Physical activity among older adults has multiple benefits but is first essential to determine the recommended activity. Physical activity may reduce inflammation and oxidative stress through multiple mechanisms, including the reduced formation of ROS and the accelerated production of DNA-repairing enzymes and antioxidant proteins; furthermore, it can moderate oxidative stress by decreasing TNF-α, C-reactive protein, and interleukin-6. There is conflicting evidence on the effect of physical activity and hormone levels, depending on the kind and length of activity, which can increase the levels of dehydroepiandrosterone and testosterone, but there is no clear evidence.
- aging
- hormones changes
- physical activity
- the Mediterranean diet
1. Introduction
In daily practice with outpatients or inpatients, it is pretty common to find a growing number of multipathological, polymedicated, and frail elderly patients; it could be said that this is the rule [1]. Therefore, if there is agreement that aging successfully is the goal to follow, the fundamental question will become how to achieve it. Knowing this reality, it can be of great help in the discussions with our patients and their families (the criteria of the original definition by Rowe and Kahn) [2] to consider what defines successful aging, that is: (1) low probability disease and associated disability; (2) elevated cognitive and physical functioning; and (3) being actively engaged in life. Therefore, the critical question will be how this could be achieved.
Increasing life expectancy is celebrated as one of humanity’s most outstanding successes. However, the other side of the coin is that aging is the leading risk factor for the most common chronic non-communicable diseases (cardiovascular diseases, strokes, dementia, cancers, among others). In other words, the advance in life expectancy was not accompanied by a similar improvement in health expectancy. Consequently, over the past decades, the gain in life years has been followed by additional years of chronic and poor health. As a result, the most significant proportion of the total health care budget expenditure is now concentrated in the last years of life [3].
The population’s aging is a global, medical, and sociodemographic problem. In response to this, the World Health Organization (WHO) promotes healthy aging, a concept that encompasses maintaining the functional capacity to achieve well-being in old age and operates through the Decade of Healthy Aging 2020–2030 program [4].
This entry will address the metabolic, hormonal, and pathophysiological changes that age brings and the benefits of the Mediterranean diet and physical exercise for active and successful aging to add life to the years ahead.
2. Endocrine Changes in the Elderly Subject
Age-related hormonal changes occur in human physiology and have been well described; however, how these changes impact health and/or disease is not yet completely known.
Like the ones involved in nutrition, reproduction, growth, and metabolism, most endocrine axes show a decrease in hormonal secretion with aging. Also, a reduction in tissues sensitivity to their action (e.g., insulin resistance) changes circadian rhythms and biological availability (e.g., sex hormones, corticosteroids). It is responsible for the clinical manifestations identified in this population [5].
Interestingly, these complex alterations in hormonal networks are related to the decline in physiologic functions and the onset of chronic diseases such as frailty, sarcopenia, osteoporosis, obesity, hypertension, diabetes, hyperlipidemia, atherosclerosis, and immune functions reduction. (Table 1) Additionally, these endocrine alterations are prevalent in patients that develop degenerative brain disease with cognitive impairment and a general decline in brain function [6].
3.Chronic Inflammation and Oxidative Stress in the Elderly Subject
The homeostatic loss and misfunctioning systems result from injury caused by the high reactivity of the free radicals and reactive oxygen species (ROS) produced on the biomolecules of these cells. The mitochondria DNA (mtDNA) is the first target of this oxidation. Because of this damage, the post-mitotic cells could not repair nor regenerate these “broken” organelles [7]. Also, age epigenetic changes include alterations in DNA methylation patterns, post-translational alteration of histones, and chromatin remodeling [8].
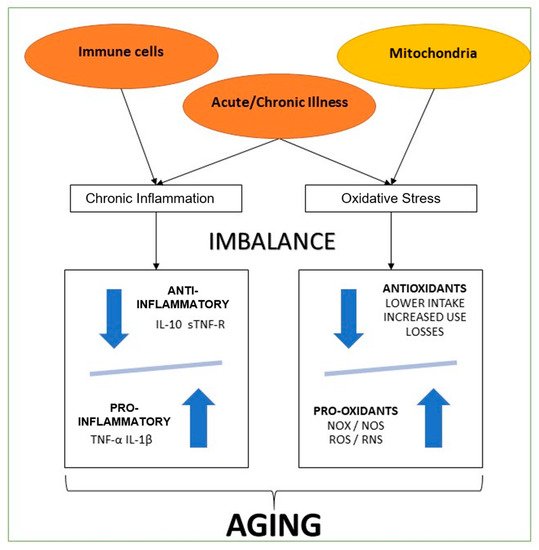
As we had explained before, an essential part of the aging process is a chronic imbalance between pro-inflammatory and anti-inflammatory networks [8]. In several studies, levels of pro-inflammatory mediators were high in the elderly, even in the absence of acute inflammatory response (e.g., acute infection) or other systemic stress events. This chronic inflammation situation is common in many aging-related conditions. Thus, inflammatory and oxidative stress leads to severe injury of cellular components (e.g., proteins, lipids, and DNA), contributing to the age-related decline of physiological capacities. This process is more evident in cells from different systems (nervous, endocrine, and immune). It undoubtedly describes their functional losses observed during aging, resulting in higher morbidity and mortality [8]. It is necessary to highlight that physiological organismal, and cellular integrity loss are the primary risk factor for several pathologies, such as cancer, T2D, CVD, and the well-known metabolic syndrome [8]. A continuing accumulation of impairment originated from oxidative stress, like glycated products, oxidized proteins, and lipid peroxidation, leading to neurons' degeneration, which is frequently found in neurological diseases. Vascular lesions also characterize cerebrovascular diseases, which are recognized for cognitive decline and dementia in old age. Moreover, in brain tissue cells, ROS are produced by microglia and astrocytes and might lead to neuroinflammation and cell death, triggering neurodegeneration and memory loss [9].
C-reactive protein activates anti-inflammatory cytokines in circulating monocytes and suppressing pro-inflammatory cytokines in tissue macrophages. The primary cytokines in this inflammatory cascade are tumor necrosis factor α (TNF-α) and interleukin 1β (IL-1β) [10], which are considered pro-inflammatory cytokines. TNF-α and IL-1β stimulate interleukin 6 (IL-6) generation, which has both actions a pro-inflammatory and an anti-inflammatory cytokine. IL-6 blocks the production of TNF-α and IL-1β, inducing the discharge of soluble TNF-α receptors (sTNF-R), and might be the main inducer of hepatocyte-derived acute-phase proteins, several of which have anti-inflammatory qualities. TNF-α and IL-1β represent classic pro-inflammatory cytokines, although sTNF-R and interleukin 10 are anti-inflammatory cytokines [11].
As we can see, multiple systems are affected by chronic inflammatory states and oxidate imbalance ( Figure Figure 1 1 ). So, they are key factors in understanding aging. This knowledge is also crucial to be considered a potential intervention measure in future therapies for these inflammatory and oxidative states [12].
4. Metabolic and Physical Performance Decline of Aging-Related to Hormone Changes and Lifestyle Changes
4.1. Physical Activity in the Elderly
Physical activity among older adults has multiple benefits but is first essential to determine the recommended activity. Physical activity may reduce inflammation and oxidative stress through multiple mechanisms, including the reduced formation of ROS and the accelerated production of DNA-repairing enzymes and antioxidant proteins; furthermore, it can moderate oxidative stress by decreasing TNF-α, C-reactive protein, and interleukin-6 [13][14][15]. There is conflicting evidence on the effect of physical activity and hormone levels, depending on the kind and length of activity, which can increase the levels of dehydroepiandrosterone and testosterone, but there is no clear evidence. Moreover, growth hormone is increased in older persons in response to short-term exercise. However, in the postexercise recovery period, the reversal of this hormone back to resting values occurs quicker in older individuals than in younger individuals, and the increase during exercise appears to be dependent on exercise intensity [6]. In more recent years, the beneficial effect of exposure to low-grade potentially damaging conditions or very low doses of toxic compounds has been conceptualized as “preconditioning” and “hormesis” [2][16]. In this respect, healthy nutrition, mainly as given by the Mediterranean diet and planned physical exercises, are determinant resources to deeply modify this systemic inflammatory balance on a long-term scale by decreasing the age-related increment of inflammatory molecules and by promoting adaptive anti-inflammatory changes [2][16][3][17].
The positive effect of physical exercises among the elderly is not only shown to improve physical status. In a meta-analysis with people diagnosed with Alzheimer's disease where 13 controlled trials were analyzed, it was revealed that physical activity improved the Mini-Mental State Examination (Standard Media Deviation = 1.12; CI = 0.66–1.59; p = 0.000) [18]. In a cross-sectional study with 2345 healthy participants, physical activity was related to life satisfaction (measured with a single question) and happiness (also measured with a simple question) [19]. Also, in the same study, satisfaction and happiness associated with physical activity increased [19]. Interestingly in this study, most of the older adults had high active physical activity (44.01%), and having this amount of activity was a predictor of life satisfaction and happiness [19]. In a 2016 survey, 1504 elders aged 65 to 80 years with or without disabilities were assessed for stress and depression using the Patient Health Questionnaire-9 and quality of life using the EuroQol-5 Dimension [20]. Patients without disabilities had more physical activity, and stress and anxiety/depression levels were higher in those with disabilities [20]. In an experimental randomized controlled study, patients were interviewed before and after a physical activity program. There was a decrease in the depression scale and in eight sub-categories and two aspects of the SF 36 Quality of Life Questionnaire [21]. Additionally, there are reports where regular exercises diminish the probability of developing chronic conditions like CVD, T2D, and some cancers [21][22]. In the same survey, in 6933 adults, it was observed that there were 0.32% longer leukocyte telomeres with an increment of one hour per week [23].
Finally, in a cross-sectional study between 1999–2002 (NHANES) including 5823 adults, the leukocyte telomere length was calculated [24]. It was shown that telomeres were 15.6 base pairs smaller for every year of chronological age. Adults with high activity had higher telomere base pairs than those with moderate, low, or sedentary activity [24].
Getting older adults to do physical activity is not easy, and there are multiple barriers described in the literature for them to do so. A systematic review observed that the most critical barriers included physical problems, time limits, fear of falling, lack of interest or motivation, physical limitations to walking, pain, and exercising alone [25].
4.2. Mediterranean Diet in the Elderly
The Mediterranean diet (MD) includes legumes, vegetables, fruits, whole grains, seeds, olive oil, and fish and also involves low meat ingestion and regular drinking of wine [26]. These foods contain vitamins E and B, oleic acid, polyphenolic, and Omega-3 fatty acids [26]. According to the components of MD, in the analysis of two large studies, poultry, fish, dairy, vegetables, fruit, potatoes, cereals, olive oil, and alcohol had a positive association with the Successful Aging Index, whereas meat had an inverse association with successful aging [15][27]. Adherence to this kind of diet has multiple benefits for the elderly.
This kind of diet seems to modulate inflammation. It has been evidenced that patients with low adherence to MD had prolonged length of stay, higher levels of inflammation markers, interleukin-6, and tumor necrosis alpha [28]. Adherence to MD also has effects on mortality. In a meta-analysis that involved 30 studies where the association between adherence to MD and mortality was studied, it was shown that mortality was inversely related to alcohol ingestion, fruit, and vegetable consumption. In contrast, a positive association between meat consumption and mortality was evidenced [29].
Also, MD has positive effects on cognition and dementia. Several studies have demonstrated that greater adherence to MD was related to less chance of mental distress [30][31][32]. Besides the positive effect on cognition and dementia, the MD seems to affect neurodegenerative diseases positively [33][34][35].
Additionally, it seems to have a positive association with quality of life [36]. Moreover, MD has cardiovascular effects. A meta-analysis evidenced that adherence to this diet reduced systolic and diastolic blood pressure levels in individuals with normal arterial pressure or mild hypertension [37]. Moreover, it was shown that a higher level of adherence to the MD is associated with a higher level of successful aging, and this effect is independent of age, sex, smoking habits, cognitive function, and inflammation levels [38].
5.Practical Recommendations on Physical Activity and the Mediterranean Diet in the Elderly
Elevating the role of nutrition and exercise at the individual and population levels is central to keeping our lives healthy and addressing issues of global concern to an aging society. Prevention is ultimately more cost-effective than treating preventable chronic diseases. Although it is never too late, prevention begins long before one becomes an older adult. Healthy eating and regular physical exercise are among the most cost-effective, accessible, and efficient interventions capable of preventing and managing the burden of chronic diseases, reducing overall mortality, and promoting healthy aging [4].
The “Pyramid of the MD”, a worldwide recognized nutritional guide, summarizes the recommendations and characteristics of the Mediterranean diet. Foods at the pyramid base should be eaten more frequently and in larger portions than those in higher tiers. The approximate distribution of macronutrients is 55% to 60% carbohydrates, with less than 10% simple sugars, 10%to 15% proteins, and 25% to 30% fats (mainly monounsaturated (MUFA) and polyunsaturated (PUFA), with extra virgin olive oil being the primary source). The MD pyramid is not only a balanced way of eating, which shows indications on the proportions and frequencies of food consumption, but also implies and highlights the elements of coexistence, both ecological and cultural, without forgetting the importance of drinking an adequate amount of water and undertaking physical activity [39].
Although the MD represents the general dietary and lifestyle patterns of a small proportion of the world’s population, some of the basic principles that shape it, such as the preference for local and seasonal foods, the daily consumption of vegetables, fruits, whole grains, and healthy fats can be applied and adapted to other territories and cultures. This flexibility has led to its promotion in regions and its addition to dietary guidelines of countries far from its geographical origin [40].
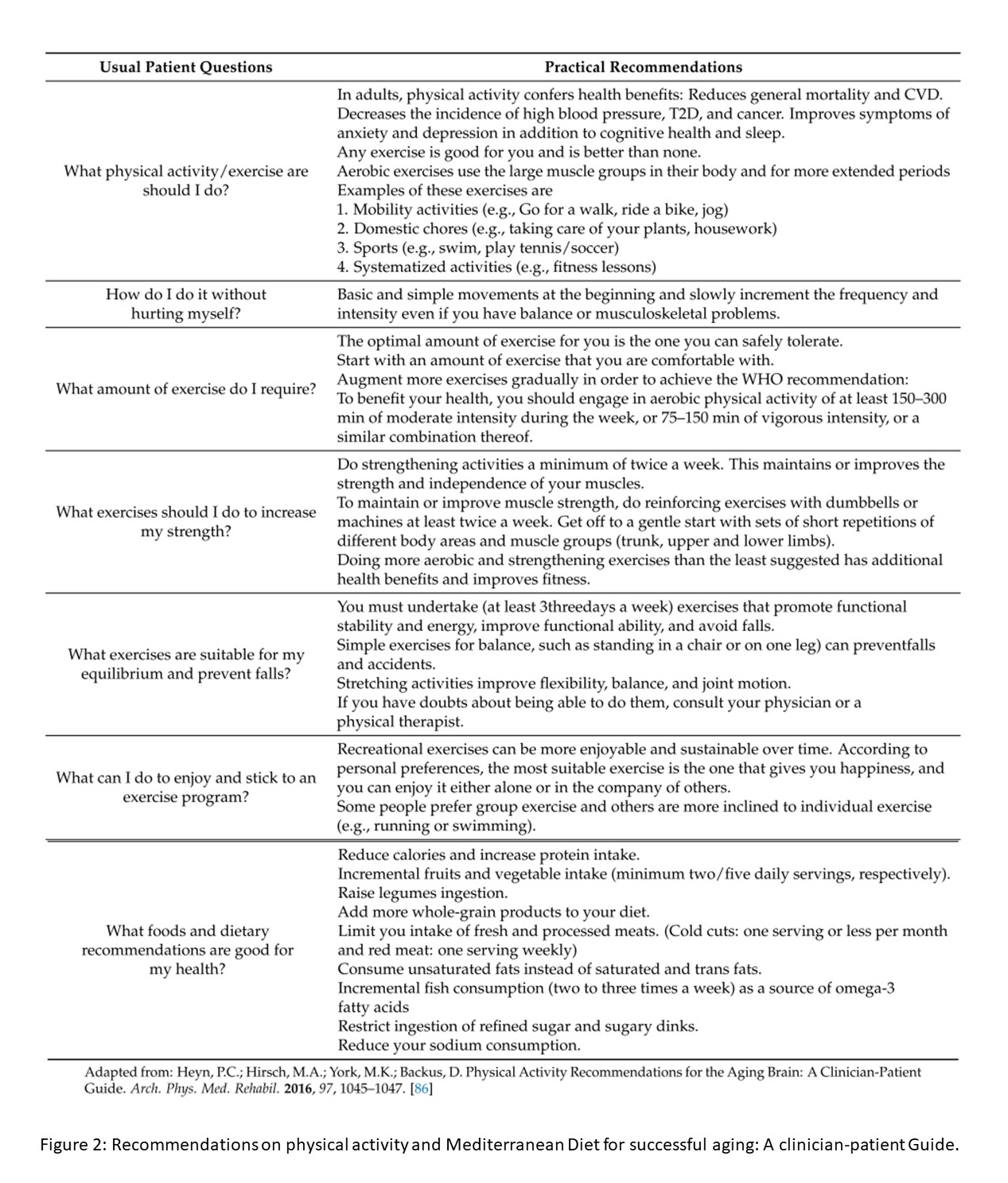
The “Global Recommendations on Physical Activity for Health” were promoted by WHO in 2010 to achieve greater physical activity levels to reduce obesity and chronic diseases. These recommendations have recently been revised. This document added specific instructions for physical activity for people with chronic diseases or disabilities to the suggested physical activity levels for three age groups (5 to 17, 18 to 64, and over 65 years of age). They are based on solid scientific evidence that shows a strong relationship between the characteristics (type, duration, and intensity) of physical activity and health outcomes [41]. From a practical perspective of the usual dialogue in the consultation, the recommendations of physical activity and MD are summarized in Figure 2 [42].
6. Conclusions
Aging can be a very challenging stage of life for patients and physicians. However, all of these expected changes are better controlled with early management and prevention of the common complications and health issues related to the aging process.
This entreviewy proposed practical recommendations on physical activity and the MD in the elderly. It very specifically presented safe and effective exercises that will contribute to healthier aging. In this respect, the MD has demonstrated to be superior to other diets with regard to preventing and managing common chronic diseases in aged patients.
References
- Dino Moretti; Martin G Buncuga; Carlos D Laudanno; Nadia D Quiñones; Carlos M Scolari Pasinato; Francisco E Rossi; [PROFUND index and global subjective assessment. Prognostic value in hospitalized pluripathological patients].. null 2019, 80, 622-632.
- Rowe J.W., Kahn R.L.,; Successful Aging. Gerontologist 1997, 37, 433-440.
- Manfred Ruthsatz; Vanessa Candeias; Non-communicable disease prevention, nutrition and aging. Acta bio-medica : Atenei Parmensis 2020, 91, 379-388.
- Ewa Rudnicka; Paulina Napierała; Agnieszka Podfigurna; Błażej Męczekalski; Roman Smolarczyk; Monika Grymowicz; The World Health Organization (WHO) approach to healthy ageing. Maturitas 2020, 139, 6-11, 10.1016/j.maturitas.2020.05.018.
- Diamanti-Kandarakis E., Dattilo M., Macut D., Duntas L., Gonos E.S., Goulis D., Gantenbein C.K., Kapetanou M., Koukkou E., Lambrioudaki; MECHANISMS IN ENDOCRINOLOGY: Aging and anti-aging: A Combo-Endocrinology overview.. Eur. J. Endocrinol 2017, 176, R283-R308.
- Mark W. Pataky; William F. Young; K. Sreekumaran Nair; Hormonal and Metabolic Changes of Aging and the Influence of Lifestyle Modifications. Mayo Clinic Proceedings 2021, 96, 788-814, 10.1016/j.mayocp.2020.07.033.
- Monica Fuente; Jaime Miquel; An Update of the Oxidation-Inflammation Theory of Aging: The Involvement of the Immune System in Oxi-Inflamm-Aging. Current Pharmaceutical Design 2009, 15, 3003-3026, 10.2174/138161209789058110.
- Fuente M., Miquel J.,; From inflammaging to healthy aging by dietary lifestyle choices: Is epigenetics the key to personalized nutrition?. Curr. Phar. Des. 2009, 15, 3003-3026.
- Marília Baierle; Sabrina N. Nascimento; Angela M. Moro; Natália Brucker; Fernando Freitas; Bruna Gauer; Juliano Durgante; Suelen Bordignon; Murilo Ricardo Zibetti; Clarissa M. Trentini; et al.Marta M. M. F. DuarteTilman GruneNicolle BreusingSolange C. Garcia Relationship between Inflammation and Oxidative Stress and Cognitive Decline in the Institutionalized Elderly. Oxidative Medicine and Cellular Longevity 2015, 2015, 1-12, 10.1155/2015/804198.
- Busra Can; Ozgur Kara; Muhammet Cemal Kizilarslanoglu; Gunes Arik; Gozde Sengul Aycicek; Fatih Sumer; Ramazan Civelek; Canan Demirtas; Zekeriya Ulger; Serum markers of inflammation and oxidative stress in sarcopenia. Aging Clinical and Experimental Research 2016, 29, 745-752, 10.1007/s40520-016-0626-2.
- David De Gonzalo-Calvo; Kim Neitzert; María Fernández; Ignacio Vega-Naredo; Beatriz Caballero; Beatriz Caballero García; Francisco Manuel Suárez; María Josefa Rodríguez-Colunga; Juan José Solano; Ana Coto-Montes; et al. Differential inflammatory responses in aging and disease: TNF-α and IL-6 as possible biomarkers. Free Radical Biology and Medicine 2010, 49, 733-737, 10.1016/j.freeradbiomed.2010.05.019.
- Yoshiaki Tamura; Takuya Omura; Kenji Toyoshima; Atsushi Araki; Nutrition Management in Older Adults with Diabetes: A Review on the Importance of Shifting Prevention Strategies from Metabolic Syndrome to Frailty. Nutrients 2020, 12, 3367, 10.3390/nu12113367.
- Larry A. Tucker; Physical activity and telomere length in U.S. men and women: An NHANES investigation. Preventive Medicine 2017, 100, 145-151, 10.1016/j.ypmed.2017.04.027.
- David B. Bartlett; Leslie H. Willis; Cris A. Slentz; Andrew Hoselton; Leslie Kelly; Janet L. Huebner; Virginia B. Kraus; Jennifer Moss; Michael J. Muehlbauer; Guillaume Spielmann; et al.William E. KrausJanet M. LordKim M. Huffman Ten weeks of high-intensity interval walk training is associated with reduced disease activity and improved innate immune function in older adults with rheumatoid arthritis: a pilot study. Arthritis Research & Therapy 2018, 20, 1-15, 10.1186/s13075-018-1624-x.
- David B. Bartlett; Cris A. Slentz; Leslie H. Willis; Andrew Hoselton; Janet L. Huebner; Virginia B. Kraus; Jennifer Moss; Michael J. Muehlbauer; Guillaume Spielmann; Deborah M. Muoio; et al.Timothy KovesHelena WuKim M. HuffmanJanet M. LordWilliam E. Kraus Rejuvenation of Neutrophil Functions in Association With Reduced Diabetes Risk Following Ten Weeks of Low-Volume High Intensity Interval Walking in Older Adults With Prediabetes – A Pilot Study. Frontiers in Immunology 2020, 11, ., 10.3389/fimmu.2020.00729.
- Morena Martucci; Rita Ostan; Fiammetta Biondi; Elena Bellavista; Cristina Fabbri; Claudia Bertarelli; Stefano Salvioli; Miriam Capri; Claudio Franceschi; Aurelia Santoro; et al. Mediterranean diet and inflammaging within the hormesis paradigm. Nutrition Reviews 2017, 75, 442-455, 10.1093/nutrit/nux013.
- Aurelia Santoro; Morena Martucci; Maria Conte; Miriam Capri; Claudio Franceschi; Stefano Salvioli; Inflammaging, hormesis and the rationale for anti-aging strategies. Ageing Research Reviews 2020, 64, 101142, 10.1016/j.arr.2020.101142.
- Rui-Xia Jia; Jing-Hong Liang; Yong Xu; Ying-Quan Wang; Effects of physical activity and exercise on the cognitive function of patients with Alzheimer disease: a meta-analysis. BMC Geriatrics 2019, 19, 1-14, 10.1186/s12877-019-1175-2.
- Hsin-Yu An; Wei Chen; Cheng-Wei Wang; Hui-Fei Yang; Wan-Ting Huang; Sheng-Yu Fan; The Relationships between Physical Activity and Life Satisfaction and Happiness among Young, Middle-Aged, and Older Adults. International Journal of Environmental Research and Public Health 2020, 17, 4817, 10.3390/ijerph17134817.
- Kyujin Lee; Wi-Young So; Differences in the Levels of Physical Activity, Mental Health, and Quality of Life of Elderly Koreans with Activity-Limiting Disabilities. International Journal of Environmental Research and Public Health 2019, 16, 2736, 10.3390/ijerph16152736.
- Neslihan Lok; Sefa Lok; Muammer Canbaz; The effect of physical activity on depressive symptoms and quality of life among elderly nursing home residents: Randomized controlled trial. Archives of Gerontology and Geriatrics 2017, 70, 92-98, 10.1016/j.archger.2017.01.008.
- Dustin Scott Kehler; Olga Theou; The impact of physical activity and sedentary behaviors on frailty levels. Mechanisms of Ageing and Development 2019, 180, 29-41, 10.1016/j.mad.2019.03.004.
- Elisa F. Ogawa; Suzanne G. Leveille; Julie A. Wright; Ling Shi; Sarah M. Camhi; Tongjian You; Physical Activity Domains/Recommendations and Leukocyte Telomere Length in U.S. Adults. Medicine & Science in Sports & Exercise 2017, 49, 1375-1382, 10.1249/mss.0000000000001253.
- Soudabeh Yarmohammadi; Hossein Mozafar Saadati; Mohtasham Ghaffari; Ali Ramezankhani; A systematic review of barriers and motivators to physical activity in elderly adults in Iran and worldwide. Epidemiology and Health 2019, 41, e2019049, 10.4178/epih.e2019049.
- Maria Justine; Azliyana Azizan; Vaharli Hassan; Zoolfaiz Salleh; Haidzir Manaf; Barriers to participation in physical activity and exercise among middle-aged and elderly individuals.. Singapore Medical Journal 2013, 54, 581-586, 10.11622/smedj.2013203.
- Aurelio Lo Buglio; Francesco Bellanti; Cristiano Capurso; Annalisa Paglia; Gianluigi Vendemiale; Adherence to Mediterranean Diet, Malnutrition, Length of Stay and Mortality in Elderly Patients Hospitalized in Internal Medicine Wards. Nutrients 2019, 11, 790, 10.3390/nu11040790.
- Alexandra Foscolou; Efi Koloverou; Antonia-Leda Matalas; Stefanos Tyrovolas; Christina Chrysohoou; Labros Sidossis; Loukianos Rallidis; Demosthenes B. Panagiotakos; Decomposition of Mediterranean Dietary Pattern on Successful Aging, Among Older Adults: A Combined Analysis of Two Epidemiological Studies. Journal of Aging and Health 2018, 31, 1549-1567, 10.1177/0898264318780608.
- Marialaura Bonaccio; Augusto Di Castelnuovo; Simona Costanzo; Alessandro Gialluisi; Mariarosaria Persichillo; Chiara Cerletti; Maria Benedetta Donati; Giovanni de Gaetano; Licia Iacoviello; Mediterranean diet and mortality in the elderly: a prospective cohort study and a meta-analysis. British Journal of Nutrition 2018, 120, 841-854, 10.1017/s0007114518002179.
- Ana Hernández-Galiot; Isabel Goñi; Adherence to the Mediterranean diet pattern, cognitive status and depressive symptoms in an elderly non-institutionalized population. Nutrición Hospitalaria 2017, 34, 338-344, 10.20960/nh.360.
- Maria Mantzorou; Konstantinos Vadikolias; Eleni Pavlidou; Christina Tryfonos; Georgios Vasios; Aspasia Serdari; Constantinos Giaginis; Mediterranean diet adherence is associated with better cognitive status and less depressive symptoms in a Greek elderly population. Aging Clinical and Experimental Research 2020, 33, 1033-1040, 10.1007/s40520-020-01608-x.
- Maria F. Masana; Josep Maria Haro; Anargiros Mariolis; Suzanne Piscopo; Giuseppe Valacchi; Vasiliki Bountziouka; Foteini Anastasiou; Akis Zeimbekis; Dimitra Tyrovola; Efthimios Gotsis; et al.George MetallinosAnna PolystipiotiJosep-Antoni TurAntonia-Leda MatalasChristos LionisEvangelos PolychronopoulosLabros S. SidossisStefanos TyrovolasDemosthenes B. Panagiotakos Mediterranean diet and depression among older individuals: The multinational MEDIS study. Experimental Gerontology 2018, 110, 67-72, 10.1016/j.exger.2018.05.012.
- Costas A. Anastasiou; Mary Yannakoulia; Mary Kosmidis; Efthimios Dardiotis; Giorgos M. Hadjigeorgiou; Paraskevi Sakka; Xanthi Arampatzi; Anastasia Bougea; Ioannis Labropoulos; Nikolaos Scarmeas; et al. Mediterranean diet and cognitive health: Initial results from the Hellenic Longitudinal Investigation of Ageing and Diet. PLoS ONE 2017, 12, e0182048-e0182048, 10.1371/journal.pone.0182048.
- José Enrique De La Rubia Ortí; María Pilar García-Pardo; Eraci Drehmer; David Sancho Cantus; Mariano Julián Rochina; Maria Asunción Aguilar; Iván Hu Yang; Improvement of Main Cognitive Functions in Patients with Alzheimer’s Disease after Treatment with Coconut Oil Enriched Mediterranean Diet: A Pilot Study. Journal of Alzheimer's Disease 2018, 65, 577-587, 10.3233/JAD-180184.
- Diane E. Hosking; Ranmalee Eramudugolla; Nicolas Cherbuin; Kaarin J. Anstey; MIND not Mediterranean diet related to 12‐year incidence of cognitive impairment in an Australian longitudinal cohort study. Alzheimer's & Dementia 2019, 15, 581-589, 10.1016/j.jalz.2018.12.011.
- L. Cherian; Y. Wang; K. Fakuda; S. Leurgans; N. Aggarwal; M. Morris; Mediterranean-Dash Intervention for Neurodegenerative Delay (MIND) Diet Slows Cognitive Decline After Stroke. null 2018, 6, 267-273.
- Isabel Morales-Ivorra; Montserrat Romera-Baures; Blanca Roman-Viñas; Lluis Serra-Majem; Osteoarthritis and the Mediterranean Diet: A Systematic Review. Nutrients 2018, 10, 1030, 10.3390/nu10081030.
- Mariela Nissensohn; Blanca Roman-Viñas; Almudena Sanchez Villegas; Suzanne Piscopo; Lluis Serra-Majem; The Effect of the Mediterranean Diet on Hypertension: A Systematic Review and Meta-Analysis. Journal of Nutrition Education and Behavior 2015, 48, 42-53.e1, 10.1016/j.jneb.2015.08.023.
- Alexandra Foscolou; Nathan M. D’Cunha; Nenad Naumovski; Stefanos Tyrovolas; Christina Chrysohoou; Loukianos Rallidis; Evangelos Polychronopoulos; Antonia-Leda Matalas; Labros S. Sidossis; Demosthenes Panagiotakos; et al. The association between the level of adherence to the Mediterranean diet and successful aging: An analysis of the ATTICA and MEDIS (MEDiterranean Islands Study) epidemiological studies. Archives of Gerontology and Geriatrics 2020, 89, 104044, 10.1016/j.archger.2020.104044.
- Anna Bach-Faig; Elliot M Berry; Denis Lairon; Joan Reguant; Antonia Trichopoulou; Sandro Dernini; F Xavier Medina; Maurizio Battino; Rekia Belahsen; Gemma Miranda; et al.Lluís Serra-Majem Mediterranean diet pyramid today. Science and cultural updates. Public Health Nutrition 2011, 14, 2274-2284, 10.1017/s1368980011002515.
- Cristina-Mihaela Lăcătușu; Elena-Daniela Grigorescu; Mariana Floria; Alina Onofriescu; Bogdan-Mircea Mihai; The Mediterranean Diet: From an Environment-Driven Food Culture to an Emerging Medical Prescription. International Journal of Environmental Research and Public Health 2019, 16, 942, 10.3390/ijerph16060942.
- Fiona C Bull; Salih S Al-Ansari; Stuart Biddle; Katja Borodulin; Matthew P Buman; Greet Cardon; Catherine Carty; Jean-Philippe Chaput; Sebastien Chastin; Roger Chou; et al.Paddy C DempseyLoretta DiPietroUlf EkelundJoseph FirthChristine M FriedenreichLeandro GarciaMuthoni GichuRussell JagoPeter T KatzmarzykEstelle LambertMichael LeitzmannKaren MiltonFrancisco B OrtegaChathuranga RanasingheEmmanuel StamatakisAnne TiedemannRichard P TroianoHidde P Van Der PloegVicky WariJuana F Willumsen World Health Organization 2020 guidelines on physical activity and sedentary behaviour. British Journal of Sports Medicine 2020, 54, 1451-1462, 10.1136/bjsports-2020-102955.
- Patricia C. Heyn; Mark A. Hirsch; Michele K. York; Deborah Backus; Physical Activity Recommendations for the Aging Brain: A Clinician-Patient Guide. Archives of Physical Medicine and Rehabilitation 2016, 97, 1045-1047, 10.1016/j.apmr.2016.02.003.
