Arterial hypertension is strongly related to overweight and obesity. In obese subjects, several mechanisms may lead to hypertension such as insulin and leptin resistance, perivascular adipose tissue dysfunction, renal impairment, renin-angiotensin-aldosterone-system activation and sympathetic nervous system activity. Weight loss (WL) seems to have positive effects on blood pressure (BP).
- hypertension
- weight loss
- obesity
1. Introduction
2. Mechanisms Linking Obesity to Hypertension
2.1. Visceral Adipose Tissue
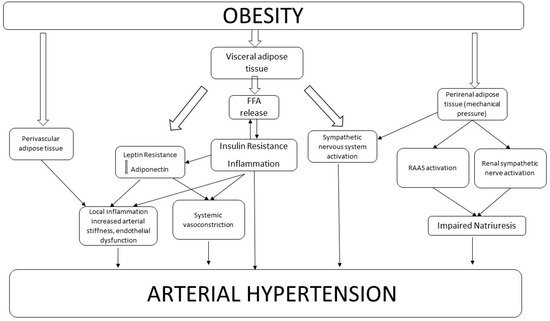
2.2. Perivascular Adipose Tissue
2.3. Renal Adipose Tissue
2.4. Renin Angiotensin Aldosterone System
2.5. Sympathetic Nervous System Activation
3. Weight Loss and Blood Pressure
|
Author, Year |
Number of Participants |
WL Intervention |
Mean WL ΔBW (Kg) ΔBMI (kg/m2) |
Mean BP Reduction (mmHg) |
Median Follow up |
|
|---|---|---|---|---|---|---|
|
Diet and Lifestyle modification |
||||||
|
Blumenthal, 2010 (ENCORE study) |
144 |
DASH diet alone |
−0.3 |
11.2 (SBP) 7.5 (DBP) |
4 months |
|
|
DASH diet plus weight management |
−8.7 |
16.1 (SBP) 9.9 (DBP) |
||||
|
Rocha-Goldberg, 2010 |
17 |
behavioral intervention |
1.5 ± 3.2 lb |
10.4 ± 10.6 (SBP) |
6 weeks |
|
|
Rothberg, 2017 |
344 |
behavioral intervention |
−6 ± 3 |
8 (SBP) * |
6 months |
|
|
170 |
behavioral intervention |
−5 ± 4 |
8 (SBP) * |
2 years |
||
|
Straznicky, 2011 |
59 |
dietary and moderate-intensity aerobic exercise |
−7.1 ± 0.6 (dietary) |
−2.4 ± 0.2 (dietary) |
10±2 (SBP) |
12 weeks |
|
−8.4 ± 1.0 (dietary + exercise) |
−2.8± 0.3 (dietary + exercise) |
|||||
|
Wing, 2011 (look AHEAD study) |
5154 |
intensive lifestyle intervention or diabetes support and education |
−4.8 ± 7.6 |
2.40 (DBP) 4.76 (SBP) |
1 year |
|
|
Pharmacological intervention |
||||||
|
Marso, 2016 |
9340 |
Pharmacologic (liraglutide vs placebo) |
2.3 kg higher in Liraglutide group |
1.2 (SBP) lower in Liraglutide group |
36 weeks |
|
|
Wijkman, 2019 |
124 |
Pharmacologic (liraglutide vs placebo) |
>3% |
9.2 (SBP) |
24 weeks |
|
|
Bariatric Surgery |
||||||
|
Ghanim, 2018 |
15 |
Surgery (RYGB) |
−11.7 |
11 (SBP) |
6 months |
|
|
Hallersund, 2013 (SOS study) |
2473 (277 gastric bypass, 1064 purely restricted proedures, 1132 control) |
Surgery (GBP, VBG/B) |
−10.1 (GBP group) |
−5.1 (SBP) −5.6 (DBP) (GBP group) |
10 years |
|
|
Seravalle, 2014 |
20 (10 surgery + 10 control) |
Surgery (vertical sleeve gastrectomy) |
−9.1 ± 1.4 |
10.2 ± 4.5 (SBP) |
6 months |
|
|
−10.8 ± 1.6 |
13.9 ± 5.0 (SBP) |
1 year |
||||
4. Possible Mechanisms Involved in BP Reduction after Weight Loss
References
- Hall, J.E.; Do Carmo, J.M.; Da Silva, A.A.; Wang, Z.; Hall, M.E. Obesity-Induced Hypertension: Interaction of Neurohumoral and Renal Mechanisms. Circ. Res. 2015, 116, 991–1006.
- Seravalle, G.; Grassi, G. Obesity and hypertension. Pharmacol. Res. 2017, 122, 1–7.
- Bramlage, P.; Pittrow, D.; Wittchen, H.U.; Kirch, W.; Boehler, S.; Lehnert, H.; Hoefler, M.; Unger, T.; Sharma, A.M. Hypertension in overweight and obese primary care patients is highly prevalent and poorly controlled. Am. J. Hypertens. 2004, 17, 904–910.
- Zhou, B.; Bentham, J.; Di Cesare, M.; Bixby, H.; Danaei, G.; Cowan, M.J.; Paciorek, C.J.; Singh, G.; Hajifathalian, K.; Bennett, J.E.; et al. Worldwide trends in blood pressure from 1975 to 2015: A pooled analysis of 1479 population-based measurement studies with 19·1 million participants. Lancet 2017, 389, 37–55.
- Kannel, W.B. Framingham study insights into hypertensive risk of cardiovascular disease. Hypertens. Res. 1995, 18, 181–196.
- Shihab, H.M.; Meoni, L.A.; Chu, A.Y.; Wang, N.Y.; Ford, D.E.; Liang, K.Y.; Gallo, J.J.; Klag, M.J. Body mass index and risk of incident hypertension over the life course: The Johns Hopkins Precursors Study. Circulation 2012, 126, 2983–2989.
- Williams, B.; Mancia, G.; Spiering, W.; Rosei, E.A.; Azizi, M.; Burnier, M.; Clement, D.L.; Coca, A.; De Simone, G.; Dominiczak, A.; et al. 2018 ESC/ESH Guidelines for the management of arterial hypertension. Eur. Heart J. 2018, 39, 3021–3104.
- Lavie, C.J.; Milani, R.V.; Ventura, H.O. Obesity and Cardiovascular Disease. Risk Factor, Paradox, and Impact of Weight Loss. J. Am. Coll. Cardiol. 2009, 53, 1925–1932.
- Fantuzzi, G.; Mazzone, T. Adipose tissue and atherosclerosis: Exploring the connection. Arterioscler. Thromb. Vasc. Biol. 2007, 27, 996–1003.
- Bosello, O.; Zamboni, M. Visceral obesity and metabolic syndrome. Obes. Rev. 2000, 1, 47–56.
- Caballero, A.E. Endothelial dysfunction in obesity and insulin resistance: A road to diabetes and heart disease. Obes. Res. 2003, 11, 1278–1289.
- Kuboki, K.; Jiang, Z.Y.; Takahara, N.; Ha, S.W.; Igarashi, M.; Yamauchi, T.; Feener, E.P.; Herbert, T.P.; Rhodes, C.J.; King, G.L. Regulation of endothelial constitutive nitric oxide synthase gene expression in endothelial cells and in vivo—A specific vascular action of insulin. Circulation 2000, 101, 676–681.
- Rahmouni, K. Obesity-associated hypertension: Recent progress in deciphering the pathogenesis. Hypertension 2014, 64, 215–221.
- Ruan, C.C.; Gao, P.J. Role of Complement-Related Inflammation and Vascular Dysfunction in Hypertension. Hypertension 2019, 73, 965–971.
- Lohn, M.; Dubrovska, G.; Lauterbach, B.; Luft, F.C.; Gollasch, M.; Sharma, A.M. Periadventitial fat releases a vascular relaxing factor. FASEB J. 2002, 16, 1057–1063.
- Szasz, T.; Webb, R.C. Perivascular adipose tissue: More than just structural support. Clin. Sci. 2011, 122, 1–12.
- Almabrouk, T.A.M.; Ewart, M.A.; Salt, I.P.; Kennedy, S. Perivascular fat, AMP-activated protein kinase and vascular diseases. Br. J. Pharmacol. 2014, 171, 595–617.
- Fernández-Alfonso, M.S.; Gil-Ortega, M.; García-Prieto, C.F.; Aranguez, I.; Ruiz-Gayo, M.; Somoza, B. Mechanisms of perivascular adipose tissue dysfunction in obesity. Int. J. Endocrinol. 2013, 2013, 402053.
- Xia, N.; Li, H. The role of perivascular adipose tissue in obesity-induced vascular dysfunction. Br. J. Pharmacol. 2017, 174, 3425–3442.
- Szasz, T.; Bomfim, G.F.; Webb, R.C. The influence of perivascular adipose tissue on vascular homeostasis. Vasc. Health Risk Manag. 2013, 9, 105–116.
- Van Dam, A.D.; Boon, M.R.; Berbée, J.F.P.; Rensen, P.C.N.; van Harmelen, V. Targeting white, brown and perivascular adipose tissue in atherosclerosis development. Eur. J. Pharmacol. 2017, 816, 82–92.
- Mikolajczyk, T.P.; Nosalski, R.; Szczepaniak, P.; Budzyn, K.; Osmenda, G.; Skiba, D.; Sagan, A.; Wu, J.; Vinh, A.; Marvar, P.J.; et al. Role of chemokine RANTES in the regulation of perivascular inflammation, T-cell accumulation, and vascular dysfunction in hypertension. FASEB J. 2016, 30, 1987–1999.
- Guzik, T.J.; Hoch, N.E.; Brown, K.A.; McCann, L.A.; Rahman, A.; Dikalov, S.; Goronzy, J.; Weyand, C.; Harrison, D.G. Role of the T cell in the genesis of angiotensin II–induced hypertension and vascular dysfunction. J. Exp. Med. 2007, 204, 2449–2460.
- Nosalski, R.; Guzik, T.J. Perivascular adipose tissue inflammation in vascular disease. Br. J. Pharmacol. 2017, 174, 3496–3513.
- Chandra, A.; Neeland, I.J.; Berry, J.D.; Ayers, C.R.; Rohatgi, A.; Das, S.R.; Khera, A.; McGuire, D.K.; De Lemos, J.A.; Turer, A.T. The relationship of body mass and fat distribution with incident hypertension: Observations from the dallas heart study. J. Am. Coll. Cardiol. 2014, 64, 997–1002.
- Chughtai, H.L.; Morgan, T.M.; Rocco, M.; Stacey, B.; Brinkley, T.E.; Ding, J.; Nicklas, B.; Hamilton, C.; Hundley, W.G. Renal sinus fat and poor blood pressure control in middle-aged and elderly individuals at risk for cardiovascular events. Hypertension 2010, 56, 901–906.
- Xiong, X.Q.; Chen, W.W.; Zhu, G.Q. Adipose afferent reflex: Sympathetic activation and obesity hypertension. Acta Physiol. 2014, 210, 468–478.
- Thatcher, S.; Yiannikouris, F.; Gupte, M.; Cassis, L. The adipose renin-angiotensin system: Role in cardiovascular disease. Mol. Cell. Endocrinol. 2009, 302, 111–117.
- Jia, G.; Aroor, A.R.; Sowers, J.R. The role ofmineralocorticoid receptor signaling in the cross-talk between adipose tissue and the vascular wall. Cardiovasc. Res. 2017, 113, 1055–1063.
- Schütten, M.T.J.; Houben, A.J.H.M.; de Leeuw, P.W.; Stehouwer, C.D.A. The Link Between Adipose Tissue Renin-Angiotensin-Aldosterone System Signaling and Obesity-Associated Hypertension. Physiology 2017, 32, 197–209.
- Grassi, G.; Mark, A.; Esler, M. The Sympathetic Nervous System Alterations in Human Hypertension. Circ. Res. 2015, 116, 976–990.
- Grassi, G.; Seravalle, G.; Brambilla, G.; Buzzi, S.; Volpe, M.; Cesana, F.; Dell’Oro, R.; Mancia, G. Regional differences in sympathetic activation in lean and obese normotensive individuals with obstructive sleep apnoea. J. Hypertens. 2014, 32, 383–388.
- Grassi, G.; Pisano, A.; Bolignano, D.; Seravalle, G.; D’Arrigo, G.; Quarti-Trevano, F.; Mallamaci, F.; Zoccali, C.; Mancia, G. Sympathetic nerve traffic activation in essential hypertension and its correlates systematic reviews and meta-analyses. Hypertension 2018, 72, 483–491.
- Fonkoue, I.T.; Le, N.A.; Kankam, M.L.; DaCosta, D.; Jones, T.N.; Marvar, P.J.; Park, J. Sympathoexcitation and impaired arterial baroreflex sensitivity are linked to vascular inflammation in individuals with elevated resting blood pressure. Physiol. Rep. 2019, 7, e14057.
- Ho, A.K.; Bartels, C.M.; Thorpe, C.T.; Pandhi, N.; Smith, M.A.; Johnson, H.M. Achieving Weight Loss and Hypertension Control among Obese Adults: A US Multidisciplinary Group Practice Observational Study. Am. J. Hypertens. 2016, 29, 984–991.
- Richard, R.N. Obesity-Related Hypertension. Ochsner J. 2009, 9, 133–136.
- Wijkman, M.O.; Dena, M.; Dahlqvist, S.; Sofizadeh, S.; Hirsch, I.; Tuomilehto, J.; Mårtensson, J.; Torffvit, O.; Imberg, H.; Saeed, A.; et al. Predictors and correlates of systolic blood pressure reduction with liraglutide treatment in patients with type 2 diabetes. J. Clin. Hypertens. 2019, 21, 105–115.
- Zanoli, L.; Di Pino, A.; Terranova, V.; Di Marca, S.; Pisano, M.; Di Quattro, R.; Ferrara, V.; Scicali, R.; Rabuazzo, A.M.; Fatuzzo, P.; et al. Inflammation and ventricular-vascular coupling in hypertensive patients with metabolic syndrome. Nutr. Metab. Cardiovasc. Dis. 2018, 28, 1222–1229.
- Wing, R.R.; Lang, W.; Wadden, T.A.; Safford, M.; Knowler, W.C.; Bertoni, A.G.; Hill, J.O.; Brancati, F.L.; Peters, A.; Wagenknecht, L. Benefits of modest weight loss in improving cardiovascular risk factors in overweight and obese individuals with type 2 diabetes. Diabetes Care 2011, 34, 1481–1486.
- Rothberg, A.E.; McEwen, L.N.; Kraftson, A.T.; Ajluni, N.; Fowler, C.E.; Nay, C.K.; Miller, N.M.; Burant, C.F.; Herman, W.H. Impact of weight loss on waist circumference and the components of the metabolic syndrome. BMJ Open Diabetes Res. Care 2017, 5, e000341.
- Blumenthal, J.A.; Babyak, M.A.; Hinderliter, A.; Watkins, L.L.; Craighead, L.; Lin, P.H.; Caccia, C.; Johnson, J.; Waugh, R.; Sherwood, A. Effects of the DASH diet alone and in combination with exercise and weight loss on blood pressure and cardiovascular biomarkers in men and women with high blood pressure: The ENCORE study. Arch. Intern. Med. 2010, 170, 126–135.
- Werner, N.; Nickenig, G. From Fat Fighter to risk factor: The zizag trek of leptin. Arterioscler. Thromb. Vasc. Biol. 2004, 24, 7–9.
- Mahmud, A.; Feely, J. Adiponectin and arterial stiffness. Am. J. Hypertens. 2005, 18 Pt 1, 1543–1548.
- Ghanim, H.; Monte, S.; Caruana, J.; Green, K.; Abuaysheh, S.; Dandona, P. Decreases in neprilysin and vasoconstrictors and increases in vasodilators following bariatric surgery. Diabetes Obes. Metab. 2018, 20, 2029–2033.
- Liu, B.X.; Sun, W.; Kong, X.Q. Perirenal Fat: A Unique Fat Pad and Potential Target for Cardiovascular Disease. Angiology 2018, 9, 3319718799967.
