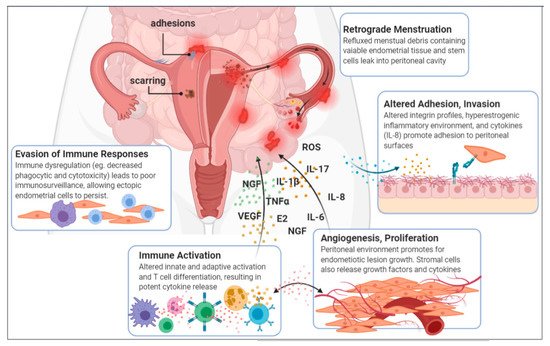
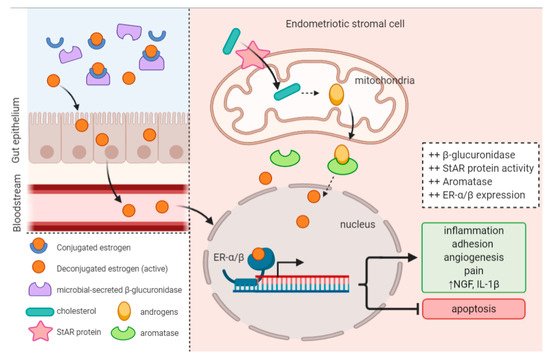
A recent study by Ata et al. sought to compare the vaginal, cervical and gut microbiota composition of women with Stage III/IV endometriosis to healthy controls
[67][95]. Remarkably, they did indeed detect a difference at the genus level. In the cervical microbiota of endometriotic women, they found increased abundance of potentially pathogenic species including
Gardnerella,
Streptococcus,
Escherichia,
Shigella and
Ureaplasma. Stool microbiota of the endometriotic group were
Shigella and
Escherichia dominant. Interestingly, they found a complete absence of
Atopobium, a gynaecologic pathogen, in the vagina and cervix of the endometriotic group. Another study reported high incidence of
Atopobium vaginae in women with endometrial cancer, and suggested that
Atopobium can facilitate intracellular
Porphyromonas infection, leading to disrupted cell regulatory functions and carcinogenic trigger
[68][96]. Conversely, they found
A. vaginae to have lower incidence in women with benign gynaecologic pathologies, suggesting a possible connection through a different mechanism of action, since endometriosis is also a benign gynaecologic pathology
[68][96]. Several other studies have also found that uterine microbiota composition is altered in women with uterine diseases, including endometriosis, demonstrating its clinical relevance
[69][70][71][97,98,99]. For example, researchers found an elevated abundance of Streptococcaceae, Moraxellaceae, Staphylococcaceae and Enterobacteriacea, and lowered Lactobacillacae in endometriotic women
[69][97]. Recently, Hernandes et al. found that, compared with eutopic endometrium, ectopic lesions have higher microbial diversity
[70][98]. In Wei et al.’s attempt to characterize microbiota composition and distribution along the FRT in endometriotic women, they found in conformance that the lower FRT was
Lactobacillus dominant, and significant differences in community diversity appeared and increased from the cervix up into the endometrium and PF
[71][99].
In general, studies to date have consistently found increases in BV-associated bacteria and opportunistic pathogens, and a decrease in
Lactobacillus in the reproductive tract of endometriotic women
[72][73][67][82,83,95].
2.2. Endometriosis Induces Gut Microbiota Alterations
In a study where mice were injected with intraperitoneal endometrial tissue to induce endometriosis, it was demonstrated that after 42 days of endometriotic lesion persistence, a distinct gut microbiota develops
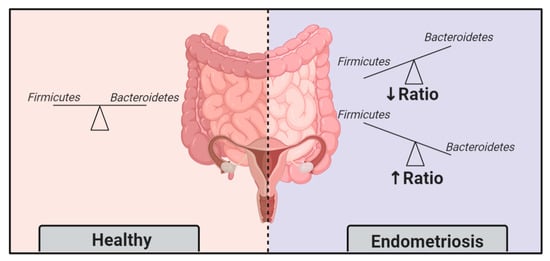
[74][100]. In other words, endometriosis progression was able to change the gut microbiota. Among the observed differences, the nearly doubled Firmicutes/Bacteroidetes ratio in endometriotic mice was discriminative and concrete
[74][100]. A previous study in 2002 also found similar differences in microbiota profiles in rhesus monkeys
[75][101]. Compared to healthy controls, monkeys with endometriosis had lower Lactobacilli and higher gram-negative bacteria
[75][101]. The ratio Firmicutes/Bacteroidetes is widely accepted as a feature of dysbiosis (); hence, these momentous findings support that endometriosis induces gut microbiota alterations.
Figure 3. The Firmicutes/Bacteroidetes ratio, an indicator of dysbiosis, is altered in endometriosis patients.
2.3. Faecal Microbiota Transfer Induces Endometriosis
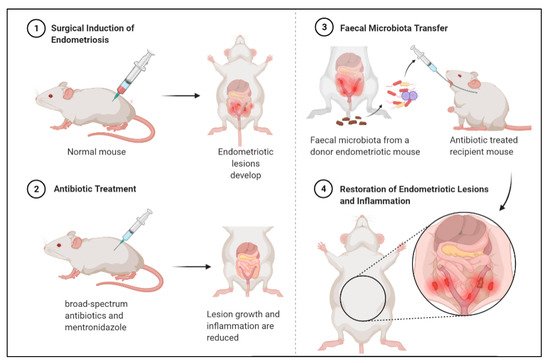
Findings from a compelling mouse model study () support that a distinct gut microbiota promotes endometriosis
[76][102]. In this study, mice were subjected to surgical induction of endometriosis, and then treated with antibiotics which reduced lesion size. Subsequently, they received faecal microbiota transfers from endometriotic mice, which restored lesion growth and associated inflammation
[76][102].
Figure 4. Antibiotic treatment can reduce endometriotic lesion growth and peritoneal inflammation, and subsequent faecal microbiota transfer from diseased mouse can restore lesion growth and inflammation. Mouse experiment demonstrates bidirectional relationship between endometriosis and gut microbiota.
2.4. Diet-Induced Gut Microbiota Changes Reduce Endometriosis Risk
Another interesting finding is that women with a high omega-3 polyunsaturated fatty acids (PUFAs) intake have lower risk for endometriosis
[77][78][103,104]. A similar diet showed anti-inflammatory effects and suppressed endometriotic lesion formation in murine models
[79][80][105,106]. It is reasonable to speculate that this can be at least partially attributed to diet-induced modification of the gut flora. Research has shown that diets high in PUFAs and probiotic supplements may alter the gut flora, and may contribute to the prevention and treatment of various diseases, including osteoporosis and obesity
[81][82][107,108].
3. What Can This Mean for Endometriosis Care
It is evident that bacterial presence in both the gut and uterus plays a major role in endometriosis. But what does this mean for patients? Could the modulation of the microbiota be a therapeutic or preventative approach? Could certain microbial compositions or the presence of microbiota-based biomarkers be used as screening or diagnostic tools?
3.1. Gynaecologic and Obstetric Applications of Microbiota Modulation
Microbiota modulation through antibiotics is already broadly applied in the field of gynaecology and obstetrics
[83][128]. In accordance with ample research showing uterine dysbiosis threatens fertility and pregnancy outcomes, it is suggested that intervention options such as uterine lavage or antibiotics to eradicate microbes or pro/prebiotics and improve the microbiota could be valuable
[83][128]. In a recent clinical setting, broad-spectrum antibiotics and pre/probiotics were employed to achieve
Lactobacillus-dominant uterine microbiota (from previously non-
Lactobacillus-dominant), which led to higher pregnancy rates
[84][129] — A hopeful finding that encourages further investigation of this approach. Moreover, many studies show that treatment of chronic endometritis with antibiotics leads to improved reproductive outcomes
[85][86][87][88][130,131,132,133].
3.2. Treating Endometriosis with Antibiotics
Antibiotics may be a promising approach for treating endometriosis. In fact, broad-spectrum antibiotic treatments have demonstrated efficacy for treating endometriosis in animal models
[76][102]. A recent study found that use of broad-spectrum antibiotics inhibited ectopic lesions, while treatment with metronidazole significantly decreased inflammation and reduced lesion size, possibly by lessening Bacteroidetes presence
[76][102]. Peculiarly, treatment with neomycin did not produce the same results, indicating that lesion-growth-promoting bacteria are metronidazole-sensitive and neomycin-resistant
[76][102].
3.3. Treating Endometriosis with Probiotics
Alternatively, probiotic intervention, the administration of live microorganisms, could be another effective approach
[83][128]. For instance, in randomised, placebo-controlled trials, oral administration of
Lactobacillus has been shown to ameliorate endometriosis-associated pain in women
[89][90][134,135], and reduce endometriotic lesions in mice by increasing IL-12 concentration and NK cell activity
[83][91][92][128,136,137]. Dysbiosis and endometriotic-inflammation leads to impaired NK cell activity, and the probiotic treatment reversed this immune dysregulation.
Lactobacillus probiotic treatment not only improved endometriosis, but is also capable of preventing its growth in rats
[91][136]. These impressive frontiers warrant research and testing.
3.4. A Mechanism for Known Treatments
New research is showing that some known endometriosis treatments may have actually been working through gut microbiota modulation
[93][138]. For example, letrozole, an aromatase inhibitor that reduces estrogen levels and Shaofu Zhuyu decoction (SFZYD), a traditional Chinese medicine that inhibits cellular proliferation, promotes apoptosis, and reduces angiogenesis in ectopic endometrial tissues, have been shown to inhibit the progression of endometriosis and reduce inflammation in mice
[93][138]. In a 2020 study, it was found that both letrozole and SFZYD exert their therapeutic effect in part through restoration of the gut microbiota; they both attenuated the Firmicutes/Bacteroidetes ratio, which was elevated in the untreated endometriotic group, and restored α-diversity and Ruminococcaceae abundance in the gut microbiota
[93][138]. Loss of Ruminococcaceae may exacerbate peritoneal inflammation, as it may be negatively correlated to apoptosis of intestinal epithelial cells and murine IL-6 levels
[94][139]. Therefore, these treatments at least partially function through restoring gut microbiota health.
3.5. Side Effects and Challenges
Unfortunately, the use of antibiotics is known to introduce off-target effects and new interventions such as pre/probiotics have poorly understood pharmacological mechanisms with difficult-to-prove clinical efficacy. For example, it is well known that routine or overuse of antibiotics increases the risk of antimicrobial resistance, which is currently one of the largest threats to global human health
[95][140]. Furthermore, the core FRT microbiota should be well characterised before applying any such treatments
[83][128]. Antibiotics are widely effective in treating infection by reducing abundance or eliminating pathogenic species, however its use alters microbial community profiles, and can create lasting disruptions to healthy microbiotas
[96][97][141,142]. A study examining the urinary microbiotas of women during BV and after oral administration of metronidazole found that while it was an effective treatment, it also significantly decreased the Shannon diversity, an effect which persisted for up to 28 days
[96][141]. Furthermore, the antibiotic treatment created complex changes to the microbiota composition, and healthy microbial community could not be restored
[96][141]. Compared to BV, dysbiosis related to endometriosis is far less studied, and therefore the convoluted effects of antibiotic treatments are largely undetermined. The use of antibiotics to treat endometriosis still requires extensive research, but is an area of potential. There are many unanswered questions regarding its practical use, and the diagnosis of an “abnormal uterine microbiota” to indicate such treatment remains a major hurdle to overcome.
3.6. Opportunities for Diagnostics
Another appealing application of the microbiota-endometriosis relationship lies in diagnostics. So much is yet to be learned about the mechanisms involved, but based on the current state of knowledge that there is an appreciable difference in the microbiotas of women with endometriosis, we can imagine its potential value as a diagnostic or screening tool. Significant differences in the community diversity of cervical microbiota in endometriotic women indicate that cervical samples may be used as an endometriosis risk indicator
[71][99]. A recent study found for the first time that vaginal microbiome profiles could be successfully used to predict the rASRM (revised American Society for Reproductive Medicine) stage of endometriosis
[98][143]. These exciting findings hopefully incite further research into non-invasive diagnostics and screening tools, as traditional techniques are limited and remain a challenge today.