Alginic acids or alginates, i.e., the salts of alginic acid, are the main polysaccharides in brown seaweeds
[24], reaching up to 16.9% DW in
S. vulgare, 20% DW in
S. longicruris, 24% DW in
A. nodosum, 32% DW in
Sargassum carpophyllum, 40% DW in
Laminaria hyperborean [25], 41% in
Sargassum siliquosum and even to 59% DW in
F. vesiculosus [26]. Within the cell wall, these polysaccharides are known to be partially responsible for the seaweed’s flexibility
[3] and therefore, expectedly, brown seaweeds grown under turbulent conditions usually have superior alginate contents than those of calm waters. In terms of structure, alginic acids or their corresponding extracted salts consist of α-
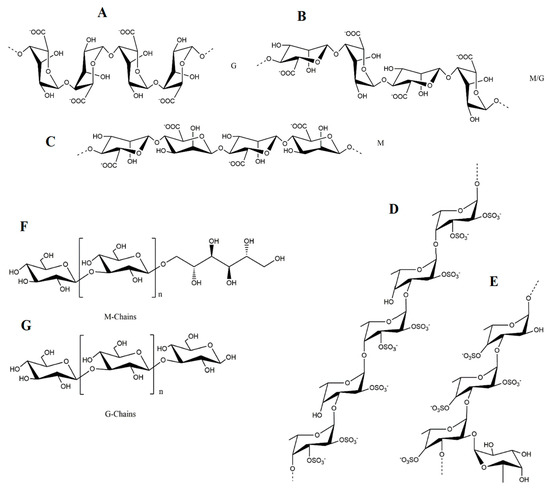
l-guluronic acid (G) and β-
d-mannuronic acid (M) (1→4)-linked residues arranged either in heteropolymeric (MG) and/or homopolymeric (M or G) blocks (
Figure 1A–C). Regardless, the variations caused by diverse factors (e.g., algae species, seasonability, parts of the algae) are expected
[16]. Noteworthy, alginates are considered one of the most important food colloids, with many applications in several industries such as foods, paper, pharmaceutical or cosmetics
[27]. In fact, G-blocks in the presence of ions, such as Ca
2+ form is the so-called egg-box, thus granting stiffness to the overall structure and conferring gel-forming properties to these polysaccharides
[28]. Therefore, they are usually used as thickeners, gels, emulsifiers and stabilizers in order to improve quality parameters, especially in food grade products
[29]. In addition to their wide applications, more recently, dietary alginates are being associated with positive health benefits in the gastrointestinal tract and appetite regulation, as well as antihypertensive and anti-diabetic effects
[30]. Alginates are also considered great prebiotics as they were demonstrated to significantly promote the growth of several bacteria, including
Bifidobacterium bifidum,
Bifidobacterium longum and
Lactobacilli, alongside with the increase of acetic acid, propionic acid and several short chain fatty acid metabolites, while decreasing deleterious metabolites, including faecal sulphide, phenol,
p-cresol, indole, ammonia and skatole
[31].
Figure 1. The structure of representative polysaccharides found in brown algae: (A–C) alginic acids; (D–E) fucoidans from A. nodusum/F. vesiculosus and S. latissima, respectively; (F–G) laminarins M and G chains.
Fucoidans i.e., metabolites belonging to the fucans family, also have a structural role in brown algae, mostly preventing dehydration
[3]. Their reported content in
Phaeophytae is variable, ranging from approximately 6–8% DW in
L. japonica, 3.2–16% DW in
U. pinnatifida, and 3.4–25.7% DW in
F. vesiculosus [10][32][10,32]. These polysaccharides are mainly composed of fucose and sulphate, although the presence of other types of monosaccharides (glucose, galactose, mannose, xylose and uronic acids), acetyl groups and proteins also occur
[33]. Despite being molecules with high structural diversity, the representative backbone of fucoidans consists of (1→3)- and (1→4)-linked α-
l-fucopyranose residues, and these polysaccharides are commonly divided in two types, the first being characterized by long chains of (1→3)-linked α-
l-fucopyranose residues (mainly present in
L. saccharina,
L. digitata,
C. okamuranus, and
Chorda filum) and the second consisting of alternating (1→3)- and (1→4)-linked α-
l-fucopyranose residues (characteristic from
A. nodosum and
Fucus spp.) (
Figure 1D,E)
[24][34][24,34].
2.2. Phlorotannins
Phlorotannins are phenolic compounds characteristic from
Phaeophytae and also represent their main phenolic pool. In brown seaweeds, they are associated with a myriad of functions, ranging from structural cell wall components, to biosyinthetic percursors and defensive mediators against natural enemies, acting as herbivore deterrents, inhibitors of digestion and agents against bacteria
[11]. Phlorotannins are known to accumulate mostly in physodes (i.e., specialized membrane-bound vesicles of the cell cytoplasm), with levels that might represent up to 25% of seaweed’s DW, despite variations which occur depending on distinct factors
[35][47]. For example, the higher levels of phlorotannins in
Fucus spp. are associated with high salinity waters and solar exposure during summer
[10].
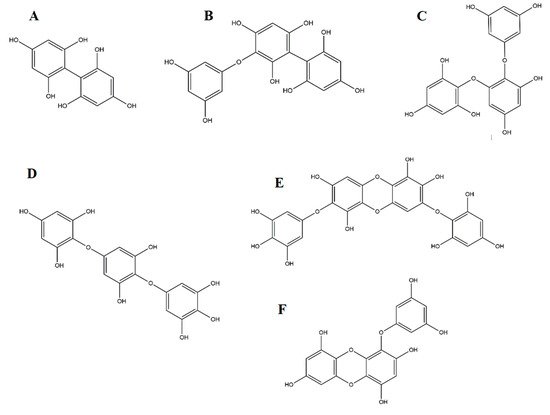
Being part of the tannins group, phlorotannins present a polymeric structure derived from several phloroglucinol (1,3,5-trihydroxybenzene) units and possess a high number of hydroxy groups, thus conferring them solubility in water
[36][48]. Depending on the linkage between phloroglucinol monomer units, a wide range of compounds with different molecular weights can be obtained
[37][49], which overall, are divided in four categories for each type of linkage: Fuhalols and phlorethols based on ether linkage, fucols based on C-C linkage, fucophlorethols for a combination of the previous ones, and, finally, eckols and carmalols, based on dibenzodioxin linkage (
Figure 2).
Figure 2. Some representative phlorotannins from brown seaweeds: (A) Fucol; (B) Fucophlorethol; (C) Phlorethol; (D) Fuhalol; (E) Carmalol; (F) Eckol.
Phenolic extracts from brown seaweeds have been demonstrated to exhibit various biological activities, including antioxidant, antidiabetic, anti-inflammatory and others
[11][38][39][11,50,51]. In this regard, O’Sullivan et al.
[40][52] observed the augment of glutathione levels in Caco-2 cell models when incubated with
A. nodosum, F. vesiculosus and
F. serratus phlorotannins extracts, while also highlighting the protective effects of the latter on the same model pretreated with H
2O
2. In vivo experiments have even demonstrated that the oral administration of 200 mg/kg/day of
F. vesiculosus polyhenol-rich extracts over 4 weeks to Sprague-Dawley rats could increase the blood plasma reducing power, paraoxonase/arylesterase 1 (PON-1) activity and O
2•− scavenging activity by 29%, 33% and 25%, respectively
[41][53]. Likewise, the antidiabetic properties of
A. nodosum and
F. vesiculosus phenolic-rich extract were observed in vivo as the postprandial blood glucose levels and insulin peak decreased 90% and 40%, respectively, on rats under hyperglycemic diets supplemented with 7.5 mg/kg compared to the unsupplemented group
[42][54]. In fact, the ingestion of 500 mg of this mixture containing
A. nodosum and
F. vesiculosus 30 min prior to the consumption of carbohydrates was shown to reduce the insulin incremental area of the curve and an increase in insulin sensitivity in a human clinical trial
[43][55]. Human trials have also been carried out to evaluate the potential antiobesity effect of polyphenolic-rich extracts of
A. nodosum (100 mg/day for 8 weeks). Although the treatment did not exhibit any significant benefits (no significant changes in C-reactive protein, antioxidant status or inflammatory cytokines), with the exception of a modest decrease of the DNA damage in the obese group, several phlorotannin metabolites were detected in the subjects plasma and urine, indicating that these compounds are metabolised and absorbed into the systemic circulation
[44][56]. These observations are in line with those reported by Corona et al.
[45][57] who also described the appearance of phlorotannin metabolites in urine and plasma collected from humans after consuming a capsule of
A. nodosum extract containing about 100 mg of polyphenols.
2.3. Fucoxanthin
In opposition to red and green macroalgae,
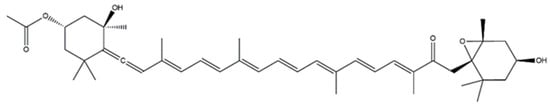
Phaophytae are characterized by the presence of the carotenoid fucoxanthin, which is responsible for their specific coloration. Fucoxanthin is a xanthophyll belonging to the tetraterpenoid family with a structure consisting of an unusual allenic bond and a 5,6-monoepoxide in its molecule (
Figure 3). The content of this pigment is highly variable amongst different species, as well as dependent on extrinsic factors, with a large range being even described within the same species. The reported levels comprise in 171 mg/kg (
Fucus spiralis), 224 mg/kg (
Fucus distichus), 364 mg/kg (
Fucus evanescens), 172–660 mg/kg (
A. nodosum), 178–468 mg/kg (
Laminaria spp.)
[46][47][41,58].
Figure 3. Structure of fucoxanthin.
Recently, this xanthophyll has earned particular attention mainly because of its promising effects in terms of antidiabetic, anti-obesity and antioxidant activities
[48][49][59,60], with claims being supported by in vivo studies. For instance, the administration of
U. pinnatifida lipids rich in fucoxanthin to male diabetic mice were associated with insulin resistance amelioration and the reduction of blood glucose levels
[50][61]. Moreover, fucoxanthin isolated from the same macraolgae species was also shown to inhibit the differentiation of 3T3-L1 preadipocytes into adipocytes by down-regulating peroxisome proliferator-activated receptor gamma (PPARγ)
[51][62]. Furthermore, a diet based on
U. pinnatifida fucoxanthin was capable of inducing uncoupling protein 1 (UCP1) expression in white adipose tissue (WAT) of obese mice. When added as a supplement to rats fed with a high-fat diet, it prompted a decrement of the mRNA expression of significant enzymes associated with lipid metabolism, such as fatty acid synthase, acyl-CoA cholesterol acyltransferase, hepatic acetyl-CoA carboxylase, glucose-6-phosphate dehydrogenase, hydroxy-3-methylglutaryl coenzyme A and SREBP-1C
[52][53][63,64].
2.4. Minerals
Due to their structural and physiological features, brown macroalgae are recognized for their superior ability to accumulate minerals. Although the content of minerals like calcium, magnesium, phosphorus, potassium, sodium and iron is usually high within the macroalgae matrix, one of the standout aspects, comparatively to plants in general, are both their low Na/K ratios and high iodine levels
[4]. In fact, it is well accepeted that low Na/K ratios are an important aspect for good maintenance of cardiovascular health
[54][65]. Therefore, according to the World Health Organization (WHO), the recommended value for this should be close to one, so consumption of food products with this proportion or below should be considered for healthy cardiovascular purposes
[55][66]. In fact, several studies point to a Na/K ratio ranging between 0.3 and 1.5 in brown seaweeds, with particular interest for
Laminaria spp. (0.3–0.4) from Spain origins, wich are significantly lower than diverse food products, such as cheddar cheese (8.7), olives (43.6), and sausages (4.9)
[4][56][57][58][59][4,67,68,69,70]. Additionally,
Phaeophyceae seaweeds, due to the rich composition in alginates and sulphated polysaccharides coupled with the presence of haloperoxidases in the cell walls, allow the accumulation of iodine to more than 30,000 times over its concentration in the surrounding environment which is even higher than any edible plant
[60][71]. The major contents of iodine were documented for
L. digitata,
A. nodosum,
H. elogata and
U. pinnatifida exhibiting concentrations of 70, 18.2, 10.7 and 3.9 mg/100 g wet weight, respectively
[61][72]. Moreover, other studies also highlight the particular affinity of Laminarales to accumulate iodine, particularly
L. digitata, in which values are known to reach 9014 and 8122 mg/kg DW, in spring and autumn, respectively
[23].
3. Use of Brown Macroalgae as Food Ingredient
Being considered as a rich and balanced source of nutrients and bioactive compounds, consumers and food industries have a growing interest to introduce macroalgae, including
Phaeophytae, into the dietary habits of the western countries, with new products already being launched in the markets at high rates in Europe. The usage of brown species as food ingredients has, however, to overcome huge challenges, that go from the guarantee of enough biomass to sustain the market development, to the gain of consistent knowledge of their physicochemical features, as well as understanding the extension of their impact when used as ingredients in foods. This section highlights some of the developed foods in the field of seaweed-fortified products, categorized by the respective incorporated algae species, considering the authorized seaweeds for human consumption in France/Europe
[14], and finalising with the influence this incorporation has on the foods’ chemical, functional and structural behaviour.
3.1. Fucus vesiculosus
F. vesiculosus has found application as a functional ingredient in many different food matrices, mostly as a source of phlorotannins and antioxidant compounds, aiming to prevent food spoilage resultant from oxidative deterioration (
Table 1). Fish and fish-derived products are one of the main matrices where several studies with this seaweed have been conducted. In this context, Dellarosa et al.
[62][73] reported that neither aqueous nor 80% ethanol extracts from
F. vesiculosus had significant effects on the lipid oxidation of fish cakes enriched with omega-3 polyunsaturated fatty acids, throughout a 28-days refrigerate storage. Nevertheless, the authors showed that no off flavour was detected in any samples tested, with low scores of rancid odour and flavour being registered in the sensory analysis. On the other hand, some studies conducted on cod fish muscle and/or protein indicated that the incorporation of
F. vesiculosus extracts could indeed prevent the lipid peroxidation events and even improve some of their sensorial aspects. In fact, the effects of the incorporation of 1% and 2% of the antioxidant dietary fibre extracted from
F. vesiculosus into minced horse mackerel revealed a significant reduction of the fish mince lipid oxidation throughout the 5 months of storage at –20 °C. These factors reduced the total drip after thawing and cooking the horse mackerel mince up to 3 months of frozen storage, a fact that could be due to the water holding capacities of the fibre. Furthermore, although the addition of 2% (but not 1%) of antioxidant dietary fibre caused changes in the fish mince flavour compared to the control, these were actually considered positive by the sensory panellists
[63][74].
Table 1. Selected studies reporting the effects of the incorporation of F. vesiculosus or isolates as ingredients in different food matrices.
| Functional Food |
Functional Ingredient |
Results |
Ref. |
| Fish cakes |
F. vesiculosus extracts:
100% H2O, 80% EtOH |
No off-flavours and lower rancid odour and flavour
None of the extracts had influence on lipid oxidation nor quality of the products |
[62][73 |
Table 2). One of the most exploited attributes of this seaweed species is perhaps its wealthy mineral composition, which makes
H. elongata a good candidate to be used as a salt replacer, contributing to the reduction of salt consumption and related health complications typical of western high-NaCl diets. It also increases the consumption of other elements, such as calcium potassium or iodine, which are usually lacking or below recommended levels in regular diets
[4].
Table 2. Selected studies reporting the effects of the incorporation of H. elongata or isolates as ingredients in different food matrices.
| Functional Food |
Functional Ingredient |
Results |
Ref. |
| Poultry steaks |
3% dry matter H. elongata | ] |
| ↑ Purge loss slightly |
| ↓ Cooking loss
↑ Levels of total viable counts, lactic acid bacteria, tyramine and spermidine
No important changes observed during chilled storage
Positive overall acceptance by a sensory panel |
[79][90] |
Cod muscle and protein isolates |
F. vesiculosus 80% EtOH
extract and further fractions (EtOAc + Sephadex LH-20) |
↓ Lipid oxidation in both fish muscle and protein isolates
300 mg/kg of the oligomeric phlorotannin fractions exhibited an effect comparable to that of 100 mg/kg propyl gallate |
[64][75] |
| Pork gel/emulsion systems |
2.5% and 5% dry matter H. elongata |
↑ Water and fat binding properties
↑ Hardness and chewiness of cooked products
↓ Springiness and cohesiveness |
[1] |
Cod mince |
EtOAc fraction of
F. vesiculosus 80%
EtOH extract |
↓ Lipid oxidation in fish muscle |
[65][76] |
| Low-salt pork emulsion systems |
5.6% dry matter H. elongata |
↑ Content of n-3 PUFA
↓ n-6/n-3 PUFA ratio
↓ Thrombogenic index
↑ Concentrations of K, Ca, Mg and Mn |
[80][91] |
Cod protein hydrolysates |
EtOAc fraction of
F. vesiculosus 80%
EtOH extract |
↓ Lipid hydroperoxide and TBARS formation during protein hydrolyzation
↑ Antioxidant activity of the final protein hydrolysates |
[66][78] |
| Cod protein hydrolysates |
EtOAc fraction of
F. vesiculosus 80%
EtOH extract |
↓ Lipid oxidation during protein hydrolysates freeze drying
↑ Antioxidant activity of the final protein hydrolysates
Improved sensorial aspects (bitter, soap, fish oil and rancidity taste) |
[67][77] |
| Minced horse mackerel |
F. vesiculosus antioxidant dietary fibre |
↓ Lipid oxidation during 5 months of storage at −20 °C
↓ Total dripping after thawing and cooking after up to 3 months of frozen storage
Improved fish mince flavour |
[63][74] |
| Granola bars enriched with fish oil emulsion |
F. vesiculosus 100% H2O, 70% acetone and 80% EtOH extracts |
↓ Oxidation products after storage
↓ Iron-lipid interactions
Acetone and EtOH extracts provided additional lipid oxidation protection
↑ Phenolic content, radical scavenging activity and interfacial affinity of phenolic compounds
Possible tocopherol regeneration |
[68 |
,101]. Other effects resultant from
H. elongata fortification in these matrices included the reduced cooking loss and increase in the Kramer shear force in restructured poultry meat
[79][90]; increased water and oil retention in pork meat batter
[81][92]; increased dietary fibre content in frankfurters
[90][101]; and increased phenolic content and antioxidant activity in meat emulsions
[80][91]. Overall, these products were well-accepted in the sensory analysis, with exception of frankfurters that were reported unpleasant mainly due to the increase of the dryness feeling and seaweed-like taste.
Cox and Abu-Ghannam
[85][96] also reported that
H. elongata-fortified beef patties (10–40%
w/
w) were very well accepted in the sensory analysis, particularly those with 40% of seaweed, getting even better scores than the control samples. This was mainly due to the improvements on the samples’ texture and overall mouthfeel, which resulted from the decrease in the cooking loss (associated to the incremented fibre content) and the increase in tenderness for more than 50%. Furthermore, a significant enhancement of the phenolic content and antioxidant activity (in a dose-dependent manner), as well as a lowered microbiological count and lipid oxidation before the chilling stage and after 30 days of storage, were observed in all patties containing seaweed. In fact, at the end of the experiment, the samples containing above 20% of
H. elongata, showed no bacterial growth at all, as well as considerably low levels of the lipid oxidation marker.
In vivo studies on rat models revealed that the introduction of restructured pork meat enriched with 5%
H. elongata (RPS) in the animals’ hypercholesterolemic diet significantly lowered the serum cholesterol levels that were augmented in the group under a non-RPS supplemented hypercholesterolemic diet. Moreover, a significant increase in SOD and GPx, alongside with a decrease of glutathione reductase (GR) expressions, were observed in both groups under hypercholesterolemic and regular RPS-supplemented diets, although increased glutathione reductase activity was also verified. Interestingly, the combined cholesterol and seaweed diet predisposed an increase in the expression of GR, SOD and liver cytochrome P450 7A1 (CYP7A1), i.e., a gene that encodes for the enzyme responsible for the elimination of cholesterol through the production of bile acids, but a decrease in the expression of CAT and GPx, suggesting a possible blocking effect of the hypercholesterolemic agent induced by seaweed incorporation
[82][93]. In a similar study, rats under RPS-supplemented hypercholesterolemic diets, not only exhibited lower plasma cholesterol levels but also lower liver apoptosis markers, namely cellular cycle DNA, caspase-3 and cytochrome c
[91][102]. Supporting these results, González-Torres et al.
[83][94] confirmed that the administration of
H. elongata-fortified restructured pork meat (at 5%) to rats under cholesterol-rich diets, partially blocked the hypercholesterolemic effect of the dietary pattern while changing the lipogenic/lipolytic enzyme expression (decreasing hormone-sensitive lipase and fatty acid synthase while increasing acetyl CoA carboxylase expressions compared with subjects under hypercholesterolemic diet) and reducing the wasting effect of hypercholesterolemia on adipose tissue in rats.
Apart from meat products,
H. elongata powder has also been used to enrich breadsticks in order to enhance their nutritional properties. From the 10 formulations tested (with seaweed concentrations of 2.63 to 17.07%
w/
w), the highest was reported to have the most significant influence on the chemical properties of breadsticks. Furthermore, this sample also had higher levels of total dietary fibre, while the total phenolic content and antiradical activity were maximized at 138.25 mg GAE/100 g dry basis and 61.01%, respectively, maintaining an acceptable edible texture and colour of the samples. Therefore, since no significant difference was seen between the control and seaweed enriched breadsticks in terms of sensory analysis, this product could have great acceptability, especially to non-seaweed consumers
[86][97]. The augmented phenolic content as well as the enhanced antioxidant activity were also described on functional breads developed with 8% of
H. elongata flour
[87][98]. On the other hand, an attempt to supplement yogurt and quark with dehydrated
H. elongata (0.25–1%
w/
w) turned out to negatively affect almost all the sensory parameters analysed, which makes this seaweed not very suitable for application in these two dairies, at least in these conditions
[88][99].
3.3. Undaria pinnatifida
Similar to
H. elongata, the applications of
U. pinnatifida as functional ingredients have mostly been reported in meat and meat-derived products (
Table 3). For instance, the incorporation of
U. pinnatifida (1–4%) into pork beef patties increased their ash content as well as their juiciness due to the lower cooking losses compared to the control
[92][103]. In a similar approach, the reformulation of low-salt (0.5%) and low-fat (<10%) beef patties by the addition of 3% of
U. pinnatifida and partial or total replacement of pork backfat with olive oil-in-water emulsion, significantly affected the frozen storage characteristics of the products. This presented enhancements in terms of technological, sensory and nutritional properties, as well as improvements in their physiological benefits. These reformulated patties demonstrated less thawing and cooking losses, and were texturally softer than the samples without seaweed, most likely due to the microstructural changes caused by the formation of alginate chains.
Table 3. Selected studies reporting the effects of the incorporation of U. pinnatifida or isolates as ingredients in different food matrices.
| Functional Food |
Functional Ingredient |
Results |
Ref. |
| Beef patties |
3% dry matter U. pinnatifida |
↑ Binding properties and cooking retention values of, fat, fatty acids and ash
Replacement of animal fat with olive-in-water emulsion and/or seaweed was reportedly healthier.
↓ Thawing and↑ softer texture
Changes on the microstructure due to formation of alginate chains
Overall acceptable products and fit for consumption |
[93][94][104,105] |
| Chicken breast |
200 mg/kg U. pinnatifida |
↑ Redness and yellowness
↓ Lipid oxidation in chilling storage and after cooking
Overall appearance and shelf-life were enhanced |
[95][106] |
| Pork gel/emulsion systems |
2.5% and 5% dry matter U. pinnatifida |
↑ Water and fat binding properties
↑ Hardness and chewiness of cooked products
↓ Springiness and cohesiveness |
| ] |
| [ |
| 79 |
| ] |
| Granola bars enriched with fish oil emulsion |
F. vesiculosus 100% H2O, 70% acetone and 80% EtOH extracts |
↓ Lipid oxidation during storage
↑ Effectiveness for lower concentrations of EtOH and acetone extracts
↑ Phenolic content, radical scavenging activity and interfacial affinity of phenolic compounds
Possible tocopherol regeneration |
[69][80] |
| Fish-oil-enriched milk and mayonnaise |
F. vesiculosus: EtOAc fraction from an 80% EtOH extract, 100% H2O |
↑ Lipid stability and ↓ oxidation of EPA and DHA and subsequent secondary degradation products in both foods—H2O extract at 2.0 g/100 g exerted higher inhibitory effects on mayonnaise’s peroxide formation. |
[70][81] |
| Fish-oil-enriched mayonnaise |
F. vesiculosus 100%
H2O, 70% acetone, and 80% EtOH extracts |
Dose-dependent inhibition of lipid oxidation exhibited by EtOH and acetone extracts
H2O extract increased peroxide formation |
[71][82] |
| Pork liver pâté |
F. vesiculosus commercial extract |
Decrease in lightness values after storage
Redness and yellowness maintained after storage
Protection against oxidation comparable to BHT samples
↓ Total volatile compounds |
[72][83] |
| Pork patties |
F. vesiculosus 50% EtOH extracts |
↓ TBARS slightly
Did not improve colour, surface discoloration or odour attributes
No significant differences between seaweed and control samples in sensory analysis |
|
| Milk |
F. vesiculosus 60% EtOH extracts |
↑ Milk lipid stability and shelf-life characteristics
Appearance of greenish colour and fishy taste
Overall sensory attributes were worsened |
[73][84] |
| Yoghurts |
F. vesiculosus 60% EtOH extracts |
No influence on chemical and microbiological characteristics
↑ Yogurts lipid stability and shelf-life characteristics
Overall sensory attributes were worsened |
[74][85] |
| Pasteurized apple beverage |
F. vesiculosus fucoidan extract |
Dose-, time- and temperature-dependent bacteriostatic and bactericidal effects against L. monocytogenes and S. typhimurium
S. typhimurium showed higher sensitivity to the extract |
[75][86] |
| Bread |
F. vesiculosus powder |
↑ Dough viscosity and wheat dough consistency
↓ Porosity
↑ Density, crumb firmness and green colour of crust
4% seaweed powder was considered optimal |
[76][87] |