Inert microspheres, labeled with several radionuclides, have been developed during the last two decades for the intra-arterial treatment of liver tumors, generally called Selective Intrahepatic radiotherapy (SIRT). The aim is to embolize microspheres into the hepatic capillaries, accessible through the hepatic artery, to deliver high levels of local radiation to primary (such as hepatocarcinoma, HCC) or secondary (metastases from several primary cancers, e.g., colorectal, melanoma, neuro-endocrine tumors) liver tumors. Several types of microspheres were designed as medical devices, using different vehicles (glass, resin, poly-lactic acid) and labeled with different radionuclides, 90Y and 166Ho.
1. Introduction
Liver radioembolization (RE) is commonly used for the treatment of hepatocellular carcinoma and secondary liver malignancies. This treatment is performed by injection of radioactive microspheres in the liver artery after transarterial catheterization. Radioactive microspheres are trapped in the microvasculature (arterioles) of tumors and of the liver parenchyma. Unlike the liver parenchyma, where blood supply is almost obtained by the portal vein, liver tumors are preferentially vascularized by the liver arteries. This preferential perfusion allows us to achieve good targeting of hypervascularized tumors with a limited radiation of the non-tumoral liver [1]. The technique was initially developed using iodine-131 (131I) Lipiodol, a radiolabeled ethiodized oil [2]. Thereafter, radiolabeled microspheres have emerged using Yttrium-90 (90Y) and holmium-166 (166Ho). These radionuclides emit beta radiations of high energy, resulting in a high delivery of energy to the tumors (absorbed doses) in the range of 100 to 1000 Gy [3]. In comparison, the total tumor dose is limited to a maximum of 70 Gy in external beam radiotherapy to avoid liver damage [4].
Three types of microspheres are commercially available: 90Y-resin microspheres (Sir-Spheres®, Sirtex Medical Ltd., Sydney, Australia), 90Y-glass microspheres (Therasphere®, Boston Scientific, Boston, MA, USA) and 166Ho-poly-L-lactic acid (PLLA) microspheres (QuiremSpheres®, Quirem Medical B.V., Deventer, The Netherlands). Other microspheres have been used but did not reach the level of marketing authorization in Europe, e.g., 188Re-microspheres [5].
2. Preparations and Labeling (Sir-Spheres
®
, Therasphere
®
, QuiremSpheres
®
)
SIR-Spheres® are cation exchange resin microspheres labeled with 90Y phosphate. The resin is supplied as symmetrical microspheres ranging from 30 to 50 µm in diameter, made of sulphuric acid groups attached to a styrene divinylbenzene copolymer resin (Aminex 50W-X4, Bio-Rad, Hercules, CA, USA). First, stable yttrium oxide (III) (89Y) is activated in a neutron beam to its radioisotope 90Y according to the nuclear reaction 89Y (n,γ) 90Y. The radioactive yttria (Y2O3) is then dissolved in sulphuric acid. 90Y is adsorbed onto the resin matrix by adding newly formed 90Y sulphate solution to the aqueous slurry of microspheres. To immobilize and stably incorporate the 90Y into the lattice, the radionuclide is precipitated as an insoluble phosphate salt by adding a tri-sodium phosphate solution. Finally, the microspheres are washed with a phosphate buffer solution and resuspended with water for injection [9][6].
The second commercially available product labeled with
90
Y is TheraSphere
®
. It consists of radioactive yttrium oxide-aluminosilicate glass microspheres. Inactive yttria is mixed with aluminum and silicone oxide. The resulting mixture is melted and stirred in a furnace at 1500 °C. The melt is then removed from the heat and rapidly quenched to a glass. The resultant glass frit is crushed to fine powder and filtered through a sieve. The powder is spheroidized by introducing it into a flame: the particles are melted and formed into spherical liquid drops by surface tension. The newly formed microspheres are quickly cooled and screened. The 20- to 30-µm fraction is rinsed and dried. The
89
Y embedded glass microspheres are eventually activated by neutron bombardment to the radioisotope
QuiremSpheres
®
are poly-L-lactic acid (PLLA) microspheres labeled with
166
Ho. The microspheres are synthesized by the solvent-evaporation technique. First, non-radioactive
165
Ho is complexed with acetylacetonate. Complex
165
Ho-acetylacetonate is then incorporated into the PLLA matrix by adding
165
Ho-actylacetonate and poly-L-lactic acid to a continuously stirred chloroform solution. The resulting solution is then added to an aqueous polyvinyl alcohol solution and stirred until complete evaporation of the chloroform. The newly formed
165
Ho-loaded microspheres are collected by centrifugation, washed and fractionated according to their size. The 20- to 50-μm fraction is dried and packed in polyethylene vials. Finally,
165
Ho is activated to
166
Ho in a nuclear reactor by neutron irradiation with a thermal neutron flux of 5 × 10
12
cm
−2
s
−1
(
165
Ho + n→
166Ho, cross section 64 barn) [14,15,16]. The final product contains 19% (w/w) holmium (essentially stable which is of interest in view of the paramagnetic properties of Ho, cross section 64 barn) [10][11][12]. The final product contains 19% (w/w) holmium (essentially stable which is of interest in view of the paramagnetic properties of 165
Ho, see paragraph 2) and a maximum
166Ho specific activity per microsphere of 450 Bq [17]. QuiremSpheres Ho specific activity per microsphere of 450 Bq [13]. QuiremSpheres ® is supplied as a ready-to-use, tailored dose: the dispatched activity is patient-specific and matches the activity at treatment time as ordered by the customer. There is no need for patient-dose preparation on-site [18]. is supplied as a ready-to-use, tailored dose: the dispatched activity is patient-specific and matches the activity at treatment time as ordered by the customer. There is no need for patient-dose preparation on-site [14].
3. Radionuclide Properties and Clinical Applications
Table 1 summarizes the main physical characteristics of 90Y and 166Ho radionuclides.
Physical characteristics of radionuclides.
], Bult W. [31] and Gupta et al. using iron labeled glass microspheres [32].
Resin and PLLA microspheres have a density comparable to blood (1.06 g/mL). Glass microspheres have a higher density (3.3 g/mL) three-fold more than blood. Regarding the specific activity per microsphere and the number of microspheres injected, resin, glass and PLLA microspheres differ highly (
was made by rescaling graphs derived from the analyses of Bakker et al. [19], Bult W. [20] and Gupta et al. using iron labeled glass microspheres [21].
Resin and PLLA microspheres have a density comparable to blood (1.06 g/mL). Glass microspheres have a higher density (3.3 g/mL) three-fold more than blood. Regarding the specific activity per microsphere and the number of microspheres injected, resin, glass and PLLA microspheres differ highly (
Physical characteristics of radioactive microspheres.
| Microspheres |
Diameter (Mean) |
Density |
Approximative Number of Micro-Spheres Per GBq * |
Activity Per Microsphere |
| 90 | Y-Resin |
32 μm |
1.6 g/mL |
13 × 10 | 6 |
50 Bq |
| 90 | Y-Glass |
25 μm |
3.3 g/mL |
0.4 × 10 | 6 |
2500 Bq |
| 166 | Ho-PLLA |
30 μm |
1.4 g/mL |
10 × 10 | 6 |
450 Bq |
| Mean: 2.5 mm (Max: 11 mm) |
| 1.77 MeV (49%), 1.86 MeV (50%) |
| Mean: 2.5 mm (Max: 8.7 mm) |
These beta particles generate free radical species in the presence of oxygen inducing DNA breaks and cell killing. Many factors influence the killing efficacy, and particularly the rate of beta particles emission (correlated to half-life) and the number of beta particles (correlated to the radioactive activity) [21]. The biological effect of radiations is demonstrated by the absorbed dose (Gy), defined by the energy (J) deposited per mass of tissue or tumor (kg). This absorbed dose is directly correlated to the cell survival fraction and tumor response [22].
The precise localization of these beta particles in the tumor is also an important factor for tumor response. Indeed, the very short range of beta emission (few millimeters) induce a biologic effect only in the neighborhood of the beta particles and consequently, their distribution in the tumor must be the as homogeneous as possible for killing a maximum of cells and induce a tumor response [23].
These radionuclides have also the ability to be detected by nuclear imaging systems. An accurate detection is important for evaluating the distribution in the targeted tissues and for a quantitative assessment of the dose deposition. Beta particles induce interactions with the surrounding tissues and generate a continuous spectrum of X-ray photons known as bremsstrahlung.
These beta particles generate free radical species in the presence of oxygen inducing DNA breaks and cell killing. Many factors influence the killing efficacy, and particularly the rate of beta particles emission (correlated to half-life) and the number of beta particles (correlated to the radioactive activity) [15]. The biological effect of radiations is demonstrated by the absorbed dose (Gy), defined by the energy (J) deposited per mass of tissue or tumor (kg). This absorbed dose is directly correlated to the cell survival fraction and tumor response [16].
The precise localization of these beta particles in the tumor is also an important factor for tumor response. Indeed, the very short range of beta emission (few millimeters) induce a biologic effect only in the neighborhood of the beta particles and consequently, their distribution in the tumor must be the as homogeneous as possible for killing a maximum of cells and induce a tumor response [17].
These radionuclides have also the ability to be detected by nuclear imaging systems. An accurate detection is important for evaluating the distribution in the targeted tissues and for a quantitative assessment of the dose deposition. Beta particles induce interactions with the surrounding tissues and generate a continuous spectrum of X-ray photons known as bremsstrahlung.
4. Radioactive Microspheres Properties
Ho-poly-L-lactic-acid [PLLA] microspheres differ from their physical characteristics, summarized in
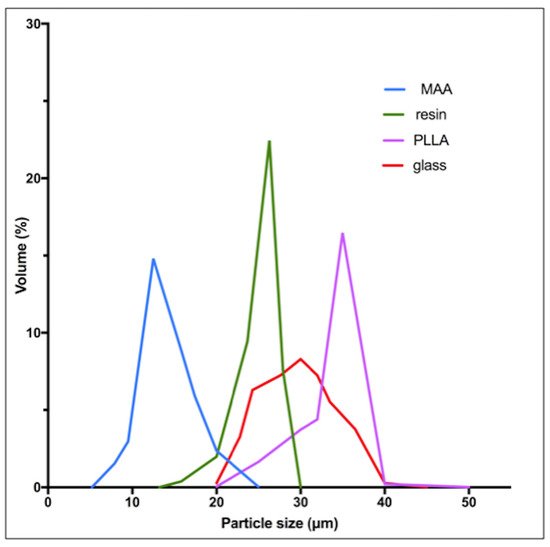
Table 2 [10,29]. Globally, all three devices have a similar diameter around 30 μm, but some differences appear in the size spectrum distribution ( [7][18]. Globally, all three devices have a similar diameter around 30 μm, but some differences appear in the size spectrum distribution ( ). MAA particles have a significantly lower size (mean: 15 μm).
Particle size distribution for resin, glass, PLLA microspheres and MAA particles.
Figure 1 was made by rescaling graphs derived from the analyses of Bakker et al. [30
* On the day of calibration (approximately). PLLA: poly-(L-lactic-acid).
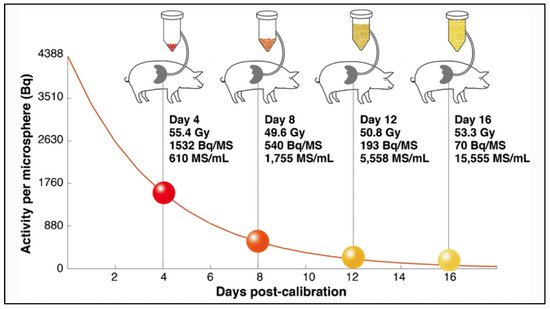
In a very interesting in-vivo study in pigs, Pasciak et al. confirmed in-vivo correlation between the number of injected microspheres and the degree of homogeneity of the absorbed dose at a microscopic level [25]. In this study, four pigs received lobar infusions of Y glass microspheres with an activity reaching a similar target liver dose of 50 Gy. In each pig, the injected glass microspheres differ in specific activity and number (from 1532 Bq/microsphere & 610 microspheres/mL of tissue to 70 Bq/microsphere & 15,555 microspheres/mL of tissue), as illustrated in
Amount of glass microspheres injected in pig livers as a function of time from microsphere calibration (4, 8, 12 or 16 days), resulting in spheres of different specific activity and concentrations. Reprinted from European journal of nuclear medicine and molecular imaging with permission of Springer Nature (Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 816–827).
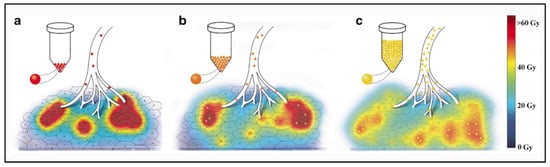
The analysis of the microdosimetry data demonstrated important differences in absorbed doses deposition at a microscopic level. Each pig received the same average dose of 50 Gy, but the distribution at a microscopic level was clearly different, being very inhomogeneous when the number of microspheres was lower. The percentage of the non-target liver receiving at least a potentially toxic dose of 40 Gy was only 24% for microspheres in relatively small number (e.g., 610 microspheres/mL of tissue) and 53% using more numerous microspheres (e.g., 15,555 microspheres/mL of tissue). This effect is well illustrated in
Artistic rendering of microscopic microsphere deposition at day 4 post-calibration (
) and 12 post-calibration (
). Increased homogeneity of absorbed dose is apparent for glass microspheres injected at day 12 post-calibration (e.g., 193 Bq/microsphere, 5558 microspheres/mL). The proportion of the liver receiving a dose higher or equal to 40 Gy (red and yellow colors) was higher for glass microspheres injected at day 12 post-calibration (
). Reprinted from European journal of nuclear medicine and molecular imaging with permission of Springer Nature (Eur. J. Nucl. Med. Mol. Imaging 2020, 47, 816–827).
Using microspheres in limited number induces a more heterogeneous liver distribution, but also permits it to be less toxic, avoiding reaching a toxic dose in a large part of the normal liver. These dosimetric concepts could be extrapolated to any type of microspheres, in order to evaluate to what extent the number of microspheres and their specific activity can influence the efficacy and toxicity of liver radioembolization.
Ho- PLLA microspheres could have an embolic effect because a high number of particles are injected in the liver arterial tree (
). However, this effect may be very limited in particular because they have a very small size, permitting to reach the terminal microvasculature [26].
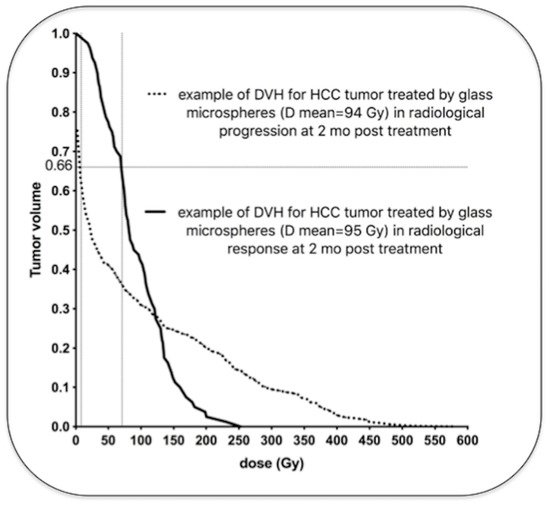
Previous data demonstrated a continuous relationship, a sigmoid correlation, between the tumor absorbed dose and the radiological response [31][36]. HCC patients treated with resin microspheres had better disease control and overall survival when tumor absorbed doses were above 100 Gy. HCC patients treated with glass microspheres also had a better outcome with a tumor-absorbed dose of at least 205 Gy [37][38]. For colorectal metastases, a dose response relationship was also demonstrated with the three types of microspheres. A significant metabolic response was achieved with a tumor cutoff dose of 50–60 Gy with resin microspheres [39][40], 139 Gy using glass microspheres [41] and 90 Gy using PLLA microspheres [42]. Then, to be efficient, the tumor absorbed dose cutoff is approximately twice with glass microspheres as compared to resin microspheres. A recent comparison of patients treated with glass and resin microspheres demonstrated similar outcome using a tumor dose cutoff of 61 Gy with resin microspheres and 118 Gy with glass microspheres [43]. But interestingly, the analysis of the tumor dose at a microscopic level did not demonstrate a two-fold difference between both types of microspheres. Indeed, patients treated with glass or resin microspheres achieved similar outcome with the same minimal dose in most of the tumor volume. A minimum dose of 40 Gy in 66% of the tumor volume was associated with similar PFS and OS with both types of microspheres. The tumor dose distribution can be accurately studied with Y PET/CT using dose volume histogram (DVH).
demonstrates examples of HCC tumors treated with glass microspheres with similar mean absorbed doses, but wide differences in intratumoral dose distributions.
Examples of two HCC tumors treated with glass microspheres with similar mean absorbed doses (94 and 95 Gy) but very different dose distributions (personal data). One tumor (dotted line) received a very low dose in a large part of the tumor volume (more than 5 Gy in 66% of the tumor volume), and was in radiological progression two months later. The other tumor (solid line) received a high dose in a large part of the tumor volume (more than 69 Gy in 66% of the tumor volume) and was in radiological response two months later. Note that the cluster with the highest local dose was found in the non-responding tumor (>500 Gy) because of the major heterogeneous distribution.