] |
||||
