Cytokine removal can attenuate dysregulated immune response caused vasoplegia, leading to quicker hemodynamic stabilization and shock reversal. The most frequently used criteria to define shock reversal include normalization of serum lactate (<2.2 mmol/L) coupled with a significant (≥90%) reduction in norepinephrine dose requirements. The current paper has summarized the available data, which indicate the important contribution of early hemoadsorption in achieving rapid hemodynamic stabilization in patients with refractory vasoplegic shock.
- shock
- hemodynamic stabilization
- hemoadsorption
- cytosorb therapy
- hypeinflammation
- decatecholaminization
1. Introduction
Regardless of the initiating insult, vital organ functions, not necessarily affected primarily, fall victim to a dysregulated host response [1]. The cytokine storm originating from the immune over-response determines impairment of the vascular tone and systemic vasodilation, which manifests as hemodynamic instability. In its most serious form as vasoplegic circulatory shock, hemodynamic instability can be life-threatening; consequently, reversing shock as soon as possible is a lifesaving measure of utmost importance to avoid the devastating effects of hypoxemic organ damage [2]. The therapy has the potential to effectively remove cytokines originating from the cytokine storm [3][4], and thus can mitigate systemic hyperinflammation, contribute to early shock reversal, and last but not least, improve clinical outcomes.
Shock is currently defined by the task force from the European Society of Intensive Care Medicine (ESICM) as a “life-threatening, generalized form of acute circulatory failure associated with inadequate oxygen utilization by the cells” [5]. Generally, this means an imbalance between oxygen delivery (DO2) and oxygen consumption (VO2). DO2 depends on cardiac output (CO) and the arterial oxygen content (CaO2), and VO2 depends on mixed venous oxygen content (SvO2):
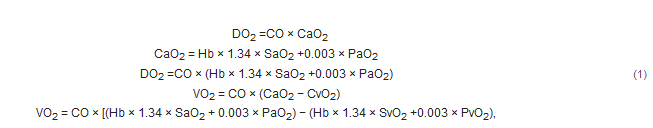
Adequate oxygen supply is paramount for preserving organ viability and is dependent on adequate tissue perfusion. The latter is commonly assessed by mean arterial pressure (MAP), which is mainly determined by vascular tone (systemic vascular resistance—SVR). The relationship between SVR, MAP, central venous pressure (CVP) and CO is described below, based on Ohm’s law:
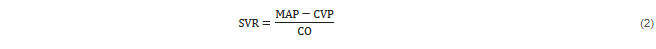

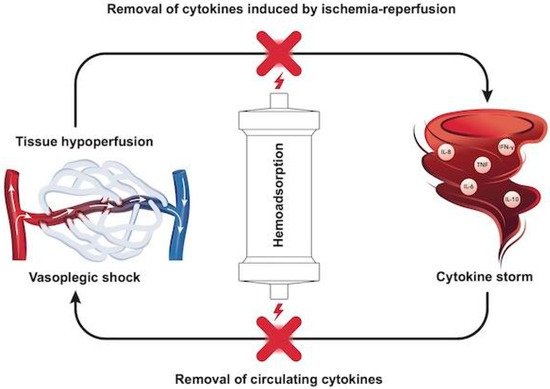
Loss of vascular tone (i.e., sepsis, hyperinflammation) results in low SVR, low MAP and preload deficit (i.e., vasoplegic shock). The different underlying mechanisms of hemodynamic instability also correspond to potential therapeutic options to be targeted, including fluids, inotropes, oxygen supplementation, and vasopressors to increase vascular tone, hence tissue perfusion. The differential diagnosis of hemodynamic instability or shock requires a skilled assessment of the complete clinical picture, which ranges from a simple measurement of vital signs such as heart rate and blood pressure, to complex, advanced hemodynamic monitoring [5]. The connection of inflammatory response–vasoplegia–tissue hypoperfusion–cytokine removal is depicted in Figure 1.
Different types of shock require different management strategies. However, hemodynamic stabilization always represents a main goal due to its role in reestablishing adequate aerobic metabolism in the cells and tissues, and in regaining control over the oxygen debt. Oxygen debt also accumulates during the resuscitation period, suggesting that shorter resuscitation times translate into lesser oxygen debts. Experimental studies suggest that both the severity and duration of hemodynamic instability are associated with poor outcomes [6].
In the past, the clinical and biochemical characteristics of vasoplegic shock were often defined within the domain of “septic shock”. However, similar features are also observed in non-infective inflammatory states, such as in acute necrotizing pancreatitis, after major trauma, major surgery, and in other conditions without an obvious infectious component. Interestingly, very similar observations were made many years before by Sir William Osler in the context of bacterial infections [7], and Hans Janos Selye in the context of stress [8]. Based on our current understanding, the term “hyperinflammatory shock” is preferred over “septic shock”, since it describes the pathophysiology more accurately and is applicable to both infectious and non-infectious etiologies.
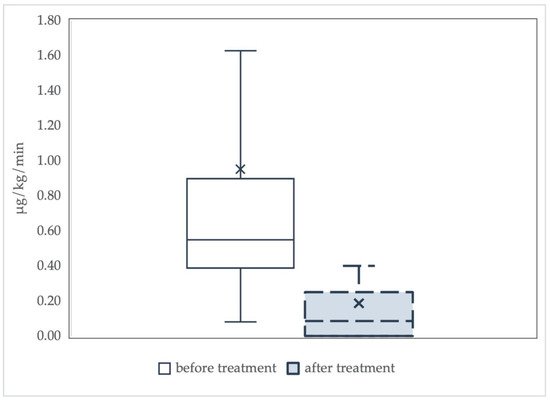
The term “refractory shock” is commonly used to describe the most severe cases of hyperinflammatory shock. Although there is no clear consensus as to the exact definition for refractory shock, it is generally intended as shock persisting for more than 6 h despite initiation of full standard therapy, and is indicated by the following:Elevated lactate levels (>2.9 mmol/L) [9];High norepinephrine (NE) requirements ( >0.3 µg/kg/min)
Up to one-third of patients admitted to the intensive care unit (ICU) are in circulatory shock [10]. As noted already, the expert community now recommends the terms “hyperinflammatory” or “vasoplegic” shock over “septic shock” to better reflect the underlying pathophysiology of a “dysregulated host response” [1][4]. Accordingly, current Surviving Sepsis Guidelines recommend early and aggressive resuscitation within the first hours of the onset of sepsis and septic shock [11]. However, especially in vasoplegic shock, which is characterized by low SVR and consequently low diastolic blood pressure, a fluid challenge alone is often insufficient to improve tissue perfusion [12][13].
Vasopressors exert their effect by either mimicking the effect of the sympathetic nervous system (sympathomimetic amines) or by raising extracellular ionized calcium concentrations (calcium chloride). Sympathomimetic amines can be divided into either catecholamines or non-catecholamines. Commonly used catecholamines with a prominent agonist activity include epinephrine (also known as adrenaline), norepinephrine (noradrenaline) and dopamine. Norepinephrine is recommended as first line treatment of septic shock by the Surviving Sepsis campaign, but the combined use of vasopressors including both vasopressin and norepinephrine is also suggested to limit adrenoceptor desensitization due to sympathetic hyperactivation [14][15].
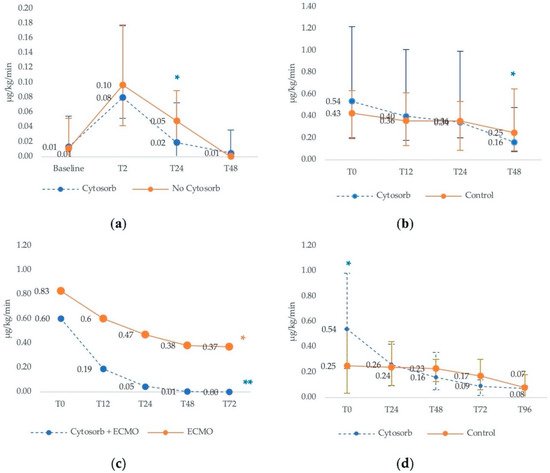
In patients with severe hyperinflammatory shock, neither vasopressors nor fluid resuscitation are effective in quickly reversing shock. Given the pathophysiological background, cytokine removal through hemoadsorption might be beneficial for patients showing resistance to resuscitation, i.e., not stabilized after 6 h of resuscitation and organ support. Cytokine removal can attenuate hyperinflammation and hence vasoplegia, leading to quicker hemodynamic stabilization and shock reversal. The most frequently used criteria to define shock reversal include normalization of serum lactate (<2.2 mmol/L) coupled with a significant (≥90%) reduction in norepinephrine dose requirements [9][16].
2. Results from Clinical Articles
2.1. Analysis of Studies with Control Cohorts
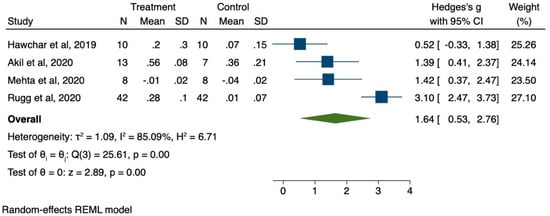
| Study | Design | Indication | Cytosorb, n | Control, n | Total |
|---|---|---|---|---|---|
| Mehta et al. [68] | Observational | Aortic surgery | 8 | 8 | 16 |
| Hawchar et al. [55] | Randomized | Septic shock | 10 | 10 | 20 |
| Akil et al. [66] | Observational | Septic shock | 13 | 7 | 20 |
| Rugg et al. [70] | Observational | Septic shock | 42 | 42 | 84 |
| Total | - | - | 73 | 67 | 140 |
2.2. Pooled Analysis
-
Small effect = 0.2;
-
Medium effect = 0.5;
-
Large effect = 0.8.
3. Insights into Cytosorb
| Vasopressor | Dose | Potential Side Effects |
|---|---|---|
| Norepinephrine (noradrenaline) | 0.05–0.1 mcg/kg/min | Acute glaucoma; anxiety; arrhythmias; asthenia; cardiomyopathy; confusion; dyspnea; extravasation necrosis; gangrene; headache; heart failure; hypovolemia; hypoxia; injection site necrosis; insomnia; ischemia; increased myocardial contractility; nausea; palpitations; peripheral ischemia; psychotic disorder; respiratory failure; tremor; urinary retention; vomiting |
| Dopamine | Up to 20 mcg/kg/min | Angina pectoris; anxiety; arrhythmias; azotemia; cardiac conduction disorder; dyspnea; gangrene; headache; hypertension; mydriasis; nausea; palpitations; piloerection; polyuria; tremor; vasoconstriction; vomiting |
| Epinephrine (adrenaline) | 0.01–0.1 mcg/kg/min | Angina pectoris; angle closure glaucoma; anxiety; appetite decreased; arrhythmias; asthenia; CNS; hemorrhage; confusion; dizziness; dry mouth; dyspnea; headache; hepatic necrosis; hyperglycemia; hyperhidrosis; hypersalivation; hypertension (increased risk of cerebral hemorrhage); hypokalemia; injection site necrosis; insomnia; intestinal necrosis; metabolic acidosis; mydriasis; myocardial infarction; nausea; pallor; palpitations; peripheral coldness; psychosis; pulmonary edema (on excessive dosage or extreme sensitivity); renal necrosis; soft tissue necrosis; tremor; urinary disorders; vomiting |
| Vasopressin | 0.01–0.07 units/min | Abdominal pain; angina pectoris; bronchospasm; cardiac arrest; chest pain; diarrhea; pain; flatulence; fluid imbalance; gangrene; headache; hyperhidrosis; hypertension; musculoskeletal chest pain; nausea; pallor; peripheral ischemia; tremor; urticaria; vomiting; vertigo |
| Dobutamine | 2.5–10 mcg/kg/min | Arrhythmias; bronchospasm; chest pain; dyspnea; eosinophilia; fever; headache; localized inflammation; ischemic heart disease; nausea; palpitations; platelet aggregation inhibition (on prolonged administration); skin reactions; urinary urgency; vasoconstriction |
Appendix A
Table A1 reports details of studies included in this review.| Reference | Type of Patients | Num of pts | Avg. Num of Adsorbers | Time Frame for Use | End Points | Results |
|---|---|---|---|---|---|---|
| Mitzner et al. [49] | Septic shock | 1 | 1 | 24 h | None stated | 86.67% reduction in norepinephrine use in 24 h |
| Hetz et al. [50] | Septic Shock | 1 | 3 | 24 h each | None stated | 83.05% reduction in norepinephrine use in first 24 h |
| Frimmel et al. [51] | Viral shock, ALF | 1 | 1 | 24 h | None stated | 58.33% reduction in norepinephrine use in 24 h |
| Hinz et al. [52] | Septic shock, ALF | 1 | 3 | 1. 24 h, 2. 2.6 h, 3. 5 days later 24 h | None stated | 76.25% reduction over 3 days |
| Traegar et al. [53] | Septic shock, ARDS | 1 | 3 | 1. 20 h, 2. 35 h, 3. 29 h | None stated | 50% increase in norepinephrine use after 1st treatment, 66.6% decrease on 2nd treatment, 100% decrease day 3. |
| Van der Linde et al. [54] | Septic shock, ARDS | 1 | 1 | 24 h | None stated | 100% reduction in norepinephrine use |
| Marek et al. [55] | Cardiogenic shock, post cardiac arrest | 1 | 4 | 4 × 24 h | None stated | 36% reduction in norepinephrine on day 1, 31% reduction day 2, 34% reduction day 3 |
| Friesecke et al. [12] | Refractory septic shock | 20 | 3 | 3 × 24 h | Primary endpoint; change in norepinephrine requirement after 6 and 12 h of treatment with CytoSorb compared with start | 51% reduction in norepinephrine in first 24 h |
| Napp et al. [56] | Acute poisoning/intoxication | 1 | 2 | 2 × 24 h | None stated | 50% reduction in norepinephrine day 1, 100% reduction day 2 |
| Steltzer et al. [57] | Septic Shock | 1 | 6 | 12 h each | None stated | 36.6% reduction in norepinephrine requirement on day 1 |
| Eid et al. [58] | Necrotizing fasciitis | 1 | 2 | 2 × 24 h | None stated | 95% reduction in norepinephrine requirements over 48 h |
| Nemeth et al. [59] | Cardiac transplant | 24 | 24 | Intra-op use | Primary outcome: hemodynamic stability and vasopressor demand during first 48 h post-op; magnitude of postoperative inflammatory response (PCT and CRP) | 57% reduction in norepinephrine requirements on day 1 |
| Nemeth et al. [60] | Septic shock, cardiogenic shock | 1 | 1 | 1 × 24 h | None stated | 100% reduction in norepinephrine in 24 h |
| Dogan et al. [61] | Cardiogenic shock | 1 | 9 | 9 × 24 h with 23 day pause | None stated | 61.54% reduction in norepinephrine on day 1, 12.5% increase day 2, 33.3% reduction day 3, 8.33% reduction day 4 |
| Leonardis et al. [62] | Pneumococcal Sepsis Pt 1 |
1 | 4 | Over 68 h | None stated | Initial increase in norepinephrine on day 1 (150%), 20% decrease day 2, 50% decrease day3, 75% decrease day 4 |
| Leonardis et al. [62] | Meningococcal Sepsis Pt 3 |
1 | 2 | Over 32 h | None stated | 60% decrease in norepinephrine on day 1 and 100% decrease on day 2 |
| Leonardis et al. [62] | Meningococcal Sepsis Pt 4 |
1 | 4 | 4 × 24 h | None stated | 20% decrease on day on of norepinephrine, 37.5% decrease day 2, 20% decrease day 3, 100% decrease day 4 |
| Leonardis et al. [62] | Meningococcal Sepsis Pt 4 |
1 | 4 | 4 × 24 h | None stated | 20% decrease on day on of norepinephrine, 37.5% decrease day 2, 20% decrease day 3, 100% decrease day 4 |
| Hawchar et al. [63] | Septic shock | 10 | 1 | 1 × 24 h | Organ dysfunction and inflammatory response | 37% reduction seen in norepinephrine on day 1 |
| Kuhne et al. [64] | Intra-op cardiac surgery | 10 | 1 | Intra-op use only | None stated | 37.5% reduction in norepinephrine use |
| Kuhne et al. [64] | Intra- and post-op cardiac Surgery | 10 | 1 | Intra- and post-op use for 72 h | None stated | 75% reduction in norepinephrine use |
| Poli et al. [65] | Septic shock | 1 | 4 | 1 × 9 h, 3 × 24 h | None stated | 0% reduction norepinephrine use day 1, 80% day 2, 50% on day 3, 100% on day 4 |
| Perez et al. [66] | Pediatric cardiogenic shock | 1 | 1 | 1 × 72 h | None stated | 27% reduction norepinephrine use day 1, 45% reduction day 2, 14% reduction day 3, 100% reduction day 4 |
| Frimmel et al. [67] | Septic shock, ALF, HLH. Pt 1 | 1 | 1 | 24 h | None stated | 58% reduction in norepinephrine |
| Frimmel et al. [67] | Septic shock, HLH Pt 2 |
1 | 2 | 2 × 24 h | None stated | 0% reduction in norepinephrine on day 1, 28.6% reduction on day 2, 100% reduction day 3 |
| De Schryven et al. [68] | Acute poisoning/intoxication | 1 | 1 | Not stated | None stated | 92.3 reduction in norepinephrine over 3 days |
| Klinkmann et al. [69] | Fungal sepsis | 1 | 1 | 20 h | None stated | 78% reduction in norepinephrine use on first day |
| Dimski et al. [70] | Septic shock | 11 | 1 | 1 × 24 h | Primary endpoint: feasibility of combined CytoSorb/CVVHD treatment with RCA |
66% reduction in norepinephrine use on first day |
| Traegar at al. [71] | Cardiogenic shock post cardiac surgery | 23 | 2 | Various lengths of time | None stated | 87% reduction in norepinephrine use on day 1, 80% reduction on day 2 |
| Stahl et al. [72] | Cytokine release syndrome | 1 | 5 | Various lengths of time | None stated | 47% decrease in norepinephrine on day 1, 57% reduction on day 2 |
| Dilken et al. [73] | Myoglobinemia | 1 | 3 | 12-hourly | None stated | 8% reduction in norepinephrine on day 1, 34% reduction on day 2 |
| Akil et al. [74] | Septic shock, ARDS | 13 | 2 | 24 h | 30 day mortality | 92.54% reduction in norepinephrine on day 1, 89% reduction on day 2, 100% reduction on day 3 |
| Wallet et al. [75] | Cytokine release syndrome | 1 | 2 | 24 h | None stated | 100% reduction in norepinephrine on day 1 |
| Mehta et al. [76] | Major aortic surgery | 8 | 1 | Intr-op use only | Changes in inflammatory markers | 77% reduction in norepinephrine on day 1, 74% reduction on day 2 |
| Alharthy et al. [77] | COVID-19, acute kidney injury | 50 | 2 | 2 × 24 h | None stated | 100% reduction in norepinephrine over 2 days in survivors |
| Rugg et al. [78] | Septic Shock | 42 | 1 | 24 h | None Stated | 52% reduction in norepinephrine on day 1, 54% reduction on day 2 |
| Rieder et al. [79] | ARDS and ECMO | 9 | 3 | 3 × 24 h | None Stated | 37.5% reduction in norepinephrine on day 1, 50% day 2 and 100% by day 3. |
| Boss et al. [80] | Septic shock after cardiac surgery | 98 | 1 | 24 h | None Stated | 51.02% reduction in norepinephrine use, average use at least 15 h |
References
- Cecconi, M.; De Backer, D.; Antonelli, M.; Beale, R.; Bakker, J.; Hofer, C.; Jaeschke, R.; Mebazaa, A.; Pinsky, M.R.; Teboul, J.L.; et al. Consensus on circulatory shock and hemodynamic monitoring. Task force of the European Society of Intensive Care Medicine. Intensive Care Med. 2014, 40, 1795–1815.
- Mitzner, S.R.; Gloger, M.; Henschel, J.; Koball, S. Improvement of hemodynamic and inflammatory parameters by combined hemoadsorption and hemodiafiltration in septic shock: A case report. Blood Purif. 2013, 35, 314–315.
- Hetz, H.; Berger, R.; Recknagel, P.; Steltzer, H. Septic shock secondary to beta-hemolytic streptococcus-induced necrotizing fasciitis treated with a novel cytokine adsorption therapy. Int. J. Artif. Organs. 2014, 37, 422–426.
- Frimmel, S.; Schipper, J.; Henschel, J.; Yu, T.T.; Mitzner, S.R.; Koball, S. First description of single-pass albumin dialysis combined with cytokine adsorption in fulminant liver failure and hemophagocytic syndrome resulting from generalized herpes simplex virus 1 infection. Liver Transpl. 2014, 20, 1523–1524.
- Hinz, B.; Jauch, O.; Noky, T.; Friesecke, S.; Abel, P.; Kaiser, R. CytoSorb, a novel therapeutic approach for patients with septic shock: A case report. Int. J. Artif. Organs. 2015, 38, 461–464.
- Traeger, K.; Schutz, C.; Fischer, G.; Schroder, J.; Skrabal, C.; Liebold, A.; Reinelt, H. Cytokine Reduction in the Setting of an ARDS-Associated Inflammatory Response with Multiple Organ Failure. Case Rep. Crit. Care 2016, 2016, 9852073.
- Van der Linde, G.W.; Grootendorst, A. First case of toxic shock treated with haemoadsorption by CytoSorb® in the Netherlands. Neth.J. Crit. Care 2016, 24, 27–29.
- Marek, S.; Gamper, G.; Reining, G.; Bergmann, P.; Mayr, H.; Kliegel, A. ECMO and cytokine removal for bridging to surgery in a patient with ischemic ventricular septal defect—A case report. Int. J. Artif. Organs. 2017, 40, 526–529.
- Friesecke, S.; Stecher, S.S.; Gross, S.; Felix, S.B.; Nierhaus, A. Extracorporeal cytokine elimination as rescue therapy in refractory septic shock: A prospective single-center study. J Artif. Organs 2017, 20, 252–259.
- Napp, L.C.; Vogel-Claussen, J.; Schafer, A.; Haverich, A.; Bauersachs, J.; Kuhn, C.; Tongers, J. First-in-Man Fully Percutaneous Complete Bypass of Heart and Lung. JACC Cardiovasc. Interv. 2017, 10, e231–e233.
- Steltzer, H.; Grieb, A.; Mostafa, K.; Berger, R. Use of CytoSorb in Traumatic Amputation of the Forearm and Severe Septic Shock. Case Rep. Crit. Care 2017, 2017, 8747616.
- Eid, M.; Fouquet, O.; Darreau, C.; Pierrot, M.; Kouatchet, A.; Mercat, A.; Baufreton, C. Successfully treated necrotizing fasciitis using extracorporeal life support combined with hemoadsorption device and continuous renal replacement therapy. Int. J. Artif. Organs. 2018, 41, 178–182.
- Nemeth, E.; Kovacs, E.; Racz, K.; Soltesz, A.; Szigeti, S.; Kiss, N.; Csikos, G.; Koritsanszky, K.B.; Berzsenyi, V.; Trembickij, G.; et al. Impact of intraoperative cytokine adsorption on outcome of patients undergoing orthotopic heart transplantation—An observational study. Clin. Transpl. 2018, 32, e13211.
- Nemeth, E.; Szigeti, S.; Varga, T.; Daroczi, L.; Barati, Z.; Merkely, B.; Gal, J. Continuous cytokine haemoadsorption incorporated into a venoarterial ECMO circuit for the management of postcardiotomy cardiogenic and septic shock—A case report. Perfusion 2018, 33, 595–596.
- Dogan, G.; Hanke, J.; Puntigam, J.; Haverich, A.; Schmitto, J.D. Hemoadsorption in cardiac shock with biventricular failure and giant-cell myocarditis: A case report. Int. J. Artif. Organs. 2018, 41, 474–479.
- Leonardis, F.; De Angelis, V.; Frisardi, F.; Pietrafitta, C.; Riva, I.; Martino Valetti, T.; Broletti, V.; Marchesi, G.; Menato, L.; Nani, R.; et al. Effect of hemoadsorption for cytokine removal in pneumococcal and meningococcal sepsis. Case Rep. Crit. Care 2018, 2018, 1205613.
- Hawchar, F.; László, I.; Öveges, N.; Trásy, D.; Ondrik, Z.; Molnar, Z. Extracorporeal cytokine adsorption in septic shock: A proof of concept randomized, controlled pilot study. J. Crit. Care 2019, 49, 172–178.
- Kühne, L.U.; Binczyk, R.; Riess, F.C. Comparison of intraoperative versus intraoperative plus postoperative hemoadsorption therapy in cardiac surgery patients with endocarditis. Int. J. Artif. Organs 2019, 42, 194–200.
- Poli, E.C.; Simoni, C.; Andre, P.; Buclin, T.; Longchamp, D.; Perez, M.H.; Ferry, T.; Schneider, A.G. Clindamycin clearance during Cytosorb((R)) hemoadsorption: A case report and pharmacokinetic study. Int. J. Artif. Organs 2019, 42, 258–262.
- Perez, M.H.; Maitre, G.; Longchamp, D.; Amiet, V.; Natterer, J.; Ferry, T.; Schneider, A.; Plaza Wuthrich, S.; Di Bernardo, S. CytoSorb((R)) hemoadsorption and mechanical circulatory support in a newborn with refractory shock after congenital heart surgery. Int. J. Artif. Organs 2019, 42, 521–524.
- Frimmel, S.; Hinz, M.; Schipper, J.; Bogdanow, S.; Mitzner, S.; Koball, S. Cytokine adsorption is a promising tool in the therapy of hemophagocytic lymphohistiocytosis. Int. J. Artif. Organs 2019, 42, 658–664.
- De Schryver, N.; Hantson, P.; Haufroid, V.; Dechamps, M. Cardiogenic Shock in a Hemodialyzed Patient on Flecainide: Treatment with Intravenous Fat Emulsion, Extracorporeal Cardiac Life Support, and CytoSorb(R) Hemoadsorption. Case Re.p Cardiol 2019, 2019, 1905871.
- Klinkmann, G.; Stope, M.B.; Meyer, A. Cytokine adsorption as a promising option for septic shock and multiple organ failure due to Candida infection and decompensated type 1 diabetes mellitus. Artif. Organs 2020, 44, 522–525.
- Dimski, T.; Brandenburger, T.; Slowinski, T.; Kindgen-Milles, D. Feasibility and safety of combined cytokine adsorption and continuous veno-venous hemodialysis with regional citrate anticoagulation in patients with septic shock. Int. J. Artif. Organs 2020, 43, 10–16.
- Trager, K.; Skrabal, C.; Fischer, G.; Schroeder, J.; Marenski, L.; Liebold, A.; Reinelt, H.; Datzmann, T. Hemoadsorption treatment with CytoSorb((R)) in patients with extracorporeal life support therapy: A case series. Int. J. Artif. Organs 2020, 43, 422–429.
- Stahl, K.; Schmidt, B.M.W.; Hoeper, M.M.; Skripuletz, T.; Mohn, N.; Beutel, G.; Eder, M.; Welte, T.; Ganser, A.; Falk, C.S.; et al. Extracorporeal cytokine removal in severe CAR-T cell associated cytokine release syndrome. J. Crit. Care 2020, 57, 124–129.
- Dilken, O.; Ince, C.; van der Hoven, B.; Thijsse, S.; Ormskerk, P.; de Geus, H.R.H. Successful Reduction of Creatine Kinase and Myoglobin Levels in Severe Rhabdomyolysis Using Extracorporeal Blood Purification (CytoSorb(R)). Blood Purif. 2020, 49, 743–747.
- Akil, A.; Ziegeler, S.; Reichelt, J.; Rehers, S.; Abdalla, O.; Semik, M.; Fischer, S. Combined Use of CytoSorb and ECMO in Patients with Severe Pneumogenic Sepsis. Thorac. Cardiovasc. Surg. 2021, 69, 246–251.
- Wallet, F.; Bachy, E.; Vassal, O.; Friggeri, A.; Bohe, J.; Garnier, L.; Salles, G.; Allaouchiche, B. Extracorporeal cytokine adsorption for treating severe refractory cytokine release syndrome (CRS). Bone Marrow Transpl. 2020, 55, 2052–2055.
- Mehta, Y.; Singh, A.; Singh, A.; Gupta, A.; Bhan, A. Modulating the Inflammatory Response With Hemadsorption (CytoSorb) in Patients Undergoing Major Aortic Surgery. J. Cardiothorac. Vasc. Anesth. 2021, 35, 673–675.
- Alharthy, A.; Faqihi, F.; Memish, Z.A.; Balhamar, A.; Nasim, N.; Shahzad, A.; Tamim, H.; Alqahtani, S.A.; Brindley, P.G.; Karakitsos, D. Continuous renal replacement therapy with the addition of CytoSorb® cartridge in critically ill patients with COVID-19 plus acute kidney injury: A case-series. Artif. Organs 2021, 45, E101–E112.
- Rugg, C.; Klose, R.; Hornung, R.; Innerhofer, N.; Bachler, M.; Schmid, S.; Fries, D.; Ströhle, M. Hemoadsorption with CytoSorb in Septic Shock Reduces Catecholamine Requirements and In-Hospital Mortality: A Single-Center Retrospective ‘Genetic’ Matched Analysis. Biomedicines 2020, 8, 539.
- Rieder, M.; Duerschmied, D.; Zahn, T.; Lang, C.; Benk, C.; Lother, A.; Biever, P.; Bode, C.; Wengenmayer, T.; Staudacher, D.; et al. Cytokine Adsorption in Severe Acute Respiratory Failure Requiring Veno-Venous Extracorporeal Membrane Oxygenation. ASAIO J. 2021, 67, 332–338.
- Boss, K.; Jahn, M.; Wendt, D.; Haidari, Z.; Demircioglu, E.; Thielmann, M.; Ruhparwar, A.; Kribben, A.; Tyczynski, B. Extracorporeal cytokine adsorption: Significant reduction of catecholamine requirement in patients with AKI and septic shock after cardiac surgery. PLoS ONE 2021, 16, e0246299.
- Saller, T.; Hagl, C.; Woitsch, S.; Li, Y.; Niedermayer, S.; Born, F.; Luehr, M.; Kammerer, T.; Pichlmaier, M.; Scheiermann, P.; et al. Haemadsorption improves intraoperative haemodynamics and metabolic changes during aortic surgery with hypothermic circulatory arrest. Eur. J. Cardiothorac. Surg. 2019, 56, 731–737.
- Rieder, M.; Wengenmayer, T.; Staudacher, D.; Duerschmied, D.; Supady, A. Cytokine adsorption in patients with severe COVID-19 pneumonia requiring extracorporeal membrane oxygenation. Crit. Care 2020, 24, 435.
- Mezger, M.; Eitel, I.; Ensminger, S.; Pogorzalek, D.; Huang, Z.; Graf, T. Sequential Use of Hemadsorption Using Cytosorb® and Biosky® Filter-Technology in A COVID-19 Patient Suffering from Severe ARDS. Arch. Clin. Med. Case Rep. 2020, 4, 969–977.
- Prakash, A.; Garg, V.; Mittal, D.K.; Upadhyay, A.B. CytoSorb in the management of severe septic shock after coronary artery bypass graft surgery. Heart India 2020, 8, 151–153.
- Alharthy, A.; Balhamar, A.; Faqihi, F.; Nasim, N.; Noor, A.; Alqahtani, S.A.; Memish, Z.A.; Karakitsos, D. COVID-19 presenting as acute abdomen and sepsis: A rare case-report. New Microbes New Infect. 2020, 38, 100818.
- Taccone, F.S.; Gardette, M.; Creteur, J.; Brasseur, A.; Lorent, S.; Grimaldi, D. Hemoadsorption to treat severe iatrogenic intoxication with Patent Blue: A case report. J. Med. Case Rep. 2021, 15, 63.
- Zickler, D.; Nee, J.; Arnold, T.; Schroder, T.; Slowinski, T.; Eckardt, K.U.; Korner, R.; Kruse, J.M. Use of Hemoadsorption in Patients With Severe Intoxication Requiring Extracorporeal Cardiopulmonary Support-A Case Series. ASAIO J. 2021.
- Mitzner, S.R.; Gloger, M.; Henschel, J.; Koball, S. Improvement of hemodynamic and inflammatory parameters by combined hemoadsorption and hemodiafiltration in septic shock: A case report. Blood Purif. 2013, 35, 314–315.
- Hetz, H.; Berger, R.; Recknagel, P.; Steltzer, H. Septic shock secondary to beta-hemolytic streptococcus-induced necrotizing fasciitis treated with a novel cytokine adsorption therapy. Int. J. Artif. Organs. 2014, 37, 422–426.
- Frimmel, S.; Schipper, J.; Henschel, J.; Yu, T.T.; Mitzner, S.R.; Koball, S. First description of single-pass albumin dialysis combined with cytokine adsorption in fulminant liver failure and hemophagocytic syndrome resulting from generalized herpes simplex virus 1 infection. Liver Transpl. 2014, 20, 1523–1524.
- Hinz, B.; Jauch, O.; Noky, T.; Friesecke, S.; Abel, P.; Kaiser, R. CytoSorb, a novel therapeutic approach for patients with septic shock: A case report. Int. J. Artif. Organs. 2015, 38, 461–464.
- Traeger, K.; Schutz, C.; Fischer, G.; Schroder, J.; Skrabal, C.; Liebold, A.; Reinelt, H. Cytokine Reduction in the Setting of an ARDS-Associated Inflammatory Response with Multiple Organ Failure. Case Rep. Crit. Care 2016, 2016, 9852073.
- Van der Linde, G.W.; Grootendorst, A. First case of toxic shock treated with haemoadsorption by CytoSorb® in the Netherlands. Neth.J. Crit. Care 2016, 24, 27–29.
- Marek, S.; Gamper, G.; Reining, G.; Bergmann, P.; Mayr, H.; Kliegel, A. ECMO and cytokine removal for bridging to surgery in a patient with ischemic ventricular septal defect—A case report. Int. J. Artif. Organs. 2017, 40, 526–529.
- Napp, L.C.; Vogel-Claussen, J.; Schafer, A.; Haverich, A.; Bauersachs, J.; Kuhn, C.; Tongers, J. First-in-Man Fully Percutaneous Complete Bypass of Heart and Lung. JACC Cardiovasc. Interv. 2017, 10, e231–e233.
- Steltzer, H.; Grieb, A.; Mostafa, K.; Berger, R. Use of CytoSorb in Traumatic Amputation of the Forearm and Severe Septic Shock. Case Rep. Crit. Care 2017, 2017, 8747616.
- Eid, M.; Fouquet, O.; Darreau, C.; Pierrot, M.; Kouatchet, A.; Mercat, A.; Baufreton, C. Successfully treated necrotizing fasciitis using extracorporeal life support combined with hemoadsorption device and continuous renal replacement therapy. Int. J. Artif. Organs. 2018, 41, 178–182.
- Nemeth, E.; Kovacs, E.; Racz, K.; Soltesz, A.; Szigeti, S.; Kiss, N.; Csikos, G.; Koritsanszky, K.B.; Berzsenyi, V.; Trembickij, G.; et al. Impact of intraoperative cytokine adsorption on outcome of patients undergoing orthotopic heart transplantation—An observational study. Clin. Transpl. 2018, 32, e13211.
- Nemeth, E.; Szigeti, S.; Varga, T.; Daroczi, L.; Barati, Z.; Merkely, B.; Gal, J. Continuous cytokine haemoadsorption incorporated into a venoarterial ECMO circuit for the management of postcardiotomy cardiogenic and septic shock—A case report. Perfusion 2018, 33, 595–596.
- Dogan, G.; Hanke, J.; Puntigam, J.; Haverich, A.; Schmitto, J.D. Hemoadsorption in cardiac shock with biventricular failure and giant-cell myocarditis: A case report. Int. J. Artif. Organs. 2018, 41, 474–479.
- Leonardis, F.; De Angelis, V.; Frisardi, F.; Pietrafitta, C.; Riva, I.; Martino Valetti, T.; Broletti, V.; Marchesi, G.; Menato, L.; Nani, R.; et al. Effect of hemoadsorption for cytokine removal in pneumococcal and meningococcal sepsis. Case Rep. Crit. Care 2018, 2018, 1205613.
- Hawchar, F.; László, I.; Öveges, N.; Trásy, D.; Ondrik, Z.; Molnar, Z. Extracorporeal cytokine adsorption in septic shock: A proof of concept randomized, controlled pilot study. J. Crit. Care 2019, 49, 172–178.
- Kühne, L.U.; Binczyk, R.; Riess, F.C. Comparison of intraoperative versus intraoperative plus postoperative hemoadsorption therapy in cardiac surgery patients with endocarditis. Int. J. Artif. Organs 2019, 42, 194–200.
- Poli, E.C.; Simoni, C.; Andre, P.; Buclin, T.; Longchamp, D.; Perez, M.H.; Ferry, T.; Schneider, A.G. Clindamycin clearance during Cytosorb((R)) hemoadsorption: A case report and pharmacokinetic study. Int. J. Artif. Organs 2019, 42, 258–262.
- Perez, M.H.; Maitre, G.; Longchamp, D.; Amiet, V.; Natterer, J.; Ferry, T.; Schneider, A.; Plaza Wuthrich, S.; Di Bernardo, S. CytoSorb((R)) hemoadsorption and mechanical circulatory support in a newborn with refractory shock after congenital heart surgery. Int. J. Artif. Organs 2019, 42, 521–524.
- Frimmel, S.; Hinz, M.; Schipper, J.; Bogdanow, S.; Mitzner, S.; Koball, S. Cytokine adsorption is a promising tool in the therapy of hemophagocytic lymphohistiocytosis. Int. J. Artif. Organs 2019, 42, 658–664.
- De Schryver, N.; Hantson, P.; Haufroid, V.; Dechamps, M. Cardiogenic Shock in a Hemodialyzed Patient on Flecainide: Treatment with Intravenous Fat Emulsion, Extracorporeal Cardiac Life Support, and CytoSorb(R) Hemoadsorption. Case Re.p Cardiol 2019, 2019, 1905871.
- Klinkmann, G.; Stope, M.B.; Meyer, A. Cytokine adsorption as a promising option for septic shock and multiple organ failure due to Candida infection and decompensated type 1 diabetes mellitus. Artif. Organs 2020, 44, 522–525.
- Dimski, T.; Brandenburger, T.; Slowinski, T.; Kindgen-Milles, D. Feasibility and safety of combined cytokine adsorption and continuous veno-venous hemodialysis with regional citrate anticoagulation in patients with septic shock. Int. J. Artif. Organs 2020, 43, 10–16.
- Trager, K.; Skrabal, C.; Fischer, G.; Schroeder, J.; Marenski, L.; Liebold, A.; Reinelt, H.; Datzmann, T. Hemoadsorption treatment with CytoSorb((R)) in patients with extracorporeal life support therapy: A case series. Int. J. Artif. Organs 2020, 43, 422–429.
- Stahl, K.; Schmidt, B.M.W.; Hoeper, M.M.; Skripuletz, T.; Mohn, N.; Beutel, G.; Eder, M.; Welte, T.; Ganser, A.; Falk, C.S.; et al. Extracorporeal cytokine removal in severe CAR-T cell associated cytokine release syndrome. J. Crit. Care 2020, 57, 124–129.
- Dilken, O.; Ince, C.; van der Hoven, B.; Thijsse, S.; Ormskerk, P.; de Geus, H.R.H. Successful Reduction of Creatine Kinase and Myoglobin Levels in Severe Rhabdomyolysis Using Extracorporeal Blood Purification (CytoSorb(R)). Blood Purif. 2020, 49, 743–747.
- Akil, A.; Ziegeler, S.; Reichelt, J.; Rehers, S.; Abdalla, O.; Semik, M.; Fischer, S. Combined Use of CytoSorb and ECMO in Patients with Severe Pneumogenic Sepsis. Thorac. Cardiovasc. Surg. 2021, 69, 246–251.
- Wallet, F.; Bachy, E.; Vassal, O.; Friggeri, A.; Bohe, J.; Garnier, L.; Salles, G.; Allaouchiche, B. Extracorporeal cytokine adsorption for treating severe refractory cytokine release syndrome (CRS). Bone Marrow Transpl. 2020, 55, 2052–2055.
- Mehta, Y.; Singh, A.; Singh, A.; Gupta, A.; Bhan, A. Modulating the Inflammatory Response With Hemadsorption (CytoSorb) in Patients Undergoing Major Aortic Surgery. J. Cardiothorac. Vasc. Anesth. 2021, 35, 673–675.
- Alharthy, A.; Faqihi, F.; Memish, Z.A.; Balhamar, A.; Nasim, N.; Shahzad, A.; Tamim, H.; Alqahtani, S.A.; Brindley, P.G.; Karakitsos, D. Continuous renal replacement therapy with the addition of CytoSorb® cartridge in critically ill patients with COVID-19 plus acute kidney injury: A case-series. Artif. Organs 2021, 45, E101–E112.
- Rugg, C.; Klose, R.; Hornung, R.; Innerhofer, N.; Bachler, M.; Schmid, S.; Fries, D.; Ströhle, M. Hemoadsorption with CytoSorb in Septic Shock Reduces Catecholamine Requirements and In-Hospital Mortality: A Single-Center Retrospective ‘Genetic’ Matched Analysis. Biomedicines 2020, 8, 539.
- Rieder, M.; Duerschmied, D.; Zahn, T.; Lang, C.; Benk, C.; Lother, A.; Biever, P.; Bode, C.; Wengenmayer, T.; Staudacher, D.; et al. Cytokine Adsorption in Severe Acute Respiratory Failure Requiring Veno-Venous Extracorporeal Membrane Oxygenation. ASAIO J. 2021, 67, 332–338.
- Boss, K.; Jahn, M.; Wendt, D.; Haidari, Z.; Demircioglu, E.; Thielmann, M.; Ruhparwar, A.; Kribben, A.; Tyczynski, B. Extracorporeal cytokine adsorption: Significant reduction of catecholamine requirement in patients with AKI and septic shock after cardiac surgery. PLoS ONE 2021, 16, e0246299.
- StataCorp. Stata Statistical Software: Release 16; StataCorp LLC: College Station, TX, USA, 2019.
- Friesenecker, B.E.; Tsai, A.G.; Martini, J.; Ulmer, H.; Wenzel, V.; Hasibeder, W.R.; Intaglietta, M.; Dunser, M.W. Arteriolar vasoconstrictive response: Comparing the effects of arginine vasopressin and norepinephrine. Crit. Care 2006, 10, R75.
- Belletti, A.; Castro, M.L.; Silvetti, S.; Greco, T.; Biondi-Zoccai, G.; Pasin, L.; Zangrillo, A.; Landoni, G. The Effect of inotropes and vasopressors on mortality: A meta-analysis of randomized clinical trials. Br. J. Anaesth. 2015, 115, 656–675.
- Colling, K.P.; Banton, K.L.; Beilman, G.J. Vasopressors in Sepsis. Surg. Infect. (Larchmt) 2018, 19, 202–207.
- Oberbeck, R. Catecholamines: Physiological immunomodulators during health and illness. Curr. Med. Chem. 2006, 13, 1979–1989.
- Jenkins, C.R.; Gomersall, C.D.; Leung, P.; Joynt, G.M. Outcome of patients receiving high dose vasopressor therapy: A retrospective cohort study. Anaesth Intensive Care 2009, 37, 286–289.
- Auchet, T.; Regnier, M.A.; Girerd, N.; Levy, B. Outcome of patients with septic shock and high-dose vasopressor therapy. Ann. Intensive Care 2017, 7, 43.
- Dunser, M.W.; Hasibeder, W.R. Sympathetic overstimulation during critical illness: Adverse effects of adrenergic stress. J. Intensive Care Med. 2009, 24, 293–316.
- Labib, A. Sepsis Care Pathway 2019. Qatar Med. J. 2019, 2019, 4.
- Bangash, M.N.; Kong, M.L.; Pearse, R.M. Use of inotropes and vasopressor agents in critically ill patients. Br. J. Pharmacol. 2012, 165, 2015–2033.
- Lam, S.M.; Lau, A.C.; Lam, R.P.; Yan, W.W. Clinical management of sepsis. Hong Kong Med. J. 2017, 23, 296–305.
