Your browser does not fully support modern features. Please upgrade for a smoother experience.
Please note this is a comparison between Version 2 by Lindsay Dong and Version 3 by Lindsay Dong.
Chronic headache is a topical problem of neurology, psychiatry and general practice. The medication-overuse headache (MOH) is one of the leading pathologies in the structure of chronic headache. The serum and urine proteomic biomarkers of MOH can potentially help with the identification of patients with MOH development.
- headache
- chronic headache
- proteomics
- proteogenomics
- serum biomarker
- urine biomarker
1. Introduction
Chronic pain (CP) is an essential problem in healthcare. Approximately 20% of the European population is affected by CP
[1]
, which has a significant influence on their daily social and working lives. Moreover, the economic impact of CP is more than heart disease, cancer and diabetes put together
[2]
. It is worth mentioning that among chronic painful disorders, headaches are predominant. According to world literature, with an incidence of 3% per year, 4–5% of the general population suffers from chronic headache, known as headache occurrence ≥15 days per month
. Chronic forms of headache, such as chronic migraine or chronic tension-type headache, often involve a high and daily intake of combination analgesics and acute headache medications (AHMs), such as nonsteroidal anti-inflammatory drugs (NSAIDs) and triptans
[6]
. Paradoxically, they only worsen the chronic symptoms, resulting in the development of the secondary headache, so-called medication-overuse headache (MOH)
[7]
. In the latest and current Third Edition of the International Classification of Headache Disorders (IHS ICHD-3), MOH, also known as a rebound headache, is described as a headache that is present on 15 or more days per month developing as a consequence of regular overuse of acute or symptomatic headache medication. Criterions of MOH: the overuse of simple analgesics on 15 or more days per month; or else the overuse of triptans, ergotamines, analgesics, opioids, and (caffeine or codeine-containing) combined analgesics on 10 or more days per month, for more than 3 consecutive months
[8]
. Although the prevalence of MOH in the general population is around 1–2%
, MOH is still defined as a socio-economic burden worldwide, especially in lesser developed countries
[11]
, associated with significant long-term morbidity, disability, and lower quality of life. According to systematic reviews of MOH epidemiology, it is most predominant in middle-aged women from 30 to 50 years old, with the male to female ratio around 1 to 3–4
. The pathophysiological mechanisms of MOH development are still an ongoing debate. Nevertheless, frequent and regular consumption of acute headache medication does not seem enough to cause MOH, therefore individual predisposition and specifics of the medication class combined play a significant role in its development
[16]
.
The proteomic analysis and the exploration of proteomic biomarkers are crucial in understanding a lot of medical conditions, especially cancer, cardiovascular and neurodegenerative diseases
. Thus, identification of chronic pain biomarkers can be helpful and valuable for clinicians in the diagnosis of patients at risk or with an already developed disorder, reducing the time and costs, selection of rational personalized pain treatment, and understanding of the underlying pathophysiological mechanisms of chronic pain development
[20]
. Moreover, a considerable number of patients with MOH can hide the truth from the doctor about the frequency and daily amount of acute headache medications they are consuming. Besides, the MOH phenotype is almost indistinguishable from other chronic headache phenotypes
[11]
. Therefore, it becomes even more challenging to diagnose MOH and selecting and monitoring the headache treatment. Furthermore, acute headache medication overuse leads to side effects, such as nephrotoxicity and kidney damage, gastrointestinal bleeding, liver impairment
[11]
, especially in a group of patients who abuse NSAIDs
[21]
. It is not possible to prevent the development of renal impairment and acute renal failure caused by drug-induced nephrotoxicity by using traditional laboratory analyses, such as creatinine, creatinine clearance, urea, electrolytes, urine sediment
[22]
. However, urinary proteomics allows the potential risks of developing severe drug-induced kidney damage to be minimized by its detection in the early stages during a normal clinical presence, particularly in NSAIDs abusers, using a panel of protein biomarkers each informing on the integrated aspects of glomerular, tubular, and interstitial function
[23]
.
The underlying mechanisms of chronic pain pathophysiology still remain poorly understood. Proteomics is one of the most promising areas that can significantly contribute to pain chronicity research, its better understanding and management.
2. Candidate Genes and Proteomic Biomarkers in Medication-Overuse Headache
2.1. Candidate Serum Proteomic Biomarkers of Patients with Medication-Overuse Headache
Candidate serum proteomic biomarkers of MOH are shown in Table 1.
Table 1.
Candidate serum proteomic biomarkers of medication-overuse headache.
| Locus |
|---|
| Protein Main Function |
|---|
| Theor. Mass. |
|---|
| References |
|---|
| Lipocalin-type prostaglandin D2 synthase | L-PGDS | PTGDS | 9q34.3 | Prostaglandin biosynthesis process | 21,029 | [23][24] |
| Uromodulin (or Tamm-Horsfall urinary glycoprotein) | UROM | UMOD | 16p12.3 | Cellular defense response | 69,761 | [21][23][24][25] |
| Alpha-1-microglobulin | AMBP | AMBP | 9q32 | Calcium channel inhibitor activity | 38,999 | [21][23][25] |
| Zinc-alpha-2-glycoprotein | ZAZG | AZGP1 | 7q22.1 | Protein binding | 34,259 | [21] |
| Inter-alpha-trypsin heavy chain H4 | ITIH4 | ITIH4 | 3p21.1 | Acute-phase response | 103,357 | [21][23][25] |
| Ig kappa chain C region | IGKC | IGKC | 2p11.2 | Complement activation | 11,765 | [21][23][25] |
| Non-secretory ribonuclease | RNAS2 | RNASE2 | 14q11.2 | Chemotaxis | 18,354 | [21] |
| Cystatin M | CYTM | CST6 | 11q13.1 | Cystein-type endopeptidase inhibitor activity | 16,511 | [21] |
| Cystatin C | CYTC | CST3 | 20p11.21 | Cystein-type endopeptidase inhibitor activity | 15,799 | [21][24][25] |
| Serum albumin | ALBU | ALB | 4q13.3 | Metal binding | 69,367 | [23][25] |
| Alpha-1-antitrypsin | A1AT | SERPINA1 | 14q32.13 | Protease inhibitor | 46,737 | [23][25] |
| Actin, cytoplasmic 1 | ACTB | ACTB | 7p22.1 | Cell junction assembly | 41,737 | [23][25] |
| Apolipoprotein H | APOH | APOH | 17q24.2 | Heparin binding | 38,298 | [23][25] |
| Serpin B3 | SPB3 | SERPINB3 | 18q21.33 | Cystein-type endopeptidase inhibitor activity | 44,565 | [23][25] |
| Annexin A1 | ANXA1 | ANXA1 | 9q21.13 | Calcium ion binding | 38,714 | [23][25] |
| Prostaglandin-H2-D-isomerase | PTGDS | PTGDS | 9q34.3 | Prostaglandin biosynthesis process | 21,029 | [23][24][25] |
| Perlecan (fragment) | PGBM | HSPG2 | 1p36.12 | Angiogenesis | 468,830 | [23][25] |
| Transthyretin | TTHY | TTR | 18q12.1 | Protein binding | 15,887 | [23][25] |
| Proactivator polypeptide | SAP | PSAP | 10q22.1 | Enzyme activator activity | 58,113 | [23][25] |
| Nuclear transport factor 2 | NTF2 | NUTF2 | 16q22.1 | Positive regulation of protein import into nucleus | 14,478 | [23][25] |
| Fatty acid-binding protein | FABP5 | FABP5 | 8q21.13 | Fatty acid binding | 15,164 | [23][25] |
| Beta-2-microglobulin | B2MG | B2M | 15q21.1 | Antigen processing and presentation of endogenous peptide antigen via MHC class I | 13,715 | [23][25] |
| Protein S100-A11 | S10AB | S100A11 | 1q21.3 | Calcium ion binding | 11,740 | [23][25] |
| Non-secretory ribonuclease | RNAS2 | RNASE2 | 14q11.2 | Chemotaxis | 18,354 | [23][25] |
| Protein S100-A8 | S10A8 | S100A8 | 1q21.3 | Calcium ion binding | 10,835 | [23][25] |
| Protein Full Name | Entry Name | Gene Name | Locus | Protein Main Function | Theor. Mass. | References |
|---|---|---|---|---|---|---|
| Lipocalin-type prostaglandin D2 synthase | L-PGDS | PTGDS | 9q34.3 | Prostaglandin biosynthesis process | 21,029 | [23][24] |
| Apolipoprotein B100 | APOB | APOB | 2p24.1 | Cholesterol metabolism | 516,651 | [23][24] |
| Alpha-2-macroglobulin | A2MG | A2M | 12p13.31 | Enzyme binding | 164,613 | [23][24] |
| Complement factor H | CFAH | CFH | 1q31.3 | Complement activation | 143,480 | [23][24] |
| Complement C3 (fragm) | CO3 | C3 | 19p13.3 | Complement activation | 188,569 | [23][24] |
| Hemopexin | HEMO | HPX | 11p15.4 | Metal ion binding | 52,385 | [23][24] |
| Serum albumin | ALBU | ALB | 4q13.3 | Metal binding | 71,317 | [23][24] |
| Alpha-1B-glycoprotein | AIBG | A1BG | 19q13.43 | Neutrophil, platelet degranulation | 54,790 | [23][24] |
| Complement component C9 | CO9 | C9 | 5p13.1 | Complement activation | 64,615 | [23][24] |
| Kininogen-1 | KNG1 | KNG1 | 3q27.3 | Cysteine-type endopeptidase inhibitor activity | 72,996 | [23][24] |
| Vitronectin | VTNC | VTN | 17q11.2 | Heparin binding | 55,069 | [23][24] |
| Haptoglobin | HPT | HP | 16q22.2 | Acute phase response | 45,861 | [23][24] |
| Apolipoprotein A-4 | APOA4 | APOA4 | 11q23.3 | Lipid binding | 45,371 | [23][24] |
| Alpha-1-acid glycoprotein 1 | A1AG1 | ORM1 | 9q32 | Inflammatory response | 23,725 | [23][24] |
| Serum paraoxonase/arylesterase 1 | PON1 | PON1 | 7q21.3 | Hydrolase | 39,877 | [23][24] |
| Zinc-alpha-2-glycoprotein | ZA2G | AZGP1 | 7q22.1 | Protein transmembrane transporter activity | 34,465 | [23][24] |
| Alpha-1-acid glycoprotein 2 | A1AG2 | ORM2 | 9q32 | Acute phase response | 23,873 | [23][24] |
| Alpha-1-antitrypsin | A1AT | SERPINA1 | 14q32.13 | Protease inhibitor | 46,737 | [23][24] |
| Immunoglobulin heavy constant alpha 1 | IGHA1 | IGHA1 | 14q32.33 | Antigen binding | 37,655 | [23][24] |
| Retinol-binding protein | RETBP | RBP4 | 10q23.33 | Retinol binding | 23,010 | [23][24] |
| Transthyretin | TTHY | TTR | 18q12.1 | Hormone binding | 15,887 | [23][24] |
| Apolipoprotein E | APOE | APOE | 19q13.32 | Lipid transport | 36,154 | [23][24] |
| Vitamin D-binding protein | VDBP | GC | 4q13.3 | Vitamin D transport | 52,918 | [23][24] |
Protein entry name, according to the UniProtKB database. Theoretical molecular weight (Da).
2.2. Candidate Urine Proteomic Biomarkers of Patients with Medication-Overuse Headache
Candidate urine proteomic biomarkers of MOH are shown in Table 2.
Table 2.
Candidate urine proteomic biomarkers of medication-overuse headache.
| Protein Full Name |
|---|
| Entry Name |
|---|
| Gene Name |
|---|
Protein entry name, according to the UniProtKB database. Theoretical molecular weight (Da).
The summary the Biomarkers of MOH are presented in
Figure 1
.
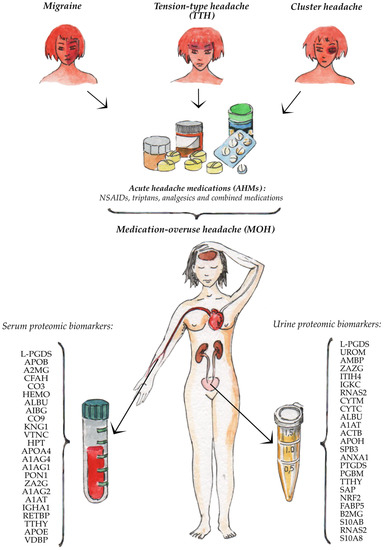
Figure 1.
Serum and urine proteomic biomarkers of the medication-overuse headache.
3. Summary
L-PGDS is expressed in various tissues, such as the brain, retina, cochlea, and male reproductive organs, and it is found in different biological fluids, such as cerebrospinal fluid (CSF), ascites, seminal plasma, serum, urine, and amniotic fluid. Alongside hematopoietic-type PGDS (H-PGDS), L-PGDS is related to a group of prostaglandin D synthases (PGDS) responsible for converting PGH2 to PGD2
[26]
, which is involved in a variety of central nervous system (CNS) functions, such as sedation, nonrapid eye movement (NREM) sleep and PGD2-allodynia. In addition to synthesizing PGD2, a potent endogenous nociceptive modulator
[27]
within the cells, in the extracellular space and body fluids L-PGDS binds various small nonsubstrate lipophilic molecules such as retinal, retinoic acid
[28]
, bilirubin, biliverdin
[29]
, gangliosides
[30]
, amyloid β peptides
[31]
. Therefore, L-PGDS can potentially be a promising proteomic biomarker as the entire prostaglandin system plays a huge role in pain and central sensitization development.
Vitamin D-binding protein (VDBP) is a monomeric glycoprotein synthesized and secreted predominantly by the liver. VDBP can be found in various body fluids, such as plasma, ascitic fluid, CSF. It was originally known as a group-specific component (GC) because of its worldwide polymorphisms
[32]
. It is also known as macrophage-activating factor (GcMAF/DBP-MAF) because it initiates macrophage activity, a key part of the host defense system
[33]
. VDBP has multiple functions, such as actin binding and neutrophil chemotaxis
[34]
. However, the main function of VDBP is to bind vitamin D and its plasma metabolites and transport them to target tissues. There are several studies showing the relation between Vitamin D deficiency or insufficiency and chronic headache development
. Possible mechanisms include the anti-inflammatory role of vitamin D, specifically, decreased production of inflammatory substances which activate trigeminal nerve, the main structure involved in migraine development
. Moreover, by inhibiting nitric oxide (NO) synthase expression, vitamin D reduces the production of NO, a key endogenous mediator in headaches, such as migraine
[39]
and tension-type headaches
[40]
, deficient levels of VDBP in MOH patients can be associated with a decreased level of vitamin D, which can be one of the mechanisms of chronic headache development.
Apolipoprotein E (APOE) is a multifunctional protein which participates in lipid metabolism and carries lipids in different tissues of the body, including both the peripheral and the central nervous system
[41]
. APOE mediates the binding of APOE-containing lipoproteins and lipid complexes to specific cell-surface receptors, such as the low-density lipoprotein (LDL) receptors, the LDL receptor-related protein (LRP), the very low-density lipoproteins (VLDL) receptor, the APOE receptor-2, gp330. Moreover, APOE demonstrates genetic polymorphisms containing three common alleles, ε2, ε3, ε4 that encode three isoforms (APOE2, APOE3, APOE4)
[42]
. According to the available literature, NO synthesis is dependent on APOE polymorphisms. Thus, the APOE4 gene is involved in the production of NO through increased uptake of arginine in the microglia compared to APOE3 gene
[43]
. APOE polymorphisms also influence the expression of cytokines which play a huge role in inflammation, pain modulation and central sensitization
[44]
.
Alpha-1 antitrypsin (A1AT), also known as alpha-1 proteinase inhibitor, is an acute phase reactant and serine protease inhibitor (serpin) whose targets are elastase, plasmin, thrombin, trypsin, chymotrypsin, plasminogen activator, and is mostly produced in the liver and expressed by hepatocytes
[45]
. It is important to note that A1AT inhibits NO production
[45]
, a key molecule in the pathophysiology of primary headaches
[46]
.
Hemopexin (HEMO) is an acute-phase plasma glycoprotein with the highest affinity to heme among all known proteins and is responsible for transporting heme from the plasma to the liver for breakdown and iron recovery. Moreover, it has intracellular antioxidant activities and, therefore, is involved in protecting cells from scavenging and oxidative stress. HEMO is expressed in various tissues such as the nervous system, skeletal muscle, retina, kidney, but mainly in the liver
[47]
. HEMO was found at significantly different levels in the CSF of patients with leptomeningeal metastases of breast cancer with neurological complications
[48]
and with other neuropathologically confirmed diseases
[49]
.
Haptoglobin (HPT) is an acute-phase protein whose main function is to bind hemoglobin (Hb) during hemolysis, forming the Hb-Hp complex, which is crucial for the elimination of free Hb by the macrophage CD163 scavenger receptor expressed on the liver Kupfer cells surface, therefore preventing kidney damage. HPT also acts as an antioxidant and plays a huge role in the neutralization of oxidative stress.
Retinol binding protein (RETBP) is specific carrier protein whose only known function is to transport retinol (vitamin A) from hepatic stores to target tissues
[50]
.
Transthyretin (TTHY) is a homotetrameric protein mostly produced in the liver and choroid plexus of the brain. The main function of TTHY, the transport of thyroxine and RETBP, is well-known. However, other functions of this protein, namely in the nervous system, have emerged.
Urinary biomarkers are increasingly used in the diagnosis, classification and prognosis of kidney diseases. Uromodulin (UROM), also known as Tamm-Horsfall protein, is the most abundant protein in urine. It can also be found in serum in lower amounts. UROM is exclusively produced by renal epithelial cells in the kidney. Amongst all the functions of UROM, the most important ones are the regulation of ion transport in the thick ascending limb, immunomodulation and protection against urinary tract infections and kidney stones
[51]
. In clinical practice, UROM is used as a valuable biomarker of tubular damage and kidney diseases, including drug-induced nephrotoxicity caused by medication overuse.
Alpha-1-microglobulin (AMBP) is one of the urinary microproteins that are becoming more and more important in clinical diagnostics and practice. AMBP is a tubular glycoprotein, mostly expressed in liver, blood and kidney, which is used for detecting acute lesions of proximal tubules. It was first discovered 40 years ago in human urine
[52]
. Functions of AMBP are still unknown. However, some reports have suggested that AMBP may be involved in oxidant-scavenging and have enzymatic reductase properties as an antioxidant
[53]
. Altered plasma and urine levels of AMBP are usually markers of impaired liver or kidney functions. Therefore, nowadays, urinary AMBP is considered as a promising inexpensive alternative biomarker for the early detection and diagnosis of urinary tract disorders
[54]
, including kidney damage, caused by acute headache medication overuse.
Zinc-alpha-2-glycoprotein (ZAZG) is a single-chain polypeptide secreted in various body fluids, such as serum and urine. Despite the fact that functions of ZAZG still remain unknown, some reports suggest that ZAZG has a lot of important functions in the human body, including fertilization and lipid mobilization, therefore it is considered as a multidisciplinary protein
. As its structural organization and folding characteristics are similar to the MHC class I antigen-presenting molecule, it may have a biological role in the immune response. ZAZG is used as a tumor biomarker for various carcinomas
[56]
. However, some immunohistochemical analyses have shown predominant expression in the kidney tubules of the human
[57]
. Therefore, urinary ZAZG may be a potential biomarker of renal damage, including drug-induced nephrotoxicity.
Inter-alpha-trypsin heavy chain H4 (ITIH4) is a liver-produced glycoprotein belonging to the liver-restricted serine protease inhibitor family. Its biological role is still unknown, as ITIH4 is cleaved in a number of different pathologies. However, it plays important role in various biological processes, such as inflammatory responses to trauma, liver formation or regeneration
[58]
.
Immunoglobulin kappa constant (IGKC) is a constant region of immunoglobulin light chains, also known as antibodies, membrane-bound or secreted glycoproteins produced by B lymphocytes. The main function of IGKC is to serve as receptors which, upon the binding of a specific antigen, trigger the clonal expansion and differentiation of B lymphocytes into immunoglobulin-secreting plasma cells. Secreted immunoglobulins play a significant role in the mediation of the effector phase of humoral immunity, which results in the elimination of bound antigens
[59]
.
Nonsecretory ribonuclease (RNAS2) is a pyrimidine specific nuclease with a slight preference for cytotoxin and helminthotoxin. RNAS2 is selectively chemotactic for dendritic cells and possesses a wide variety of biological activities.
Cystatin C (CYTC) is a nonglycosylated basic protein encoded by the CST3 gene found in all nucleated cells. CYTC was first discovered in 1961 and formally named in 1984. CYTC is a potent inhibitor of lysosomal proteinases and extracellular inhibitors of cysteine proteases that play a huge role in human vascular pathophysiology
[60]
. Nowadays, CYCT is used as a more accurate alternative to serum creatinine for measuring glomerular filtration rate (GFR), one of the main parameters in the estimation of kidney function
[61]
. CYCT is increasingly used as an earlier biomarker for acute kidney injury, a superior marker of kidney transplant function, cardiovascular disease risk and transplant failure. Therefore, CYTC may be a promising proteomic biomarker for MOH and drug-induced nephrotoxicity, caused by acute headache medication overuse.
References
- Van Hecke, O.; Torrance, N.; Smith, B.H. Chronic pain epidemiology and its clinical relevance. Br. J. Anaesth. 2013, 111, 13–18.
- Gaskin, D.J.; Richard, P. The Economic Costs of Pain in the United States. J. Pain 2012, 13, 715–724.
- Stovner, L.J.; Zwart, J.-A.; Hagen, K.; Terwindt, G.M.; Pascual, J. Epidemiology of headache in Europe. Eur. J. Neurol. 2006, 13, 333–345.
- Scher, A.I.; Lipton, R.B.; Stewart, W.F.; Bigal, M. Patterns of medication use by chronic and episodic headache sufferers in the general population: Results from the frequent headache epidemiology study. Cephalalgia 2009, 30, 321–328.
- Katsarava, Z. Medication-Overuse Headache (MOH). Comorbidities Headache Disord. 2016, 207–222.
- Diener, H.-C.; Dodick, D.; Evers, S.; Holle, D.; Jensen, R.H.; Lipton, R.B.; Porreca, F.; Silberstein, S.; Schwedt, T. Pathophysiology, prevention, and treatment of medication overuse headache. Lancet Neurol. 2019, 18, 891–902.
- Wakerley, B.R. Medication-overuse headache: Painkillers are not always the answer. Br. J. Gen. Pract. 2020, 70, 58–59.
- Headache Classification Committee of the International Headache Society (IHS) The International Classification of Headache Disorders, 3rd edition (beta version). Cephalalgia 2013, 33, 629–808.
- Westergaard, M.L.; Hansen, E.H.; Glümer, C.; Olesen, J.; Jensen, R.H. Definitions of medication-overuse headache in population-based studies and their implications on prevalence estimates: A systematic review. Cephalalgia 2014, 34, 409–425.
- Ayzenberg, I.; Katsarava, Z.; Sborowski, A.; Chernysh, M.; Osipova, V.; Tabeeva, G.; Yakhno, N.; Steiner, T.J.; Burden, O.B.O.L.T. The prevalence of primary headache disorders in Russia: A countrywide survey. Cephalalgia 2012, 32, 373–381.
- Vandenbussche, N.; Laterza, D.; Lisicki, M.; Lloyd, J.; Lupi, C.; Tischler, H.; Toom, K.; Vandervorst, F.; Quintana, S.; Paemeleire, K.; et al. Medication-overuse headache: A widely recognized entity amidst ongoing debate. J. Headache Pain 2018, 19, 1–14.
- Jonsson, P.; Hedenrud, T.; Linde, M.; Bjerkeli, P. Epidemiology of medication overuse headache in the general Swedish population. Cephalalgia 2011, 31, 1015–1022.
- Wang, S.-J.; Fuh, J.-L.; Lu, S.-R.; Liu, C.-Y.; Hsu, L.-C.; Wang, P.-N.; Liu, H.-C. Chronic daily headache in Chinese elderly: Prevalence, risk factors, and biannual follow-up. Neurology 2000, 54, 314.
- Straube, A.; Pfaffenrath, V.; Ladwig, K.-H.; Meisinger, C.; Hoffmann, W.; Fendrich, K.; Vennemann, M.; Berger, K. Prevalence of chronic migraine and medication overuse headache in Germany—The German DMKG headache study. Cephalalgia 2009, 30, 207–213.
- Kristoffersen, E.S.; Lundqvist, C. Medication-overuse headache: Epidemiology, diagnosis and treatment. Ther. Adv. Drug Saf. 2014, 5, 87–99.
- Bahra, A.; Walsh, M.; Menon, S.; Goadsby, P.J. Does Chronic Daily Headache Arise De Novo in Association With Regular Use of Analgesics? Headache J. Head Face Pain 2003, 43, 179–190.
- Suchkov, S.; Kostyushev, D.; Krynskii, S.; Gnatenko, D.; Paltsev, M.A. Proteomics as a Fundamental Tool for Subclinical Screening, Tests Verification and Assessment of Applied Therapy. Ann. Russ. Acad. Med. Sci. 2013, 68, 65–71.
- Fredriksson, S.; Horecka, J.; Brustugun, O.T.; Schlingemann, J.; Koong, A.C.; Tibshirani, R.; Davis, R.W. Multiplexed Proximity Ligation Assays to Profile Putative Plasma Biomarkers Relevant to Pancreatic and Ovarian Cancer. Clin. Chem. 2008, 54, 582–589.
- Fredriksson, S.; Dixon, W.; Ji, H.; Koong, A.C.; Mindrinos, M.; Davis, R.W. Multiplexed protein detection by proximity ligation for cancer biomarker validation. Nat. Chem. Biol. 2007, 4, 327–329.
- Kwok, Y.H. Potential Peripheral Biomarkers for Chronic Pain. Ph.D. Thesis, The University of Adelaide, Adelaide, Australia, June 2014. Available online: https://digital.library.adelaide.edu.au/dspace/bitstream/2440/88013/9/01front.pdf (accessed on 15 June 2021).
- Bellei, E.; Cuoghi, A.; Monari, E.; Bergamini, S.; Fantoni, L.I.; Zappaterra, M.; Guerzoni, S.; Bazzocchi, A.; Tomasi, A.; Pini, L.A. Proteomic analysis of urine in medication-overuse headache patients: Possible relation with renal damages. J. Headache Pain 2012, 13, 45–52.
- Liangos, O.; Perianayagam, M.C.; Vaidya, V.S.; Han, W.K.; Wald, R.; Tighiouart, H.; MacKinnon, R.W.; Li, L.; Balakrishnan, V.S.; Pereira, B.J.; et al. Urinary N-Acetyl-β-(D)-Glucosaminidase Activity and Kidney Injury Molecule-1 Level Are Associated with Adverse Outcomes in Acute Renal Failure. J. Am. Soc. Nephrol. 2007, 18, 904–912.
- Bellei, E.; Monari, E.; Cuoghi, A.; Bergamini, S.; Guerzoni, S.; Ciccarese, M.; Ozben, T.; Tomasi, A.; Pini, L.A. Discovery by a proteomic approach of possible early biomarkers of drug-induced nephrotoxicity in medication-overuse headache. J. Headache Pain 2013, 14, 6.
- Bellei, E.; Monari, E.; Bergamini, S.; Cuoghi, A.; Tomasi, A.; Guerzoni, S.; Ciccarese, M.; Pini, L.A. Validation of potential candidate biomarkers of drug-induced nephrotoxicity and allodynia in medication-overuse headache. J. Headache Pain 2015, 16, 1–9.
- Bellei, E.; Monari, E.; Bergamini, S.; Pini, L.A.; Tomasi, A.; Ozben, T. Urinary proteomics in biomarker discovery of kidney-related disorders: Diabetic nephropathy and drug-induced nephrotoxicity in chronic headache. Electron. J. Int. Fed. Clin. Chem. Lab. Med. 2018, 29, 290–297.
- Lee, S.; Jang, E.; Kim, J.-H.; Kim, J.-H.; Lee, W.-H.; Suk, K. Lipocalin-type Prostaglandin D2 Synthase Protein Regulates Glial Cell Migration and Morphology through Myristoylated Alanine-rich C-Kinase Substrate. J. Biol. Chem. 2012, 287, 9414–9428.
- Minami, T.; Okuda-Ashitaka, E.; Nishizawa, M.; Mori, H.; Ito, S. Inhibition of nociceptin-induced allodynia in conscious mice by prostaglandin D2. Br. J. Pharmacol. 1997, 122, 605–610.
- Tanaka, T.; Urade, Y.; Kimura, H.; Eguchi, N.; Nishikawa, A.; Hayaishi, O. Lipocalin-type Prostaglandin D Synthase (β-Trace) Is a Newly Recognized Type of Retinoid Transporter. J. Biol. Chem. 1997, 272, 15789–15795.
- Beuckmann, C.T.; Aoyagi, M.; Okazaki, I.; Hiroike, T.; Toh, H.; Hayaishi, O.; Urade, Y. Binding of Biliverdin, Bilirubin, and Thyroid Hormones to Lipocalin-Type Prostaglandin D Synthase†. Biochemistry 1999, 38, 8006–8013.
- Mohri, I.; Taniike, M.; Okazaki, I.; Kagitani-Shimono, K.; Aritake, K.; Kanekiyo, T.; Yagi, T.; Takikita, S.; Kim, H.-S.; Urade, Y.; et al. Lipocalin-type prostaglandin D synthase is up-regulated in oligodendrocytes in lysosomal storage diseases and binds gangliosides. J. Neurochem. 2006, 97, 641–651.
- Kanekiyo, T.; Ban, T.; Aritake, K.; Huang, Z.-L.; Qu, W.-M.; Okazaki, I.; Mohri, I.; Murayama, S.; Ozono, K.; Taniike, M.; et al. Lipocalin-type prostaglandin D synthase/beta-trace is a major amyloid beta-chaperone in human cerebrospinal fluid. Proc. Natl. Acad. Sci. USA 2007, 104, 6412–6417.
- Bouillon, R.; Schuit, F.; Antonio, L.; Rastinejad, F. Vitamin D Binding Protein: A Historic Overview. Front. Endocrinol. 2020, 10, 910.
- Gregory, K.J.; Zhao, B.; Bielenberg, D.R.; Dridi, S.; Wu, J.; Jiang, W.; Huang, B.; Pirie-Shepherd, S.; Fannon, M. Vitamin D Binding Protein-Macrophage Activating Factor Directly Inhibits Proliferation, Migration, and uPAR Expression of Prostate Cancer Cells. PLoS ONE 2010, 5, e13428.
- The Functions of Vitamin D—Binding Protein (VDBP). Available online: https://www.researchgate.net/figure/The-functions-of-vitamin-D-binding-protein-VDBP_tbl2_43534863 (accessed on 7 June 2021).
- Nowaczewska, M.; Wiciński, M.; Osiński, S.; Kaźmierczak, H. The Role of Vitamin D in Primary Headache–from Potential Mechanism to Treatment. Nutrients 2020, 12, 243.
- Ghorbani, Z.; Togha, M.; Rafiee, P.; Ahmadi, Z.S.; Magham, R.R.; Haghighi, S.; Jahromi, S.R.; Mahmoudi, M. Vitamin D in migraine headache: A comprehensive review on literature. Neurol. Sci. 2019, 40, 2459–2477.
- Levy, D.; Burstein, R.; Kainz, V.; Jakubowski, M.; Strassman, A.M. Mast cell degranulation activates a pain pathway underlying migraine headache. Pain 2007, 130, 166–176.
- Burstein, R.; Noseda, R.; Borsook, D. Migraine: Multiple Processes, Complex Pathophysiology. J. Neurosci. 2015, 35, 6619–6629.
- Moskaleva, P.; Shnayder, N.; Petrova, M.; Kaskaeva, D.; Gavrilyuk, O.; Radostev, S.; Garganeeva, N.; Sharavii, V.; Vaiman, E.; Nasyrova, R. The Role of Single Nucleotide Variants of NOS1, NOS2, and NOS3 Genes in the Development of the Phenotype of Migraine and Arterial Hypertension. Brain Sci. 2021, 11, 753.
- Shnayder, N.; Petrova, M.; Moskaleva, P.; Shesternya, P.; Pozhilenkova, E.; Nasyrova, R. The Role of Single-Nucleotide Variants of NOS1, NOS2, and NOS3 Genes in the Comorbidity of Arterial Hypertension and Tension-Type Headache. Molecules 2021, 26, 1556.
- Zhao, N.; Liu, C.-C.; Qiao, W.; Bu, G. Apolipoprotein E, Receptors, and Modulation of Alzheimer’s Disease. Biol. Psychiatry 2018, 83, 347–357.
- Liu, S.; Liu, J.; Weng, R.; Gu, X.; Zhong, Z. Apolipoprotein E gene polymorphism and the risk of cardiovascular disease and type 2 diabetes. BMC Cardiovasc. Disord. 2019, 19, 1–6.
- Gupta, R.; Kumar, V.; Luthra, K.; Banerjee, B.; Bhatia, M.S. Polymorphism in apolipoprotein E among migraineurs and tension-type headache subjects. J. Headache Pain 2009, 10, 115–120.
- The Immune System and Headache. Available online: https://www.practicalpainmanagement.com/pain/headache/immune-system-headache (accessed on 7 June 2021).
- Hunt, J.M.; Tuder, R. Alpha 1 Anti-Trypsin: One Protein, Many Functions. Curr. Mol. Med. 2012, 12, 827–835.
- Gallai, V.; Sarchielli, P. Nitric oxide in primary headaches. J. Headache Pain 2000, 1, 145–154.
- Tolosano, E.; Altruda, F. Hemopexin: Structure, Function, and Regulation. DNA Cell Biol. 2002, 21, 297–306.
- Galicia, N.; Díez, P.; Dégano, R.M.; Guest, P.C.; Ibarrola, N.; Fuentes, M. Proteomic Biomarker Identification in Cerebrospinal Fluid for Leptomeningeal Metastases with Neurological Complications. Adv. Exp. Med. Biol. 2017, 974, 85–96.
- Castaño, E.M.; Roher, A.E.; Esh, C.L.; Kokjohn, T.A.; Beach, T. Comparative proteomics of cerebrospinal fluid in neuropathologically-confirmed Alzheimer’s disease and non-demented elderly subjects. Neurol. Res. 2006, 28, 155–163.
- Retinol Binding Protein—An Overview. ScienceDirect Topics. Available online: https://www.sciencedirect.com/topics/neuroscience/retinol-binding-protein (accessed on 7 June 2021).
- Devuyst, O.; Olinger, E.; Rampoldi, L. Uromodulin: From physiology to rare and complex kidney disorders. Nat. Rev. Nephrol. 2017, 13, 525–544.
- Ekström, B.; Peterson, P.; Berggárd, I. A urinary and plasma α1-glycoprotein of low molecular weight: Isolation and some properties. Biochem. Biophys. Res. Commun. 1975, 65, 1427–1433.
- Åkerström, B.; Lögdberg, L. A1-Microglobulin. Available online: https://www.ncbi.nlm.nih.gov/books/NBK6003/ (accessed on 16 June 2021).
- Penders, J.; Delanghe, J.R. Alpha 1-microglobulin: Clinical laboratory aspects and applications. Clin. Chim. Acta 2004, 346, 107–118.
- Hassan, I.; Waheed, A.; Yadav, S.; Singh, T.P.; Ahmad, F. Zinc α2-Glycoprotein: A Multidisciplinary Protein. Mol. Cancer Res. 2008, 6, 892–906.
- Elsheikh, M.; Elhefnawy, K.A.; Emad, G.; Ismail, M.; Borai, M. Zinc alpha 2 glycoprotein as an early biomarker of diabetic nephropathy in patients with type 2 diabetes mellitus. Braz. J. Nephrol. 2019, 41, 509–517.
- Tada, T.; Ohkubo, I.; Niwa, M.; Sasaki, M.; Tateyama, H.; Eimoto, T. Immunohistochemical localization of Zn-alpha 2-glycoprotein in normal human tissues. J. Histochem. Cytochem. 1991, 39, 1221–1226.
- Pihl, R.; Jensen, R.K.; Poulsen, E.C.; Jensen, L.; Hansen, A.G.; Thøgersen, I.B.; Dobó, J.; Gál, P.; Andersen, G.R.; Enghild, J.J.; et al. ITIH4 acts as a protease inhibitor by a novel inhibitory mechanism. Sci. Adv. 2021, 7, eaba7381.
- Mcheyzerwilliams, M.G.; Okitsu, S.L.; Wang, N.; Mcheyzerwilliams, L.J. Molecular programming of B cell memory. Nat. Rev. Immunol. 2011, 12, 24–34.
- Fernando, S.; Polkinghorne, K.R. Cystatin C: Not just a marker of kidney function. Braz. J. Nephrol. 2020, 42, 6–7.
- Ferguson, T.W.; Komenda, P.; Tangri, N. Cystatin C as a biomarker for estimating glomerular filtration rate. Curr. Opin. Nephrol. Hypertens. 2015, 24, 295–300.
More
