Nuts are indehiscent dry fruits with one seed and a thick, hard pericarp. The presence of nuts in diets has notably increased due to their composition, and the presence of antioxidants and their unsaturated fatty acid profile has led to a considerable increase in their consumption. The volatile profile of nuts is important from different points of view. It affects consumer’s selection, influences raw material selection for the production of composite foods, dictates variety selection in breeding programs, and, from a quality perspective, its changes can indicate food degradation or alteration.
- nut
- volatile
- aroma
- flavor
- key odorants
- analytical methods
- solid-phase microextraction
1. Introduction
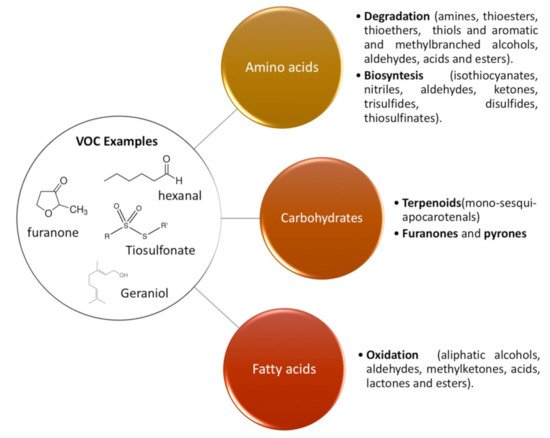
2. Main Volatile Organic Compounds (VOCs) Present in Nuts
3. Analysis of VOCs Present in Nuts
Equilibration Conditions | Extraction Conditions | |||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Type of Nut | Sample Amount (g) | Fiber | ||||||||||||||||
Nut | Main VOCs | Ref. | ||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Agitation | Time (min) | Temperature (°C) | Time (min) | Temperature (°C) | Quantification | Column | Ref. | |||||||||||||||||||||||||||||
Almond | 5.0 * | |||||||||||||||||||||||||||||||||||
Almond | 1-Hexanol, 3-methyl-1-butanol, nonanal, 2-methyl-1-propanol, 1-propanol | 1-cm 50/30 µm DVB/CAR/PDMS | No |
[ | 40 | 24 | ] | 30 | 24 | (1) | [37] | DB-Wax | (30 m × 0.25 mm × 0.25 μm) |
[15] |
[38] |
|||||||||||||||||||||
Almond | 5.0 * | |||||||||||||||||||||||||||||||||||
Benzaldehyde, hexanal, 1,2-propanediol, 1-chloro-2-propanol, 3-methyl-1-butanol, pentanal, 2-heptanone, 1-hexanol. | 1-cm 50/30 µm DVB/CAR/PDMS |
[15] |
[ | Yes | 38] | 45 | ||||||||||||||||||||||||||||||
Hexanal, 3-methyl-1-butanol, benzaldehyde, heptanal, nonanal, 1-octanol, 2-octanone |
[18] |
[22] |
5. Volatile Profile of Nuts after Thermal Treatments
Thermal Processing | Nut | Processing Conditions | Main VOCs | Ref. | |||||||||||||||||||||||||||||||||||||||||||
|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|---|
Hot-air roasting | Hazelnut | 34, 18, 13 min at 130, 140 and 150 °C | 2,3-pentanedione, 2-acetyl-1-pyrroline, dimethyl sulphide, 2-furfurylthiol, 3-methylbutanal, 2-nonenal, 2-decenal, hydroxy-2,5-dimethyl-3(2H)-furanone |
[50] |
[59] |
||||||||||||||||||||||||||||||||||||||||||
20, 25 and 30 min | at 160 °C | 2-methylpropanal, 2-methylbutanal, 3-methylbutanal, 2,5-methylpyrazine |
[51] | 40 | 45 | [ | 40 | (3) | ] | DB-Wax | (30 m × 0.25 mm × 0.25 μm) |
[16] |
[21] |
||||||||||||||||||||||||||||||||||
Almond | 0.250 * | 1-cm 50/30 µm DVB/CAR/PDMS | No | 15 | 25 | 30 | 25 | (2) | DB-Wax | ||||||||||||||||||||||||||||||||||||||
40 min at 140 °C | 2-methylbutanal, 3-methylbutanal, 2,5-methylpyrazine, furfuryl alcohol, 2-methylpropanal, ehtyl acetate, 2,3-pentanedione, 2-methylpyrazine, 2,5-methylpyrazine, furfural, 1-hydroxy-2-propanone |
[21] |
[ | (30 m × 0.25 mm × 0.25 μm) | 43] |
[17] |
[37] |
||||||||||||||||||||||||||||||||||||||||
Almond | Chestnut | 5.0 * | |||||||||||||||||||||||||||||||||||||||||||||
ϒ-terpinene, phenylaldehyde, hexanal, furfural, α-terpinene | 1-cm 50/30 µm DVB/CAR/PDMS | Almond | [35] |
[57] | Yes | 45 | 40 | 45 | 40 | ||||||||||||||||||||||||||||||||||||||
5 min at 177 °C | Benzyl alcohol, benzaldehyde, 1-octen-3-ol, toluene, dimethylpyrazine, 1-butanol, hexanal | (3) | DB-Wax | (30 m × 0.25 mm × 0.25 μm) |
[52] |
[ |
[10] |
[12] |
|||||||||||||||||||||||||||||||||||||||
] | Almond | 3.0 * | 1-cm 50/30 µm DVB/CAR/PDMS | No | Hazelnut | 10 | 40 | α-tujone, β-tujone, 2-pentanone, acetic acid, 3-methyl-2- butanol, n-decane | 30 |
[21 | |||||||||||||||||||||||||||||||||||||
] |
[43] | 40 | |||||||||||||||||||||||||||||||||||||||||||||
5–10 min at | 170–190 °C | Hexanal, 2-methyl-butanal, 2-methyl-pyrazine, 2,5-dimethyl-pyrazine, furfural |
[53] |
[ | (1) | 62] | TRB-5MS | (30 m × 0.25 mm × 0.25 μm) |
[18] |
[22] |
|||||||||||||||||||||||||||||||||||||
Beechnut, hazelnut, pistachio and walnut | Peanut | 10.0 ** | Hexanoic acid, 2-ethyl-1-hexanol, 1-hexanol, pentanal, hexanal, palmitic acid, 2-ethyl-5-methylpyrazine, heptanal | 1-cm 50/30 µm DVB/CAR/PDMS |
[24] |
[44] | Yes | 60 | 25 | 60 | 25 | No | RTx-5 (60 m × 0.25 mm × 0.25 μm) |
[19] |
[30] |
||||||||||||||||||||||||||||||||
10 min at 190 °C | 2,5-dimethyl-pyrazine, trimethylpyrazine |
[39] |
[ |
Hazelnut | 0.1 * | 1-cm 75 µm | CAR/PDMS | No | 10 | 60 | 10 | 60 | (3) | DB-Wax | (30 m × 0.25 mm × 0.5 μm) |
[20] |
[15] |
||||||||||||||||||||||||||||||
2-Propanone, α-pinene, benzene, α-terpinolene, hexanal, d-limonene |
[36] |
[29] |
Hazelnut | 1.5 * | 2-cm 50/30 µm DVB/CAR/PDMS | No | 20 | 50 | 20 | 50 | (3) º | MEGA-WAX™ (30 m × 0.20 mm × 0.20 μm) |
[21] |
[43] |
|||||||||||||||||||||||||||||||||
Peanut | 5.0 * | PDMS/DVB | No | 30 | 60 | 15 | 60 | (3) | DB-5 |
[22] |
[35] |
||||||||||||||||||||||||||||||||||||
Peanut | 5.0 * | 50/30 µm | DVB/CAR/PDMS | Yes | 1440 | 25 | 20 | 21 | |||||||||||||||||||||||||||||||||||||||
] | |||||||||||||||||||||||||||||||||||||||||||||||
33 min at 138 °C | Hexanal, benzeneacetaldehyde, 2,5-dimethyl-pyrazine, nonanal |
[15] |
[38] |
Toluene, α-limonene, γ-terpinene, p-cymene, nonanal, β, pinene, hexanoic acid |
[25] |
[39] |
|||||||||||||||||||||||||||||||||||||||||
28 and 38 min at 138 °C | 2-methylbutanal, 3-methylbutanal, hexanal, benzaldehyde, furfural, 2-phenyl acetaldehyde |
[15] |
[38] |
2,5-Dimethylpyrazine, nonanal, hexanal, 2-ethyl-5-methylpyrazine, octanal, 2,5-dimethyl-3-ethylpyrazine |
[28 | ||||||||||||||||||||||||||||||||||||||||||
] |
[33] |
||||||||||||||||||||||||||||||||||||||||||||||
28, 33 and 38 min | at 138 °C | 2-methylbutanal, 3-methylbutanal, hexanal, benzaldehyde, furfural, 2-phenyl acetaldehyde |
[54] |
[63] |
(1) | SUPELCOWAX™ 10 (30 m, 0.25 mm, 0.25 mm) |
[23] |
[25] |
|||||||||||||||||||||||||||||||||||||||
Hexanal, benzaldehyde, benzenacetaldehyde, 2,5-dimethylpyrazine, 2-heptenal, 2-ethyl-5-methylpyrazine, trimethylpyrazine, 3-ethyl-2,5-dimethylpyrazine |
[27] |
[ |
Peanut | 3.0 ** | |||||||||||||||||||||||||||||||||||||||||||
Chestnut | 26] |
||||||||||||||||||||||||||||||||||||||||||||||
25 min at 200 °C | Hexanal, butylacetate, ethylbenzene, 2-hydroxy-2-cyclopenten-1-one |
[54] |
[63] |
Pistachio | 9-Octadecenoic acid, α-pinene, 1-methyl-1H-pyrrole, α-terpinolene, limonene, dimethyl-2H-pyran-2-one, 2-octenal, 2-hexenal | 1-cm 65 µm | PDMS/DVB | Yes | 10 | 50 | 40 | 50 | (1) | DB-Wax | (30 m × 0.25 mm × 0.25 μm) |
[24 |
|||||||||||||||||||||||||||||||
Pistachio with and without salt |
[29] |
[34] | ] | [ | 44] |
||||||||||||||||||||||||||||||||||||||||||
Peanut | 5.0 ** | 2-cm 50/30 µm DVB/CAR/PDMS | |||||||||||||||||||||||||||||||||||||||||||||
90 min at 120 °C | α-pinene, limonene, 3-carene |
[55] |
[64] |
No | 30 | 80 | 10 | 80 | (1) | RTX-5MS |
α-Pinene, α-terpinolene, 1H-pyrrole, ethyl-alcohol, limonene, hexane |
[30(30 m × 0.25 mm × 0.25 μm) | |||||||||||||||||||||||||||||||||||
[ |
Cashew | ] | [ | 39] |
|||||||||||||||||||||||||||||||||||||||||||
] |
[28] |
||||||||||||||||||||||||||||||||||||||||||||||
3 and 9 min | at 143 °C | Methylbutanal, hexanal, acetaldehyde, heptane, ethanol, pentane, acetone |
[56] |
[65] |
Peanut | 5.0 ** | α-Pinene, β-pinene, 2-ethyl-1-hexanol, α-terpineol, camphene, hexanoic acid | ||||||||||||||||||||||||||||||||||||||||
Microwave roasting | 1-cm 50/30 µm DVB/CAR/PDMS |
[2] | Yes |
||||||||||||||||||||||||||||||||||||||||||||
Almond | 120 V for 2 min | Benzyl alcohol, methional, benzaldehyde, dimethylpyrazine, nonanal, undecane, 1-octen-3-ol, 1,4-butyrolactone | 30 |
[52] | 50 | [ | 30 | 50 | No | DB-17MS | (60 m × 0.25 mm × 0.25 μm) |
[26] |
[32] |
||||||||||||||||||||||||||||||||||
Peanut | Walnut | 0.2 * | Hexanal, hexanoic acid, 1-pentanol, 2-octenal, pentanal, 2-pentylfuran, propanoic acid |
[33] |
[45] |
||||||||||||||||||||||||||||||||||||||||||
] | 2-cm | DVB/CAR/PDMS | Yes | ||||||||||||||||||||||||||||||||||||||||||||
Pistachio | 480 or 640 W for 2, 3 and 4 min | α-pinene, limonene, nonanal | 8 h |
[57] |
[66] | 20 | 50 | 60 | (1) | dB5-MS semi-polar (60 m × 0.32 mm × 1 μm) |
[27] |
[26] |
|||||||||||||||||||||||||||||||||||
Peanut | 5.0 ** | ||||||||||||||||||||||||||||||||||||||||||||||
Hot air-assisted radio frequency | 2-Octenal, hexanoic acid, hexanal, 2-decenal, 1-octen-3-ol, nonanal | 1-cm 65 µm |
Almond | DVB/CAR/PDMS |
[14] |
[20] | 15 min at 120–130 °C | No | 2,5-dimethyl-pyrazine, toluene, hexanal and heptane | 20 | 80 | 60 | 80 |
[ | No | 58] | HP-5 | (30 m × 0.25 mm × 0.25 μm) | [67] |
[28] |
[33] |
||||||||||||||||||||||||||
Pistachio | 15.0 * | 50/30 µm | DVB/CAR/PDMS | Yes | 15 | 50 | 120 | 50 | No | HP-5 | (30 m × 0.32 mm × 0.25 μm) |
[13] |
[19] |
||||||||||||||||||||||||||||||||||
Deep-frying | Almond | 5 min at 135 °C | Benzyl alcohol, methional, benzaldehyde, 1-butanol, 1-octen-3-ol |
[52] |
[61] |
Pistachio | 8.0 * | 50/30 µm | DVB/CAR/PDMS | No | - | - | 60 | 83 | No | HP-5MS (30 m × 0.25 mm × 0.25 μm) |
[29] |
[34] | |||||||||||||||||||||||||||||
10–15 min at 160–200 °C | Hexanal, 2-methyl-butanal, 3-methyl-butanal, 2,5-dimethyl-pyrazine, 1-pentanol |
[53] |
[62] |
Pistachio | 10.0 * | 50/30 µm | DVB/CAR/PDMS | 15 | |||||||||||||||||||||||||||||||||||||||
] | 50 | Chestnut | 120 | 15 min at 240 °C | Hexanal, octanal, nonanal, furfural, 3-heptanone, 4-hydroxy-2-butanone |
[59] | 50 | (1) | Equity-5 (30 m × 0.25 mm × 0.25 μm) |
[30] |
[28] |
||||||||||||||||||||||||||||||||||||
[ |
Pistachio | 24.5 * | PDMS-DVB | No | 30 | 30 | 20 | 30 | No | Agilent DB-1 | (60 m × 0.320 mm × 0.25 μm) |
[31] |
|||||||||||||||||||||||||||||||||||
Pistachio | 1.5 ** | 2-cm 50/30 µm DVB/CAR/PDMS | Yes | - | - | 30 | 40 | (1) | DB-Wax | (30 m × 0.25 mm × 0.25 μm) |
[2] |
||||||||||||||||||||||||||||||||||||
Walnut | 0.5 * | 50/30 µm | DVB/CAR/PDMS | Yes | 15 | 50 | 30 | 60 | (1) | RTX-5MS | (30 m × 0.25 mm × 0.25 μm) |
[32] |
[27] |
||||||||||||||||||||||||||||||||||
Walnut | 3.0 ** (mL) | 1-cm 50/30 µm DVB/CAR/PDMS | No | 10 | 50 | 30 | 50 | No | HP-INNOWAX | (30 m × 0.25 mm × 0.25 μm) |
[33] |
[45] |
|||||||||||||||||||||||||||||||||||
Walnut | 1.0 ** | 65 µm | PDMS/DVB | No | - | - | 30 | 50 | No | CP-Wax52CB | (30 m × 0.25 mm × 0.25 μm) |
[14] |
[20] |
||||||||||||||||||||||||||||||||||
Pecan | 2.0 * | 50/30 µm | DVB/CAR/PDMS | Yes | 30 | 25 | 30 | 65 | (1) | HP-5 | (30 m × 0.25 mm × 0.25 μm) |
[34] |
[36] |
Sample pre-treatment: * Only grinding ** Grinding and oil extraction. Quantification: 1: Semi-quantification with IS, 2: (1) corrected by sample amount, 3: quantification. º: IS was employed. DVB: Divinylbenzene, CAR: Carboxen, PDMS: Polydimethylsiloxane.
